Abstract
Current practice of osteoarthritis has its insufficiencies which researchers are tackling with artificial intelligence (AI). This article discusses three kinds of AI models, namely diagnostic models, prediction models and morphological models. Diagnostic models enhance efficiency in diagnosis by providing an automated algorithm in knee images processing. Prediction models utilize behavioral and radiological data to assess the risk of osteoarthritis before symptom onset and needs to perform surgery. Morphological models detect biomechanical changes to facilitate understanding of pathophysiology and provide personalized intervention. Through reviewing present evidence, we demonstrate that AI could assist doctors in diagnosis, predict osteoarthritis and guide future research.
Introduction
Osteoarthritis
Osteoarthritis is a degenerative disorder that is getting more common as the global population continues to age. 7% of the global population are affected by osteoarthritis in 2020, and the disease burden is one of the greatest amongst non-communicable diseases. The most common site of osteoarthritis is the knee, followed by the hip and the hand joints. Patients with knee osteoarthritis has one of the highest disability-adjusted life years. 1 Yet, the treatment developed for knee osteoarthritis has not caught up the uprising trend of osteoarthritis. In fact, knee osteoarthritis is often overlooked and downplayed by health practitioners compared to other non-communicable diseases such as cardiovascular disease and diabetes mellitus. 2 As a result of uprising prevalence and oversight, healthcare burden of osteoarthritis continues to rise globally, particularly in developed countries. 3 Osteoarthritis has therefore become a major public health challenge, as we attempt to improve healthcare service for osteoarthritic patients.
Artificial intelligence, machine learning and deep learning
When it comes to artificial intelligence (AI), the terminology can be as perplexing as medical jargons, if not more confusing. Artificial Intelligence, which are computer programs with the ability to learn and reason like humans, is an emerging area of medical research. Machine Learning is under the umbrella term of artificial intelligence that specifically refer to algorithms that is able to learn by itself and generally improves with experience. Deep learning refers to machine learning programs that is built based on “neural network”, which are built based on the neural network structure of humans and they are able to adapt themselves through repetitive training to uncover patterns and generate insights. Figure 1 explains the relationships between the terms. All of them perform complex tasks through a series of statistical analysis of input and output. The input varies from behavioral pattern such as gait and health seeking behavior to medical images like X-ray images, CT Scans and MRI images. The output also varies from a simple “yes or no” in diagnosis to a quantitative numerical “risk index” in prognostication. One of the most commonly applied machine generated model is the convoluted neural network (CNN), a deep learning algorithm designed similar to human brain neurons, which has somewhat led to higher diagnostic accuracy, less inter-rater variability and more successful prediction of future symptomatology. 4 As promising as it sounds, the use of deep learning in medicine is still in the starting point, and more research in the relevant field is needed for the application of deep learning in clinical context.
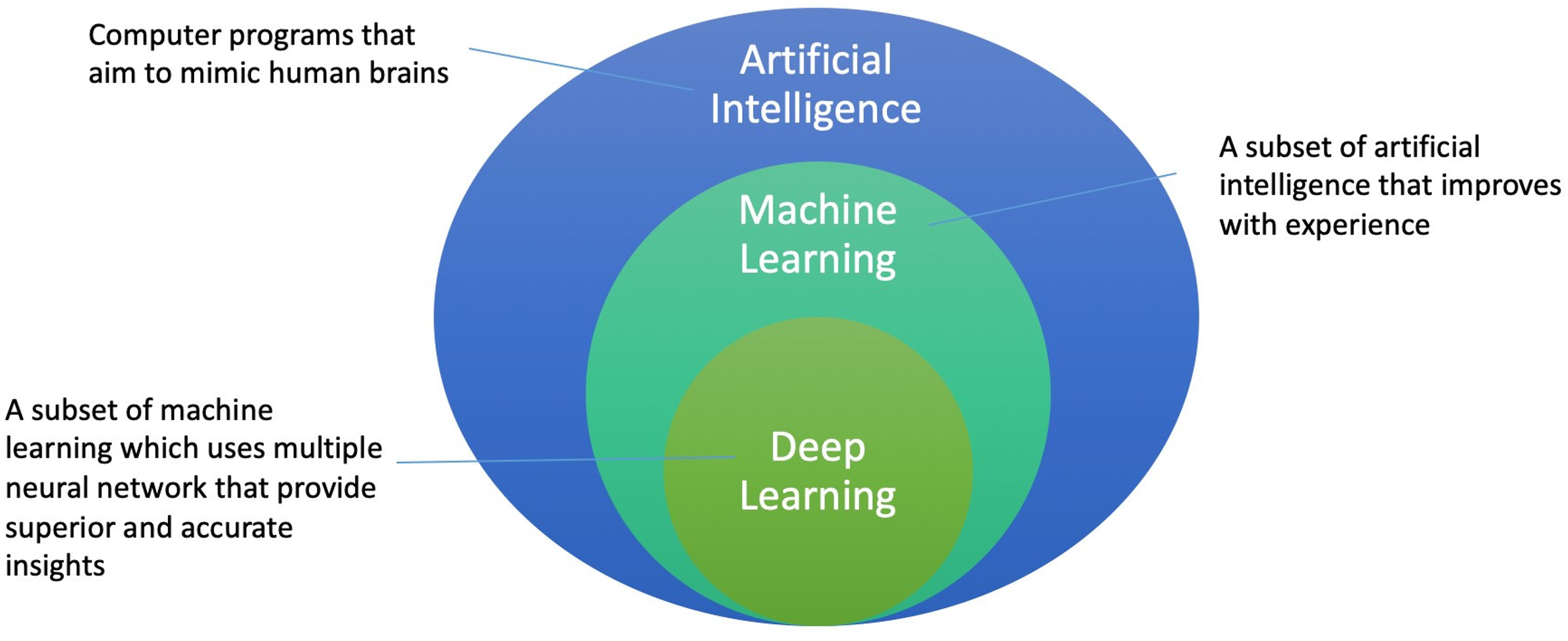
Relationship between artificial intelligence, machine learning and deep learning.
Knee osteoarthritis: current practice and its barriers
This section discusses the risk factors, diagnosis and grading, as well as management of osteoarthritis. In the discussion, current pitfalls that can be potentially tackled by artificial intelligence are also identified.
Risk factors for osteoarthritis
Osteoarthritis is a multifactorial, complex disorder with various risk factors that contribute to the end pathology. For non-modifiable risk factors, osteoarthritis is traditionally associated with age and female sex, with recent research demonstrating that genetics could also predispose to osteoarthritis. 5 Occasionally, osteoarthritis can be attributed to previous joint injury, particularly anterior cruciate ligament in knee osteoarthritis. 6 For modifiable risk factors, obesity and occupation play important roles in the development of osteoarthritis. Other possible risk factors include physical inactivity, smoking and dense bones. Early detection of osteoarthritis and prediction of disease progression can prompt non-pharmacological treatment of osteoarthritis with the intention to slow down the disease progress, which is highly important given osteoarthritis is an irreversible process.
Imaging for osteoarthritis: diagnosis and grading
Diagnosis of knee osteoarthritis are mainly based on clinical signs and symptoms as well as imaging of the knee joint. Pain related to use and relieved by rest, decreased range of motion and bony swelling are typical of knee osteoarthritis. Imaging modalities, such as Knee X-rays and magnetic resonance imaging (MRI), can detect the presence and grade the severity of osteoarthritis. To diagnose osteoarthritis, at least two views (anteroposterior and lateral view) shall be used to identify all three compartments of the knee. The typical radiological features of osteoarthritis include marginal osteophytes, subchondral sclerosis, subchondral cysts and joint space narrowing. The most widely used criteria for diagnosing OA in knee X-rays is the osteoarthritis research society international (OARSI) criteria, which includes (a) Joint space narrowing of grade 2 or higher, (b) sum of osteophyte grade ≥2 and (c) Grade 1 joint space narrowing in combination with a grade 1 osteophyte. 7
The most common grading system of osteoarthritis is the Kellgren-Lawrence (KL) System 8 (Table 1) (Figure 2). In KL system, the first radiological sign of osteoarthritis is joint space narrowing.
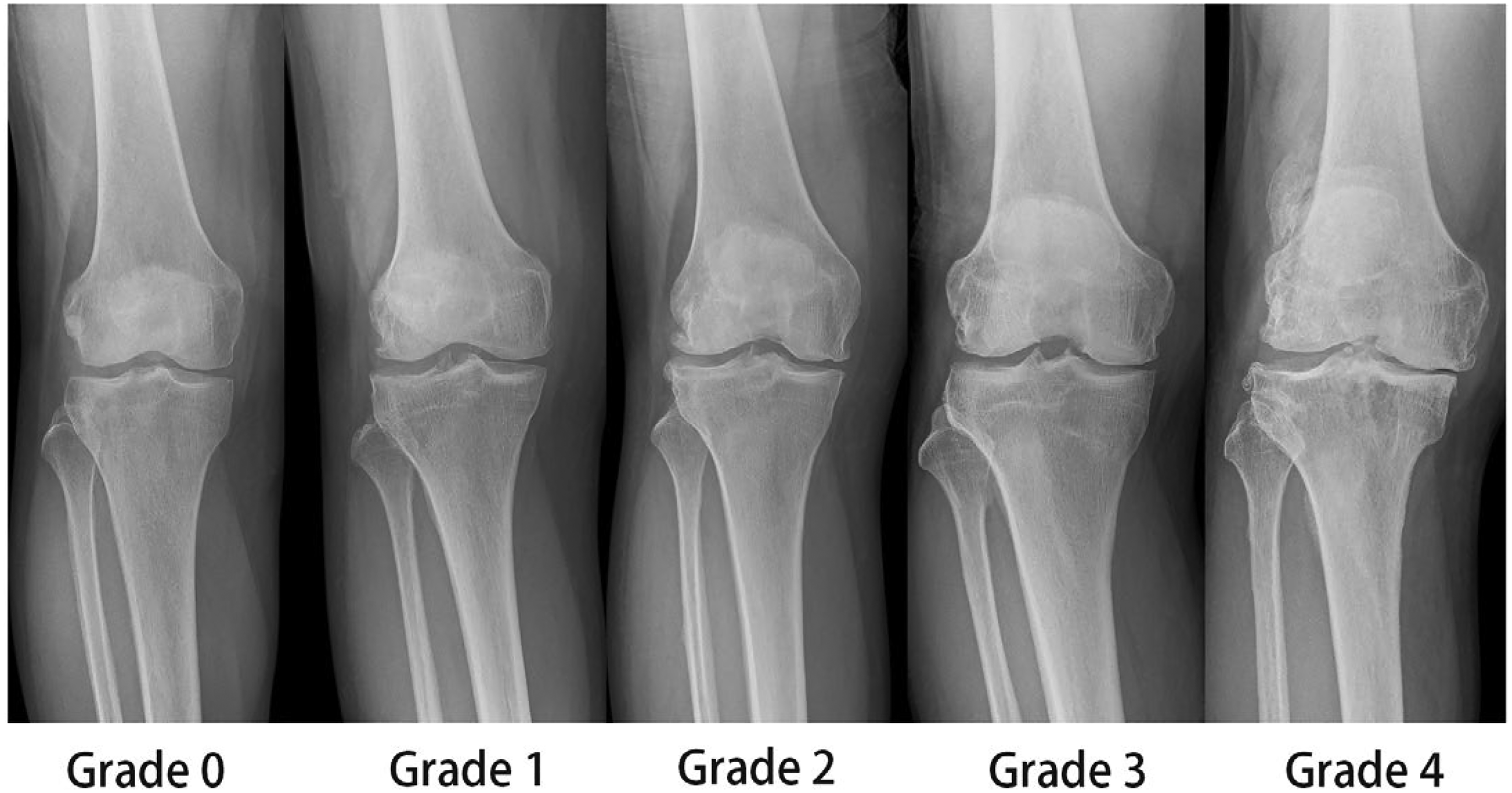
Kl grading on knee X-rays.
Kl grading for osteoarthritis.

The problem with current methods of assessing knee osteoarthritis in Knee X-rays is that it fails to provide robust and accurate prediction of disease progression and treatment outcome. The KL classification has been criticized that it is inapplicable to disease progression, with the reason behind being the assumption of progression of osteoarthritis shall be from osteophyte formation to joint space narrowing, therefore patients with joint space narrowing without osteophyte formation could not be assessed. 9 Furthermore, the KL system do not take patient's personal risk factors and behavioral pattern into account, hence failing to accurately predict the progression of knee osteoarthritis in different individuals. For example, a female obese patient with predisposing genetic factors having the same KL grade of knee osteoarthritis are more likely to have worse prognosis than a lean male patient. Kim et al. found that the assessment of joint space width together with additional patient information is preferable in monitoring the disease progression over KL classification. 10
Another research gap identified in knee osteoarthritis research is the use of MRI knee. The application of MRI knee has enabled clinicians and scientists to have better understanding of the biomechanical changes and progress in osteoarthritis. The detection of “pre-morphologic” changes in cartilage and peri-articular tissues lead to earlier diagnosis of knee osteoarthritis. MRI is also useful in classifying osteoarthritis into different phenotypes, which cannot be achieved by conventional radiography. The increasing trend of MRI application calls for more research into universal guidelines for diagnosing and grading severity of osteoarthritis on MRI, where artificial intelligence could help.
Management of osteoarthritis
Management of knee osteoarthritis include non-pharmacological which target mild osteoarthritis, pharmacological and surgical measures which target moderate and severe osteoarthritis. Non-pharmacological measures mainly include education on disease process, weight reduction, exercise and biomechanical intervention such as walking canes. Pharmacological measures consist of pain relief medications such as paracetamol, oral or topical non-steroidal anti-inflammatory drugs and intra-articular steroid injection. Intra-articular hyaluronic acid, opioids and duloxetine are also used in cases of severe pain. Surgical measures are indicated in patients with refractory knee pain, with the current gold standard treatment being Total Knee Arthroplasty. Alternatives to TKA are knee osteotomy and unicompartmental knee arthroplasty, which are viable options for younger patients and patients with unicompartmental end-stage osteoarthritis respectively.
How could deep learning for osteoarthritis tackle the current problem?
This section aims to provide reader with simple and concise concepts of deep learning in medical imaging and an overview of deep learning application in knee osteoarthritis and its benefits.
Brief summary of deep learning
Deep learning is a subgroup of machine learning, under the umbrella term of artificial intelligence. Deep learning stands out from traditional machine learning as the conclusion can be generated automatically instead of manually. It does not require “human supervision”, meaning that data can be extracted and analyzed without manual labeling. Inputs for deep learning can be stratified into traditional numerical values and medical images.
For inputs of numerical values such as demographic data, medical history, biomarkers, and kinetic variables (such as ground reaction force and first peak rotational angle), random forest regression models are the state-of-the-art statistical analysis model. To understand random forest regression, one must first comprehend decision tree system. Decision tree uses a tree diagram to delineate the process of categorization. The root (starting node) is the first categorization process, then they are split into different branches to undergo further categorizations, and finally arrive at the leafs (terminal node). The system would store prior input and output information to build a decision tree diagram, and when new input information is put into the tree diagram it would try to predict the outcome. Random Forest Regression is the summation of a large amount of decision tree diagram to generate the average outcome or the mean value.
For inputs of imaging, deep learning algorithms use pixel values directly in images as the input information, as opposed to “traditional” machine learning which uses “features” identified in the image such as contrast, size and circularity. 11 Figure 3 illustrates the difference between traditional machine learning and deep learning in a simple manner. In the case of knee imaging, the model can diagnose and grade osteoarthritis just with the statistical pixel values without applying the traditional radiological signs of osteoarthritis and classical grading system. This gives an edge to artificial intelligence over traditional imaging, as it could detect lesions that are microscopic to the human eye. Two classes of deep learning models are popular in medical imaging, which are convolutional neural networks (CNN) and massive-training artificial neural networks (MTANN). There are two significant differences between the two. Firstly, the output of CNN consists of class categories, whereas the output of MTANN consists of continuous values. 12 Secondly, CNN in general requires larger number of training samples, i.e. medical images, than the MTANN. 13 The application of CNN has covered the vast majority of medical imaging, including chest radiographs, positron-emitted computed tomography and brain MRI. 14
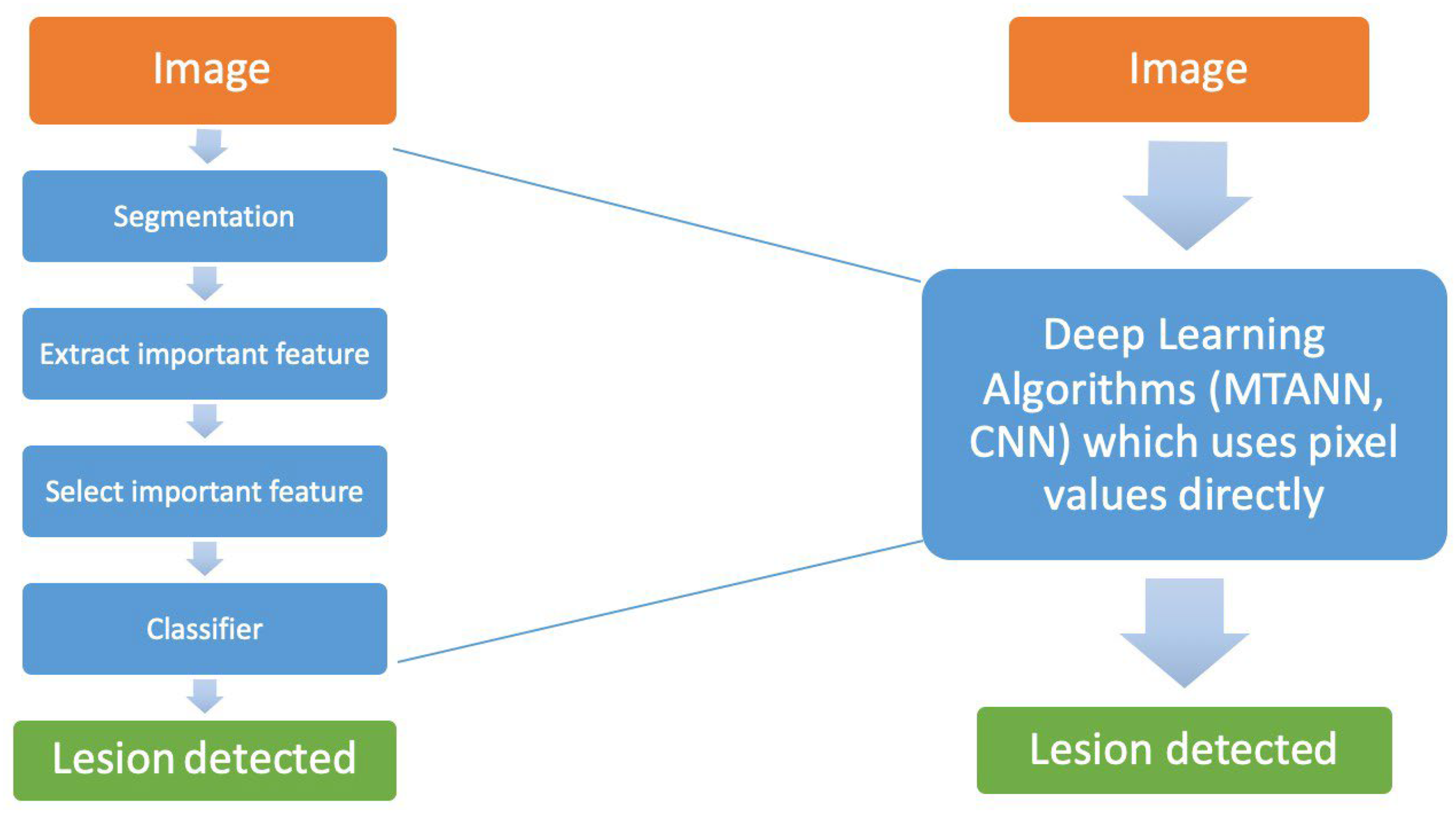
Comparison between traditional machine learning (left) and deep learning (right).
Application of deep learning in knee osteoarthritis
The application of deep learning in knee osteoarthritis can be divided into diagnostic model, prediction model, and morphological model based on the output of the algorithm. Diagnostic models are models that give accurate and rapid diagnosis of osteoarthritis. They are typically developed based on knee X-rays as it is comparably cheap and readily available. Morphological models are models that generate new “hotspots” for the progress of osteoarthritis apart from traditional knee X-rays signs. They are usually based on MRI as it is better in soft tissue differentiation. Prediction models are models that give a numerical value of “risk index” of how likely one is going to develop severe osteoarthritis or require surgical intervention. These models are often based on multiple variables, which include Knee-X Ray, Knee MRI, patient's demographics and many others.
Diagnostic models for knee osteoarthritis
The basic principle of deep learning algorithm in diagnosing knee osteoarthritis is not difficult to comprehend. (Figure 4) Most, if not all, diagnostic models rely on pixel values to classify knee X-rays into disease state or healthy state. Some more advanced models would also be able to classify according to KL grading of the knee X-rays.7,15 The input can be either Knee X-rays or MRI images. Before processing, a process called segmentation is done to extract the useful parts for analysis, which is the medial and lateral knee joint in this case. The data is then processed by deep learning models, which recognize the image by comparing to the database and generate a probability distribution. The output is therefore a suggested diagnosis or grade which has the highest probability by the computer algorithm.
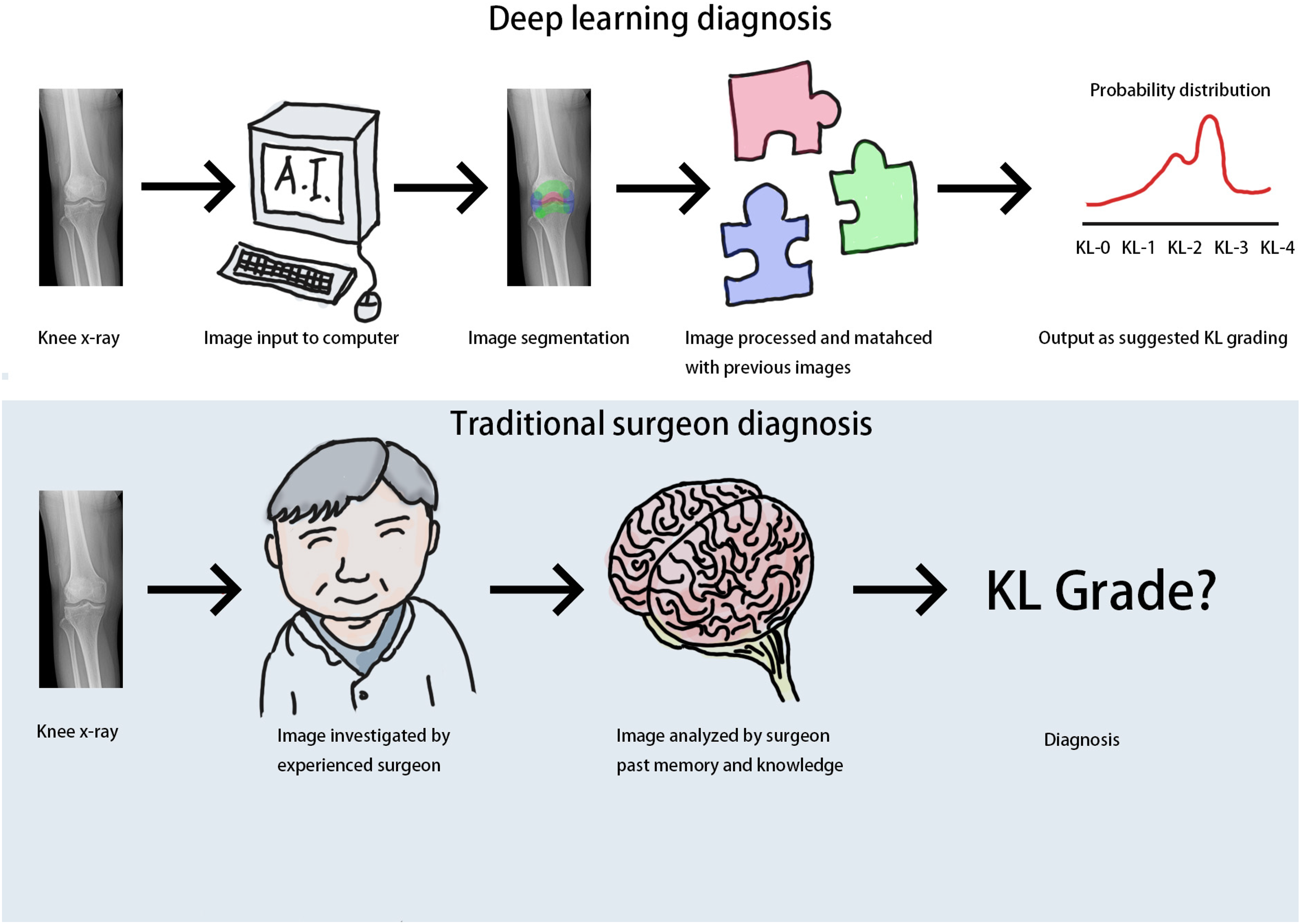
Basic principle of diagnostic model in knee osteoarthritis.
The current use of deep learning to analyze knee X-rays for diagnosing early knee osteoarthritis has been described by many. In 2018, one of the most quoted model is the automatic diagnosis method published by Tiulpin et al. 15 Using CNN, Tiulpin's model achieved a high Kappa coefficient, reflecting his model has low misclassification errors compared with the expert annotations in the MOST dataset. Liu et al. 16 further improved the accuracy and speed of the CNN, where his model only takes 0.33 s to process each knee X-rays image. Schwartz et al. 17 conducted an experiment to compare the accuracy in diagnosing osteoarthritis between the CNN and four fellowship-trained arthroplasty surgeons in the united states, with results demonstrating that a CNN can classify knee osteoarthritis just as accurate as healthcare professionals. Taking the technology into the clinical context, Nguyen et al. 18 proposed a novel method of semi-machine learning, which allows machine learning to supplement manual labeling. Tiulpin and Saarakkala 19 has recently published another model that could not only diagnose osteoarthritis on knee X-rays, but also simultaneously grade the osteoarthritis into KL and OARSI grading system. The use of machine learning in diagnosing knee osteoarthritis has several benefits. Firstly, diagnosis of knee osteoarthritis could be made quicker using an automated diagnostic method. Patients could receive non-surgical treatment and education at an earlier stage of disease, in the hope of delaying disease progression. Secondly, the use of an automated image analysis could alleviate the cost in healthcare provision, especially the person-hour of professionals. Yet, most of the current models developed are based on the osteoarthritis Initiative data, which focus on African Americans and might not be representative to all populations. 15
Prediction models for knee osteoarthritis
Deep learning-based prediction models for knee osteoarthritis is a useful tool to assess disease progression and the need for knee replacement surgery. Basic principle of prediction models varies as different researchers use different input values. Models that use medical imaging as input have similar approach as diagnostic models. Models that use health utilization behavior mostly apply principal component analysis in the deep learning algorithm processing that look at the statistical distribution of the health data. Some other models combine the both principles for more accurate prediction. The output could be binary (e.g. requirement for surgery or not) or continuous (e.g. how likely the patient develop end stage osteoarthritis).
Although there are a multitude of prediction models using different input variables amongst the current evidence, one thing in common is that they all aim to detect osteoarthritis at an earlier stage than current practice. For example, the model of Lim et al. 20 makes use of medical utilization and health behavior information as simple statistical data to predict the risk of developing osteoarthritis. Tiulpin et al. 21 predicted knee osteoarthritis progression with one single knee X-rays of each patient supplemented with clinical data, and successfully predict the outcomes even for patient with KL grade 0 or 1. The study done by Kim et al. 10 utilized additional patient information with the radiographic KL grading to demonstrate that the combination of both clinical and radiological input variables lead to a better prediction. As for MRI imaging, Ashinsky's et al. 22 model used multi-slice T2-weighted weight knee imaging on the central portion of medial femoral condyle to show early osteoarthritic changes in cartilage thickness. Kundu et al. 23 followed pre-symptomatic individuals for 3 years and successfully predict osteoarthritis with a robust accuracy of 78% by producing cartilage texture maps also on MRI imaging. More importantly, Tolpadi et al. 24 use deep learning to assess MRI images in order to predict the need for total knee replacement. Furthermore, Chan et al. 25 investigated the contributions of local and systemic risk factors of osteoarthritis through developing a novel combination of machine learning approaches. The result generated by the model is promising in predicting onset and deterioration of knee osteoarthritis, as well as validating certain risk factors such as history of injury, diabetes and smoking as key contributors of the development of knee osteoarthritis. The use of machine learning in predicting knee osteoarthritis have several benefits. Firstly, prediction models help to predict the likelihood of patients developing osteoarthritis, hence specific groups of high risk pre-symptomatic patients can receive additional checkup and timely intervention before osteoarthritis exacerbates. Secondly, personalized medicine can be provided to patients with specific risk factors, particularly modifiable risk factors that are amendable to prevent osteoarthritic development. Yet, the use of prediction models might give a false sense of reassurance to individuals that are predicted to have better prognosis. Vice versa, individuals predicted to have worse prognosis may suffer from unnecessary anxiety and excessive health seeking behavior.
Morphological models for knee osteoarthritis
Looking beyond diagnosis and prediction models, deep learning is also applied in the morphological changes in osteoarthritis. Basic Principle for morphological models involves use of CNN to analyze knee MRI and knee X-rays. Different from the diagnostic model, the morphological model often looks at multiple site known as “hotspots”. Popular hotspots include the patellar cartilage, meniscus and bone marrow. Besides, morphological models would use a technique called “data augmentation” to train the model to ignore small geometric difference by slightly rotating the samples for a few times. The image processing with deep learning model are otherwise similar to diagnostic model. As for the output, morphological models typically generate continuous figure such as cartilage thickness and degree of degenerative changes that can be stratified into different classes.
Several models have been developed to perform automated segmentation for knee structures, including meniscus and cartilage. Pedoia et al. 26 look at degenerative changes at meniscus and patellofemoral joint cartilage with the data from Osteoarthritis Initiative database, and successfully generated an automated MRI reading model which could detect degenerative changes in meniscus (80.7%) and patellar cartilage (80.2%). Ambellan et al. 27 developed a model to perform automated segmentation of knee bones and cartilage from MRI through combining statistical shape knowledge and CNN. Their model is also capable of giving semi-quantitative data of knee osteoarthritic change such as meniscal defects, presence of bone marrow lesions and radiographic scores. The application of deep learning in analyzing morphological changes in MRI leads to several advantages. With the help of deep learning, large amount of MRI data can be analyzed without much inter-rater variability and far less time-consuming. This would enable clinicians to set the benchmark for grading of MRI images. The use of deep learning in MRI images could also help identify new radiological biomarkers in knee osteoarthritis, supplementing or even replacing the conventional ones suggested by Kellgren and Lawrence. Clinicians and basic scientists, through learning the new radiological changes in different stages of osteoarthritis, could gain new insight and better understanding of the pathophysiological process of the disease. This could prompt the new development of pharmacological intervention which aims to reverse the seemingly irreversible process of osteoarthritis.
Conclusion
This article reviewed the up-to-date practice of knee osteoarthritis and the current barriers. The diagnosis of knee osteoarthritis is often made late (KL grade ≥2) due to extended follow-up time for knee pain, hindering prompt risk factors modification and non-pharmacological interventions. This could potentially be tackled deep learning models, providing automated radiological diagnosis of knee osteoarthritis as fast as 0.33 s per image. The current practice also failed to provide a robust prediction of disease progression. Deep learning models can integrate clinical data (e.g. pain score) and patient demographics together with radiological data (e.g. quantitative analysis of joint space narrowing) to accurately project the timeline for non-invasive interventions and surgical procedures needed. Knee osteoarthritis are also often overlooked in healthcare resources allocation despite its prevalence. The use of deep learning in exploring new biomarkers for knee osteoarthritis could improve our understanding towards the disease, assisting and promoting further research into newer agents to tackle osteoarthritis. All in all, deep learning could assist doctors in diagnosis, disease prediction and research of knee osteoarthritis. Future applications of deep learning into clinical context is highly favorable to clinicians.
Footnotes
Abbreviations
Authors’ contributions
HTV Yick: Conception, Design of the work, Manuscript draft. PK Chan: Conception, Analysis. C Wen: Conception. WC Fung: Analysis. CH Yan: Critical revision of content. KY Chiu: Critical revision of content
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
