Abstract
Background and objective:
Breast cancer and its treatments can have a marked impact on the patient health-related quality of life. The aim of this study was to produce and validate Finnish versions of the breast-conserving treatment, mastectomy and breast reconstruction modules of the BREAST-Q, a patient-reported outcome tool designed specifically for women undergoing treatment for breast cancer.
Methods:
The relevant BREAST-Q modules were translated to Finnish according to established guidelines. Altogether 777 preoperative women were recruited at the Breast Surgery Unit and the Plastic Surgery Unit of Helsinki University Hospital between December 2019 and March 2021. This included 541 patients scheduled for breast-conserving surgery, 86 for mastectomy, and 150 for breast reconstruction. Postoperative patients were recruited through a postal survey, approaching 500 women operated for breast cancer in 2017, including 250 women who had undergone breast-conserving surgery and 250 women who had undergone mastectomy, as well as 339 women who had undergone breast reconstruction between August 2017 and July 2019. The patients were invited to fill the BREAST-Q modules relevant to their treatment and the general health-related quality-of-life instrument SF-36. A repeat administration of the BREAST-Q was done 2 weeks later.
Results:
A total of 665 (41%) women participated in the study, 339 (44%) preoperatively and 326 (39%) postoperatively. The BREAST-Q subscales showed high internal consistency with most Cronbach’s alphas > 0.8. The repeatability of the subscales was excellent with most intra-class correlation coefficients > 0.75. Low or negligible correlation was observed between BREAST-Q subscales and SF-36 domains.
Conclusions:
The Finnish version of the BREAST-Q modules breast-conserving treatment, mastectomy, and breast reconstruction performs well in assessing the health-related quality of life of women undergoing surgery for breast cancer or breast reconstruction.
Context and Relevance
An increasing emphasis is placed on patient-reported outcomes across surgical specialties. Effective evaluation of these outcomes requires validated instruments, specifically designed to assess measures relevant to the patient group. The BREAST-Q is a modular patient-reported outcomes instrument, developed for the assessment of the health-related quality of life of women undergoing breast surgery. Here, we translated and validated Finnish versions of the BREAST-Q modules relevant to women undergoing surgery for breast cancer.
Introduction
Breast cancer is the most commonly occurring cancer, affecting 48 in 100,000 women globally every year. 1 Breast cancer treatments often have an impact on women both physically and psychologically, and it is thus important for clinicians and researchers to have pertinent tools to measure such effects.2,3 To date, no validated patient-reported outcomes instrument that specifically assesses the impact of breast surgery on the health-related quality of life of women with breast cancer is available in the Finnish language.
BREAST-Q is a modular patient-reported outcomes instrument, designed to measure the health-related quality of life of women undergoing breast surgery.4–7 The questions included address physical, psychological, and social aspects of the treatment. Three of the BREAST-Q modules are relevant to patients undergoing surgery for breast cancer, namely the Breast-Conserving Therapy (BCT) Module, the Mastectomy Module, and the Reconstruction Module. These modules have been translated to altogether over 40 languages. 8
The aim of this study was to produce a Finnish language version of the BREAST-Q modules relevant to patients with breast cancer and to assess the validity and reproducibility of the scales.
Methods
The study protocol was approved by the research ethics committee of Helsinki University Hospital. Written, informed consent was obtained from all participants.
Translation of the questionnaire
The BREAST-Q BCT, Mastectomy, and Reconstruction modules were translated into Finnish according to the ISPOR guidelines and the questionnaire developer’s instructions. 9 Two independent Finnish translations were done by native Finnish speakers familiar with the subject matter and compared to produce the Finnish forward translation. A translation back into English was done by a bilingual professional translator and then compared with the original questionnaire, to identify and correct any ambiguities. The final Finnish version was reviewed by a panel of six surgeons. A pilot study was then conducted with 15 patients invited to comment on the language and suggest any further changes. Permission for the translation of the BREAST-Q was sought from the copyright owner, and members of the Q-Portfolio team checked and approved the final translations.
Validation of the questionnaire
A total of 777 patients were recruited preoperatively at the Helsinki University Hospital Breast Surgery Unit and Plastic Surgery Unit between December 2019 and March 2021. Out of these, 627 women had newly diagnosed breast cancer and 150 women were scheduled for breast reconstruction (see Fig. 1). Postoperative questionnaires were sent to 500 patients operated for breast cancer in 2017, and 339 patients who had undergone breast reconstruction between August 2017 and July 2019 (see Fig. 1). A pre-paid return envelope was provided. Patients filled the module relevant to the treatment they were about to undergo or had undergone. The general health-related quality of life instrument SF-36 was used for comparison.10,11 To assess the reliability of the BREAST-Q questionnaire on repeat administration, the postoperative patients filled the questionnaire a second time 2 weeks later. No repeat completion was requested of the preoperative patients for the BCT and mastectomy modules since most were scheduled for surgery less than 2 weeks from the appointment at the clinic.
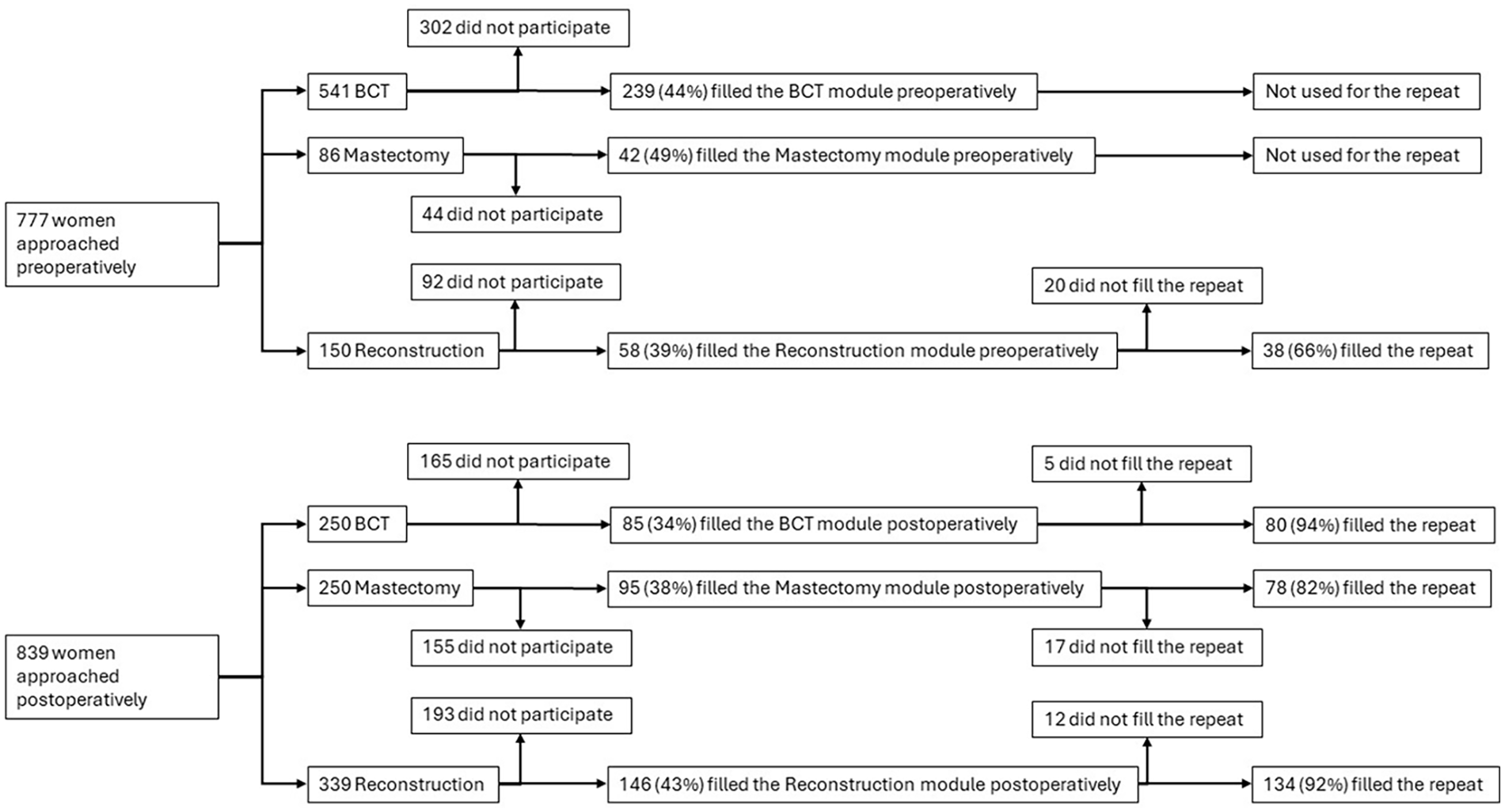
Flow chart showing the patient recruitment for the study.
Each BREAST-Q subscale was analyzed separately. The data were screened for missing answers. Questions with over 50% of missing answers were excluded. For questions with less than 50% of the answers missing, the data were completed with the mean response of the other items in the scale. The answers were then rescaled according to the original BREAST-Q scale scoring, with 0 indicating the worst possible result and 100 the best result. Exceptions were the adverse effect scales, which were not rescaled as per the original BREAST-Q scale instructions.4,6
Score distributions were assessed with mean and standard deviation. Floor and ceiling effects were explored, defined as being present when the proportion of patients scoring the minimum or maximum score, respectively, was over 15%. The internal consistency of the scales was evaluated with Cronbach’s alphas, bootstrapping with 1000 replacements. An alpha above 0.70 was considered acceptable. 12 Measurement reliability was evaluated with repeated measurements, the difference between the measurements assessed with Bland–Altman analysis using 95% limits of agreement and the Bradley–Blackwood procedure to determine the significance of the difference. The intra-class correlation coefficient (ICC) and the repeatability coefficient (CR) were calculated between the two measurements using one-way random effects model. An ICC value over 0.75 was interpreted as excellent and value over 0.6 as good reliability of the measurement. 13
Spearman correlation coefficients were calculated between the BREAST-Q scale scores and the eight SF-36 domains. The statistical significance of the correlation was estimated with Sidak-adjusted probabilities. A correlation coefficient of over 0.7 was interpreted as high, 0.5–0.7 moderate, 0.3–0.5 low, and less than 0.3 negligible.
Stata 18 statistical software was used in data processing and statistical analysis. 14
Results
Translation of the questionnaire
Translation of the BREAST-Q questionnaire subscales to Finnish was straightforward. Review of the back translations resulted in no changes in the translation. The panel review of the translation led to two grammatical changes and the selection of a synonym for the translation of the word “body.” The pilot study comments did not identify a need for further changes.
Validation of the questionnaire
A total of 665 (41%) women participated in the study (see Fig. 1). Of the women operated for breast cancer, 281 (45%) were preoperative and 180 (36%) postoperative cases, and of the women undergoing breast reconstruction, 58 (39%) were preoperative and 146 (43%) postoperative cases. The mean age was 61.2 (standard deviation (SD) 11.0) years (see Table 1). For women in the postoperative group, the mean time from surgery was 3.0 (SD 1.3) years. Out of the women in the postoperative group that had undergone breast-conserving surgery, only three (4%) had oncoplastic surgery performed and one (1%) a contralateral procedure for symmetrization. Of the women in the postoperative group operated with mastectomy, 18 (19%) underwent bilateral procedures. As per study design, the women in the preoperative group scheduled for breast-conserving surgery or mastectomy had not received neoadjuvant therapy. Of the women who had undergone breast-conserving surgery, all had received adjuvant radiotherapy, 17 (20%) had received adjuvant chemotherapy with/without targeted HER-2 therapy, and 52 (61%) received endocrine therapy. Of the women who had undergone mastectomy, 48 (51%) had received adjuvant radiotherapy, 45 (47%) had received chemotherapy with/without targeted HER-2 therapy, and 68 (72%) received endocrine therapy.
Characteristics of study participants.
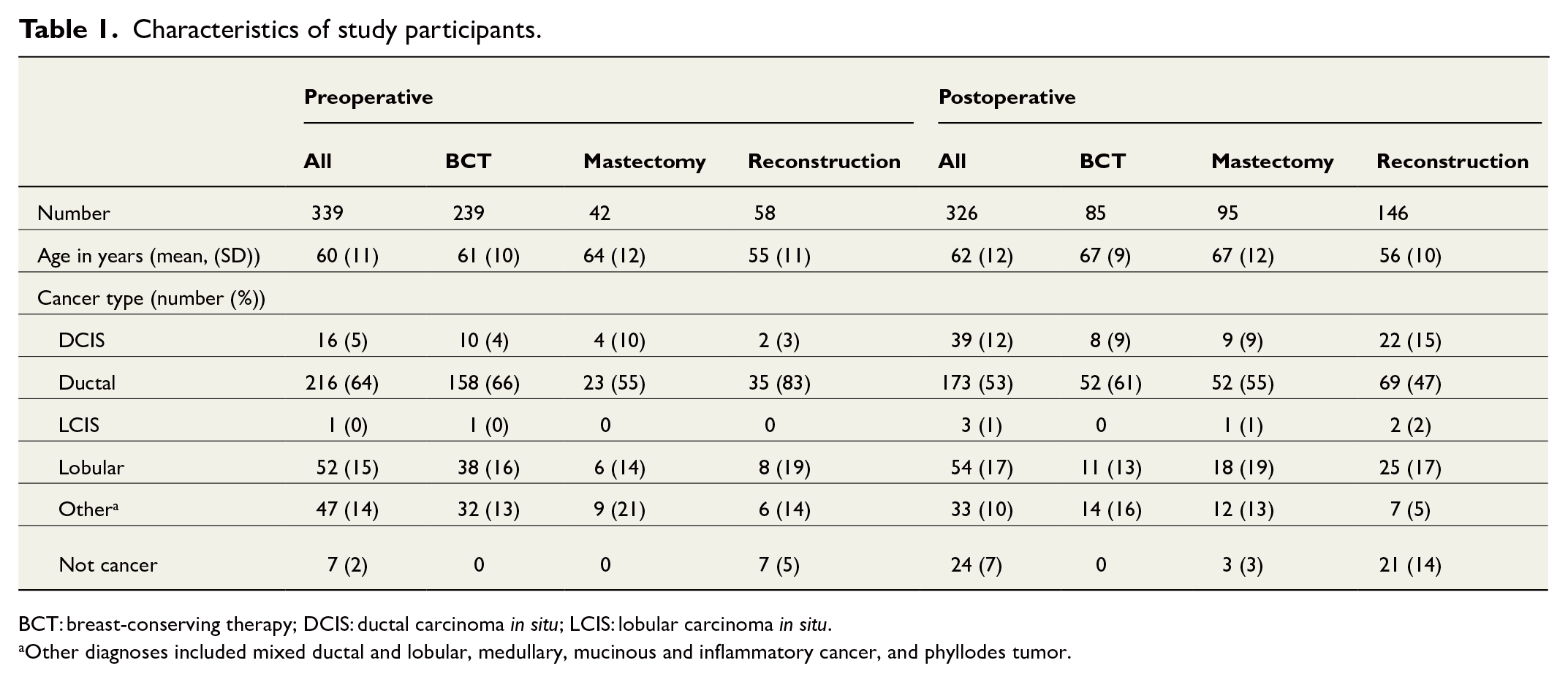
BCT: breast-conserving therapy; DCIS: ductal carcinoma in situ; LCIS: lobular carcinoma in situ.
Other diagnoses included mixed ductal and lobular, medullary, mucinous and inflammatory cancer, and phyllodes tumor.
Performance of the BREAST-Q subscales was good, showing high internal consistency with Cronbach’s alphas above 0.80 for most subscales. Exceptions were the adverse effects of radiation checklist (Cronbach’s alpha 0.74) and the preoperative “physical well-being: abdomen” subscale of the Reconstruction Module (Cronbach’s alpha 0.79), for which the observed internal consistency was still acceptable (see Table 2). A floor effect was observed for the adverse effects of radiation checklist and the preoperative “satisfaction with abdomen” question of the Reconstruction Module. A ceiling effect was observed for nine subscales: “physical well-being: chest,” “satisfaction with surgeon,” “satisfaction with medical team,” “satisfaction with office staff”; BCT module postoperative: “satisfaction with breasts” and “satisfaction with information—radiation oncologist”; Reconstruction Module preoperative: “physical well-being: abdomen”; Reconstruction Module postoperative: “satisfaction with nipple reconstruction”; latissimus dorsi postoperative: “satisfaction with back.” The ceiling effect was notably prominent in the cancer core scales assessing satisfaction with different team members (see Table 2).
Scores of the BREAST-Q subscales and assessment of the internal consistency of the subscales.
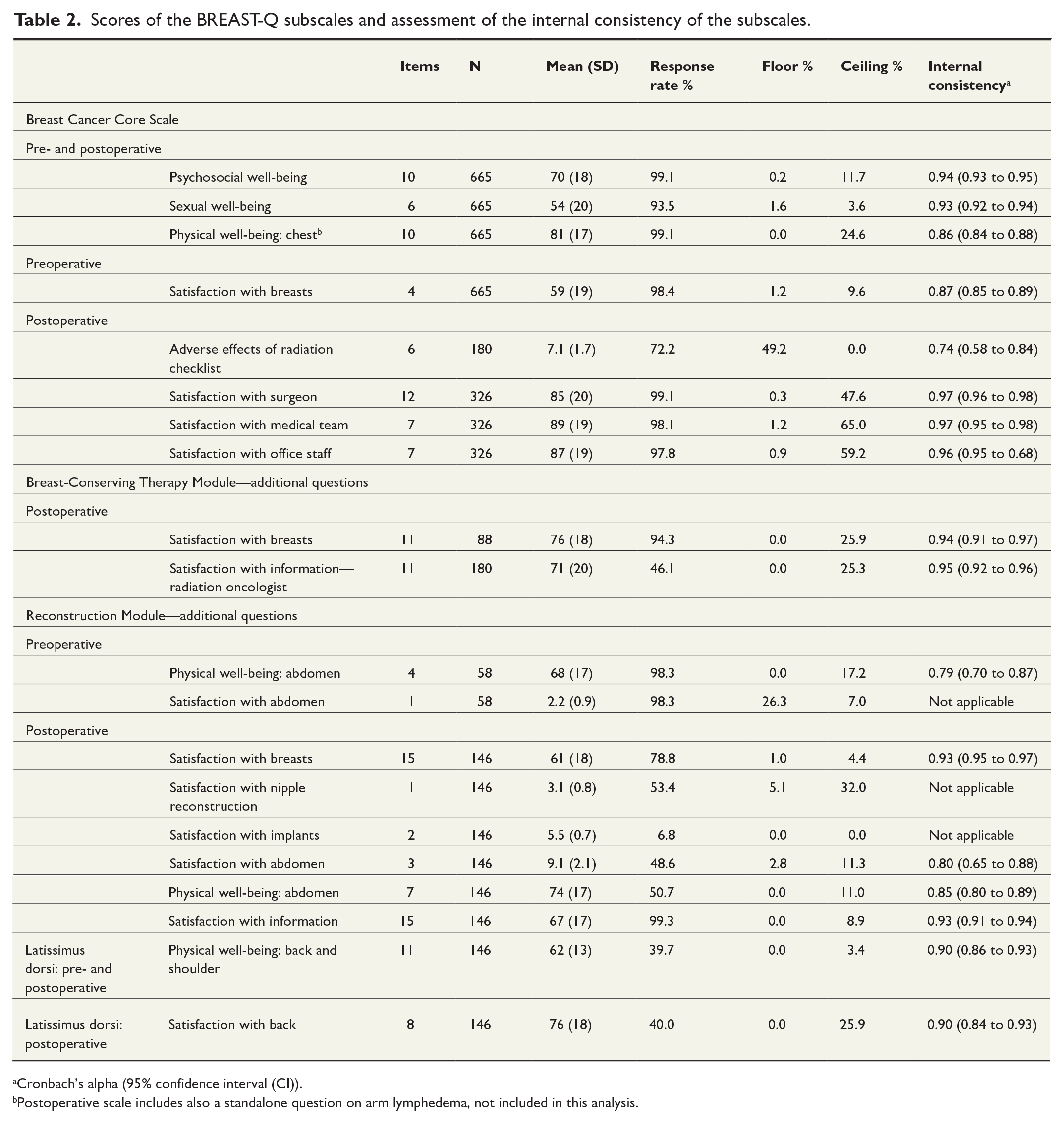
Cronbach’s alpha (95% confidence interval (CI)).
Postoperative scale includes also a standalone question on arm lymphedema, not included in this analysis.
Reproducibility of most of the subscales was excellent with ICC values above 0.75 (see Table 3). For the subscales Reconstruction postoperative: “satisfaction with nipple” and Reconstruction preoperative: “physical well-being: abdomen,” the reproducibility was good. The difference between the score for the first administration and the second administration was statistically significant for the psychosocial well-being subscale (see Table 3).
Reliability of the BREAST-Q subscales on repeat administration.
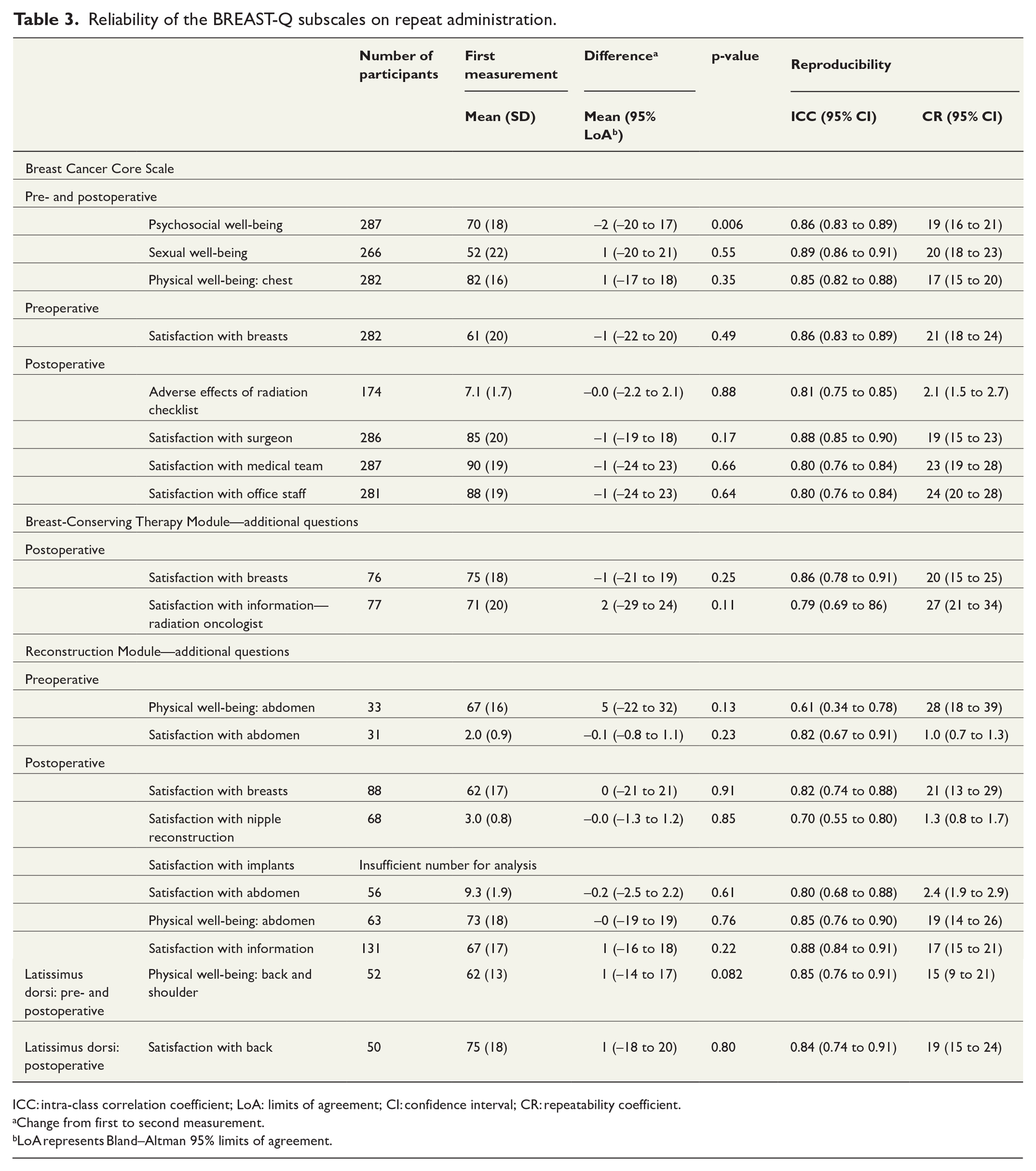
ICC: intra-class correlation coefficient; LoA: limits of agreement; CI: confidence interval; CR: repeatability coefficient.
Change from first to second measurement.
LoA represents Bland–Altman 95% limits of agreement.
The correlation between BREAST-Q subscales and most SF-36 domains was low or negligible (see Table 4). Moderate correlation was observed with the physical function domain and the subscales reconstruction preoperative: “physical well-being: abdomen” and latissimus dorsi: pre- and postoperative: “physical well-being: back and shoulder.” The back and shoulder subscale correlated moderately also with the “physical role limitations” domain and the “bodily pain” domain.
Convergence between the BREAST-Q subscales and the SF-36 domains.
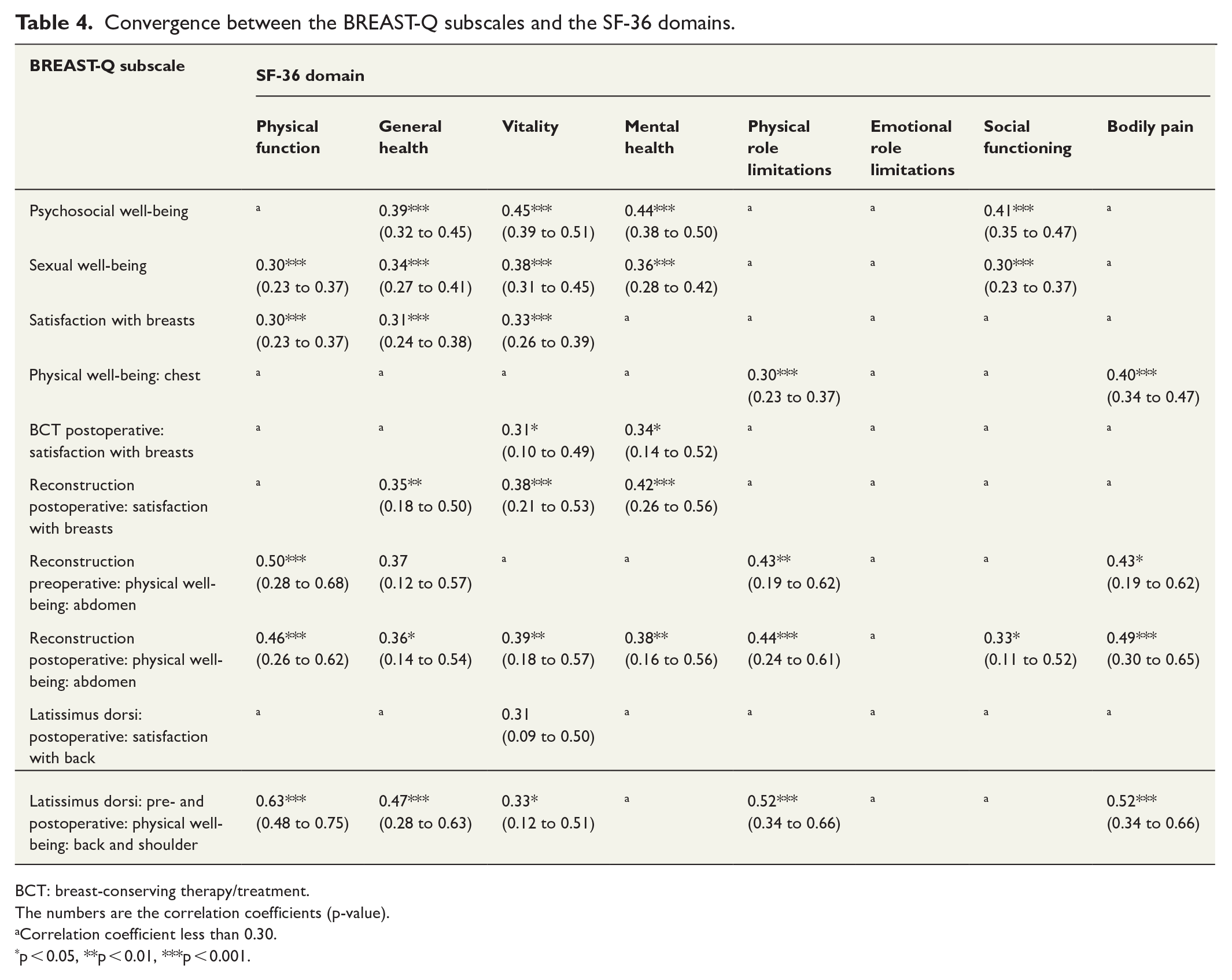
BCT: breast-conserving therapy/treatment.
The numbers are the correlation coefficients (p-value).
Correlation coefficient less than 0.30.
p < 0.05, **p < 0.01, ***p < 0.001.
Discussion
Breast cancer surgery can have a significant impact on patient health-related quality of life. 15 Understanding this impact and incorporating it into clinical decision-making is increasingly valued.16,17 Here, Finnish language versions of the BREAST-Q modules BCT, Mastectomy, and Reconstruction were produced. BREAST-Q is a patient-reported outcomes instrument designed for the assessment of patients undergoing breast cancer treatments.4–7 The validity and the reliability of the instrument in the Finnish language patient population was demonstrated through a study involving altogether 665 breast cancer patients.
The Breast Cancer Core Scales, applicable to all pre- and postoperative patients, and the additional scales of the BCT and Reconstruction modules performed well in the study population, displaying acceptable internal consistency and high test–retest reliability. A floor or a ceiling effect at the positive end of the scale was, however, present in half of the scales, suggesting that the questions may not sufficiently differentiate between patients generally satisfied with their situation. In addition, a floor effect with over a quarter of the patients expressing dissatisfaction with their abdominal region was observed in the patients awaiting breast reconstruction.
Normative values have been published for the Breast Cancer Core Scales, based on an American population of over 1000 women, with and without a history of breast cancer. 18 The mean values for the subscales psychological well-being (70), sexual well-being (54), and satisfaction with breasts (59), were similar to the normative values published for these scales, 71, 56, and 58, respectively. 18 The difference was less than the minimal important difference, 4, for all of the subscales. 19 This suggests that these subscales function appropriately in the Finnish language patient population. The difference between the mean score for the physical well-being: chest subscale, 81, and the normative value for the subscale, 93, exceeded the minimal important difference for the subscale, that is, 3.18,19 The score observed in the present population was, however, higher than the clinical reference values reported for women following breast reconstruction. 20 This may reflect the presence of women without a history of breast cancer in the population used to derive the normative values, while the study population here consisted solely of women with current or past breast cancer.
Results of the BREAST-Q subscales correlated at best moderately with domains of the general health-related quality of life instrument SF-36. Positive correlations, although low in strength, were observed between the subscales psychological well-being and sexual well-being and the SF-36 domains general health, vitality, mental health, and social functioning. Similarly, low or moderate correlations were detected between the subscales assessing physical well-being of the abdomen or the back following breast reconstruction and the SF-36 domains concerning physical function, its limitations, pain, general health, and vitality. These observed correlations further support the psychometric validity of the BREAST-Q instrument. 21 The subscales “satisfaction with breasts” and Reconstruction postoperative: satisfaction with breasts showed a correlation with the domains general health, vitality, and in the case of women with breast reconstruction, mental health. This is in line with previous studies demonstrating an impact of either breast deformity or reconstruction on both overall and psychological well-being of women.22,23
The main strength of this study is the large patient sample, altogether 665 women, invited to participate in the validation of the subscales relevant to the type and stage of their breast cancer treatment only. For most of the subscales, the response rate and the number of responses were sufficient for a reliable analysis. 24 Women with implant reconstructions, in particular, were however underrepresented, rendering the validity of the translated subscale “satisfaction with implants” uncertain. The number of oncoplastic procedures, and of contralateral surgery for symmetrization was also lower in the study population than in the authors patient population in general, which could introduce a potential source of bias. In addition, the test–retest validity of the scale was assessed in postoperative women only, limiting the sample size. The assessment of the conceptual validity, in turn, was limited by the lack of an existing breast-specific scale to use as a comparison.
The Finnish language versions of the BREAST-Q BCT, Mastectomy, and Reconstruction modules perform well at assessing the health-related quality of life of women undergoing surgery for breast cancer or breast reconstruction. Further research is needed to establish the normative values and the minimally important difference for the scales in the Finnish language population. The BREAST-Q scales are available free of charge for clinical and research use at the Q-Portfolio website.
Footnotes
Acknowledgements
The authors would like to thank representatives of the Q-portfolio for their assistance in the translation process, breast cancer nurses at the Helsinki University Hospital Breast Cancer Unit for patient recruitment, Leena Caravitis for her contribution to data processing, and Hannu Kautiainen for statistical consultation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the department of Musculoskeletal and Plastic Surgery, Helsinki University Hospital.
