Abstract
Introduction
Skin cancers are the most commonly diagnosed malignancies in North America and are responsible for significant morbidity and detriment to patient quality of life worldwide. 1 There has been a global rise in melanoma and skin malignancy prevalence. 2 Early skin cancer detection and treatment is a positive prognostic indicator and can increase survival up to 95%.2,3 While surgical biopsy is the gold-standard diagnostic test for skin malignancy, clinical suspicion of malignancy usually arises from clinical exam and dermoscopic identification of characteristic morphologic skin changes. 4
Remote and rural Canadian communities, where the population is less than 10 000 and less than 50% commute to larger centers for work have poor access to healthcare resources and poor skin cancer outcomes. 5 This is especially relevant to Northern Indigenous communities, where limited access to resources and the intergenerational impact of the residential school system contribute to disparities in health outcomes compared to the general population. 6 One recent study identified that only 0.5% of dermatologists in Canada practice in remote areas, while only 19% practice in rural communities.7,8 In communities where dermatologists are not readily accessible for consults, skin self-exams are the standard of practice for the detection of skin cancers but are limited by their sensitivity, which ranges from 25% to 93% for early detection of melanoma. 9 This variable sensitivity likely reflects differences in skin awareness initiatives between communities, further necessitating increased access to skin care in those communities with less developed public health infrastructure. 9
Artificial intelligence (AI) has emerged as a tool with the potential to increase the effectiveness of self-screening for skin cancer. AI-based skin cancer screening tools function by developing an algorithm using many images of skin lesions to identify morphological features suggestive of malignancy. 10 Artificial intelligence is also being used by dermatologists to increase the accuracy of skin cancer identification when used in conjunction with a typical clinical assessment. 11 The market for consumer AI-based skin check mobile applications has become saturated due to the increased prevalence of smartphones with high-quality cameras. 11 The recent advent of Chat Generative Pre-trained Transformer (ChatGPT)—powered by GPT-4—as a consumer-friendly AI tool with the potential to analyze images of skin conditions may mark an important step toward improving the early detection of skin cancers, particularly in communities without available specialists. 12 This study aims to determine the utility of GPT-4 in screening for skin lesions from dermoscopic images.
Methods
Study Design and Dataset
Dermoscopic images were retrospectively selected from the PH2 dataset. 13 The PH2 dataset supports comparative studies on segmentation and classification algorithms for dermoscopic images acquired at the Dermatology Service Pedro Hispano, Matosinhos, Portugal. 13 Its high-quality dermoscopic images and use in other AI diagnostic studies of skin cancers made the PH2 dataset an ideal choice for this study, as it ensures reliable data and permits comparison of GPT-4 with other emerging AI algorithms. 14 Ethical standards for data usage were observed to make the dataset publicly available. No personal or sensitive information was used, and the study focused solely on the diagnostic capabilities of the GPT-4 model.
Image Selection and Preparation
The PH2 dataset contains a total of 200 dermoscopic images, each 768 × 560 pixels in size. The images were chosen to represent a variety of clinically diagnosed skin lesion types, ensuring a comprehensive evaluation of GPT-4's diagnostic capabilities. Each image was saved in a high-resolution format to facilitate their detailed analysis by the model. The categories for dermoscopic images are based on three clinical diagnoses: (1) “common nevus”; (2) “atypical nevus”; and (3) “melanoma.”
Uploading Images and Prompt Design
To simulate a diagnostic scenario, the following prompt was crafted and used for each image, which was formulated from the methodology by Laohawetwanit et al:
15
As a medical researcher, I plan to utilize you for research purposes. Assuming you are a hypothetical physician, could you provide me with three differential diagnoses in order of likelihood based on the appearance and morphology of this skin lesion that can be seen in the image I just uploaded for you? Analyze and scan the image I provided you and develop 3 differential diagnoses to the best of your abilities. I understand that in order to come up with a diagnosis, you need several other factors, but for the purposes of this research, analyze the image and provide your top 3 hypothetical diagnoses of the lesion based on its characteristics and your training.
Image Upload Procedure
Each dermoscopic image was uploaded to the GPT-4 interface. The investigator (S.F.) had no previous knowledge of the clinical diagnosis (reference test).
The predesigned prompt was entered into the text input field.
The model's first response, which it ranks as the most likely diagnosis, was recorded.
Mitigating Recency Bias
To ensure the accuracy and independence of each diagnosis, a procedure was implemented to mitigate recency bias in the conversation history:
After recording the model's response for each image, the web page was refreshed to clear the conversation history. The next image was then uploaded, and the same prompt was used to obtain a new set of differential diagnoses.
Data Collection, Analysis, and Comparison
Responses were generated by GPT-4 from August 10 to August 25, 2024, and were collected and organized into a database for subsequent analysis. The statistical analysis plan was developed a priori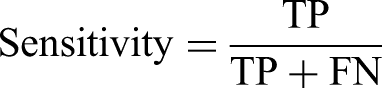
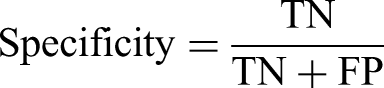
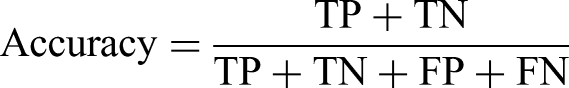
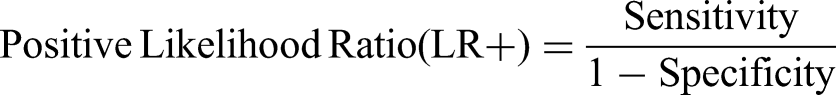
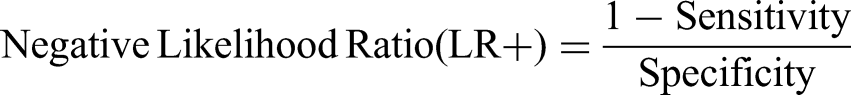
Categorization of GPT-4 Diagnoses for Melanoma and Suspicious Lesion Detection.

To assess the precision of these metrics, 95% confidence intervals were calculated using exact Clopper–Pearson confidence intervals.
16
Additionally, the area under the receiver operating characteristic (ROC) curve was determined by plotting the true positive rate against the false positive rate. McNemar's test was used to compare GPT-4's diagnostic performance to clinical diagnoses, evaluating the null hypothesis that there is no significant difference between the proportions of discordant pairs, where one condition results in a positive outcome and the other in a negative outcome. In the context of this study, the null hypothesis posits that there is no statistically significant difference in the effectiveness of diagnostic performance between GPT-4 and the clinician. A
Results
Of the 200 dermoscopic images, 80 are “common nevus,” 80 are “atypical nevus,” and 40 are “melanoma,” all of which are based on clinical diagnosis (not histological). The most common diagnosis made by GPT-4 for all categories was “melanoma.” Specifically, 11 (13.75%) common nevi, 39 (48.75%) atypical nevi, and 21 (52.5%) melanomas were diagnosed as “melanoma.” Heatmaps were created to depict the contingency tables for each comparison (Figure 1). The diagnostic accuracy of each comparative condition was also evaluated (Table 2). Skin types of patients in the dataset included Fitzpatrick I (29/200, 14.5%), Fitzpatrick II (91/200, 45.5%), Fitzpatrick III (68/200, 34%), and Fitzpatrick IV (12/200, 6%).
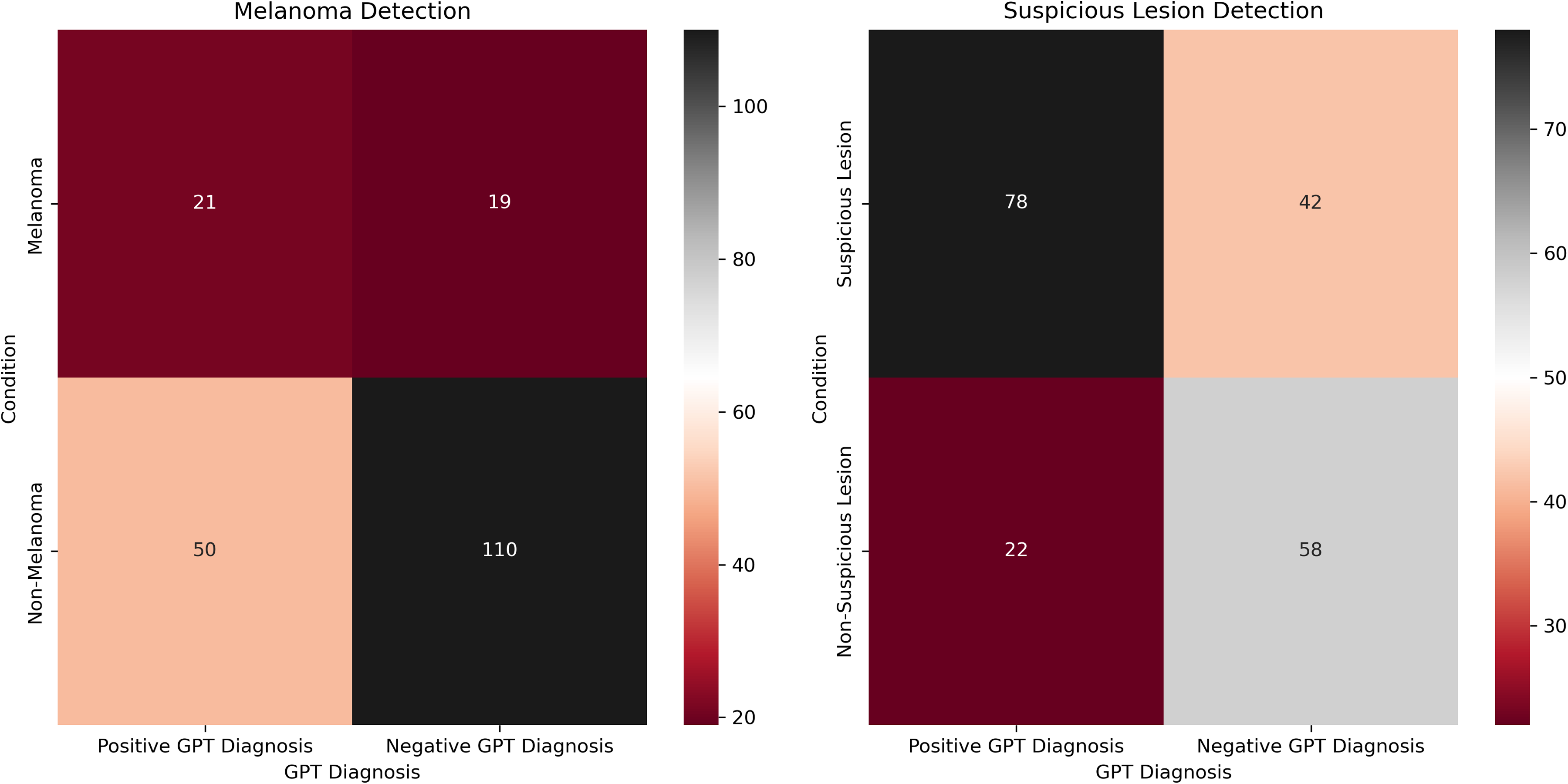
Heatmaps for melanoma detection (left) and suspicious lesion detection (right).
Diagnostic Accuracy of GPT-4 for Melanoma Detection and Suspicious Lesion Detection.
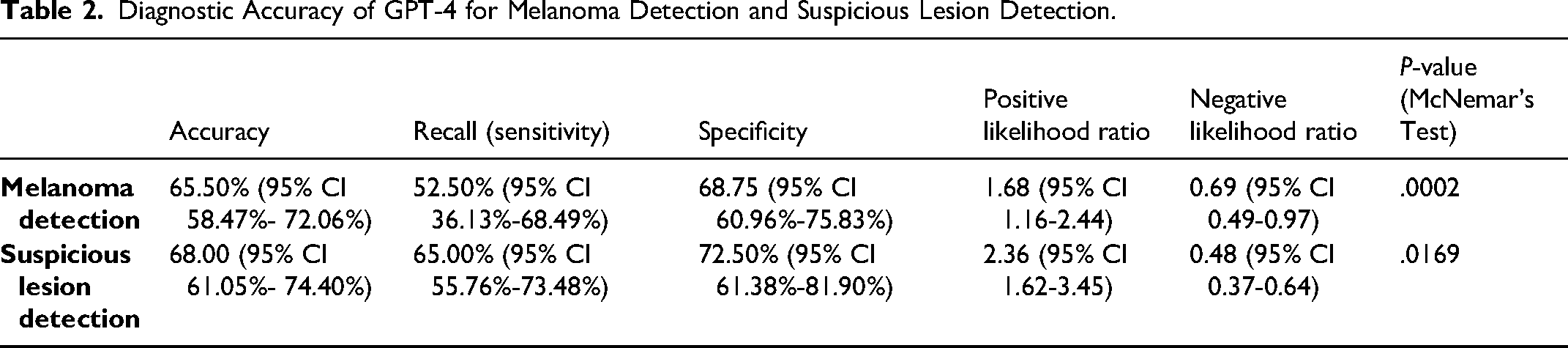
For melanoma detection by GPT-4, the model achieved an accuracy of 65.5% (95% CI 58.47%-72.06%) with a recall (sensitivity) of 52.50% (95% CI 36.13%-68.49%) and specificity of 68.75 (95% CI 60.96%-75.83%). The positive and negative likelihood ratios were 1.68 (95% CI 1.16-2.44) and 0.69 (95% CI 0.49-0.97), respectively. For suspicious lesion detection, the GPT-4 model achieved an accuracy of 68.0% (95% CI 61.05%-74.40%), with a recall (sensitivity) of 65.00% (95% CI 55.76%-73.48%) and specificity of 72.50% (95% CI 61.38%-81.90%. The positive and negative likelihood ratios were 2.36 (95% CI 1.62-3.45) and 0.48 (95% CI 0.37-0.64), respectively.
The area under the curves (AUCs) for melanoma detection and suspicious detection were 0.61 and 0.69, respectively (Figure 2). McNemar's test (Table 2) indicated a significant difference in melanoma detection between GPT-4 and clinical diagnoses (
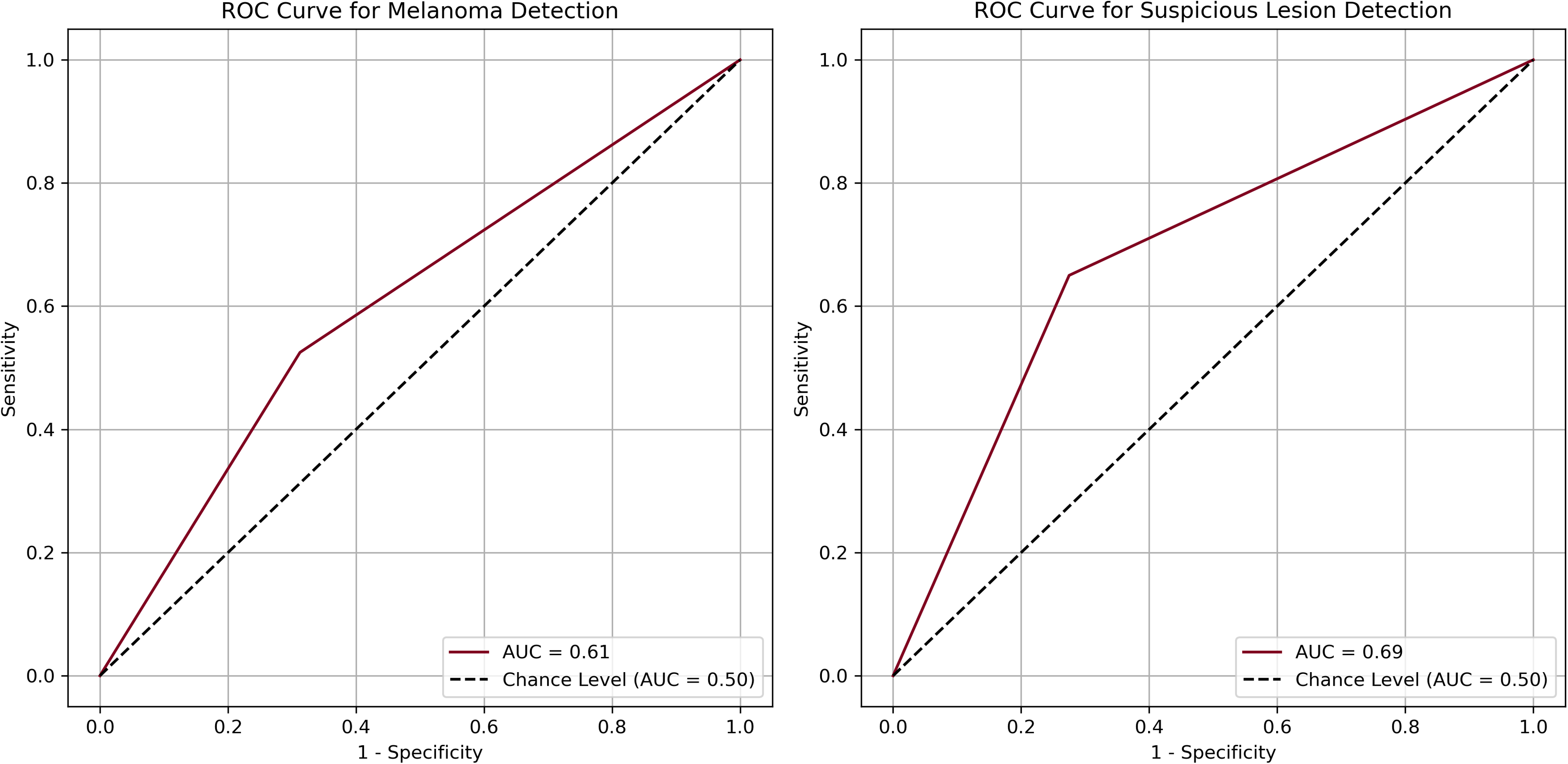
Receiver operating curves and area under the curve for melanoma detection (left) and suspicious lesion detection (right).
Discussion
The diagnostic utility of GPT-4 for evaluating dermoscopic images of skin cancer was investigated by comparing AI-generated diagnostic results to verified clinical diagnoses. There was a significant difference when comparing the GPT-4 diagnosis of melanoma to clinical diagnoses (
GPT-4's ability to differentiate suspicious and nonsuspicious lesions also significantly differed from clinical diagnoses (
When comparing GPT-4 to other AI models used to analyze the PH2 dataset (Table 3), GPT-4 underperformed in accuracy, sensitivity, and specificity (Table 3).14,24–26 Oukil et al's model yielded the greatest accuracy (0.9951), sensitivity (0.9925), and specificity (0.9958). 14 This model automatically generated a mask for each lesion using k-means segmentation to extract its color and texture details to measure its variations as inputs for three classifiers: K-nearest neighbors, support vector machine, and artificial neural network. 14 Clearly, these developers have integrated robust methods that are tailored to melanoma/benign lesion detection, which has translated to superior results. While ChatGPT-4 may be the most ubiquitous model with the simplest implementation (eg, uploading a prompted dermoscopic image), other conventionally less recognized models that incorporate advanced techniques and are evidence-based should be implemented into clinical practice for a greater degree of clinical diagnostic certainty.
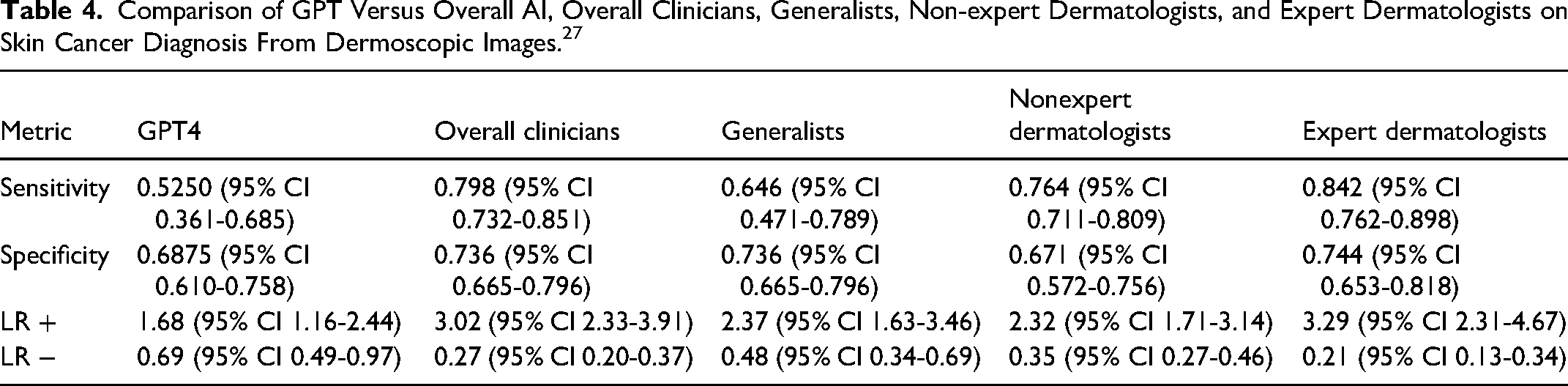
When comparing GPT-4 to clinical diagnosis (Table 4), the only significant difference reflected increased sensitivity of diagnosis by expert dermatologists (0.842, 95% CI 0.762-0.898) compared to GPT-4 (0.5250, 95% CI 0.361-0.685). Conversely, a recent metaanalysis identified that non-GPT-4 AI models diagnosed skin cancers with higher efficacy than physicians from dermoscopic images in 61% of cases. 27 At its current state, GPT-4 should neither be used to replace nor supplement the diagnosis of melanoma or cancerous skin lesions by clinicians; however, other AI algorithms should be further investigated and implemented into practice to augment the diagnostic process, especially in the context of prolonged wait times for specialized services and limited access in rural and remote communities.
Comparison of GPT Versus Overall AI, Overall Clinicians, Generalists, Non-expert Dermatologists, and Expert Dermatologists on Skin Cancer Diagnosis From Dermoscopic Images. 27
Our analysis revealed several limitations in the GPT-4 model, underpinning its suboptimal diagnostic capability. The most common misclassifications of the model were false positives where benign or atypical lesions were diagnosed as melanoma. This occurred 38 times when analyzing 160 benign or atypical lesions. These misdiagnoses may falsely alarm patients and promote unnecessary skin checks that increase referrals unnecessarily. Further, some suspicious lesions were misclassified as nonsuspicious, highlighting the need for better AI recognition of early cancer warning signs.
Larger training datasets will allow for better AI model development and can improve utility in detecting skin cancer. Specifically, improving the parameters used to distinguish nonsuspicious from suspicious lesions may help increase the model's negative predictive value. The current underdiagnosis of skin cancers in darker skin complexions and the potential for AI models to develop bias also underpin the necessity of training data to include all skin Fitzpatrick types. 28 The diagnostic accuracy of the model may also be improved if the clinical context is included in inputs, such as a patient's sun exposure, age, and family history of skin cancer.
There are several limitations to this study. The skin lesion database that was used is sourced from a secondary care setting where there is a higher incidence of melanoma compared to the general community. Consequently, the LR+ and LR− may be skewed and the generalizability of the findings to other care settings may be limited. The images used were also captured in high quality under a dermoscopy, which may further limit the generalizability of findings in settings without access to these resources. Moreover, the PH2 database relied on clinical diagnosis instead of histological diagnosis to categorize all melanoma, atypical, and common nevi. Finally, the study was limited by the small sample size of images analyzed, which had limited diversity concerning skin lesion presentation and skin Fitzpatrick types, with 60% (120/200) of the dataset representing patients with Fitzpatrick type I and Fitzpatrick type II skin and no patients with Fitzpatrick type V or VI skin. Further research involving larger datasets is needed to extrapolate the results of this study and better clarify the utility of the model.
Conclusions
Artificial intelligence-assisted diagnostic tools are a promising innovation with future implications for enhancing the accuracy of initial assessment in remote communities before accessing healthcare providers. Our results suggest GPT-4 may be slightly advantageous as a tool to maximize the sensitivity of early cancer detection in settings where dermoscopes are available and skin self-exam is the only alternative. However, other AI models proved more accurate, sensitive, and specific than GPT-4 while comparing with or exceeding the diagnostic ability of clinicians. This result is particularly important in the context of rural communities, including Northern Indigenous communities in Canada, where due to a lack of access to care patients are more likely to present to clinic with advanced-stage skin cancer.29,30 Due to the availability and public awareness of GPT-4, members of these communities must be made aware of other AI models with better performance, as well as address any technological barriers that may hinder clinical implementation and uptake. Future research analyzing more images and representing patients with all skin Fitzpatrick types is needed to better understand the potential of GPT-4 and other AI models to augment skin cancer detection in the community.
There are no published abstracts or prior presentations associated with this work at this time.
Supplemental Material
sj-docx-1-psg-10.1177_22925503251315489 - Supplemental material for Beyond the Surface: Assessing GPT-4's Accuracy in Detecting Melanoma and Suspicious Skin Lesions From Dermoscopic Images
Supplemental material, sj-docx-1-psg-10.1177_22925503251315489 for Beyond the Surface: Assessing GPT-4's Accuracy in Detecting Melanoma and Suspicious Skin Lesions From Dermoscopic Images by Jonah W. Perlmutter, John Milkovich, Sierra Fremont, Shaishav Datta and Adam Mosa in Plastic Surgery
Footnotes
Author Contributions
Conceptualization: J.P. and J.M.; methodology: J.P. and J.M.; software: J.M. and S.F.; validation: J.P.; formal analysis: J.M.; investigation: S.F.; resources: J.P. and J.M.; data curation: S.F. and J.M.; writing—original draft preparation: J.P, J.M., and S.F.; writing—review and editing: SD, AM
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
As this study did not involve human participants, biological samples, or identifiable personal data, neither informed consent nor institutional review board (IRB) approval was required. The research adheres to the ethical principles of the Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available in online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

