Abstract
Introduction
Minor burns affecting less than 10% total body surface area (TBSA), especially those resulting from scalds, are common in children and are generally managed well with outpatient care.1,2 Despite this, severe complications are still observed in some patients. Toxic shock syndrome (TSS) is one such complication that can be life-threatening. 3 TSS in children has well-defined diagnostic criteria 4 (see Table 1).
aAs per CDC, these criteria should not be used to exclude a case that is highly suspicious for toxic shock syndrome, even if all criteria are not met.
Burn injuries put patients at risk of developing TSS for several reasons. 6 Patients are immune-compromised due to destruction of the skin barrier, diminished cell-mediated immunity, and decreased serum immunoglobulin and complement levels.6,7 The physiological response to burn injuries provides ideal conditions for toxin-producing pathogens, most commonly Staphylococcus aureus. 6
Currently, literature is sparse on the association of TSS with minor burns.3,8 While awareness of pediatric TSS related to burns has grown, it varies globally. Case reports have been published in the United Kingdom, and yet the Netherlands has explicitly reported its absence. 9 Despite low incidence of TSS in minor burns, delayed recognition and management is associated with significant morbidity and mortality. 10 Therefore, the variability of TSS case reports in minor burns underscores a need to enhance awareness and understanding through additional reporting. 3
Our objective was to identify and review the clinical course of pediatric patients with minor burns treated on an outpatient basis who were subsequently diagnosed with TSS.
Methods
Study Design
This study examined a retrospective cohort of consecutive pediatric patients under the age of 18 who suffered a minor burn injury (<10% TBSA) and were treated as an outpatient at a tertiary-level pediatric hospital between May 2012 and May 2022. The primary outcome was diagnosis of TSS.
Study Setting and Participants
This tertiary-level pediatric hospital is the regional referral center for pediatric burns in the southern region of the province. 11 Smaller burns are managed in an outpatient setting by a physiotherapist with formal training in burn care and oversight from 1 of 5 plastic surgeons. This arrangement ensured consistent burn management throughout the study duration.
Participant Search Strategy
Our search strategy employed the International Classification of Disease-10 (ICD-10) system to identify relevant medical charts and electronic health records. Patients were included if they were assigned an ICD-10 code for minor burn (T31.0 “burns involving less than 10% of body surface”) and the ICD-10 code A48.3 “toxic shock syndrome” or A41.9 “sepsis.”
Participant Selection
Inclusion criteria: (i) Patient age less than 18 years at the time of treatment; (ii) patients with a medical record of hospital/emergency room visit for a minor burn (<10% TBSA) treated on an outpatient basis, as well as a record of TSS, either at the same admission or in outpatient/ED visit before this admission ; and (iii) the patient hospital visit, and episode of TSS must have occurred within the study period.
Exclusion Criteria
Exclusion criteria: (i) incomplete patient records, (ii) burn was not followed temporally by TSS diagnosis, or (iii) the TSS was not attributed to the burn.
Data Extraction
Three reviewers screened the records retrieved by the initial search. Cases that met the inclusion criteria proceeded to full medical record review. Reviewers independently extracted relevant data from the full patient records. Disagreements between reviewers on interpretations of chart data were resolved by discussion and consensus agreement.
Clinical Definitions
Toxic shock syndrome: Defined with criteria listed by Centre for Disease Control (CDC) (Table 1).4,5
Results
We identified and reviewed the medical records of 6 children with minor burns who were treated on an outpatient basis and subsequently developed symptoms consistent with TSS.
During the study period (May 2012 to May 2022) 3706 patients were treated at the tertiary-level pediatric hospital for burn injury (any% TBSA). Of these, 3526 patients were treated on an outpatient basis and 180 patients were treated as inpatients. Eight hundred records were identified for children under the age of 18 who had a burn and a diagnosis of TSS or sepsis (Figure 1). Of these records, only 6 patients had a minor burn and were subsequently admitted for TSS. The characteristics of these children are summarized in Table 2.
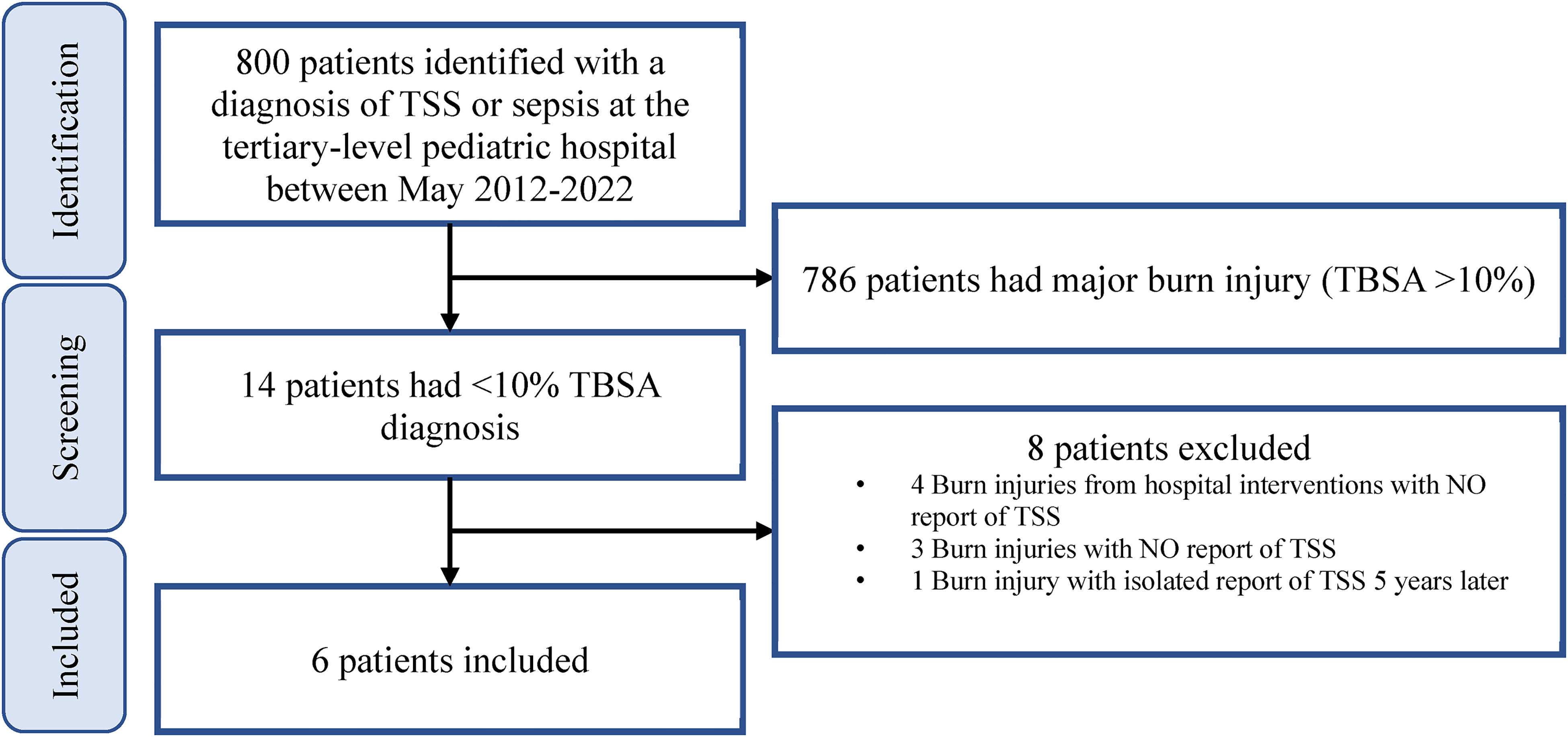
Flow diagram for chart selection process to identify pediatric patients who had a minor burn injury and developed subsequent TSS.
Characteristics of 6 Pediatric Patients Who Had a Minor Burn Injury and Developed Subsequent TSS/Sepsis.
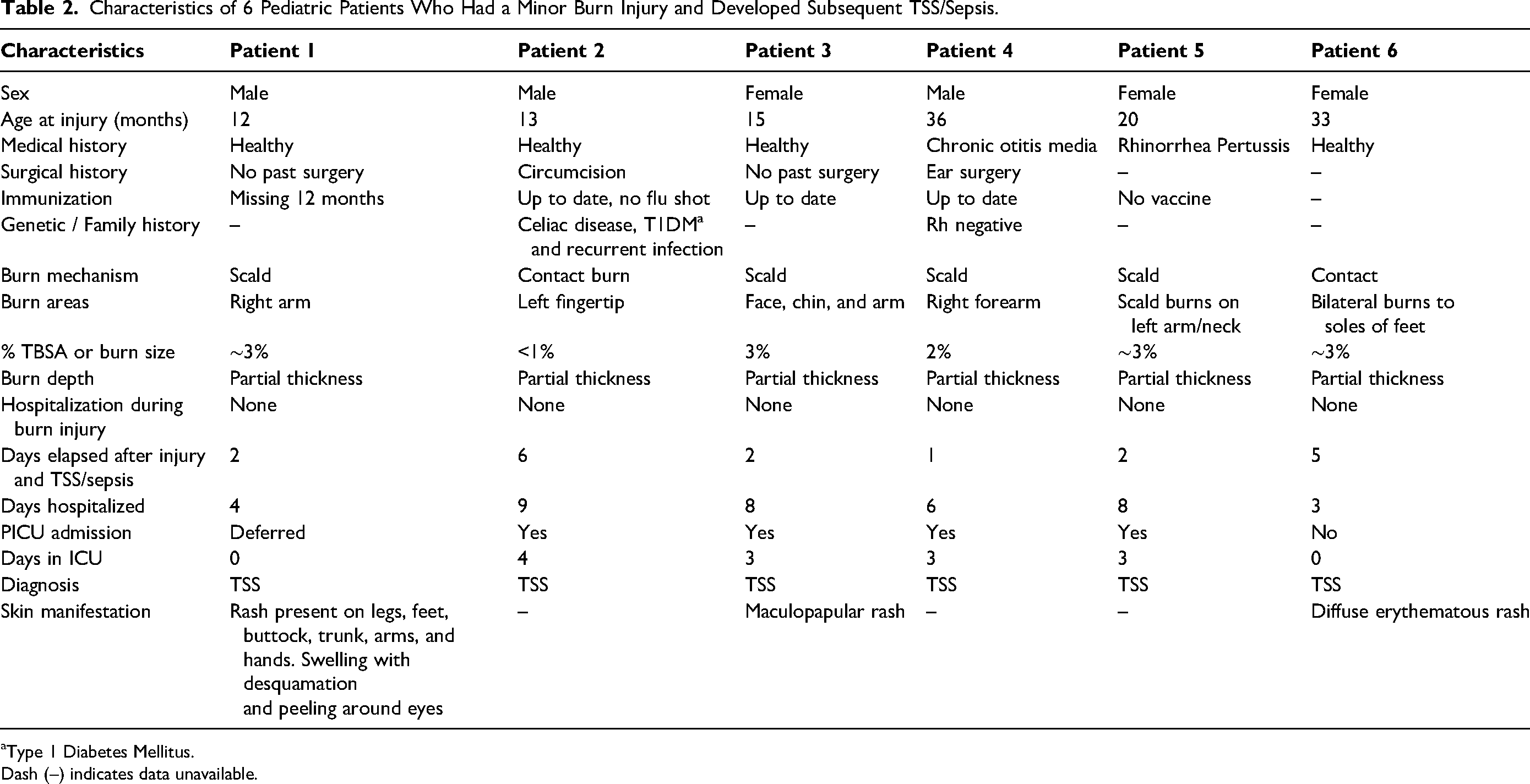
Type 1 Diabetes Mellitus.
Dash (–) indicates data unavailable.
Burn Injury Characteristics and Initial Management
Four patients sustained scald injuries, and 2 patients had contact burn injuries. While all the patients had injuries to their extremities, 2 of them (patients 3 and 5) also had chin and neck burns. In all 6 cases burn depth was documented as partial thickness with TBSA ranging between <1% and 3%. At the time of injury, one patient received burn wound care under sedation, while another patient received broad-spectrum antibiotics (refer to Supplemental Table 1). Only one patient had their burn dressing type documented, which consisted of Mepilex Ag (Mölnlycke © 2021 Mölnlycke Health Care AB).
Clinical Presentation of TSS in Burn Injury Patients
The most frequently observed early symptoms of TSS, noted at the time of burn injury, were vomiting (n = 6) and fever (n = 5) (Figure 2). Hypotension (n = 5) emerged as a later symptom of TSS, specifically during the time of admission for burn injury. Tachycardia (n = 4) and tachypnea (n = 3) were also observed.
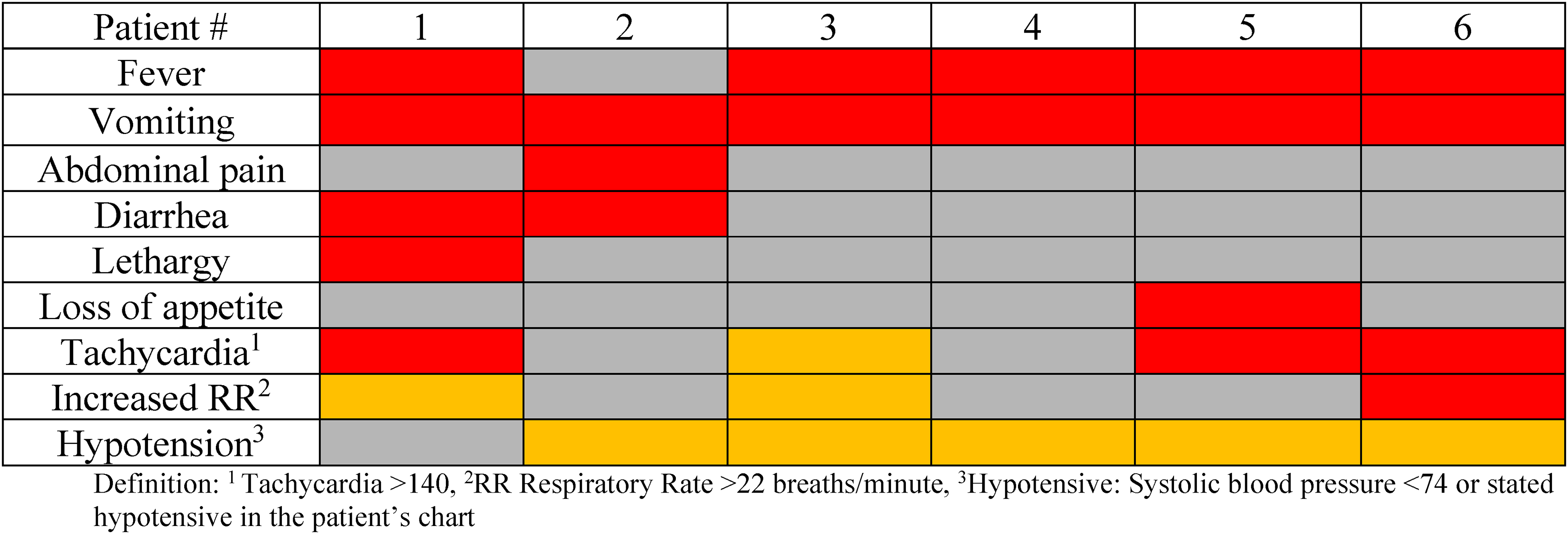
Symptoms present at the time of burn injury (red boxes) and at the time of admission for TSS after burn injury (orange boxes).
Clinical Variability and Impact of Additional Factors on TSS Admission
Two patients developed viral illnesses around the time of their burn injuries. Patient 1's journey began with 2 visits to the Emergency Department (ED) prior to admission. Despite receiving initial pain management in the ED, the patient endured distressing symptoms throughout the night. The next day, the patient had contact with dog feces and subsequently developed diarrhea, vomiting, fever, and worsened lethargy (Figure 2). This patient's condition deteriorated, culminating in the emergence of a widespread macular rash and the discovery of a COVID-positive status during the hospital stay. Notably, this patient presented with both an underlying viral illness and exposure to animal stool pathogens.
Patient 2 presented with a 1-week history of cough and gastrointestinal illness before sustaining a burn on their fingertip. Initially, the wound was considered unremarkable. However, as time progressed, symptoms resembling septic shock emerged, necessitating intensive care. Given this patient's history of gastrointestinal infection, an explorative laparotomy was carried out to investigate further. No significant findings were made.
Microbiological Findings and Antibiotic Management for TSS Patients
The primary antibiotics administered to patients for initial TSS management included vancomycin, ceftriaxone, and clindamycin to provide broad coverage pending blood cultures (Supplemental Table 2).
Blood cultures performed during the hospital admission for TSS were negative in all 6 patients. Three patients (patients 2, 4, and 6) had positive wound cultures for Staphylococcus aureus, prompting the initiation of IV Ancef (cefazolin) for 1 patient and stepped down to Keflex (cefalexin) at discharge for 2 patients.
PICU Care
Four of the 6 patients were admitted to the Pediatric Intensive Care Unit (PICU) (Supplemental Table 2). Patients 2 and 5 required intubations, and a combination of inotropes and vasopressor support to maintain adequate perfusion.
Patient 3 underwent a fluid resuscitation of 60 mL/kg over 3 h, followed by transfer to the PICU. To manage the patient's pain in the setting of hypotension (blood pressure of 70/30), they received morphine, acetaminophen, ibuprofen, and dopamine.
Patient 4 had a right femoral central line in place for 6 days and required dopamine for low blood pressure, along with stress dose steroids and IVIg while in the PICU.
Patient 5, while initially receiving 6 L of oxygen via a face mask in the ED, was weaned to 0.5 L, but experienced desaturation to 62%, prompting their transfer to the PICU and intubation. They also received ketamine, rocuronium, IVIg treatment, and steroids for 3 days while intubated.
Patient 6 underwent debridement in the operating room, and received Ringer's lactate and IVIg.
Complications
Three patients experienced complications during their admissions. Patient 5 exhibited fever, reduced intake, lethargy, and postprandial emesis following scald burns to the left arm and neck. Upon admission for TSS, the patient displayed fluctuating consciousness and decreased motor activity bilaterally, along with encephalopathy and no focal deficits. Two of the 6 patients had thrombotic complications. Patient 5 developed a deep vein thrombosis in the left femoral iliac vein, necessitating anticoagulation with enoxaparin.
Patient 2 presented with 1 week of swelling in their right lower limb, prompting ultrasound imaging which identified a non-occlusive right external iliac vein thrombus. Like Patient 5, Patient 2 was given enoxaparin for the thrombus.
Patient 3, who was admitted to ICU 2 days after their partial thickness burn injury to their face, chin, and arm suffered from iatrogenic complications. The patient had developed a firm abdomen suggestive of septic ileus which resolved upon discontinuation of morphine.
These 3 cases demonstrate the challenges of addressing additional complexities in the ICU management of TSS patients.
Timing of Injury to TSS Admission and Hospitalization Duration
Figure 3 illustrates the duration between injury and admission for TSS in relation to the total time of hospitalization for each patient along with time spent in ICU for 4 patients.
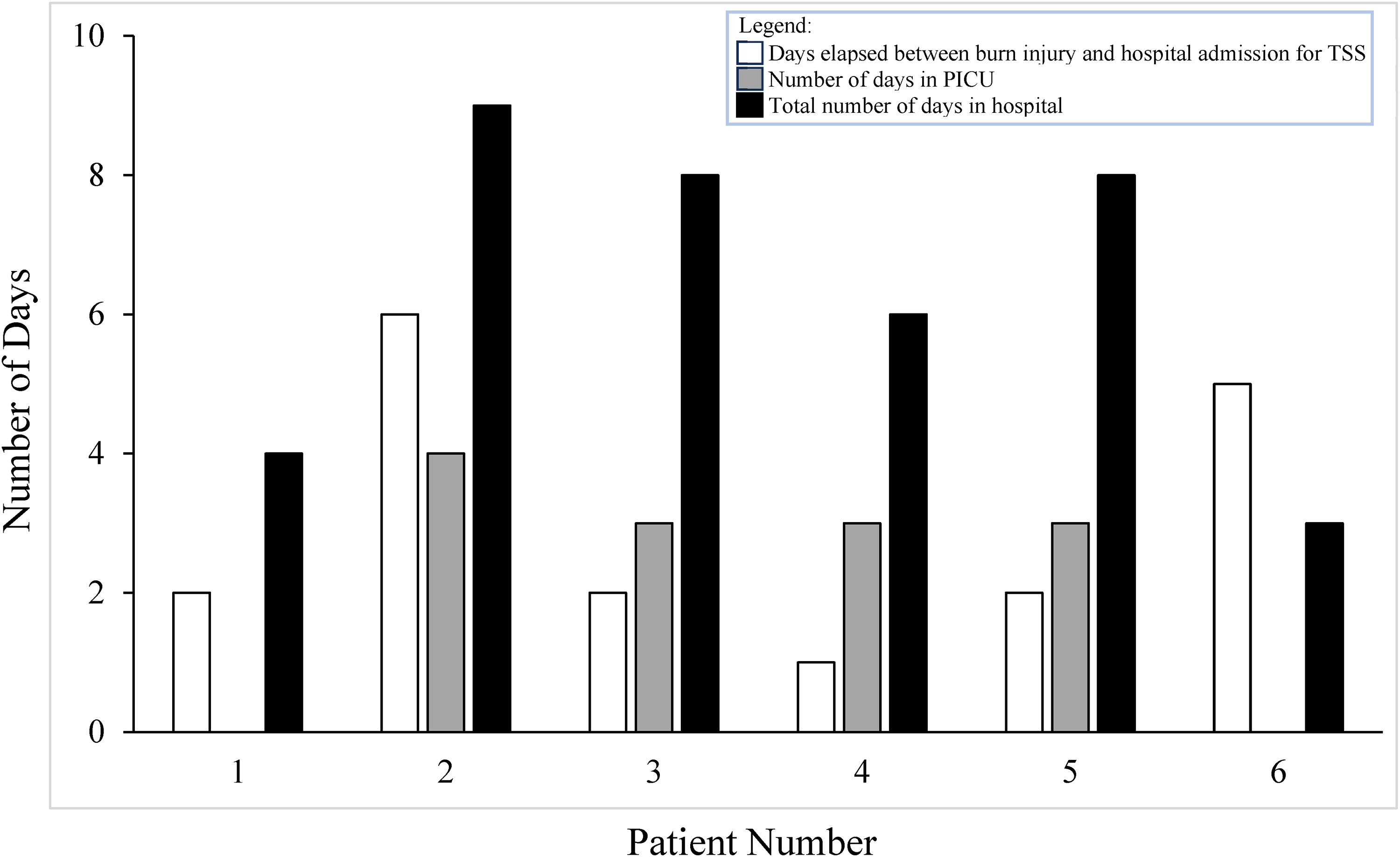
Hospital course with number of days that elapsed between burn injury and hospital admission for TSS (white), days spent in PICU (grey), and total number of days hospitalized (black) for the 6 patients who had a burn injury and subsequently developed TSS.
The interval between burn injury and TSS admission ranged from 1 to 6 days with most patients being admitted within 2 days. The total duration of hospitalization ranged from 3 to 9 days with ICU admission duration ranging between 3 and 4 days.
Patient 2 had the longest delay between injury and TSS admission (6 days), as well as the longest duration of hospitalization among all cases (9 days).
Dermatologic Presentation During TSS Admission
Three patients had dermatologic manifestation of their burn injury (Table 1). Patient 1 had a coalescing macular rash over their legs and feet including the soles of their feet, buttocks, trunk, arms, and back of hands. Their upper eyelid was edematous with some desquamation and peeling noted around the eyes.
Patient 3 had a maculopapular rash on their face that lasted 1 day.
Patient 6 had bilateral burns to the soles of their feet, initially without any associated symptoms. During their ED visit, a notable contrast was observed between the right and left foot blisters: the former had ruptured, while the latter remained intact, surrounded by serous fluid and erythema. Subsequently, the patient was discharged home with outpatient burn clinic follow-ups on post-burn days 1 and 4, where the burn wound was reported as unremarkable. However, on day 5, the patient developed a fever and experienced an episode of emesis, accompanied by facial swelling and decreased urine output. Upon admission, a wound culture revealed a significant growth of methicillin-sensitive Staphylococcus aureus, and a diffuse erythematous rash was observed on the soles of their feet. Notable laboratory findings, including lymphopenia, thrombocytopenia, and hypoalbuminemia, concurred clinically with the diagnosis of TSS.
Discussion
Here we summarize the clinical course of 6 pediatric burn patients who had TSS-complicated minor burn injuries, 4 of whom were admitted to the PICU. Concurrent viral illnesses, seen in 2 of our patients, may play an important role in pediatric patients developing TSS after a minor burn injury. Three patients identified in our study had their hospital admission complicated by other medical issues such as deep vein thrombosis and medication interventions in ICU. All patient developed TSS within 6 days of their initial minor burn. Diagnosing TSS early can been challenging and often TSS is confirmed only after the patient has gone into multi-organ failure and shock. 12 Pediatric health care providers should have a high clinical suspicion for TSS diagnosis if a burn patient presents with fever, vomiting and abdominal pain.13,14 These early symptoms were observed in our patients as shown in our chart review.
We highlighted 2 cases (patients 1 and 2) where underlying circumstances of viral illness and exposure to pathogens may have complicated patients’ admission for TSS. Additionally, we discussed cases of 3 patients (2, 3, and 5) who had thrombotic and iatrogenic complications in ICU. While not fully understood, the high rate of thrombotic complications in patients with septic shock is associated with systemic proinflammatory and procoagulant effects of infection, including both the immune response and factors released by pathogens.15,16
We also describe 2 cases of patients (patients 3 and 5) who tested positive for rhinovirus at the time of hospitalization for TSS. It is unclear whether viral illness could have contributed to the severity of their TSS. The effects of medical history preceding hospitalization for TSS from our study are described in another case report study that evaluated a pediatric patient with Down syndrome who developed TSS 4 days after discharge following a scald burn to his back and posterior head. 14 This patient experienced multiple complications and required intensive care, including 2 cardiac arrests. The authors of that study suggested that delayed diagnosis and the immunodeficiency associated with Down syndrome may have contributed to the severity of TSS. 14 Additionally, the effect of immunodeficiency may be relevant to the observations we made in the case of Patient 4 who had recurrent chronic otitis media.
Staphylococcus aureus and Streptococcus pyogenes are the most common causative agents of TSS. 17 These bacteria can cause life-threatening infections such as scarlet fever, bacteremia, pneumonia, and necrotizing fasciitis. TSS has been known to cause significant morbidity and mortality in children even in the presence of minor burns. 8 In our chart review, we found 3 patients who had wound culture positive for Staphylococcus aureus (Supplemental Table 2). Notably, children under the age of 5 years may be more vulnerable to developing TSS due to lower levels of protective antibodies to TSST-1. 18 The production of TSST-1 antibodies increases with age, with 47% and 58% of the population possessing protective antibody levels at the ages of 1 and 5, respectively. 18
Children also have a higher body surface area to body mass ratio than adults, resulting in increased fluid losses and resuscitation needs. 19 As such, many of the patients in our study required fluid resuscitation during admission to hospital for TSS (Supplemental Tables 1 and 2). Children also experience greater evaporative water loss and were more susceptible to hypothermia. This requires temperature regulation through non-shivering thermogenesis, which increases metabolic rate and lactate production. 20 Plasma lactate serves as a marker of cellular hypoxia and shock. 20 In burn patients, plasma lactate has shown to be a robust predictor biomarker of sepsis and mortality. 20
A systematic literature review conducted by Gutzler et al (2018) and a case report by Blomqvist (1997) examined case series to gain insights into the rare but potentially life-threatening complication of TSS in pediatric burn patients.3,8 While large burns are expected to have a greater degree of morbidity and mortality, some complications are disproportionately observed in small burns including TSS.6,12,21,22 This is purportedly attributed to differences in management, where larger burns receive surgical debridement and wound closure to reduce pathogen load as well as infusion of blood products that boost passive immunity.6,23Furthermore, this highlights the need to raise awareness among healthcare providers about unexpected consequences associated with seemingly minor burns. These findings align with our investigation and emphasize the importance of recognizing and understanding TSS as a potential complication in pediatric burn care. Prompt recognition and management of TSS symptoms is crucial, as delayed treatment can lead to severe complications. 22 Gutzler et al (2018) identified specific risk factors associated with the development of TSS in pediatric burn patients, such as delayed burn wound closure and the presence of necrotic tissue. 3 Close monitoring of burn patients, particularly those with risk factors, is recommended to detect early signs of TSS and initiate appropriate interventions. Additionally, the studies stress that TSS should be considered as a potential complication in all burn patients, regardless of burn size, and call for further research to enhance understanding of underlying mechanisms and optimize management strategies.
Strengths and Limitations
In our retrospective chart review design, we constructed a comprehensive timeline spanning from the initial burn injury to the point of admission for TSS for each pediatric patient. This approach, utilizing pre-existing medical records, afforded a cost-effective means of investigating the profile of patients at our centre who developed TSS following a burn injury. Reliance on electronic medical records can limit data availability. We encountered challenges in determining if blood culture investigations were carried out during the initial visit, along with ascertaining specific details such as burn dressing protocols, including dressing types and intervals, in 5 cases.
Although our small sample size speaks to the rarity of TSS in minor burns and limits generalizability, it enabled an in-depth exploration of individual patient cases, highlighting the unique circumstances contributing to TSS development. Furthermore, our study identified avenues for further investigation: examining the role of dressing types, dressing change frequency, symptoms at initial presentation, and the interval between burn injury and TSS onset.
Significance and Future Directions
We anticipate that pediatric burn care providers will find these cases stimulating, prompting further investigation into complications arising from minor pediatric burn injuries. Growing knowledge in this area could enhance patient outcomes and burn research. For instance, a registry of burn injuries with structured data collection could greatly enhance our understanding of burn care and support future studies. With the implication of lower levels of TSTT-1 antibodies in children, future protocols can test for TSTT-1 toxins as part of the blood work. 18 Detailed reports of burn injuries, including mechanism, extent, and depth, are important predictors of infection risk. 24 These data can be used to determine risk factors, and better understand pathophysiology of infections in pediatric burns.
In summary, we have presented 6 cases of pediatric patients who developed TSS after minor burn injuries. A range of outcomes was observed. Four patients required PICU admission which included interventions like intubation, vasopressor support and fluid resuscitation, and 3 developed complications. One patient underwent surgical debridement of their wounds, and no other surgical procedures were performed. All patients were eventually discharged home. The variable duration between burn injury and TSS admission and length of hospital stay reflects the diverse clinical course of TSS in pediatric burn patients. Future directions include carrying out a larger-scale prospective study that examines the background, risk factors, and management of children who go on to develop TSS.
Supplemental Material
sj-docx-1-psg-10.1177_22925503241249758 - Supplemental material for Toxic Shock Syndrome as an Unexpected Complication in Outpatient Pediatric Burn Treatment: A Chart Review
Supplemental material, sj-docx-1-psg-10.1177_22925503241249758 for Toxic Shock Syndrome as an Unexpected Complication in Outpatient Pediatric Burn Treatment: A Chart Review by Reshma Sirajee, Altay Baykan, Frankie O. G. Fraulin, Thomas Cawthorn, Kimberly Sass, Alan Robertson Harrop, Rebecca L. Hartley and Donald Ford McPhalen in Plastic Surgery
Footnotes
Acknowledgments
We would like to thank Doug Baron for providing us with data on the total number of inpatient and outpatient pediatric burn patients between 2012 and 2022.
Authors’ Note
Author Contributions
Conceptualization: R.S, F.F, T.C, A.H, D.M; writing—original draft preparation: R.S; writing—review and editing: R.S, A.B, F.F, T.C, K,S, A.H, R.H, D.M; investigation: R.S, D.M; methodology: R.S, D.M; visualization: R.S; all authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
IRB Approval Statement
This study was approved by the University of Calgary Conjoint Health and Research Ethics Board (REB22-0679).
Helsinki Statement
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

