Abstract
Purpose:
The management of bone defects following simple curettage of bone tumors is controversial and in light of the numerous emerging substitutes for bone grafts, we wished to review and report our experience with the use of cancellous allograft bone in the treatment of benign and borderline bone tumors.
Methods:
We reviewed the medical records of 164 consecutive patients with benign or borderline bone tumors treated with simple curettage at our orthopedic oncology center between 2009 and 2013. Postoperative radiological changes were evaluated by a modified Neer’s classification in defects that were subsequently reconstructed with allograft bone (n = 133).
Results:
Simple curettage with subsequent defect filling using allograft bone was the surgical procedure performed in the majority of our patients (81%) and was associated with a low overall 2.5-year local recurrence (LR; 9.8%) and complication rate (7.5%). The radiological appearance of the grafted defects was deemed satisfactory in 85% of cases, with signs of either complete or partial healing present 6–12 months postoperatively. With respect to pathology, we found high rates of LR in giant cell tumors (GCTs) of bone, simple cysts (SCs) in children, and preexisting local recurrent disease. We did not observe any allograft-related complications.
Conclusions:
Simple curettage and bone defect reconstruction with bone allograft is a sufficient treatment for most benign bone lesions and is associated with a low complication rate. For high-risk entities, such as GCTs of bone, SCs in children, and recurrent disease, additional adjuvant treatment could be considered to avoid LR.
Keywords
Introduction
Simple curettage is the most common surgical treatment for benign bone tumors 1 –3 and has been reported as an acceptable method associated with relatively low recurrence rates, even for the treatment of low-grade chondrosarcomas (CS). 4,5 The management of subsequent bone defect, however, remains controversial and treatment options vary greatly from no treatment to use of various filling materials. 6 While satisfactory clinical results have also been shown in defects without grafting, 7,8 residual fracture risk remains a concern as natural bone healing may not be sufficient for all bone defects beyond a certain size. Autogenous cancellous bone is still widely considered the gold standard for the reconstruction of bone defects, due to its potential osteogenetic, osteoinductive, and osteoconductive abilities. However, due to its well-known disadvantages, including donor site morbidity, prolonged operating time, and limited availability, it is often augmented with—or replaced by—allografts, bone cements, or bone graft substitutes, particularly for larger reconstructions. 9 –11 Traditionally, cancellous allograft bone has been the graft material of choice for bone defect reconstruction at our institution for many years, and although curettage and bone grafting is commonly used for the treatment of contained defects in orthopedic tumor surgery in general, only limited data on clinical results and complication rates have been published during recent years (Figure 1). With a view to eventually compare the performance of newer bone graft substitutes to conventional bone grafting in the future, we wished to review and report our experience with the use of simple curettage and cancellous allograft bone in the treatment of benign and borderline bone tumors and cysts.

Upper row: (a) Preoperative X-ray of a GCT in the proximal tibia of a 29-year-old female. (b) Immediate postoperative X-ray confirms complete defect filling with bone graft. (c) X-ray 1 year postoperatively displays graft integration with small radiolucent areas below the lateral subchondral plate and the tibial eminence (MNC grade 2). (d) 5 years postoperatively, the defect appears completely healed (MNC grade 1). Lower row: (e) Preoperative X-ray of FD in the proximal ulna of a 15-year-old girl. (f) Immediate postoperative X-ray shows complete defect filling. (g) X-ray 1 year postoperatively displays complete graft integration (MNC grade 1) and beginning cortical consolidation. (h) X-ray 2 years postoperatively shows signs of progressive graft integration and remodeling. GCT: giant cell tumor; FD: fibrous dysplasia. MNC: Modified Neer’s Classification.
Materials and methods
Study design and patient population
We performed a retrospective review of all patients treated with curettage for benign or borderline bone tumors and cysts in our orthopedic oncology department between January 1, 2009, and December 31, 2013. All relevant data (age, gender, histological diagnosis, grade, size and anatomical location of the lesion, details of the surgical procedure, use of orthopedic implants, weight bearing status, occurrence of postoperative complications, and local recurrence (LR)) were collected from the medical records, the national pathology database, 12 and the national countrywide electronic patient file system. We identified 164 patients (M/F = 79/85, median age = 29 (2–70) years) with a minimum follow-up of 2.7 years (median 5.0 (2.7–7.6) years), who had undergone simple curettage for their primary (n = 151/164 (92%)) or recurrent (n = 13/164 (8%)) bone lesion. We calculated the time to LR, defined as the time from the date of the initial intervention to the date of histological verification of the LR, and time to unrestricted weight bearing (UWB), defined as the time from operation until permission of UWB by the surgeon, based on clinical and/or radiological examination as a surrogate measure of clinical healing.
Bone lesion characteristics and surgical intervention
The reasons for initial presentation were either local symptoms (n = 148/164 (90%)) or a pathologic fracture (n = 16/164 (10%)). With respect to anatomical location, approximately two-thirds of the bone tumors in our cohort were situated in the lower extremity (n = 105/164 (64%)). The most common histopathological diagnosis encountered was enchondromas, comprising almost half of the study population, followed by simple (unicameral bone) cysts (SCs), aneurysmal bone cysts (ABCs), fibrous dysplasia (FD), and giant cell tumors (GCTs) of bone. The cohort also included four cases of low-grade CS treated by simple curettage (Table 1). We calculated the size of the lesion by approximating measures from computed tomography (CT)/magnetic resonance imaging to a spherical shape (height × width × length × 0.52). 7 All lesions were graded with respect to radiological appearance and deemed structural integrity using a modified Campanacci classification. 3 Accordingly, from a radiological and structural perspective, the residual walls of the defects were intact (grade 1) in 72 of 164 (45%) lesions, thinned (grade 2) in 74 of 164 (45%) lesions, and deficient or breached (grade 3) in 18 of 164 (11%) lesions, with an estimated mean residual bone defect volume after tumor curettage of 11.5 (0.1–94) mL. Cancellous allograft was used for bone defect reconstruction in the majority of the cases (n = 133/164; 81%). In four cases of small metacarpal or phalangeal bone defects, autograft harvested from the distal radius was chosen as filling material, and a bone graft substitute (HydroSet™, Stryker Corp, MA, USA: n = 5 or Cerament™|Bone Void Filler, Bonesupport AB, Lund, Sweden: n = 1) was used in six other locations due to small defect size or nonavailability of allograft bone. In 21 small lesions, deemed structurally irrelevant (modified Campanacci grade 1, n = 13), located in the hand (n = 9), or other non-weight bearing locations (n = 3), the bone defect was left empty at the discretion of the individual surgeon. In lesions that were deemed structurally impaired or at risk, mechanical augmentation with internal fixation, using either plates (n = 16/164 (10%)) or intramedullary nails (n = 4/164 (2%)), was performed in addition to the bone defect reconstruction.
Histology and number of LRs of the surgically treated bone tumors and cysts.

LR: local recurrence; SC: simple cyst; ABC: aneurysmal bone cyst; FD: fibrous dysplasia; GCT: giant cell tumor; CS: chondrosarcoma.
Radiological evaluation
X-rays of the bone defects were classified according to a modified Neer’s classification of radiological bone cyst appearance (Table 2). 13,14
Modified Neer classification of radiological evaluation of bone defect healing.

a With or without small nonprogressive radiolucent area(s) less than 1 cm in size.
Statistical analysis and approvals
We applied Kaplan–Meier survival analysis to estimate the cumulative incidence function (time to event) for the outcome of interest (LR) with calculation of 95% confidence intervals (95% CI) and log-rank test using SPSS software (version 22). Approval for the study was obtained from the Danish Data Protection Agency (J.no. 2014-41-3021) and Department of Health Agency (J.no. 3-3013-705/1). For this type of study, formal consent was not required.
Results
Local recurrence
LR occurred in 19 of 164 patients (11.6%), most commonly among GCT (n = 4/12 (33%)) and SC (n = 4/10 (40%)) in children (Table 1). Most recurrences (n = 14/19 (74%)) occurred within the first 2.5 years and the overall cumulative incidence was 8.5% (95% CI: 4.1–12.9%; Figure 2). For patients whose defects were treated with allograft bone, the overall 2.5-year cumulative incidence was 9.8% (95% CI: 4.6–15%). Among SC in adults or enchondromas, no LR was observed within the first 2.5 years, whereas SC in children had a high (p = 0.002) 2.5-year cumulative incidence of 40% (95% CI: 9.0–71.0%; Figure 2). As expected, LR was more common (p = 0.002) for preexisting recurrent disease compared to primary disease with 2.5-year cumulative incidences of 30.8% (95% CI: 5.2–56.4%) and 6.6% (95% CI: 2.6–10.6%), respectively (Figure 2). It is worth mentioning that LR was least common in adult SC (0%), ABC (0%), and benign or borderline cartilaginous tumors, that is, enchondromas (n = 2/57 (4%)) or low-grade CS (0%).

Kaplan–Meier survival analyses displaying LR-free survival following curettage of benign bone tumors and cysts. Left: displaying overall LR-free survival. Middle: comparing recurrence-free survival, SC in children (blue) and SC in adults (green). Right: comparing recurrence rate for primary disease (blue) with recurrent disease (green). LR: local recurrence; SC: simple cyst.
Time to UWB
Immediate postoperative UWB could be permitted in the majority of cases (n = 100/164; 61%). For the remaining patients (n = 64/164; 39%), UWB was allowed after a median of 5.9 (2–21) and 6.0 (2–21) weeks for patients treated with allograft (n = 53/164; 32%). Restriction of UWB beyond 6 weeks was typically recommended to patients with larger tumors (mean volume: 24 (0.1–94) mL) located in the lower extremity (n = 26/31). Plate or nail augmentation was used in 20 cases, most commonly (n = 15/20) in the proximal femur, and in 13 of these cases, immediate postoperative UWB was permitted. For the remaining seven patients augmented with plate or nail, UWB was allowed after a mean of 10 (6–21) weeks.
Radiological evaluation
Postoperative follow-up X-rays were available in 131 of the 133 patients treated with allograft bone. Mean radiological follow-up was 19 (1–84) months, however, only 73 patients with either larger tumors than average (mean volume: 19.1 (0.5–95) mL), tumor location in the lower extremity (n = 55), and entities with tendency to LR (n = 46) were followed radiologically for more than 6 months. Evaluation of the immediate postoperative X-rays revealed incomplete defect filling in 24 of 131 cases (18%) and complete filling in 107 cases (82%). After 6–12 months postoperatively (n = 73), 38 defects appeared completely filled (grade 1), 24 had minor defects (grade 2), 6 had larger defects (grade 3), and 5 had recurrent disease (grade 4; Figure 3). While the grade of filling remained unchanged in 45 cases during the follow-up period, incomplete postoperative filling further consolidated to complete filling in only 3 cases or decreased in other 3 cases. Deterioration of complete initial defect filling due to graft resorption was observed in 17 cases (grade 2: n = 14 and grade 3: n = 3) and due to LR in 5 cases (Figure 4). Of the 21 empty defects, only four patients had radiological follow-up for more than 6 months and in two of these cases (both small chondroblastomas in the distal femur of children), the defect displayed signs of bone healing with defect filling >50% (grade 2).

Radiological evaluation based on a modified Neer’s classification (Table 2) in patients treated with bone allograft and more than 6 months of radiological follow-up.
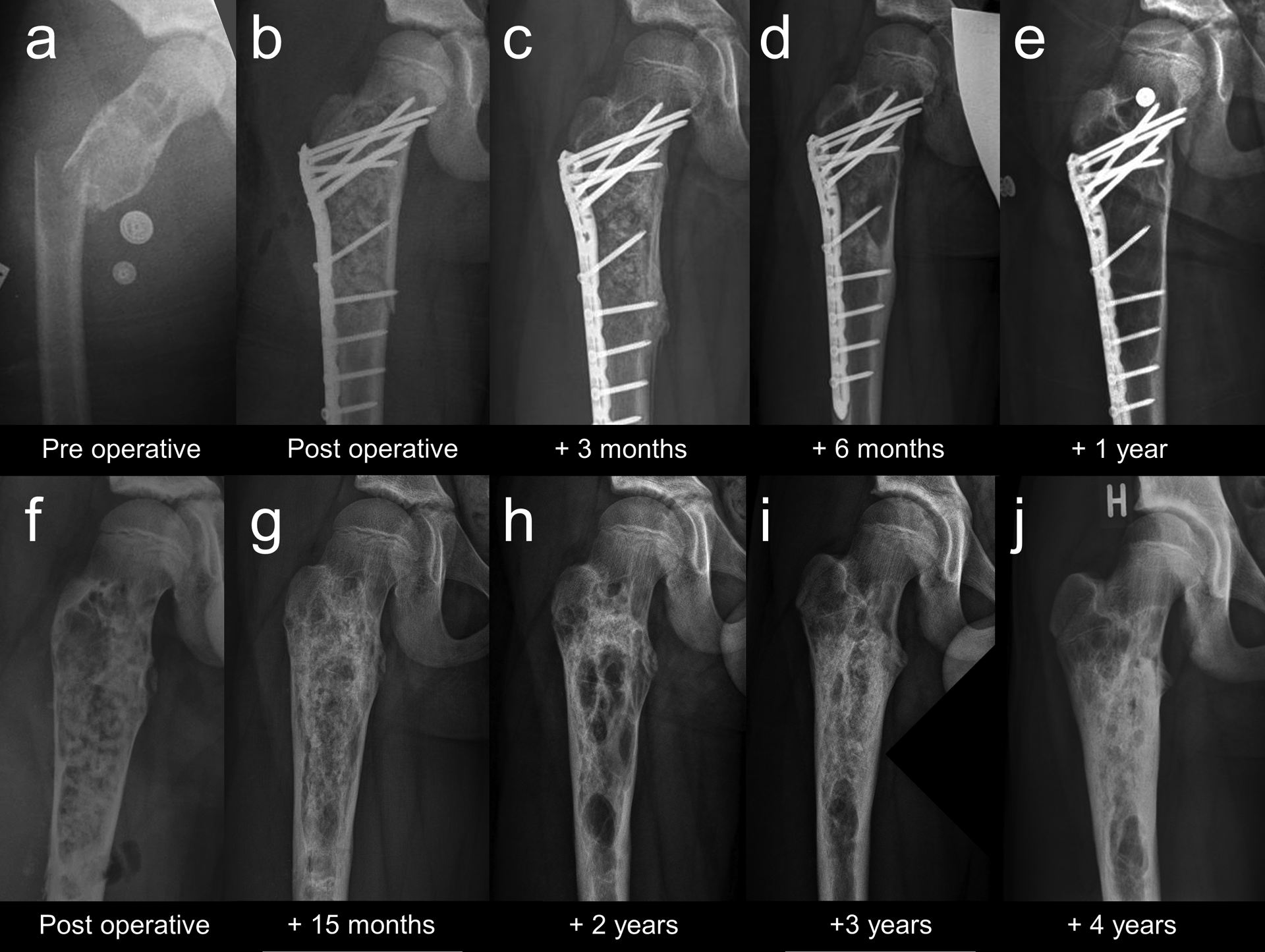
Upper row: (a) Preoperative X-ray of a pathologic fracture through a large bone cyst affecting the proximal femur in a 10-year-old boy. (b) Immediate postoperative X-ray after ORIF and bone grafting displays anatomical reduction, locking plate fixation, and complete defect filling. (c) X-ray 3 months postoperatively confirms consolidation of the fracture. (d) X-ray 6 months postoperatively shows significant graft resorption and early signs of LR in the medial subtrochanteric region with increasing radiolucency and cortical thinning. (e) 1 year postoperatively, complete resorption of all previously implanted allograft is evident. Due to substantial progression of local cyst recurrence, reoperation with implant removal and repeat grafting was indicated. Lower row: (f) Immediate post reoperation X-ray shows complete defect filling after implant removal. (g) 15 months postoperatively, graft integration appears radiographically complete (MNC grade 1) and both cortices show signs of consolidation. (h) X-ray 2 years postoperatively displays significant graft resorption over 50% (MNC grade 3). (i) 3 years postoperatively, there is evidence of progressive graft integration and remodeling with only small radiolucent areas in the central portions of the defect (MNC grade 2). (j) 4 years postoperatively, the natural course of graft integration and remodeling appears almost complete without residual lucent areas (MNC grade 1) and substantial consolidation of all previously involved cortices. LR: local recurrence; ORIF: open reduction and internal fixation.
Postoperative complications
Complications occurred in 11 of 164 patients (7%) with postoperative infection (n = 3), postoperative fracture (n = 2), and nerve palsy (n = 2) being the most common (Table 3). All but one complication (transient peroneal nerve palsy after curettage of a proximal fibula intraosseous ganglion filled with a bone graft substitute; Hydroset™, Stryker) occurred in patients treated with allograft bone (n = 10/133 (8%)). One deep infection occurred after plate augmentation of a SC (16 mL) in the proximal femur. Two superficial infections occurred after simple curettage and grafting of a small (4 mL) enchondroma in the distal femur and curettage and grafting of a SC in the proximal femur, treated with temporary external fixation for stabilization of a pathological fracture. Postoperative fracture occurred after removal of a large (22 mL) intraosseous myofibroma in the calcaneus and after removal of an enchondroma (11 mL) in the distal femur. The latter patient also developed postfracture deep venous thrombosis. Transient neurapraxia of the peroneal nerve followed curettage of a recurrent intraosseous ganglion in the proximal fibula, and persistent radial nerve damage followed curettage of a large (75 mL) humeral diaphyseal bone cyst that had previously fractured and healed in malalignment. Minor pulmonary embolism occurred following curettage of FD affecting the proximal femur of a 40-year-old woman. All wound healing–related complications occurred in the foot, delayed wound healing following curettage of a SC in the calcaneus and a GCT in talus, respectively, and one skin necrosis following curettage of osteoid osteoma in the talus.
Complications.

Discussion
Although the differential treatment of benign bone lesions has evolved considerably during the past two decades, the goals of treatment remain to establish a correct diagnosis, completely remove the lesion, and to relieve pain if present, while preserving and/or restoring function and minimizing complications. While emerging minimally invasive techniques such as radiofrequency ablation, embolization, and percutaneous injection techniques are gaining importance for certain entities, our results indicate that simple curettage remains an adequate treatment option for a wide spectrum of indications, particularly those with low risk of LR, such as enchondromas, where the expected rate of LR is almost nonexistent even without the use of any local adjuvants. 4,5 Despite the promising surgical results, nonoperative treatment and radiological follow-up should be considered for patients with non-symptomatic enchondromas, hereby avoiding the risks of open surgery. 15 Extended curettage with adjuvant treatment, on the other hand, should be considered in high-risk entities such as GCTs of bone, where the risk of LR could be as high as 50% if treated without adjuvants. 16 Our results also confirm previous reports that SCs in children display recurrence rates comparable to GCT’s, 17,18 which is why this particular patient group deserves also special attention and consideration of suitable adjuvant treatment.
Surprisingly, little data are published to guide surgeons whether reconstruction of a bone defect resulting from surgical removal of a benign bone lesion is indicated and how this is technically best accomplished. Furthermore, no precise objective criteria exist to determine when a bone defect is sufficiently healed to withstand the risk of fracture. Consequently, these decisions are based on clinical judgment, rather than objective factors. Although Wolff’s law indicates that healthy bone naturally adapts to load-bearing, it cannot predict the speed of bone healing or determine which bone defects are too large for spontaneous bone healing. Therefore, the degree of radiographic consolidation observed in a bone defect may well be important to guide the physician to evaluate a persisting risk of fracture, especially in case of insufficient spontaneous bone healing. Two recent articles demonstrate that most patients treated without bone defect reconstruction regain full weight bearing capacity and that bone defects consolidate without augmentation with bone graft. 7,8 While the clinical results reported in these two studies are comparable with those observed in our cohort, the reported fracture rates (3.8% and 10%, respectively) are considerably higher than the fracture rate observed in our study (1.5%). There was a tendency in both studies that larger defects were associated with higher complication rates, which indicates that the omission of bone defect reconstruction may not lead to an increase in absolute fracture risk per se, but depends on other factors such as the relative defect size, anatomical location, as well as the extent of presence or absence of spontaneous bone healing of the individual defect. As the compressive strength of bone allograft is poor, the potential increase in structural integrity by grafting a bone defect is unlikely to be the explanation for the observed differences in fracture rates.
Our rationale for bone grafting was mainly to enhance bone healing by providing an osteoconductive scaffold. A study that examined untreated cavitary bone defects with CT 1 year postoperatively found evidence of compensatory cortical thickening, but no or very little evidence of spontaneous bone formation within the actual cavity itself. 8 In contrast, results from another study on treatment of metaphyseal bone defects with impaction grafting using cancellous allograft bone as an adjunct to osteosynthesis of tibial plateau fractures in osteoporotic patients confirmed that the majority of the bone graft (mean: 55%) showed evidence of remodeling into trabecular bone with a normal radiological appearance on X-ray and CT after 1 year, while 37% remained unremodeled, and only 8% had resorbed. 19 These results are comparable to our radiological findings, which may indicate that unchanged X-ray appearance could be perceived as an indirect sign of at least partial graft integration. Furthermore, these results also support the rationale for the use of osteoconductive scaffolds, such as bone allografts or bone graft substitutes, which may contribute to a more complete bone cavity remodeling. Bearing in mind the relatively rare complications associated with bone allografts, such as immunogenic reactions, disease transmission, and risk of infection, 9 –11,20 it is not surprising that we did not observe any true allograft-related complications; however, our study sample is of course far too small for any meaningful conclusion in this regard.
The main limitation of this study is the lack of complete clinical and radiological follow-up in all patients and the use of an indirect outcome measure (UWB) as a surrogate for clinical bone healing, which of course does not reflect actual bone healing. Although the study is retrospective, our nationwide electronic patient file system made it possible to detect admissions and treatments at other hospitals to complement our own patient file, leading to very little or no loss of information regarding serious complications, recurrences, and reoperations, which otherwise could have led to underestimation of complication and recurrence rates.
Conclusion
Simple curettage and bone defect reconstruction with cancellous allograft bone is a good treatment option for many benign bone lesions and is associated with a low complication rate. Most commonly, the majority of allograft bone remains clearly visible on X-ray at 6–12 months postoperatively, which may indicate at least partial graft integration. For high-risk entities, such as GCTs of bone, recurrent disease, and SCs in children, additional adjuvant treatment should be considered to avoid LR.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: An institutional research grant from Bonesupport AB (Sweden) was used to fund part of Peter Horstmann’s PhD work. None of the other coauthors have any conflicts of interest related to this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
