Abstract
Background:
The ability of fracture healing in Tibetans is significantly superior to Chinese Hans, which may attribute to factors from diet, altitude to physical conditions. Conjugated linoleic acid (CLA) is an important ingredient in Tibetan diet, playing a role in antioxidation, antiatherosclerosis, and decrease in body fat accumulation.
Methods:
This study further quantified CLA effect in fracture healing in rats using combined structural evaluation (X-ray and micro-computed tomography), biomechanical test, and histological examination.
Results:
CLA could promote fracture healing with quicker development of trabecular connection, network and thickening and were more active at the stage of bony union and remodeling. The load to failure could reach 78.12 ± 10.03 N, 41.4% stronger than the control by week 6 (p = 0.0209).
Conclusions:
CLA improved the quality and mechanical strength of fracture healing in rats callus. The information may offer insight in development of new therapeutic strategies of fracture healing for general populations beyond Tibetans.
Introduction
It has been observed clinically that fracture healing in Tibetans is significantly better than Chinese Hans. 1 –3 Understanding the underlying mechanism may offer insight in development of new therapeutic strategies for fracture healing in Hans and other non-Tibetans.
The possible reasons of the superior fracture healing in Tibetans may be summarized as follows: stronger physical conditions in both musculoskeletal and immune systems, genetic factors, high elevation environment (extra ultraviolet intake, hypoxia, and significant outdoor activities), and diet. Muscle power of hand grip and legs has been shown to be positively associated with the increase of bone mineral density (BMD) at the lumbar spine and hips. 4 Studies have reported that fracture healing potential is affected by the patient genome, and variations in the IL1B and NOS2 genes may contribute to delayed fracture-healing. 5 Ultraviolet radiation B produces significant alteration to vitamin D levels via serum 25(OH)D, which in turn increases BMD and reduces the risk of fracture. 6,7 Activation of the hypoxia inducible factor, a pathway in bone development, increases bone modeling events through cell-nonautonomous mechanisms to coordinate the timing, direction, and degree of new blood vessel formation in bone. 8 Therapeutic exercise is often recommended as a non-pharmacologic treatment of vertebral fractures to reduce pain and restore functional movement. 9 To date, however, it remains undefined if the diet can affect the fracture healing significantly.
The daily diet of Tibetans differs from that of Hans. The primary differences include yak butter (73.0 g/day in Tibetans vs. 3.7 g/day in Hans), beef and lamb (226.2 g/day in Tibetans vs. 115.7 g/day in Hans), vegetables and fruits (217.8 g/day in Tibetans vs. 514.3 g/day in Hans). 10 As the main portion of daily food intake of Tibetans, yak butter, beef and lamb are all high in the content of multifunctional fatty acids. 11 For example, yak butter consists of conjugated linoleic acid (CLA, 17.2 mg/g), linolenic acid and arachidonic acid (9.07 mg/g), eicosapentaenoic acid (0.56 mg/g), and docasahexaenoic acid (0.28 mg/g). 12
It has been known that CLA functions include anticancer, antioxidation, antiatherosclerosis, lessening markers of catabolism, enhancement of the immune system, and decrease in body fat accumulation. 13 Studies suggested that dietary CLA suppressed the production of prostaglandin E2 (PGE2) in serum and spleen, 14 CLA might reduce PGE2 level by limiting arachidonic acid. 15 Anticarcinogenic effects of CLA indicated a modifying role in peroxisome proliferator–activated receptor α (PPARα) action. 16 Moreover, CLA actions on fat and energy metabolism could, in part, be directed through changes in both PPARα and PPARγ. 17,18 Recent studies further indicated the role that CLA played in osteoblast-like cells. CLA increased levels of osteocalcin and activity of alkaline phosphatase (markers of bone formation) in murine MC3T3-E1 osteoblast-like cells in culture, providing the involvement in osteoblast-like cells. 15,19 –21
Fracture healing is a complicated biological repairing process involving the coordination of a variety of different processes. Healing is typically characterized by four overlapping stages: the initial inflammatory response, soft callus formation, hard callus formation and initial bony union, and remodeling. 22 Studies have shown that PGs are multifunctional regulators of bone metabolism stimulating both bone resorption and formation. 23 Evidences indicate an important role of PPARγ in bone metabolism. Heterozygous PPARγ-deficient (PPARγ +/–) mice exhibit enhanced bone formation with increased osteoblastogenesis. 24
As CLA is closely related to PGs, PPARγ, and osteoblast-like cells, we hypothesized that CLA can significantly regulate fracture healing. In this study, we developed a rat fracture model to examine this hypothesis, and used structural, biomechanical and histological techniques to evaluate the outcomes of fracture healing.
Materials and methods
Animal model
The experiment was performed on Sprague-Dawley rats (n = 30, 200 ± 15 g in weight; approved by the institutional animal committee). The specimens were divided randomly into two groups equally: A. CLA (10 g CLA/kg diet) with the basal diet, and B. control with the basal diet only. The rats were harbored individually in cages at 20 °C with access of water and foods.
Surgical technique
The tibial fracture was created following a standard procedure. 25,26 In brief, after anesthesia via the intramuscular injection of 10% chloral hydrate (0.3 ml/100 g), rats were placed in supine position. The left lower extremity was shaved, cleaned, and then properly rotated to abduction position to ensure superior migration of fibula. Infrapatellar tibia was palpated and accessed via anteromedial incision. After the skin and cutaneous maximus muscle were dissected, the tibia was reached through the tibialis cranialis, peroneus longus, and brevis muscles. The tibial tuberositas caudal to the patellar joint was palpated and a 0.8-mm K-wire advanced until the tarsal joint was delivered. The osteotomy line was performed manually using 2-mm osteotome from 15 mm caudal to the patellar joint resulting in fracture (Figure 1). After surgery, rats were assessed with respect to wound site infection and dehiscence daily.

Illustration of the rat fracture model (the arrow pointed to the fracture site).
Structural evaluation
Fracture healing process was first evaluated radiologically at weeks 2, 4, and 6. The degree of union was scored using a five-point system: 0—no callus, 1—little-to-moderate callus, 2—profuse callus tissue, 3—bridging periosteal callus, 4—mature callus with interfragmentary bridging, and 5—callus resorption after solid union. 27 The scores were evaluated by three surgeons independently and averaged.
The healing process was also examined by micro-CT scanning of 17.51 μm resolution at 4 and 6 weeks postoperation (Skyscan 1176, Bruker, Kontich, Belgium). Acquired tomographic images were at the energy settings of 65 kV and 385 mA, integration time of 250 ms, with 0.7° rotation between frames .The fracture line was identified after reconstruction by simultaneously viewing multiple orthogonal slices. The analysis determined the average cross-sectional area (CSA) of the callus and the corresponding BMD. The bone strength index (BSI) of the fracture callus was then calculated from 3.5 mm proximal to 3.5 mm distal of the fracture line as CSA × BMD. 28
Biomechanical test
Fracture healing strength was tested at week 6. 29 The rats were killed using high-dose anesthesia of 10% chloral hydrate, and the k-wire in each tibia was kept to protect the callus tissue. The entire tibia was removed by being detached from its surrounding soft tissue. Between steps, the tibia was soaked in saline. The mechanical properties of the tibia were measured using a three-point bending test and the load to failure was documented. 30
Histological examination
Fracture healing was further examined at week 6 histologically. Fracture calluses were decalcified and embedded in paraffin as described previously. 31 Sagittal sections (5 mm) were cut in standardized plane using a microtome (Eclipse 80i, Nikon, Japan) and stained with hematoxylin and eosin for basic morphology, or alcian blue and nuclear fast red for analysis. A scoring system was used for quantification. The system scored zero for nonunion, one for healing with complete fibrous callus, two for some calcification with fibrous tissue, and three for completely calcified callus or new bone formation. 32 The scores were evaluated by a pathologist independently.
Statistical analysis
Mean and standard deviation in each group were calculated. The Wilcoxon signed-rank test was performed with a significant difference among two groups (p < 0.05).
Results
Structural evaluation
Radiological results indicated that fracture lines were clearly visible and fracture was not bridged in both CLA and control groups at week 2 with the evaluation scores of 1.3 ± 0.9 and 1.0 ± 0.5, respectively (p = 0.4058; Figure 2). At week 4, the fracture line became obscure in both groups. The CLA group appeared to have more callus formation with connection than the control, they were different statistically in evaluation scores (3.7 ± 0.7 in CLA and 3.0 ± 0.5 in the control, p = 0.0197). At week 6, the fracture line in the CLA group vanished, and 40% specimens had complete callus absorption. The evaluation score was 4.4 ± 0.6. In contrast, the control group still had the vague fracture lines with matured callus and significant callus bridges. The evaluation score was 3.4 ± 0.5 (p < 0.001).
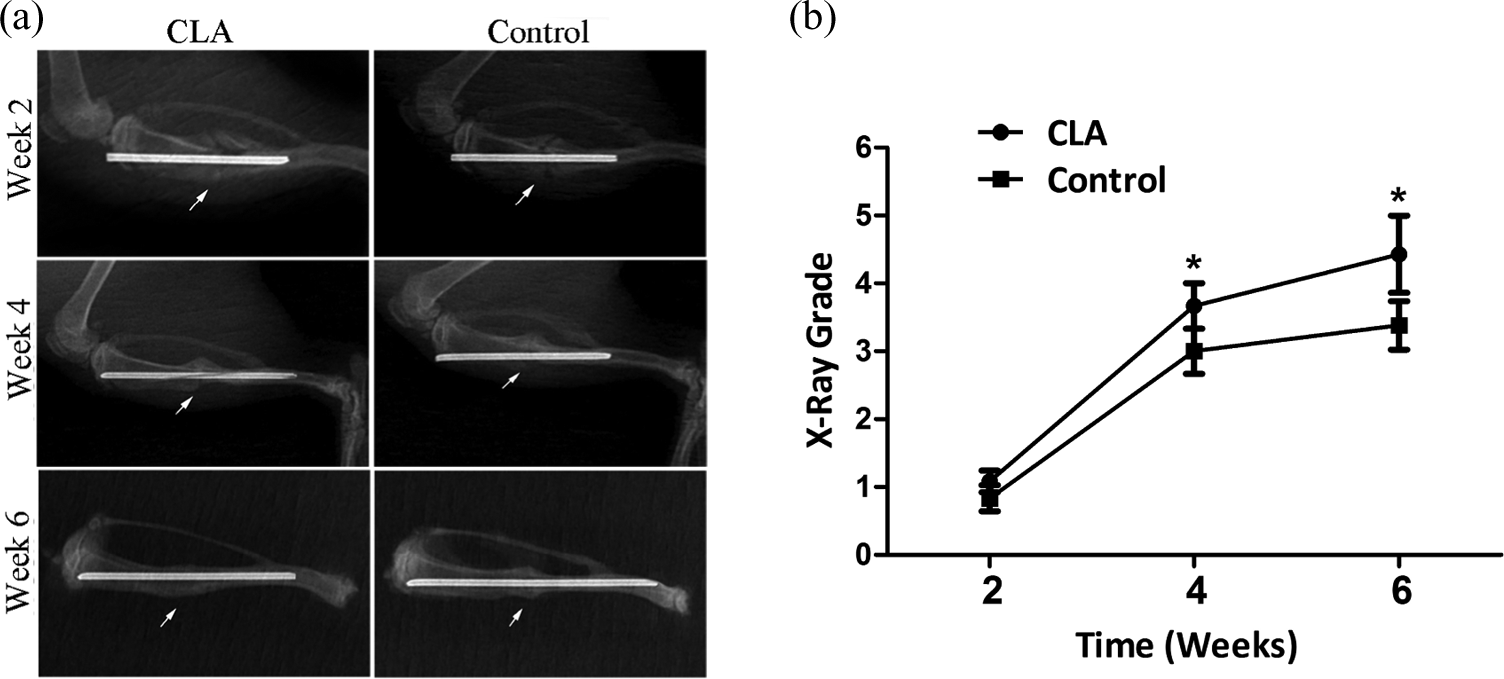
Radiographic evaluation at weeks 2, 4, and 6. (a) A typical radiographic image showing the fracture healing with CLA intervention at week 6 (the arrow pointed to the fracture site). (b) The radiographic grade showing significant difference between the CLA and control groups at weeks 4 and 6 (p < 0.05). CLA: conjugated linoleic acid.
The micro-CT images indicated that at week 4, both groups had no significant difference in CSA (p = 0.1172), BMD (p = 0.6015) except for BSI (p = 0.0472; Figure 3). However, at week 6, the CLA group had 14.05 ± 3.13 mm2 in CSA, 0.98 ± 0.04 mg/mm3 in BMD and 13.73 ± 2.67 mg/mm in BSI, which were significantly greater than the control (9.38 ± 1.87 mm2 in CSA, p = 0.0163, 0.86 ± 0.78 mg/mm3 in BMD, p = 0.0163, and 8.07 ± 1.66 mg/mm in BSI, p = 0.0039).
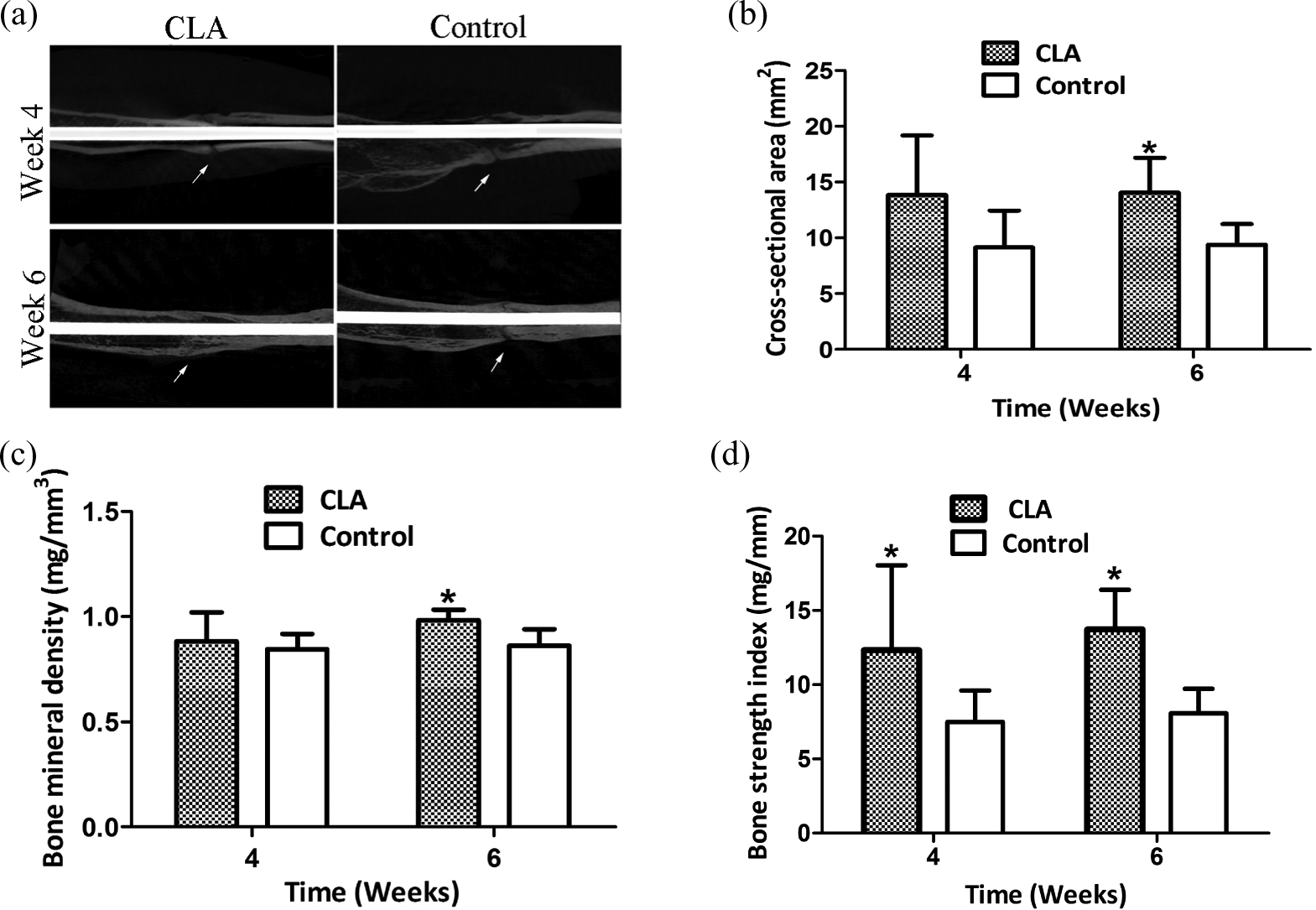
Micro-CT examination at weeks 4 and 6. (a) A typical micro-CT image revealing the fracture healing with CLA intervention at week 6 (the arrow pointed to the fracture site). (b) The average CSA of callus. (c) The BMD of callus. (d) The BSI of callus. CLA: conjugated linoleic acid; CSA: cross-sectional area; BMD: bone mineral density; BSI: bone strength index.
Biomechanical test
The three-point bending test indicated that the load to failure of the CLA and the control groups was 78.12 ± 10.03 N and 32.34 ± 8.99 N, respectively. The difference was statistically significant (p = 0.0209; (Figure 4)).
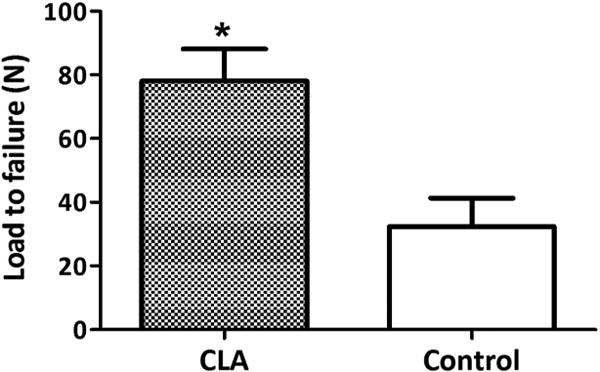
Biomechanical strength of fractured tibia 6 from three-point bending test at week 6.
Histological examination
Histological results demonstrated the evidence of fracture healing in the CLA group at week 6 (Figure 5). Under 4× microscope, trabeculae were densely reconnected and medullary canal was redeveloped. The control group still showed disorganized and loose trabeculae. Under 10× microscope, the CLA group had matured callus, and woven bone spicules were observed predominantly with trabeculae significantly thicker than the control. The averaged score was 2.2 ± 0.75 in the CLA group and 0.8 ± 0.75 in the control group (p = 0.041).
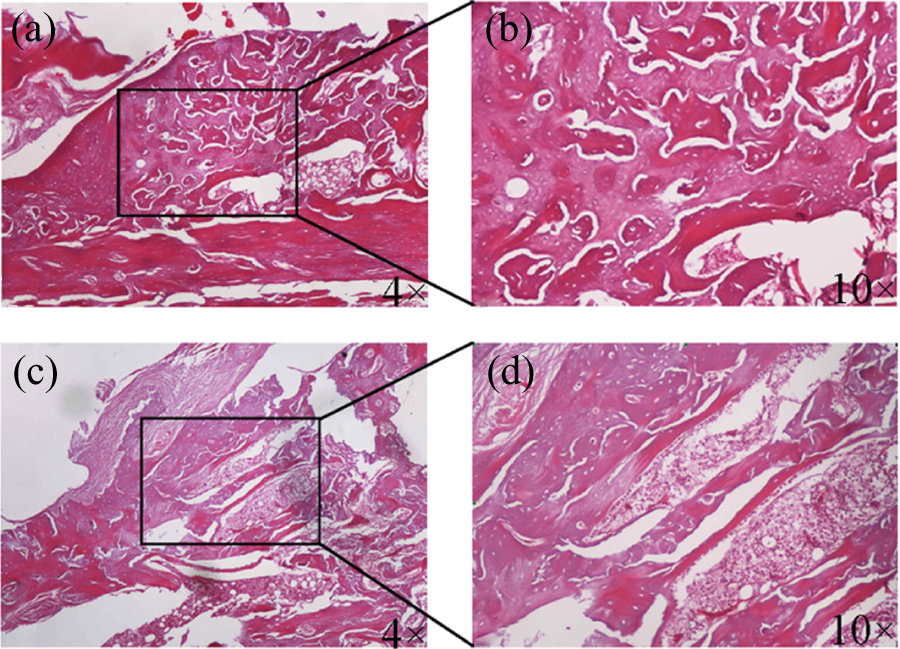
Histological evaluation at week 6. (a) and (b) were with the CLA intervention, and (c) and (d) were the control. CLA: conjugated linoleic acid.
Discussion
In the present study, we demonstrated that dietary supplementation with CLA benefited to fracture healing in rats. Structural evaluation of X-ray indicated that fracture was healed in 6 weeks with the CLA intervention; and the evaluation scores were all between 4 and 5, significantly higher than those in the control (p = 0.002). Micro-CT examination further showed fracture healing at the microstructural level; and the BSI was significantly greater than the control at week 6, suggesting the improvement of overall strength being achieved via reestablishing trabecular connections. Biomechanical test of three-point bending at week 6 also revealed the improvement of bending strength after CLA intervention. Histologically, the CLA group, in comparison with the control, showed the maturity of healing with solid network connection and thickening of trabeculae.
The rationale of this study needs to be discussed: 1. The animal model. The fracture model of rats has been used routinely in the laboratory. 25 As reported here, the model served the purpose of showing CLA role in promoting fracture healing. 2. CLA dose. We used 10 g CLA/kg diet for intervention. This dose was similar to the typical daily intake dose of Tibetans. 11 CLA intake was confirmed on one rat with higher serum high density lipoprotein cholesterol and triacylglycerol levels (0.55 and 0.94 mmol/L) comparing to the control (0.38 and 0.53 mmol/L) following the protocol in the literature. 33 3. Measurement timing. During fracture healing of rats, the inflammatory response takes 2 weeks, callus formation another 2 weeks, and bony union and remodeling additional 2 weeks. 22 Our measurement was therefore chosen at weeks 2, 4, and 6.
CLA is a collective term describing a mixture of positional and geometric isomers of linoleic acid with conjugated double bonds, which may be of cis or trans configuration at positions 9 and 11 or 10 and 12. 34,35 The cis 9-trans 11 and trans 10-cis 12 isomers of CLA are routinely found in animal tissues. 36 CLA isomers occur naturally in ruminant food products (beef, lamb, and dairy) during the process of bacterial biohydrogenation of linoleic acid in the rumen. 34,37,38 The cis 9-trans 11 and trans 10-cis 12 isomers of CLA have been shown to possess biological activity. 39,40 The trans 10-cis 12 isomer of CLA reduces adipose mass in mice and humans, while the cis 9-trans 11 isomer has anti-inflammatory properties. All of the studies conducted to date on the functional effects of CLA have used mixed preparations of CLA with approximately equal amounts of the bioactive 9, 11 and 10, 12 isomers. In our study, this mixture of CLA was therefore adopted.
Fracture healing is a process that recapitulates certain aspects of skeletal development and growth involving a complex interplay of cells, growth factors, and extracellular matrix. Repair is typically characterized by four overlapping stages. The initial inflammatory response is coordinated by and involves the secretion of a range of cytokines and growth factors including platelet-derived growth factor, fibroblast growth factor-2, vascular endothelial growth factor, macrophage colony stimulating factor, interleukins-1 and -6 39 –43 in the initial inflammatory response. These factors facilitate the migration and invasion of multipotent mesenchymal stem cells. 44,45 The soft callus formation stage is dominated at the cellular level by chondrocytes and fibroblasts although the relative proportions of the different cell types can vary between fractures. The hard callus formation is characterized by high levels of osteoblast activity and the formation of mineralized bone matrix, which arises directly in the peripheral callus in areas of stability, it represents most active period of osteogenesis. In the final stage of fracture reunion remodeling, the key cell type involved with resorption of mineralized bone is the osteoclast. Cytokines that are secreted by osteoblasts, growth factors, and cytokines are critical for the induction, survival, and competency of osteoclasts. Our structural evaluation suggested that the CLA intervention was the most effective in the stage of bony union and remodeling from week 4 to 6, followed by the (soft and hard) callus formation stage from week 2 to 4 and the initial inflammatory stage up to 2 weeks (Figure 2). In addition, CLA have various effects besides fracture healing. A diet containing CLA reduced body weight and body fat mass in the animal model as well as in human trials. 46,47 CLA administration with 1% dietary calcium significantly improved total ash percentage in femurs, suggesting the CLA potential to improve bone mass. 48
As the first step toward systematic investigation of CLA regulation on fracture healing, this study was limited to the animal model study. CLA may accelerate healing through osteoblast differentiation and function, promotion of calcium and phosphate absorption to improve bone strength. These questions need to be clarified in the future.
Conclusions
The unusual ability of fracture healing in Tibetans stems from multiple intrinsic and extrinsic factors. This study suggested the involvement of CLA, a major dietary ingredient in Tibetans, in promoting the fracture healing in rats. The heal was achieved by quicker development of trabecular connection, network, and thickening. The information offered insight in development of new therapeutic strategies of fracture healing for general populations beyond Tibetans.
Footnotes
Acknowledgement
This work is partly supported by the Natural Science Foundation of China under grants of 81320108018, 31270995, 31570943, and 81071450, and the Priority Academic Program Development of Jiangsu Higher Education Institutions. Authors also thank Dr. Lei Wang for histological analysis.
Author’s Note
Zhongshu Shan and Zong-Ping Luo—these authors contribute equally.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
