Abstract
Purpose:
Feeding difficulties are common among patients with cerebral palsy (CP) and neuromuscular (NM) scoliosis. We theorize that posterior spinal fusion (PSF) reduces intra-abdominal pressure, resulting in improved feeding and subsequent weight gain. We hypothesized that, among nonambulatory patients with CP and NM scoliosis, we would observe significant gain in weight following PSF.
Methods:
Fifty subjects with nonambulatory CP who underwent PSF for NM scoliosis were included. Age and weight were recorded for the preoperative year; on the day of surgery; and at 6-month, 1-year, and 2-year follow-up. Weights were converted to weight percentiles using CP-specific growth charts. The weight percentile distributions were compared between time points using descriptive statistics as well as regression analysis.
Results:
The average change in weight from the day of surgery to 2-year follow-up was +3.4 percentiles. Patients who started out under the 50th percentile gained an average of 17.3 percentiles in the first year after PSF (p = 0.009). Regression analysis showed that patients with baseline weight <50th percentile tended to gain in weight percentile over the first postoperative year (β = 1.990, p = 0.001). No trend was present among this group prior to surgery (p = 0.692) or during the second postoperative year (p = 0.945). No trends were noted prior to or after surgery for patients with baseline weights ≥50th percentile. No significant association was observed between curve severity (measured by preoperative Cobb angle) and weight change.
Conclusions:
This series is the first to document significant weight gain after PSF for NM scoliosis, supporting the theory that spinal correction improves digestive function.
Level of Evidence:
Prognostic level II.
Introduction
The most severely affected pediatric patients with cerebral palsy (CP) commonly suffer from other comorbidities including undernutrition or malnutrition due to a loss of pharyngeal tone, gastroesophageal reflux, impaired gastrointestinal (GI) motility, or a combination of these. Many such patients require a gastrostomy, gastrojejunostomy, or jejunostomy to maintain caloric intake, and many have considerable spasticity or hyperkinetic movements which may increase caloric demands. In one series, 74% of patients with CP had chronic constipation suggestive of decreased enteric motility, 1 and another study showed that 76% of those with severe/profound CP had moderate/severe oral motor dysfunction limiting oral feeding. 2
In addition to the aforementioned nutritional problems, over 60% of nonambulatory patients with CP develop significant neuromuscular (NM) scoliosis. 3 As the degree of spinal deformity progresses, worsening head balance and pelvic obliquity can make comfortable seating for transportation, upright feeding, and hygiene increasingly difficult. As pelvic obliquity and scoliosis increase in severity, abdominal and intrathoracic volume decreases, which we speculate may exacerbate feeding difficulties. 4 Posterior spinal fusion (PSF) is thought to improve seating posture and thereby ease the difficulty of daily care. 5 As orthopedic practitioners, we have anecdotally noted increased reflux and decreased tolerance of feeds in patients with CP and significant NM scoliosis. As these issues seemingly improve after PSF, we have come to believe that NM scoliosis may have an additive effect on these patients’ nutritional difficulties; we theorize that the untreated spinal deformity decreases the functional volume available in the peritoneal cavity, thereby raising intra-abdominal pressure, contributing to early satiety, and interfering with normal enteric motility. While PSF is known to arrest the progression of the spinal deformity and correct spinal imbalance and pelvic obliquity, there is little-to-no published evidence regarding GI symptoms, body mass, or nutritional variables following PSF. The purpose of this study was to examine patient weights before and after PSF in patients with nonambulatory CP. We hypothesized that patients would gain weight percentiles on the CP growth charts in the first 2 years following PSF.
Methods
After appropriate institutional review board approval, we identified 50 consecutive patients with nonambulatory CP (Gross Motor Function Classification System (GMFCS) IV and V) who had undergone PSF for NM scoliosis at our institution between January 2009 and February 2015. Patients had a minimum of 1 year of clinical follow-up. Subject age, gender, weight, and GMFCS status at the time of surgery were recorded from an electronic health record database. Weight and age data were further cataloged for visits in the year prior to surgery and over the first 25 months postoperatively. Duplicate data entries were removed. The regular use of a gastrostomy tube (G-tube) for enteric feeding leading up to surgery was also recorded. With reference to CP-specific growth charts, 6 weight, age, gender, GMFCS level, and G-tube status were used to define the weight-for-age percentile for each weight measure. Preoperative Cobb angle was measured by two raters (KDB and DJM).
Beginning with all of the weight percentile recordings for each patient, the data set was reduced to leave each patient with at most one datum for each of the following periods: the year prior to surgery, the day of surgery, 6-month follow-up, 1-year follow-up, and 2-year follow-up. The earliest recording in the year prior to surgery was retained as the datum for the year prior to surgery. Each patient had a day-of-surgery weight. The recording from the latest visit ≤7 months postoperatively was retained as the datum for 6-month follow-up. The recording from the latest visit >7 months but ≤13 months postoperatively was retained as the datum for 1-year follow-up. The recording from the latest visit >13 months but ≤25 months postoperatively was retained as the datum for 2-year follow-up. Each subject had four to five weight percentile measures over the study period in the final data set. The weight trend was assessed with descriptive statistics, and linear regression was subsequently performed to assess the temporal trends in weight percentile over the year prior to surgery, the first postoperative year, and the second postoperative year. This was repeated in a stratified fashion based upon weight percentile on the day of surgery.
Stata 14.2 (College Station, Texas, USA) was used for all statistical analysis in this study. A type I error rate of 0.05 was used as a cutoff for statistical significance.
Results
Fifty patients met the inclusion criteria for this study. Eight patients functioned at GMFCS level IV and 42 patients functioned at GMFCS level V. 7 Patients underwent surgery at a median age of 14.5 years (range 6.6–18.3 years). Other baseline demographics are shown in Table 1. Considering weight on the day of surgery, median weight percentile was 70.6 (range 17.1–99). Thirty percent (15 of 50) of patients were below the 50th percentile for weight on the day of surgery. The remainder were at or above the 50th percentile.
Baseline characteristics of the patient sample.
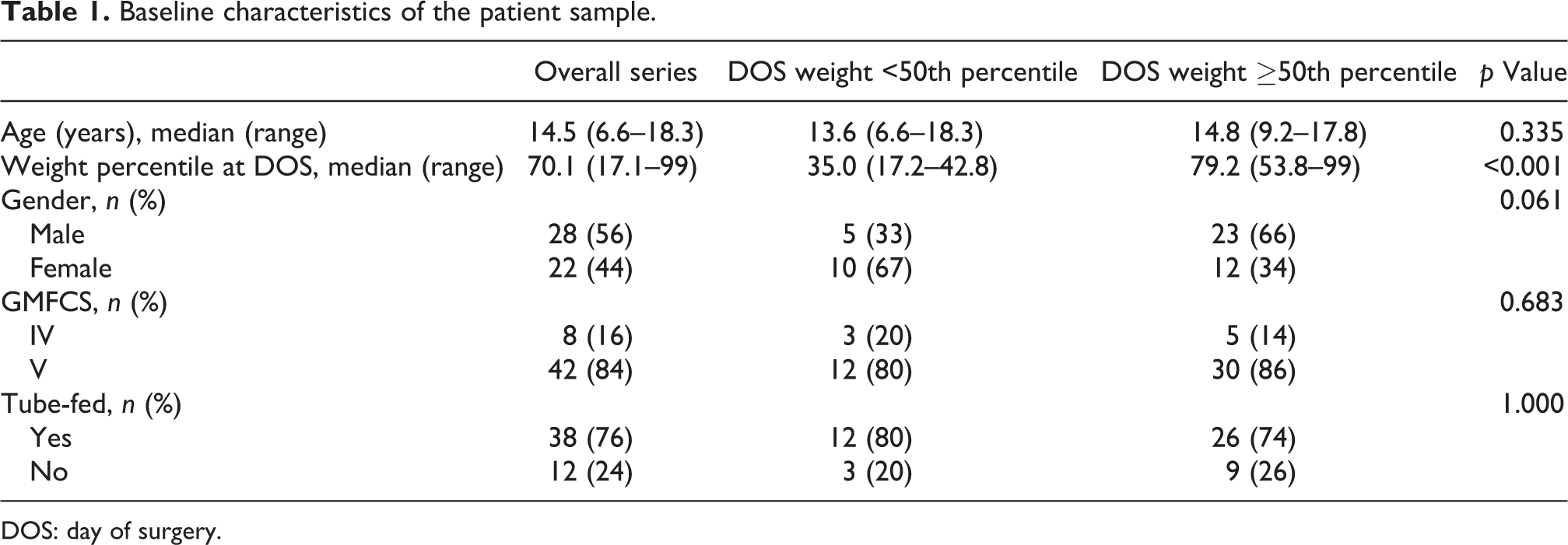
DOS: day of surgery.
At 1-year follow-up, 67% (30 of 45) of patients had gained in weight percentile from the day of surgery, and only 20% (9 of 45) of patients were below the 50th percentile for weight. The average change in weight from the day of surgery to 1-year follow-up was +6.5 percentiles. At 2-year follow-up, 65% (32 of 49) of patients had gained in weight percentile from the day of surgery, and only 24.5% (12 of 49) of patients were below the 50th percentile for weight. The average change in weight from the day of surgery to 2-year follow-up was +3.4 percentiles. Among those who started below the 50th weight percentile, patients gained an average of +17.3 percentiles by 1-year follow-up (p = 0.009) and +13.2 percentiles by 2-year follow-up (p = 0.043). Within this subgroup, regression analysis revealed no significant weight gain in the year prior to surgery (p = 0.692), but there was significant weight gain in the first postoperative year (p = 0.001) which did not persist through the second postoperative year (p = 0.945). Among those patients who started at or above the 50th weight percentile, there was no significant weight gain in the first preoperative, first postoperative, or second postoperative year. These trends are illustrated in Figure 1. The stratified groups (≥50th percentile vs. <50th percentile at baseline) were statistically similar in age, gender, preoperative Cobb angle, GMFCS level, and G-tube feeding status (Table 1). Backward stepwise regression was performed using age, preoperative Cobb angle, G-tube status, and group membership (≥50th percentile vs. <50th percentile) as independent variables. Only group membership in the <50th percentile group was a significant independent predictor of postoperative weight percentile gain (β = 13.4, p = 0.006).

This graph illustrates the trend lines for the two stratified groups during the three defined perioperative time periods. The only statistically significant trend found was during the first postoperative year among patients with baseline weight <50th percentile.
Discussion
Pediatric spine deformity surgeons have anecdotally noted improvements in weight and GI symptoms among patients with CP and NM scoliosis following PSF. While we know that no previously published evidence supports this theory, studies of patients with early onset scoliosis have associated orthopedic intervention using growing rods 8 or Vertical Expandable Prosthetic Titanium Ribs (VEPTRs; Synthes Spine Co., West Chester, PA, USA) 4 with improvement in weight after surgery. In this study, we provide the first published evidence associating PSF for NM scoliosis with weight gain after surgery.
Patients with CP and NM scoliosis routinely receive a preoperative evaluation by a nutritionist and/or gastroenterologist in preparation for surgery and continue to be followed up postoperatively as appropriate. While many of our patients therefore receive additional caloric intake, leading to weight gain, and research indeed shows that enteric feeding leading up to surgery can improve weight status in preparation for spinal surgery, 9 we did not observe an upward trend in weight percentile over the year prior to surgery in our series. This suggests that the postoperative trends in weight observed in our series should not be simply attributed to nutritional supplementation, and physiological changes after surgery may—at least partly—account for postoperative weight changes in the most vulnerable patients.
While this study lacked a true control group that did not receive PSF, by evaluating the weight in the year prior to surgery, each patient served as his/her own control. We noted in our series a postoperative upward trend in weight among patients who started out below the 50th percentile on the CP growth charts. While this weight gain was not observed in the year prior to surgery, it presented in the year after surgery and seemed to “level off” in the second year, suggesting that patient weight had re-equilibrated by this time. As aforementioned, this suggests that realignment of the trunk—possibly resulting in increased peritoneal volume and decreased intra-abdominal pressure—may potentially result in increased space available for food and improved intestinal peristalsis. In theory, this would allow for less regurgitation of abdominal contents and less reflux, although we currently lack the data to state this definitively.
The upward trend in weight percentile observed in the first postoperative year did not reach statistical significance when examining the entire cohort. However, the correlation was readily apparent when restricting analysis to those patients whose weights were below the 50th percentile on the day of surgery. This subset gained an average of over 10 percentiles on the CP growth charts over the first two postoperative years. Our finding that patients with more severe nutritional deficits stand a greater chance to make significant weight gain with surgery complements prior evidence documenting better outcome metrics for patients with lower functional status prior to surgery. 10 We found postoperative gain in weight percentile (compared to the day of surgery) in similar proportions of subjects at 1- and 2-year follow-up, suggesting that—despite no apparent trend in weight percentile during the second postoperative year—weight gain in the first postoperative year did seem to persist beyond this initial time period. Figure 2 details the perioperative courses of two patients, illustrating significant weight gain after PSF.

These two panels (A and B) detail the perioperative courses for two subjects with weight <50th percentile on the day of surgery. These patients exhibited marked increase in weight percentile after surgery, as shown in the graphs at the right. Pre- and postsurgical radiographs are included on the left.
This study has several limitations. First, we detail experience from an urban tertiary care center, limiting the generalizability of our findings. Our sample was also relatively small, which may have limited our statistical power in searching for trends in weight percentile. We believe that using weight percentile instead of raw weight as the primary outcome measure decreased the likelihood that observed increases in weight simply reflected the natural history of growth. However, using percentiles may have opened the project to a ceiling effect bias partially accounting for the greater improvement seen among patients with lower baseline weights (subjects at higher weight percentiles inherently had lower potential for gain in the outcome measure). Recognizing that the majority of patients, especially those felt to be undernourished, received consultation from a nutritionist or gastroenterologist, we were unable to control for treatments they received, the type of practitioner treating them, or the targets and lengths of nutritional supplementation. With respect to other possible outcome variables, although we have anecdotally noted in our practice less complaints of reflux after PSF, such symptoms were too inconsistently documented in the health record to allow for them to be retrospectively examined as outcomes. It is further important to note that outcome data available for this study were limited to weight and not other markers of nutrition such as body composition, serum albumin, proton pump inhibitor use, or volume of oral versus G-tube intake. Finally, while the raw weight of implants in PSF may account for some perioperative weight gain, it cannot account for the different temporal trends seen in the stratified groups.
In conclusion, we believe that PSF itself leads to altered enteric physiology that partially accounts for the weight gain observed in the first postoperative year for our series of patients. In a similar way that spinal intervention in thoracic insufficiency syndrome can increase thoracic volume allowing for improved lung function, we propose that lengthening and straightening of the thoracolumbar spine increases abdominal space allowing for improved enteric motility and peristalsis. We suspect that along with improved mechanical activity in the gut comes improved nutrient digestion and absorption, leading to weight gain. While this study documents significant postoperative weight gain in patients with CP and NM scoliosis who undergo PSF, further study is needed to discern the true mechanisms accounting for this phenomenon. We also look forward to replication of our results in other series in order to better understand the generalizability of our findings.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
