Abstract
Introduction
Thoracic outlet syndrome (TOS) is defined by compression of subclavian vessels and brachial plexus as they traverse the thoracic outlet, leading to symptoms such as upper-extremity pain, paresthesia, weakness, edema, heaviness, coolness, pallor, and aggravation of the aforementioned symptoms with physical activity. Compression can occur anywhere within the thoracic outlet region, including between the clavicle and first rib (costoclavicular space), between the anterior and middle scalene muscles (interscalene triangle), and deep to the pectoralis musculature near their insertion on the humerus (retro-coraco-pectoral space). There are three subtypes: arterial (aTOS), venous (vTOS), and neurogenic (nTOS)—of which nTOS is the most common, representing over 90% of cases that are diagnosed and subsequently treated. Though compression is the mechanism for development of TOS, multiple predisposing factors have been identified. These include, but are not limited to: congenital anomalies, such as the presence of cervical ribs, aberrant scalene musculature, and prominent C7 transverse processes; and acquired factors, such as repetitive strain, trauma, and subsequent scarring. 1
When considering acquired, or secondary factors, breast hypertrophy and breast surgery have been identified independently as predisposing factors for the development of TOS.1–3 In one clinical observation, primary breast surgery with radical mastectomy and secondary surgery with implant-based breast reconstruction were recognized as potential etiologies of TOS. 2 This is thought to be due to the anatomical interplay of the myofascial and neurovascular structures, which are subsequently changed in some way, such as by scarring or myofascial shortening, as a result of surgery or even postmastectomy radiation treatment.3,4 Additionally, altered cervicothoracic biomechanics are theorized to influence nTOS onset through similar mechanisms seen in macromastia. This includes changes in the frontal plane, such as scapular protraction and shoulder line angles, as well as anteroposterior disturbances such as increased thoracic kyphosis.3,5–8
Unfortunately, one complicating factor for the diagnosis of TOS after ablative or reconstructive breast surgery is often the imprecise diagnosis of postmastectomy pain syndrome (PMPS) applied to all patients presenting with persistent pain after treatment. As such, PMPS typically encompasses a wide range of pain disorders with varying etiologies and potentially distinct treatments, such nTOS, neurogenic pectoralis minor syndrome (nPMS), and even cervical radiculopathy. 3 Additionally, multiple tools including use of physical exam and provocative maneuvers, radiographic and electrophysiological assessments, and minor procedures such as anesthetic nerve blocks, are used to aid diagnosis of TOS and other pain-related syndromes. As such, TOS diagnosis lacks a unified, standardized protocol, potentially leading to misdiagnosis and imprecise treatment as alluded to above.9–11 The diagnostic complexity of TOS is further illustrated in patients with a history of chemotherapy, radiation, or lymphedema—all conditions that can confound the clinical presentation of TOS and potentially delay diagnosis.3,12,13
Despite clinical suspicion of TOS after breast surgery, there are no studies that have elucidated this connection as it relates specifically to breast reconstruction. Additionally, no studies have attempted to determine incidence of TOS after breast reconstruction based on reconstructive plane (ie, subpectoral or prepectoral), despite evidence that chronic pain is more prevalent in patients who have undergone subpectoral reconstruction.14–16 As such, this study aims to further investigate the relationship between implant-based reconstruction and TOS, with consideration for the plane of reconstruction and what implications this may have on diagnosis and subsequent treatment.
Methods
Data Collection
This study was performed with approval by the Institutional Review Board (IRB 20-1056). A retrospective review using the Informatic for Integrating Biology and the Bedside (i2b2) platform was performed, identifying all adult female patients presenting for evaluation of TOS between April 2014 and December 2022.
Cohort Definition
The study cohort consisted of patients whose visit diagnoses included International Classification of Diseases (ICD) 9 and 10 codes for TOS and brachial plexopathies; patients who underwent procedures for treatment of TOS or brachial plexopathy with relevant current procedural terminology (CPT) codes; and patients who had undergone extirpative and reconstructive surgery after breast cancer diagnosis. Patients with diagnoses of acute brachial plexus injuries, metastases to the brachial plexus as a manifestation for primary oncologic process (ie, Pancoast tumors), radiation-induced brachial plexopathy, and chemotherapy-induced brachial plexopathy were excluded from the study. Diagnosis of TOS was based on physician-documented conclusions following comprehensive diagnostic evaluations. Only cases that were explicitly identified by physicians as TOS postevaluation were classified as such. Conversely, cases where evaluations had negative diagnostic evidence of TOS or yielded inconclusive results were categorized under “TOS-Like Symptoms Without Formal Diagnosis.”
Outcomes
Of the subjects who underwent implant-based breast reconstruction, the following outcomes were considered: medical history, oncologic history, breast reconstruction technique, symptomatology, and TOS diagnostic and treatment history.
Statistical Methods
All data analyses were conducted using IBM Statistical Package for the Social Sciences (SPSS), version 28.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were used to summarize key diagnostic and treatment characteristics. Categorical variables, including breast cancer stage, ablative surgery type, additional oncologic treatment, and TOS diagnosis, were summarized using frequencies and percentages. Continuous variables, such as days from implantation to symptom onset, age at symptom onset, and the number of diagnostic procedures per patient, were expressed as means with standard deviations. If a subject underwent multiple reconstructions, the most recent episode of reconstruction was used for data analysis, with the day of implant device, whether tissue expander or implant, placement being used to determine the time to onset of symptoms.
Results
Of the patients presenting to our institution for evaluation of TOS during the study period, a total of 319 patients were included in our study. Sixteen of 319 (5.0%) had a history of breast cancer treated with subsequent ablative surgery and immediate implant-based breast reconstruction. Fourteen of 16 patients underwent mastectomies for primary treatment of their cancer. In addition to mastectomy, 6 patients received additional oncologic treatment, including chemotherapy and radiation therapy (Table 1). Overall, average time to TOS symptom onset following implant-based reconstruction is 421 ± 446.9 days. Eleven of 16 patients reported acute onset of symptoms, while 5 of 16 reported acute-on-chronic symptoms (Table 2).
Oncologic and Reconstructive Data (Median ± Std Dev and n (%)).
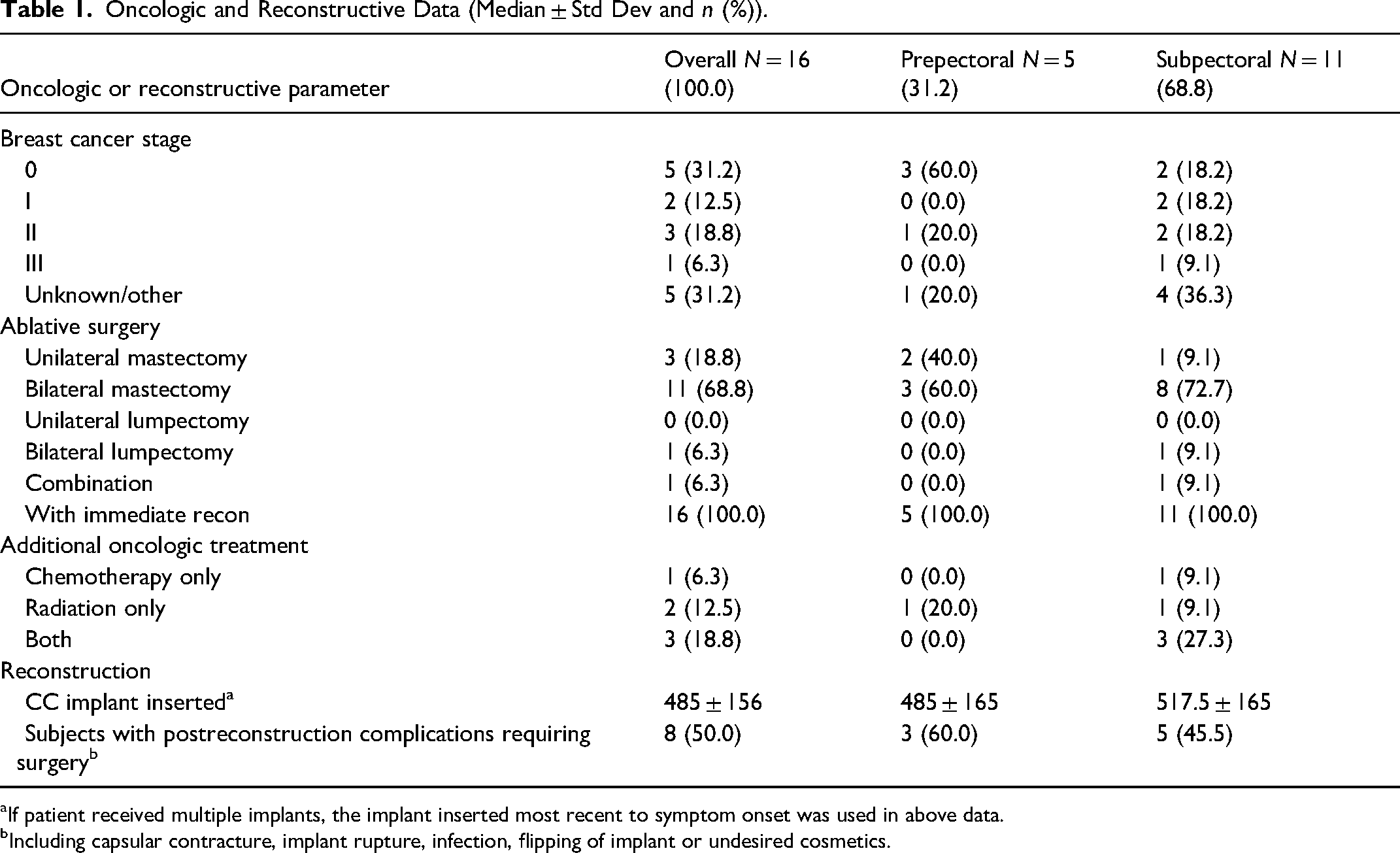
aIf patient received multiple implants, the implant inserted most recent to symptom onset was used in above data.
bIncluding capsular contracture, implant rupture, infection, flipping of implant or undesired cosmetics.
Diagnostic and Treatment Data (Median ± Std Dev and n (%)).
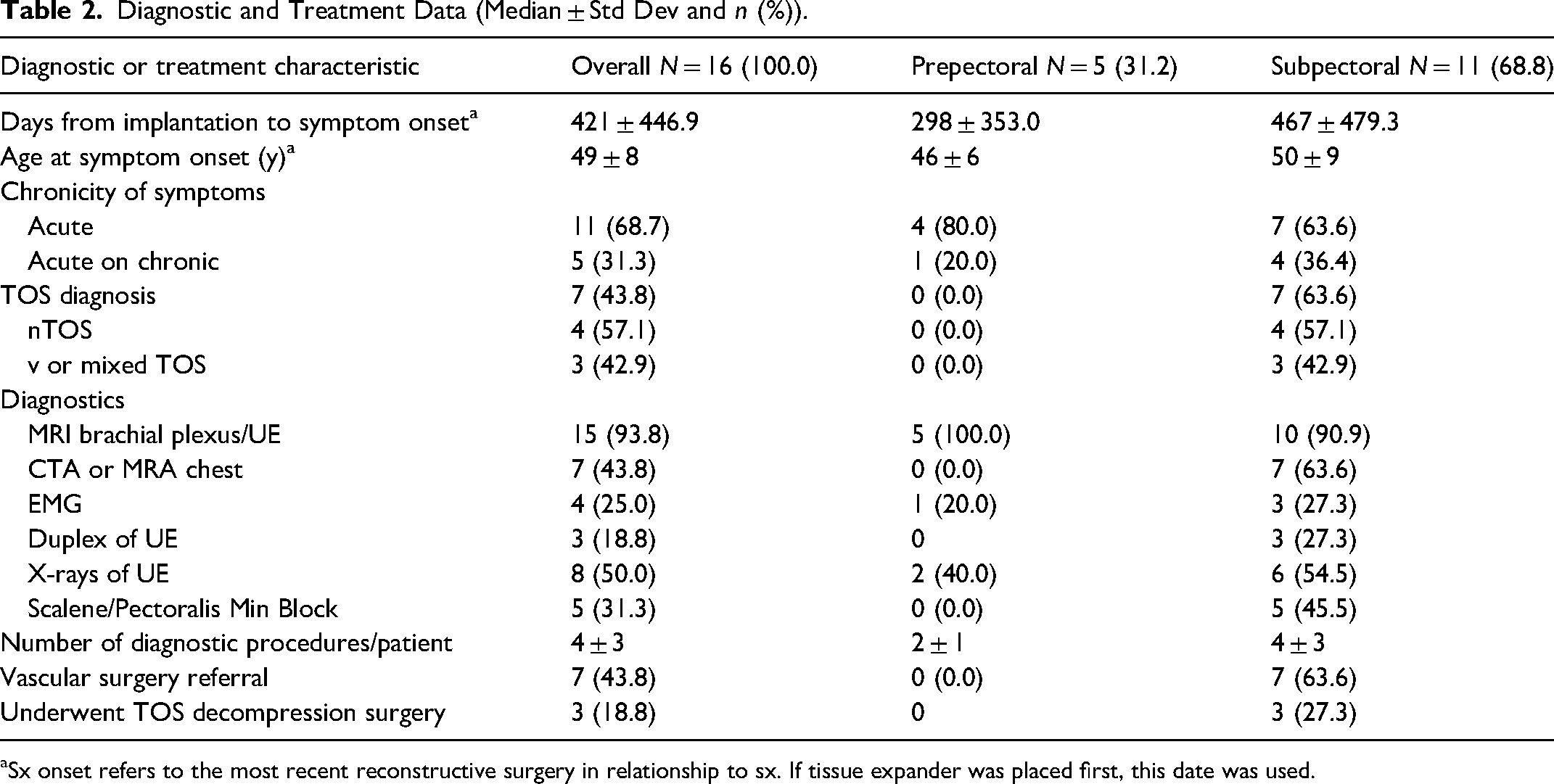
aSx onset refers to the most recent reconstructive surgery in relationship to sx. If tissue expander was placed first, this date was used.
Patients With Prepectoral Reconstruction
Five of 16 patients (31.2%) underwent implant-based reconstruction in the prepectoral plane. The average size of implant inserted at the timing of reconstruction was 485 ± 165 milliliters (mL). Three of 5 patients subsequently had complications postreconstruction requiring additional surgery (Table 2). All patients reported either acute or acute-on-chronic symptom onset roughly 298 ± 353 days after implantation. All patients received MRI brachial plexus for diagnostic evaluation, with one patient additionally receiving an electromyography (EMG) and two patients receiving plain film evaluation of the upper extremity. An average of 2 ± 1 diagnostic procedures was performed per patient. Despite extensive work up, no patients in this cohort received a formal diagnosis of TOS.
Patients With Subpectoral Reconstruction
Eleven of 16 patients (68.8%) underwent implant-based reconstruction with subpectoral implant placement. The average size of implant inserted at the timing of reconstruction was 517.5 ± 165 mL. Five of 11 patients had postreconstruction complications which required additional surgery (Table 2). Patients reported symptom onset 467 ± 479.3 days after implant placement. Ten of 11 patients received MRI brachial plexus for diagnostic evaluation, in addition to other studies including CTA or MRA of chest, venous duplex of the upper extremity, plain radiographic evaluation of the upper extremity, EMG, as well as scalene and pectoralis minor blocks. After extensive work up, 7 of 11 patients (63.6%) received a formal diagnosis of TOS (Table 2). The majority (4) of those diagnosed with TOS had the neurogenic subtype, while with 3 patients were diagnosed with vTOS or mixed TOS. All 7 patients subsequently were referred to Vascular Surgery for additional management, with 3 of 7 patients (27.3%) requiring thoracic outlet decompression surgery for treatment of the symptoms.
Discussion
Over 9 years, 319 individuals were evaluated for TOS, 16 (5.0%) of whom had implant-based breast reconstruction. Nearly half (43.81%) of these patients were diagnosed with TOS, and also had subpectoral implant-based breast reconstruction. Despite symptoms, the remaining patients did not receive a formal diagnosis as their work up either lacked conclusive evidence or findings were not consistent with TOS. These results hint at a correlation between subpectoral implants and TOS, indicating a pressing need for refining diagnostic strategies in patients presenting with pain postmastectomy and specifically postreconstruction. The authors of this study concur that TOS manifestation in this subset is likely multifactorial with several variables potentially contributing to development of TOS.
While subpectoral reconstruction was observed in all patients diagnosed with TOS, it is likely not the sole contributor to pathogenesis. Several congenital and acquired factors are known to increase the risk of TOS. Congenital anomalies such as cervical ribs, elongated C7 transverse processes, and aberrant scalene musculature have been widely documented in the literature as structural contributors to neurovascular compression. 17 Acquired risk factors include repetitive strain, trauma to the neck and shoulder, and altered postural biomechanics—particularly scapular protraction, thoracic kyphosis, and increased cervical lordosis—all of which can distort the thoracic outlet dimensions.17,18 Adjuvant therapies such as radiation may also lead to regional fibrosis and soft tissue contracture, creating secondary compression points. 19 These factors may act independently or synergistically to predispose patients to TOS following breast reconstruction.
The role of PMPS warrants initial consideration. PMPS, like TOS, is a non-specific and complex neuropathic pain syndrome that is thought to stem from intraoperative nerve injury. Its prevalence ranges from 12% to 72% in postmastectomy patients and there have been numerous attempts to better characterize the syndrome, starting with terminology and etiology.3,20 Worth highlighting are the six individuals who underwent additional oncologic treatment in the form of chemoradiation, either in combination or each alone. Adjuvant therapies such as chemotherapy and particularly radiation are known to cause neuropathic pain, with documented cases of radiation-induced fibrosis mimicking TOS.19,21 Given the higher incidence of these conditions compared to TOS, there exists a potential diagnostic overshadowing or availability bias further complicating TOS diagnosis.
Additionally, 8 of 16 patients (50%) in our study, about half of each respective cohort, encountered complications postreconstruction that required additional surgery. These complications included: mastectomy skin flap necrosis, infections, or capsular contracture. Acquired TOS, though less common, can develop from repetitive stress or trauma, leading to scarring and fibrotic alterations causing compression of neurovascular structures. 22 This mechanism might explain the acute-on-chronic symptomatology in almost one-third (5 of 16) of patients in our study. Breast reconstruction and additional breast surgeries may act as inciting events for fibrosis in adjacent anatomical structures and progression to being symptomatic is likely in those with predisposing anatomic factors, such as a cervical ribs. In our cohort, we have one case that highlights this, as cervical ribs were incidentally detected on radiography prior to reconstruction. Following breast surgery, the patient experienced functional impairment and arm numbness, culminating in a TOS diagnosis after thorough investigation.
Existing literature provides limited insight into postsurgical TOS, with isolated cases reported postchondrosarcoma resection of the manubrium and following minimally invasive pectus excavatum correction. These postoperative cases noted structural changes of the chest wall and this was thought to be contributory.23,24 Two instances postbreast surgery have been documented, one being a contralateral occurrence postmastectomy and another following cosmetic augmentation. Interestingly, both patients exhibited elongated C7 transverse processes, deemed by Emlakcioglu et al as a potential risk factor, exacerbated by postmastectomy cervicothoracic biomechanical shifts.25,26
The connection between mastectomy and biomechanical changes is further evidenced by studies demonstrating a markedly higher rate of postural failures in women postmastectomy (82.3%) compared to those in healthy individuals (35.1%), accompanied by statistically significant evidence of disruptions in proper posture.27,28 Specific biomechanical alterations documented in the literature encompass the frontal plane, such as scapular retraction and an increased shoulder line angle, and anteroposterior plane changes, specifically increased thoracic kyphosis, that may result in increased cervical lordosis.8,28–30 In addition, breast hypertrophy has been hypothesized to modify the thoracic outlet, shifting gravitational center, inducing cervical lordosis, and prompting coracoid process anterior tilt, thereby constricting the thoracic outlet. 5 Analogously, reconstruction, particularly utilizing implants deviating from the patient's natural breast dimensions, could cause comparable thoracic outlet anatomical shifts. Currently, the only factor identified in studies to positively influence the preservation of proper body posture after mastectomy is immediate reconstruction.31,32
In this study, all diagnosed patients had subpectoral implants. In general, patients with subpectoral implants often report prolonged postoperative pain and increased incidence of chronic pain, with scant research on the precise etiology of their long-term discomfort.14–16,33 Additionally, subpectoral implant placement is associated with animation deformity from repetitive pectoralis muscle contractions. 33 This muscular interplay, relevant in TOS pathology, may hint at a connection between TOS and subpectoral implants, warranting further exploration. Consequently, some patients have pursued conversion to prepectoral implants, with preliminary findings suggesting chronic pain reduction based on BREAST-Q scores.34–37
Of the individuals included in our study, seven were diagnosed with TOS, leaving nine with undetermined symptomatology. The latter group underwent fewer diagnostic procedures, averaging two compared to the former's four procedures. Of the patients with undetermined symptomatology, only one was referred to Vascular Surgery despite all having positive provocative tests (ie, Adson's and Roos) on exam. This discrepancy between presentation and diagnosis underscores the necessity for heightened TOS awareness, appropriate referrals, and expanded diagnostic tools. Current reliance on brachial plexus or upper-extremity MRI may overlook contributory breast-centric factors and surrounding myofascial pathologies. Despite the hypothesized role of the pectoralis muscle in development of postreconstruction TOS symptoms, none of undiagnosed individuals in our study received a pectoralis minor block for work up of TOS, although this minor procedure is suggested by the Society for Vascular Surgery.10,38 Recent advancements in diagnostic imaging, such as contrast-enhanced magnetic resonance angiography (CE-MRA), short inversion time inversion recovery sampling perfection with application-optimized contrasts using different flip angle evolutions (T2-STIR-SPACE), and volumetric interpolated breath-hold examination (VIBE) sequence have demonstrated efficacy in detailed neurovascular bundle evaluation, including fibromuscular anomalies and posture-related factors, both relevant to breast reconstruction outcomes and TOS manifestations. 39 These innovative imaging strategies could enhance TOS diagnostic accuracy in postreconstruction patients, although dependent on prompt symptom identification and appropriate specialist referral.
Nonetheless, this study is not without limitations. With a limited sample size, it is difficult to extrapolate enough information to draw conclusions about true causality with regards to implant-based reconstruction and subsequent development of TOS symptoms. Its retrospective design is subject to confounders, such as the effect of additional surgical procedures to address complications and need for adjuvant therapies like chemotherapy and radiation, especially when considering prolonged timing between breast reconstruction and onset of symptoms. While existing literature sporadically acknowledges breast reconstruction as a potential TOS etiology, this paper is the first to our knowledge to report plausible interlinking mechanisms through retrospective analysis.2,34 Future initiatives aim to broaden the research demographic, aspiring that these preliminary insights will heighten awareness of the possible TOS linkage, thereby facilitating timely referrals and comprehensive diagnostic evaluations for affected individuals, ultimately paving the way for effective TOS alleviation.
Conclusions
This study attempts to explore the relationship between implant-based breast reconstruction surgery and the subsequent development of TOS symptoms, as determined by temporality of symptom onset relative to reconstruction and diagnostic work up. We postulated that breast surgery, reconstruction or otherwise, due to iatrogenic trauma and subsequent fibrosis in an anatomic area wherein neurovascular structures are closely associated, was the main etiology leading to compression of the thoracic outlet structures. Interestingly, a formal diagnosis of TOS was received by those patients who underwent subpectoral implant placement, suggesting there may be additional determinants for patients postreconstruction.
Footnotes
Acknowledgments
i2b2 software was used in conducting this study. i2b2 is the flagship tool developed by the i2b2 (Informatics for Integrating Biology and the Bedside) Center, an NIH funded National Center for Biomedical Computing based at Partners HealthCare System. The i2b2 instance at the University of North Carolina is supported by the National Center for Advancing Translational Sciences (NCATS), National Institutes of Health, through Grant Award Number UL1TR002489. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author Contributions
Dr Carr represents this work's senior author and primary investigator. Dr Ndem and Ms. Longfellow represent co-first authorship of this manuscript, involved in inception, data collection and analysis, literature review, and manuscript draft and editing. Dr Kapsalis is next author as she was instrumental in literature review and manuscript draft and editing. Ms. Cook and Mehta were involved in data collection and literature review. Drs. Kugar and Lopez were instrumental in inception of project as well as initial literature review.
Consent to Participate
The IRB waived informed consent for research [45 CFR 46.116 (f)] and waiver of HIPAA authorization [45 CFR 164.512 (i) (2) (ii)] due to minimal risk.
Consent for Publication
Not applicable. This study does not include identifiable individual participant data.
Data Availability Statement
Data are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent Statements
This study received ethical approval from the University of North Carolina at Chapel Hill IRB (approval #20-1056) on July 23, 2023. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Presented
This article was presented at North Carolina Society of Plastic Surgeons Annual Meeting, Kiawah Island, SC, on November 2023; University of North Carolina Nathan A Womack Surgical Society Research Day, Chapel Hill, NC, on May 2024.
