Abstract
Introduction
Breast cancer is the most common malignancy in women, with treatment typically requiring surgical resection with or without systemic or focused neoadjuvant or adjuvant therapies. 1 While women who undergo breast cancer surgery generally demonstrate favorable outcomes with respect to health-related quality of life (HRQL), disabling symptoms associated with surgery and concomitant treatment may persist after the treatment period. 2 Specifically, patient HRQL concerns pertaining to fear of cancer progression/recurrence,2,3 chronic fatigue 4 as well as impaired return to work or pretreatment activities are commonly reported in the literature. 5
Despite known sequelae of breast cancer and its treatment, there exists a paucity of evidence which seeks to prognosticate which patients are likely to struggle following a breast cancer diagnosis. As such, efforts by clinicians and researchers to identify patients at high-risk of poor HRQL outcomes following a breast cancer diagnosis can aid resource allocation to ensure these individuals are adequately supported. Specifically, Park et al 6 advocate for targeted survivorship care in breast cancer survivors to improve HRQL outcomes.
To understand the impact of breast cancer and its treatment on patient HRQL, condition-specific patient-reported outcome measures (PROMs) are required.7,8 To date, the BREAST-Q is the most frequently used PROM to measure HRQL outcomes in this patient population. 8 Specifically, the BREAST-Q Breast Cancer module utilizes independently functioning scales to measure the outcomes that matter to patients with a breast cancer diagnosis. Specifically, our research team developed and validated three novel BREAST-Q Breast Cancer module scales that measure HRQL outcomes specific to cancer worry, fatigue, and impact on work.9,10 These scales are designed for patients who have undergone either breast reconstruction, mastectomy without reconstruction, or breast-conserving therapy.
In this study, the authors sought to perform an exploratory analysis using patient data collected from the PROM field-test validation study of the BREAST-Q Cancer Worry, Fatigue, and Impact on Work scales. The primary aim was to identify which patient clinical and demographic characteristics are associated with poor HRQL outcomes following a breast cancer diagnosis.
Methods
This cross-sectional study was approved by two respective research ethics boards sites and the Love Research Army (LRA; formerly known as the Army of Women) Scientific Advisory Committee.
Sample and Recruitment
A convenience sample of participants were recruited through the LRA as part of a larger field-test study to validate the novel BREAST-Q Breast Cancer Worry, Fatigue and Impact on Work scales. LRA members in the United States were sent an electronic recruitment email (ie, e-blast) describing the study and given access to a REDCap survey link (hosted at participating hospital). Data collection took place between October and November 2019. Participants were included if: (1) they were 18 years or older; (2) English-speaking; and (3) had a prior breast cancer diagnosis. Women who self-selected to meet the inclusion criteria completed demographic and breast cancer-specific questions alongside the Cancer Worry, Fatigue, and Impact on Work scales. All participants completed the Cancer Worry scale. The Fatigue scale was only administered to participants who endorsed fatigue related to breast cancer and/or its treatment, and the Impact on Work scale was only completed by participants who indicated they worked in a job (for pay) in the past six months.
Scoring of BREAST-Q Scales
All three BREAST-Q scales are continuous outcomes, scored from 0 to 100. For the Cancer Worry scale (ie, 10 items), higher scores reflect more cancer worry, whereas for the Fatigue (ie, 10 items) and Impact on Work (ie, 8 items) scales, higher scores correspond to better HRQL outcomes. Each scale takes approximately 1-4 min to complete.
Data Analysis
Descriptive statistics were used to evaluate participant demographic and clinical characteristics. Predictor variables hypothesized to be clinically relevant to the Breast Cancer Worry, Fatigue, and Impact on Work scales were selected a priori by the study authors. A univariable regression analysis was used to evaluate the existence of a linear relationship between each predictor variable and the BREAST-Q scales. Statistical significance was determined using a Wald test (ie, continuous variables) or Partial F-Test (ie, categorical variables). Predictors demonstrating a statistically significant linear relationship from this univariable analysis were included in a multivariable linear regression model for each scale. Standardized coefficients (β*) were used to compare the relative importance of predictor variables. The total variability explained through this model was summarized using the coefficient of determination (R2). An a priori Variance Inflation Factor (VIF) > 5 was used to denote definite multicollinearity. Given a sample size rule of thumb of 10-20 participants per predictor established by Harrell,11,12 the sample size for each scale was considered sufficient. Pairwise deletion (ie, available case analysis) was performed to address missing data within regression analyses. Statistical significance was considered at P ≤ .05. All analyses were performed using SPSS ® version 26.0 (IBM Corporation, Armonk NY, USA for Windows®).
Results
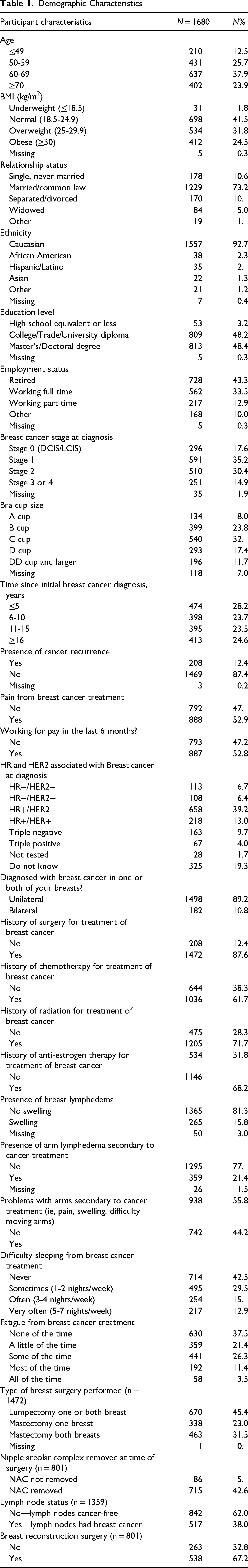
Of the 1717 participants who opened the REDCap link, n = 1680 (97.8%) completed at least one scale. The Cancer Worry, Fatigue, and Impact on Work scales were completed by n = 1680, n = 1037, and n = 873 participants, respectively. Participants had a mean (± standard deviation, SD) age of 61.9 (±10.1) years and body mass index (BMI) of 26.9 kg/m2 (±5.8). Common characteristics of the cohort included respondents being married/common law (73.2%), Caucasian (92.7%), had started or obtained a College/University diploma (48.2%) or Master's/Doctoral degree (48.4%), and were working full time (33.5%) or retired (43.5%). With respect to their oncologic status, most participants had surgery (89.2%) and/or radiation (71.7%) for treatment of their breast cancer and had not experienced a cancer recurrence (87.4%). Participant demographic characteristics are reported in Table 1.
Demographic Characteristics
Cancer Worry Scale
The participant sample (n = 1680) demonstrated a normal distribution of BREAST-Q Cancer Worry scores with a mean of 45.4 (±17.7) (Figure S1 and Table 2).
BREAST-Q Cancer Worry, Fatigue and Work-Life Scale Scores

N, frequency; SD, standard deviation.
Following a univariable linear regression analysis (Table 3), more Cancer Worry (ie, higher scores) was significantly associated with younger participant age [β −0.55, 95% CI −0.64 to −0.48, P < .01], increasing BMI [β 0.20, 95% CI 0.05 to 0.35, P < .01], shorter duration since breast cancer diagnosis (P < .01) and working full- or part-time relative to being retired (P < .01). Additionally, participants who reported ongoing pain secondary to breast cancer [β 9.44, 95% CI 7.80 to 11.08, P < .01], a higher breast cancer stage at diagnosis (P < .01), triple positive disease (P < .01), breast cancer recurrence [β 4.01, 95% CI 1.45 to 6.58, P < .01], treatment with chemotherapy [β 4.61, 95% CI 2.88 to 6.34, P < .01], radiation [β 3.62, 95% CI 1.74 to 5.50, P < .01], or anti-estrogen therapy [β 3.80, 95% CI 1.98 to 5.61, P < .01], ongoing breast lymphedema [β 10.46, 95% CI 8.18 to 6.65, P < .01], positive lymph nodes [β 4.21, 95% CI 2.32 to 6.10, P < .01], and those who underwent a lumpectomy relative a unilateral or bilateral mastectomy (P < .01) reported more cancer worry.
Univariable and Multivariable Linear Regression of BREAST-Q Cancer Worry Scale on Participant Variables
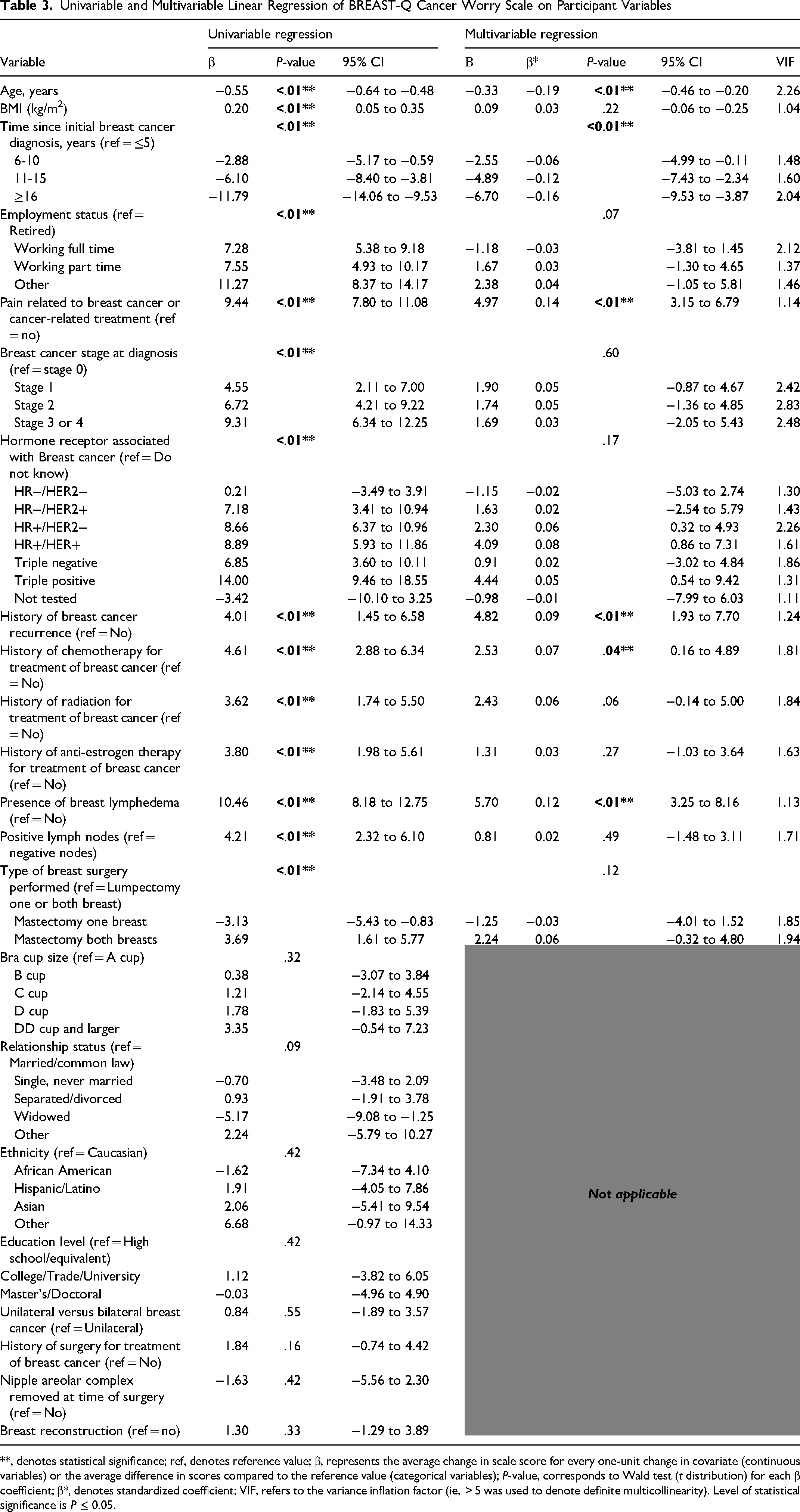
**, denotes statistical significance; ref, denotes reference value; β, represents the average change in scale score for every one-unit change in covariate (continuous variables) or the average difference in scores compared to the reference value (categorical variables); P-value, corresponds to Wald test (t distribution) for each β coefficient; β*, denotes standardized coefficient; VIF, refers to the variance inflation factor (ie, > 5 was used to denote definite multicollinearity). Level of statistical significance is P ≤ 0.05.
When statistically significant variables were included in a multivariable linear regression model, only younger participant age [β −0.33, 95% CI −0.46 to −0.20, P < .01], shorter duration of time since initial breast cancer diagnosis (P < .01), ongoing pain related to breast cancer or treatment [β 4.97, 95% CI 3.15 to 6.79, P < .01], history of breast cancer recurrence [β 4.82, 95% CI 1.93 to 7.70, P < .01], ongoing breast edema/lymphedema [β 5.70, 95% CI 3.25 to 8.16, P < .01], and treatment with chemotherapy [β 2.53, 95% CI 0.16 to 4.89, P = .04] remained statistically significant after adjusting for all other variables in the model (Table 3). Using standardized coefficients (β*), participant age was determined to be the most important predictor variable for the BREAST-Q Cancer Worry score [β* = −0.19] (Figure 1). Overall, the multivariable model explained 22.4% of the variance in Cancer Worry Scores [R2 = 0.22, F(27,1290) = 13.79, P < .01].
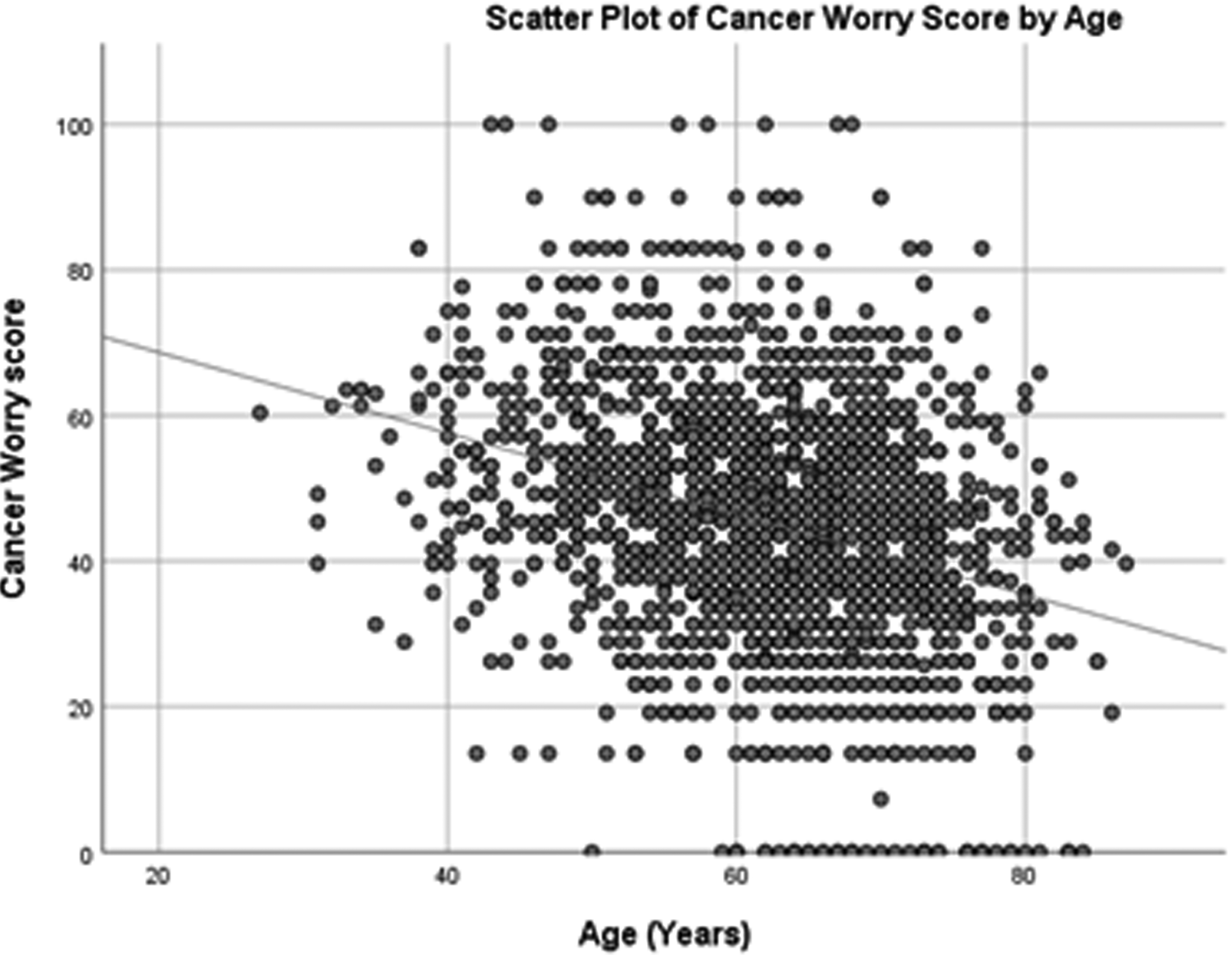
Scatter plot of cancer worry score by age (years).
Fatigue Scale
A sample of n = 1037 participants completed the BREAST-Q Fatigue scale and demonstrated a mean scale score of 69.4 (±20.0). Scale distributions were skewed (Figure S1).
Following a univariable analysis (Table 4), worse participant Fatigue (ie, lower scores) was associated with younger age [β 0.26, 95% CI 0.14 to 0.38, P < .01], increasing BMI [β −0.43, 95% CI −0.64 to −0.22 P < .01], shorter time since diagnosis (P < .01), participant ethnicity (P < .01), employment status (P < .01), self-reported pain related to breast cancer or cancer treatment [β −10.68, 95% CI −13.03 to−8.32, P < .01], higher breast cancer stage at diagnosis relative to stage 0 disease (P < .01), increased difficulty sleeping relative to no difficulty sleeping (P < .01), cancer recurrence [β −4.48, 95% CI −8.17 to −0.79, P = .02], history of surgical treatment for breast cancer [β −3.76, 95% CI −7.45 to −0.06, P = .04] or chemotherapy [β −4.31, 95% CI −6.80 to −1.81, P < .01], ongoing breast edema/lymphedema [β −8.33, 95% CI −11.66 to −5.00, P < .01], and self-reported problems with arms secondary to cancer treatment (eg, pain, swelling, difficulty moving arms) [β −7.36, 95% CI −9.78 to −4.95, P < .01].
Univariable and Multivariable Linear Regression of BREAST-Q Fatigue Scale on Participant Variables
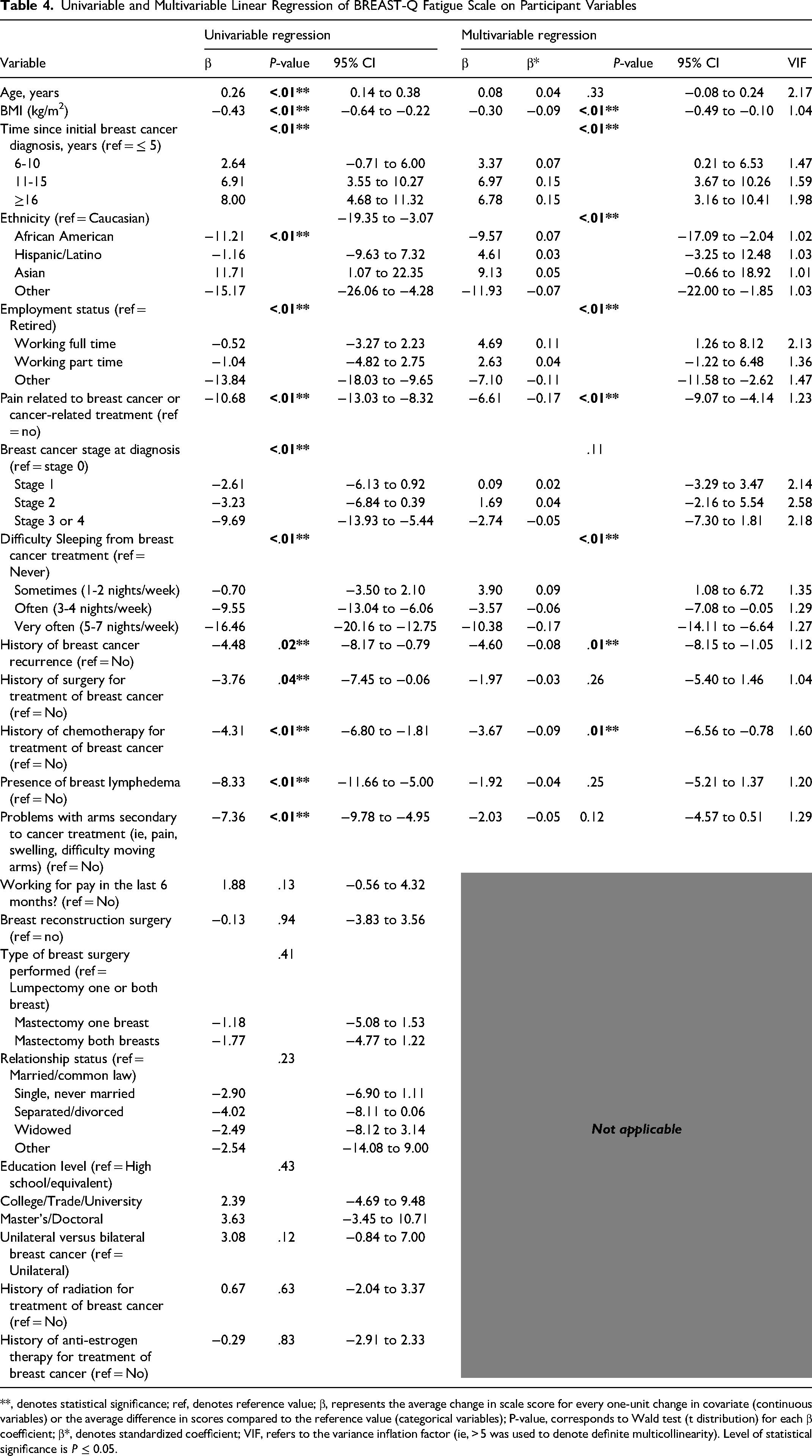
**, denotes statistical significance; ref, denotes reference value; β, represents the average change in scale score for every one-unit change in covariate (continuous variables) or the average difference in scores compared to the reference value (categorical variables); P-value, corresponds to Wald test (t distribution) for each β coefficient; β*, denotes standardized coefficient; VIF, refers to the variance inflation factor (ie, > 5 was used to denote definite multicollinearity). Level of statistical significance is P ≤ 0.05.
Again, when statistically significant variables were included in a multivariable linear regression model only increasing BMI [β −0.30, 95% CI −0.49 to −0.10, P < .01], participant ethnicity (P < .01), employment status (P < .01), ongoing pain [β −6.61, 95% CI −9.07 to −4.14, P < .01], history of cancer recurrence [β −4.60, 95% CI −8.15to −1.05, P < .01], treatment with chemotherapy [β −3.67, 95% CI −6.56 to −0.78, P = .01], and increased frequency of difficulty sleeping (P < .01) were significantly associated with BREAST-Q Fatigue scores after adjusting for other variables in the model. Using standardized coefficients (β*), difficulty sleeping “very often (5 to 7 nights/week)” and “pain related to cancer or related treatments” were determined to be the most important predictors for the BREAST-Q fatigue score [β* = −0.17] (Figure 2). Overall, the multivariable model explained 22.3% of the variance in Fatigue Scores [R2 = 0.22, F(24,973) = 11.64, P < .01].
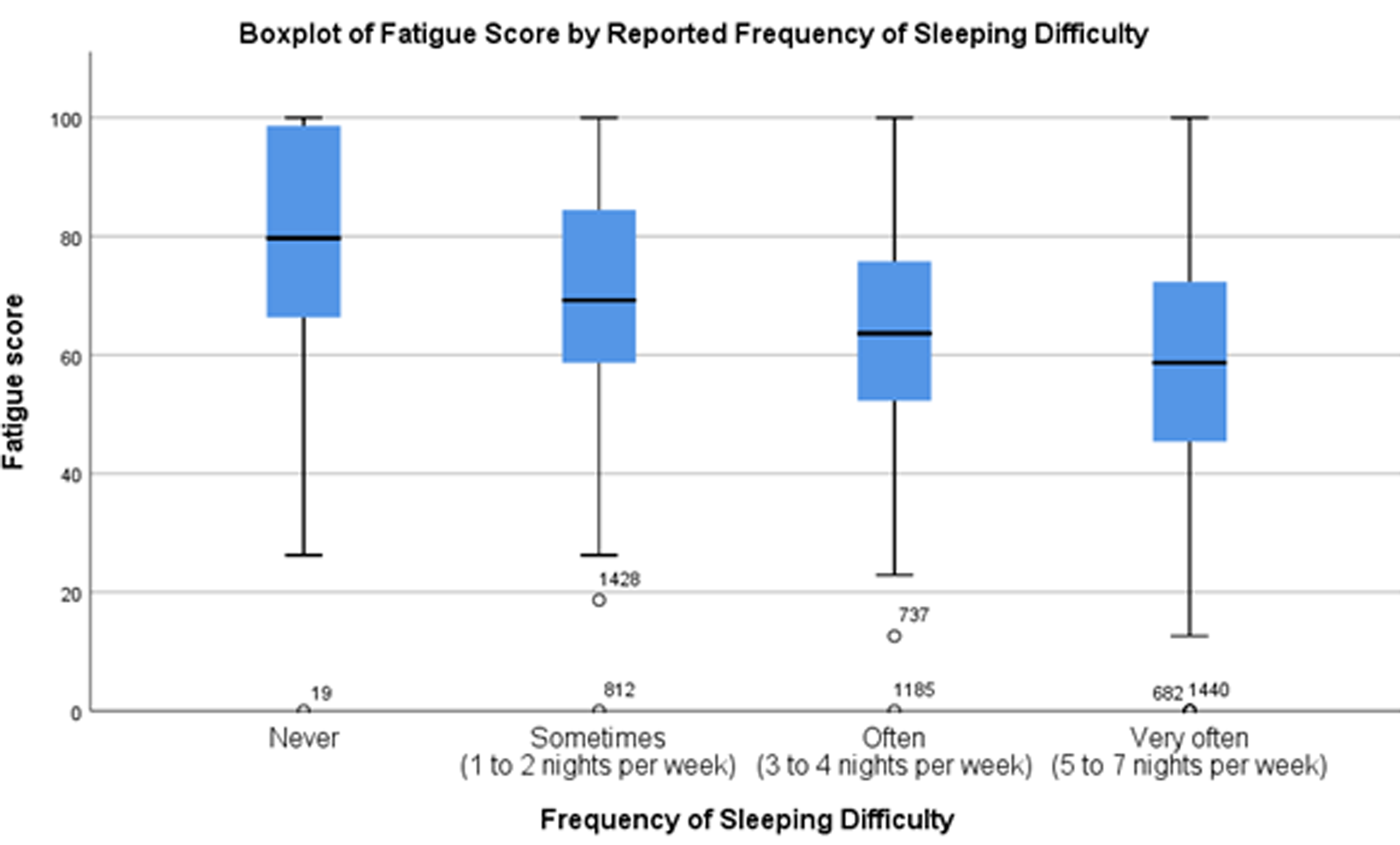
Box plot demonstrating relationship between self-reported frequency of sleeping difficulty and BREAST-Q fatigue scores.
Impact on Work Scale
A sample of n = 873 participants completed the BREAST-Q Impact on Work scale and demonstrated a mean scale score of 73.0 (±28.1). Scale distributions were skewed (Figure S1).
Following univariable regression (Table 5), worse participant Impact on Work (ie, lower scale scores) was associated with younger participant age [β 0.33, 95% CI 0.15 to 0.52, P < .01], shorter time since initial breast cancer diagnosis (P < .01, part-time employment status relative to retirement (P < .01), ongoing pain related to breast cancer or associated treatments [β −13.82, 95% CI −17.44 to −10.19, P < .01], higher cancer stage at diagnosis (P < .01), increased difficulty sleeping secondary to breast cancer/treatment (P < .01), worse self-reported fatigue frequency (P < .01), history of chemotherapy [β −8.09, 95% CI −11.89 to −4.29, P < .01], ongoing breast edema/lymphedema [β −14.09, 95% CI −11.89 to −4.29, P < .01], problems with arms secondary to cancer treatment [β −12.46, 95% CI −16.12 to −8.79, P < .01], and separated/divorced relationship status relative to married participants (P = .03).
Univariable and Multivariable Linear Regression of BREAST-Q Impact on Work Scale on Participant Variables
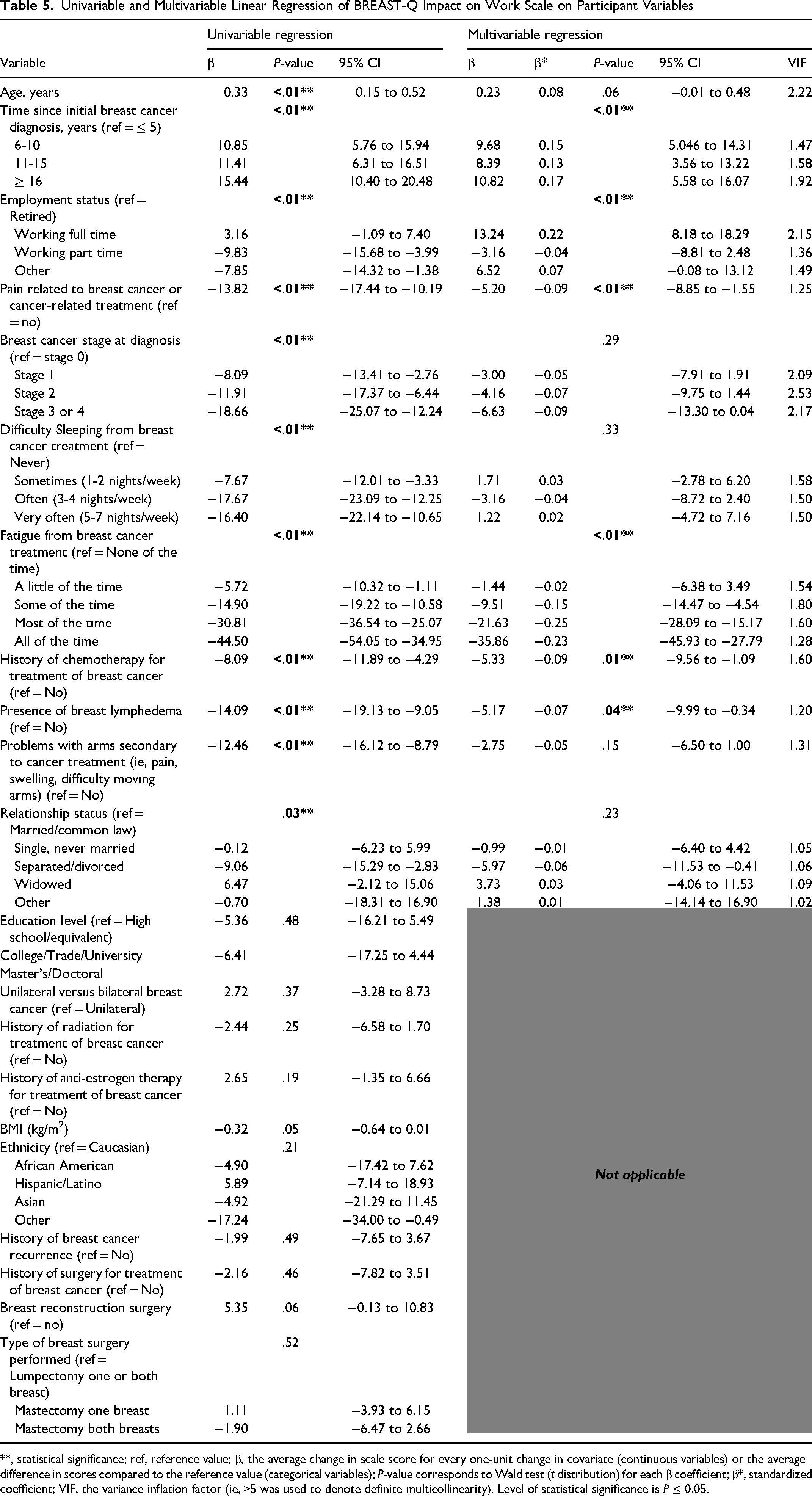
**, statistical significance; ref, reference value; β, the average change in scale score for every one-unit change in covariate (continuous variables) or the average difference in scores compared to the reference value (categorical variables); P-value corresponds to Wald test (t distribution) for each β coefficient; β*, standardized coefficient; VIF, the variance inflation factor (ie, >5 was used to denote definite multicollinearity). Level of statistical significance is P ≤ 0.05.
When these statistically significant variables were included in a multivariable linear regression model (Table 5), only time since initial breast cancer diagnosis (P < .01), employment status (P < .01), presence of pain related to breast cancer [β −5.20, 95% CI −8.85 to −1.55, P < .01], increased self-reported fatigue frequency (P < .01), treatment with chemotherapy (P = .01), and presence of breast lymphedema [β −5.17, 95% CI −9.99 to −0.34, P = .04] remained statistically significant after adjusting for all other variables in the model. Using the standardized coefficients (β*), participant self-reported fatigue described as “Most of the time” [β* = −0.25] and “All of the time” [β* = −0.23] was determined to be the most important predictor variable for the BREAST-Q Impact on Work score. Overall, this multivariable model explained 28.2% of the variance in scores [R2 = 0.28, F(25,823) = 12.04, P < .01].
Discussion
This analysis provides evidence of patient characteristics that may be associated with increased breast cancer worry, fatigue and impact on work following a breast cancer diagnosis in a well-educated and predominantly white cohort of women. For clinicians and researchers, this analysis is hypothesis generating and identifies patient characteristics that may influence HRQL outcomes in this patient population. The results of this study can be used to identify patients at high-risk of poor HRQL outcomes following a breast cancer diagnosis and can aid resource allocation to ensure these individuals are adequately supported in survivorship.
In-keeping with the findings of Lebel et al, 13 who concluded that younger patient age was associated with more fear of cancer recurrence among breast cancer patients, we identified a statistically significant association between younger participant age and breast cancer worry such that each additional 1-year increase in age was associated with a 0.55 reduction in Cancer Worry score (Table 3). This result corresponds to an average reduction of −5.55 points for a 10-year increase in participant age. When standardized coefficients are compared in this multivariable model, age has the largest absolute value (β*Age = −0.19) and suggests that a change of 1 standard deviation in participant age will result in an average change of 0.19 standard deviation units in Cancer Worry score, given all other variables are included. As such, participant age appears to have the greatest impact on Cancer Worry score after adjusting for other clinically relevant variables in our multivariable regression model. To address fear of cancer recurrence, Lebel et al 13 advocates for the implementation of clinical interventions which focus on anxiety management. Specifically, a randomized control trial comparing cognitive-behavioral group therapy (CBT) or supportive therapy versus standard treatment in reducing fear of disease progression in cancer patients by Herschbach et al 14 demonstrated a moderate reduction in fear of disease progression after four sessions over a 12-month period.
Moreover, this analysis identified increasing difficulty sleeping and pain related to breast cancer or related treatments to be the most important predictors for the BREAST-Q Fatigue scale. Specifically, relative to those participants who reported “never” for difficulty sleeping, those who selected “very often (5 to 7 nights/week)” demonstrated a 16.5-point reduction in Fatigue scale scores. Additionally, those participants who reported ongoing pain related to breast cancer or cancer-related treatment demonstrated, on average, a 10.7-point reduction in Fatigue scores. These participant variables remained statistically significant following inclusion in a multivariable linear regression model and were determined to be the most important variables following a comparison of standardized coefficients. This finding is consistent with Okuyama et al 15 who reported that fatigue in breast cancer survivors was significantly correlated with insufficient sleep, and this association was independent of other psychological factors such as depression. Furthermore, the association between pain and fatigue is supported by Schreier et al 16 who concluded that pain intensity was positively correlated with fatigue and described correlations between pain interference, sleep disturbance, and anxiety. To address fatigue in breast cancer patients, Okuyama et al 15 advocate for the treatment of concurrent symptoms (eg, dyspnea and insomnia) as well as the implementation of psychosocial interventions which have demonstrated some effectiveness—although additional research is required. 15
Finally, the authors identified participant self-reported fatigue to be the most important predictor of negative work-life HRQL such that, relative to those who reported no fatigue, those who endorsed fatigue “Most of the time” and “All of the time” scored, on average, 30.1 and 44.5 points lower on the BREAST-Q Impact on Work scale, respectively. This finding is in keeping with Schmidt et al 5 who identified an association between impaired return to work and increased fatigue at 1.5 years after surgery in a breast cancer sample. In this study, fatigue was the most frequently cited reason for impaired return to work and persistent fatigue at 1.5 years following treatment remained a significant long-term determinant for work-life impairment. These results are further supported by several studies which corroborate an association between fatigue and impaired return to work in cancer patients.17–20 Again, the implementation of psychosocial interventions may be effective at reducing fatigue strategy in breast cancer patients. 15
There are several limitations to this analysis. Firstly, there is no minimal clinical important difference established for the BREAST-Q Cancer Worry, Fatigue, and Impact on Work scales yet. As such, we are unable to precisely interpret the independent effect of predictor variables that results in a clinically meaningful difference in HRQL outcomes. Secondly, the BREAST-Q Fatigue and Impact on Work scales demonstrate a highly skewed distribution with 13.3% and 36.4% of participants scoring 100 (ie, best HRQL), respectively. To ensure the normality and homoscedasticity assumptions for linear regression were met, the authors examined residuals using visual inspection and p-p plots. Moreover, regression remains robust to violations of the normality assumption and does not noticeably impact results in large sample sizes (eg, where the number of observations per variable is >10). 21 The skewed distribution of the data is therefore unlikely to impact our results. Thirdly, as the demographic and clinical data provided by the sample are self-reported, the accuracy of the data cannot be verified. Lastly, given that our cross-sectional sample was largely Caucasian, educated, non-working, and resided in the United States, further research is needed to ensure these associations remain applicable to other ethnic and socioeconomic groups in other settings and countries. The results of this analysis should be viewed with these limitations in mind.
Conclusions
The BREAST-Q Cancer Worry, Fatigue, and Impact on Work HRQL scales are rigorously developed, and validated PROMs designed for use in patients with a breast cancer diagnosis. For outcomes researchers, this analysis is hypothesis generating and identifies patient variables that may influence scale scores in this population. Clinicians and study investigators should follow-up with patients who endorse predictors significantly associated with reduced HRQL outcomes, address areas of concern in participant scale scores, and ensure patients are offered further supports, if necessary. The results of this study can be used to identify patients at high-risk of poor HRQL outcomes following a breast cancer diagnosis and can aid resource allocation to ensure these individuals are adequately supported. Future research is necessary to confirm these associations in a diverse, prospective patient sample and establish minimal clinical important differences for these scale scores.
Supplemental Material
sj-docx-1-psg-10.1177_22925503231161067 - Supplemental material for Patient Factors Associated With Increased Cancer Worry, Fatigue, and Impact on Work Following a Breast Cancer Diagnosis: A Cross-Sectional Analysis
Supplemental material, sj-docx-1-psg-10.1177_22925503231161067 for Patient Factors Associated With Increased Cancer Worry, Fatigue, and Impact on Work Following a Breast Cancer Diagnosis: A Cross-Sectional Analysis by Sophocles H. Voineskos, Lucas Gallo, Manraj Kaur, Elena Tsangaris, Lauren Griffith, Jonas A. Nelson, Anne F. Klassen and Andrea L. Pusic in Plastic Surgery
Footnotes
Author Contributions
All authors contributed to the study design, analysis, and manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The BREAST-Q is owned by Memorial Sloan Kettering Cancer Center, Mass General Brigham, and McMaster University. Drs Pusic and Klassen are co-developers and receive royalty for use in for-profit studies.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research Ethics and Patient Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. This study was approved by the research ethics boards at Brigham and Women's Hospital (BWH, Boston, MA, USA), McMaster University (Hamilton, ON, Canada), and the Love Research Army (LRA; formerly known as the Army of Women) Scientific Advisory Committee.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
