Abstract
Introduction
Flap failure can have devastating consequences, and venous thrombosis has been cited as the leading cause. 1 Thrombosis can result from technical factors such as kinks, compression of the vessels, unrecognized intimal injury (backwall suture, inadequate coupler disk alignment), or non-technical factors such as patient co-morbidity. Numerous anti-thrombotic protocols have been described to prevent or treat thrombosis of free flaps. 2 -5 Currently, there is no clear consensus on which anti-thrombotic regime is superior. Multiple studies have proposed anti-thrombotic guidelines, but none have been shown to be superior in preventing thrombosis. 5 -11 Moreover, the diversity of flap characteristics and patient co-morbidities presents additional complexity in comparing studies and forming a standardized protocol. To compound the issue, studies such as Chang et al 7,12,13 and Zhou et al 14 suggest that anti-thrombotic agents may not improve survival rate of flaps, raising questions on the efficacy of anti-thrombotic agents.
Nevertheless, the majority of surgeons still use pharmacological agents to prevent thrombosis. 15 Hematologists may be asked to provide recommendations on prophylaxis or treatment of flap thrombosis. It is important to understand the practice variations among physicians involved in free flap reconstruction in an attempt to standardize protocols, understand agent choice, and determine optimal regimens. The purpose of this study was to determine the practice variations in Canada, taking into consideration the different perspectives of the physicians performing or caring for patients undergoing free flap surgery.
Methods
A national survey of Plastic surgeons (PS), ENT surgeons, and hematologists/thrombosis physicians caring for patients undergoing free flap reconstruction was conducted to examine current anti-thrombotic practice patterns. Seven hundred and twenty-two surveys were distributed electronically to members of Thrombosis Canada (n = 112), the Canadian Society of Otolaryngology-Head & Neck Surgery (n = 209), and the Canadian Society of Plastic Surgeons (n = 401). A reminder e-mail was sent after 2 weeks. Data were collected on physician demographics and practice, routine screening practices for hypercoagulopathy, perceptions of risk factors for flap failure, and routine pre-, intra-, and post-operative anti-thrombotic strategies (Figure 1). Physicians were also asked how their management would change if patients had a risk factor for thrombosis or if the patients developed flap thrombosis intra- or post-operatively. The survey was refined following internal pilot testing and feedback.
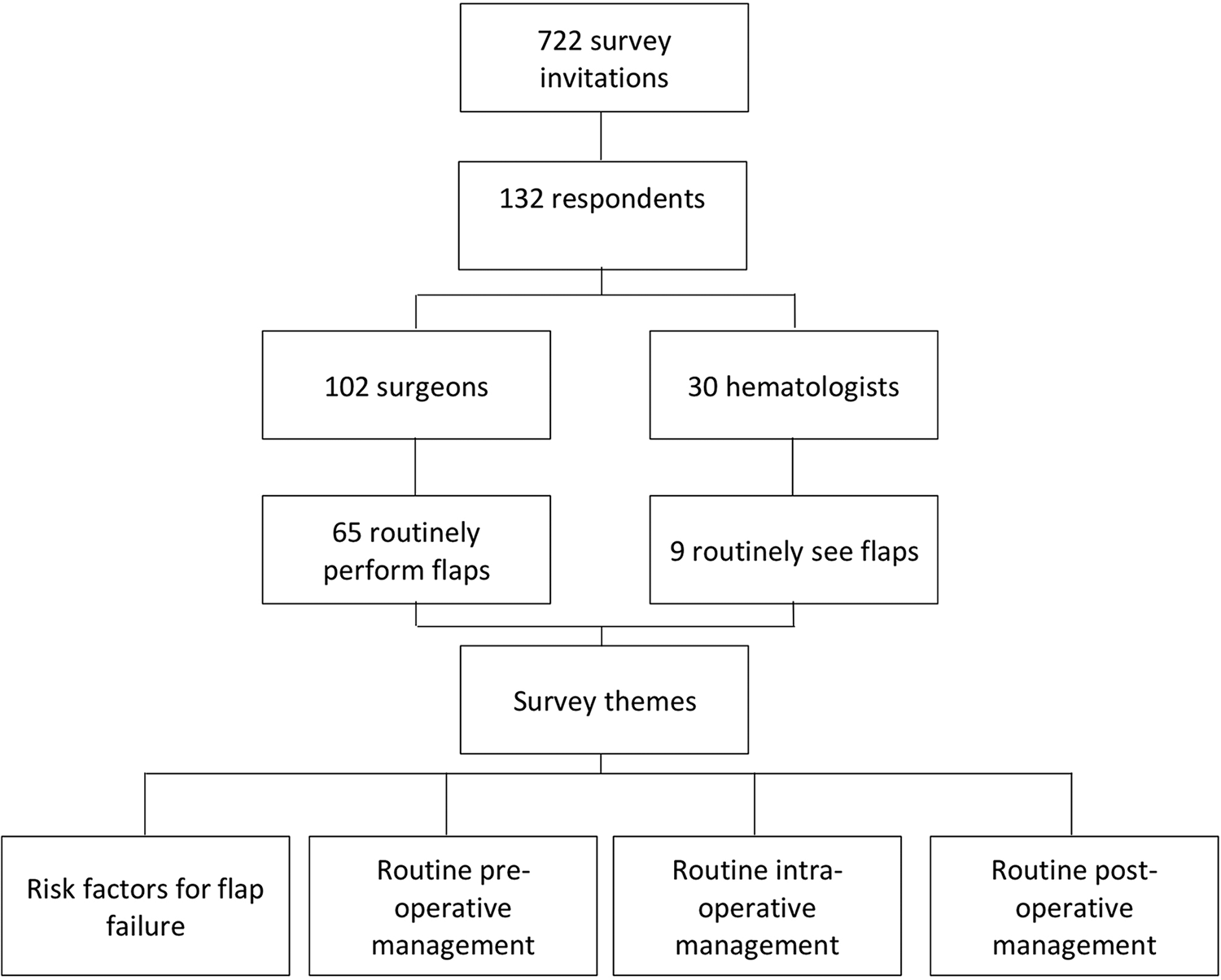
Methodology of survey distribution.
Physicians and surgeons who did not routinely perform or manage free flaps were excluded. Responses were analyzed with SPSS version 23. Descriptive statistics were performed, and continuous variables were compared using the Student t test. The study received ethics approval by the Hamilton Integrated Research Ethics Board.
Results
Demographics
Surveys were distributed to 722 potential participants across the 3 mailing lists (Figure 1). There were 132 (18%) respondents consisting of 102 surgeons and 30 hematologists. From the respondents, 65 surgeons (44 PS and 21 ENT) routinely perform free flaps and 9 hematologists routinely see patients with flaps perioperatively. Physicians and surgeons who did not routinely perform or care for free flaps were excluded from the survey (n = 67). Most respondents (85%) who manage flaps were from academic centers. The average duration in practice was 12 ± 9.2, 9 ± 8, and 15 ± 7.4 years for PS, ENT, and hematologists, respectively. Plastic surgeon and ENT surgeons performed on average 19 ± 17 and 45 ± 19 flaps per year, respectively. Hematologists reported seeing on average 3.7 ± 3.3 flaps per year (Table 1).
Respondent Demographics.
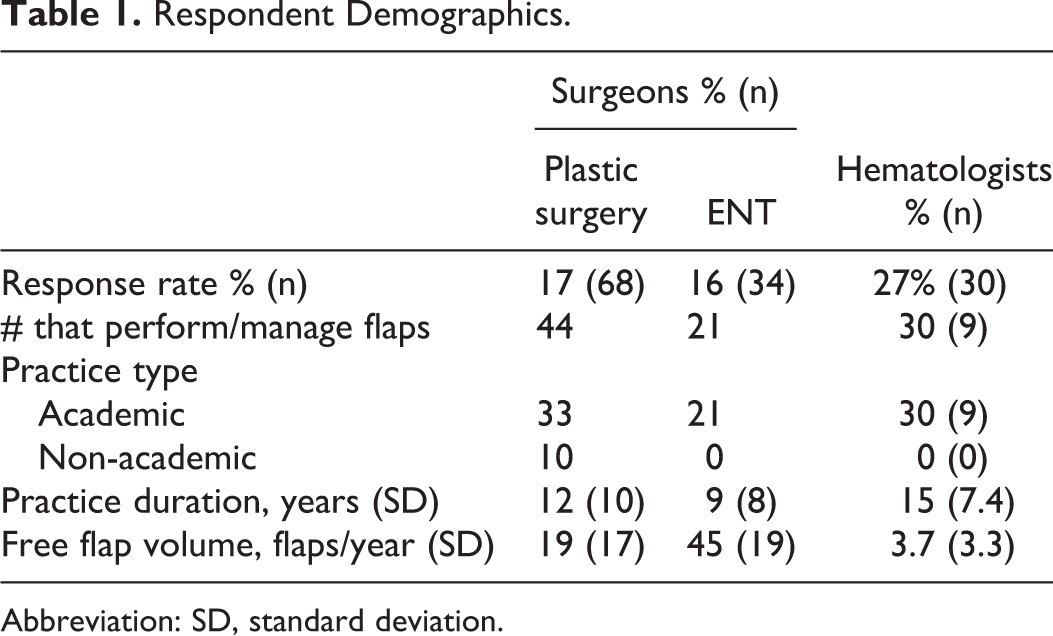
Abbreviation: SD, standard deviation.
Risk Factors for Thrombosis
The top 3 perceived patient risk factors for flap failure according to surgeons were medical co-morbidities, past arterial thrombosis younger than 50 years old, and thrombophilia. A third of the surgeons (31%) would add an anti-thrombotic agent if a patient had a known thrombophilia. Hematologists, however, reported diabetes, smoking, and medical co-morbidities as their top 3 risk factors. Hematologists did not perceive thrombophilia to be as high of a risk factor in terms of flap failure and would most likely not add an anti-thrombotic in patients with thrombophilia (n = 8/9). Not only was there a disparity between surgeons and hematologists on the perceived importance of thrombophilia as a risk factor, but there was also a significant difference of opinion on preoperative chemotherapy, preoperative radiation, and diabetes mellitus (Table 2).
Perceived Patient Risk Factors for Flap Failure Between Surgeons and Hematologists.
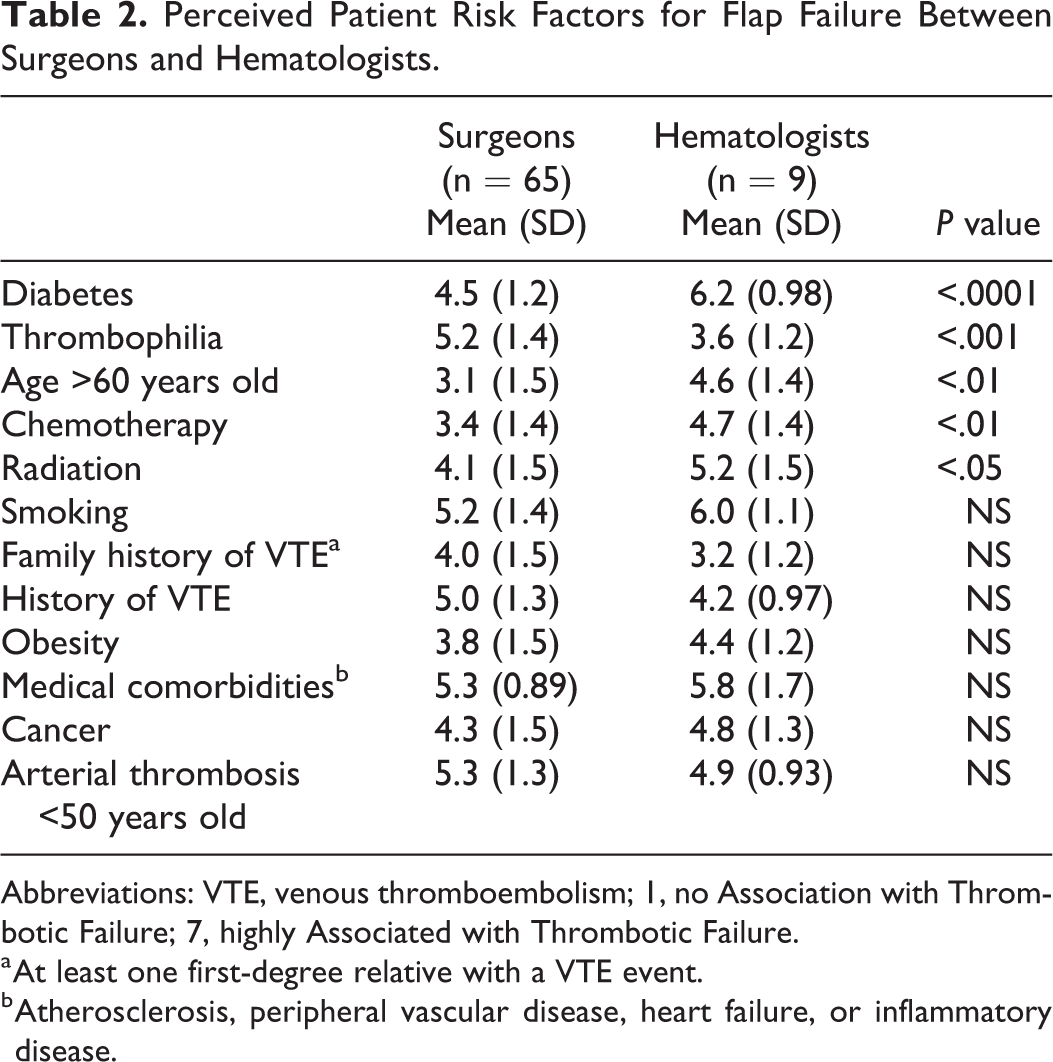
Abbreviations: VTE, venous thromboembolism; 1, no Association with Thrombotic Failure; 7, highly Associated with Thrombotic Failure.
a At least one first-degree relative with a VTE event.
b Atherosclerosis, peripheral vascular disease, heart failure, or inflammatory disease.
Anti-Thrombotic Strategies
In total, 54% of physicians routinely use an anti-thrombotic agent preoperatively: 32% use unfractionated heparin (UFH; PS = 12; ENT = 9), and 22% use low-molecular-weight heparin (LMWH; PS = 6; ENT = 8) preoperatively (Figure 2). The remaining surgeons (46%) did not routinely use anti-thrombotic agents preoperatively. Forty-four percent of hematologists use either UFH or LMWH with an even split between both agents. The remaining 56% of hematologists would not routinely recommend any preoperative agents.
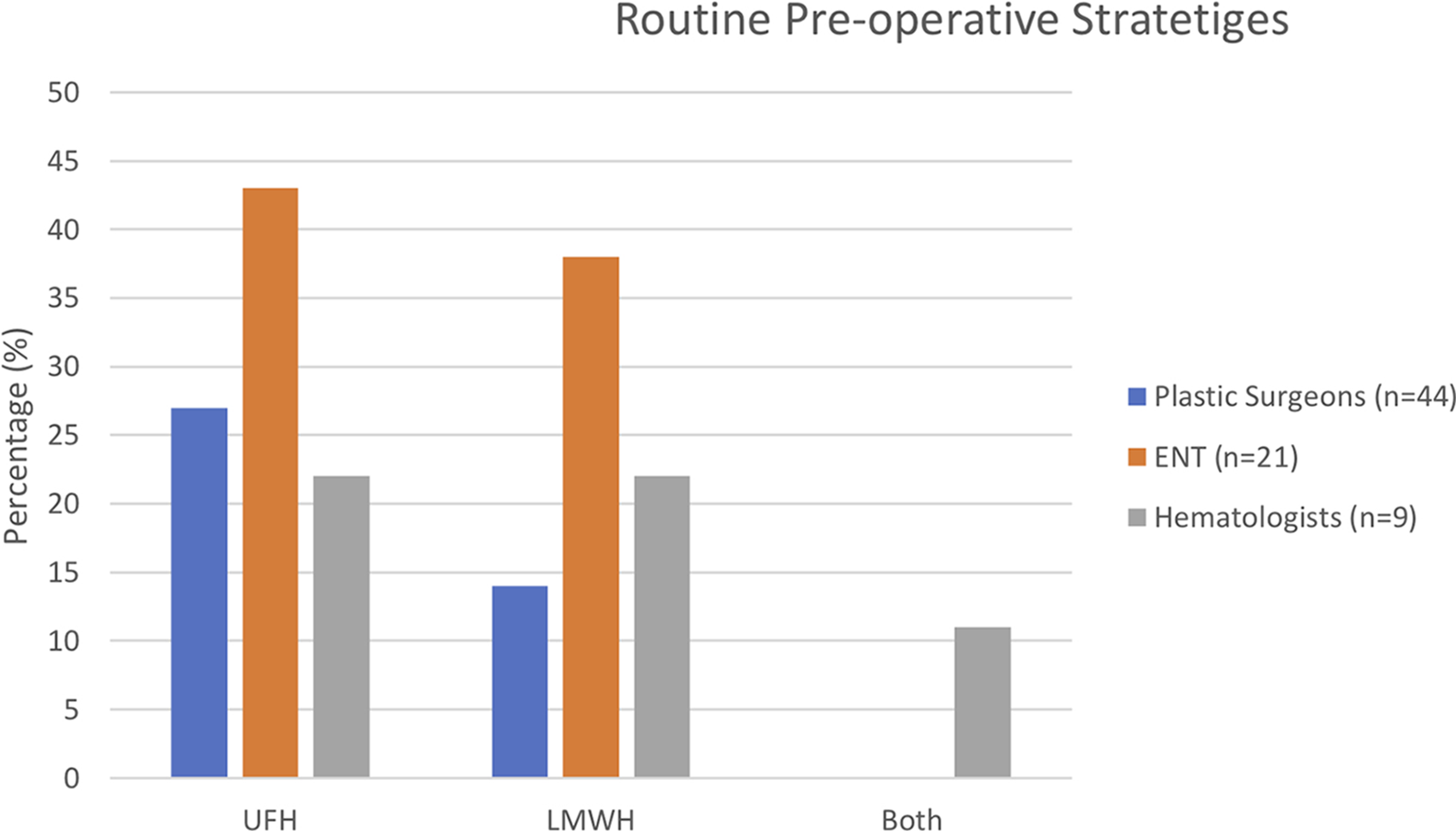
Routine preoperative strategies. LMWH indicates low-molecular-weight heparin; UFH, unfractionated heparin.
Only surgeons were mainly involved in routine intra-operative prophylaxis. Surgeons flushed the flap with heparin (38%; PS = 15; ENT = 10), UFH IV (6%; PS = 3; ENT = 1), both (8%; PS = 4; ENT = 1), or did not use any intra-operative agents (48%; Figure 3). There was a large variation in management strategies if an intra-operative thrombosis occurred. Different strategies consisted of changing recipient vessels (34%; PS = 12; ENT = 10), UFH IV (42%; PS = 19; ENT = 8), flushing the flap (54%; PS = 25; ENT = 10), adding post-operative UFH (34%; PS = 14; ENT = 8), adding post-operative ASA (42%; PS = 17; ENT = 10), or post-operative dextran (8%; PS = 5; ENT = 0). One hematologist recommended flushing the flap with heparinized saline (Figure 4).
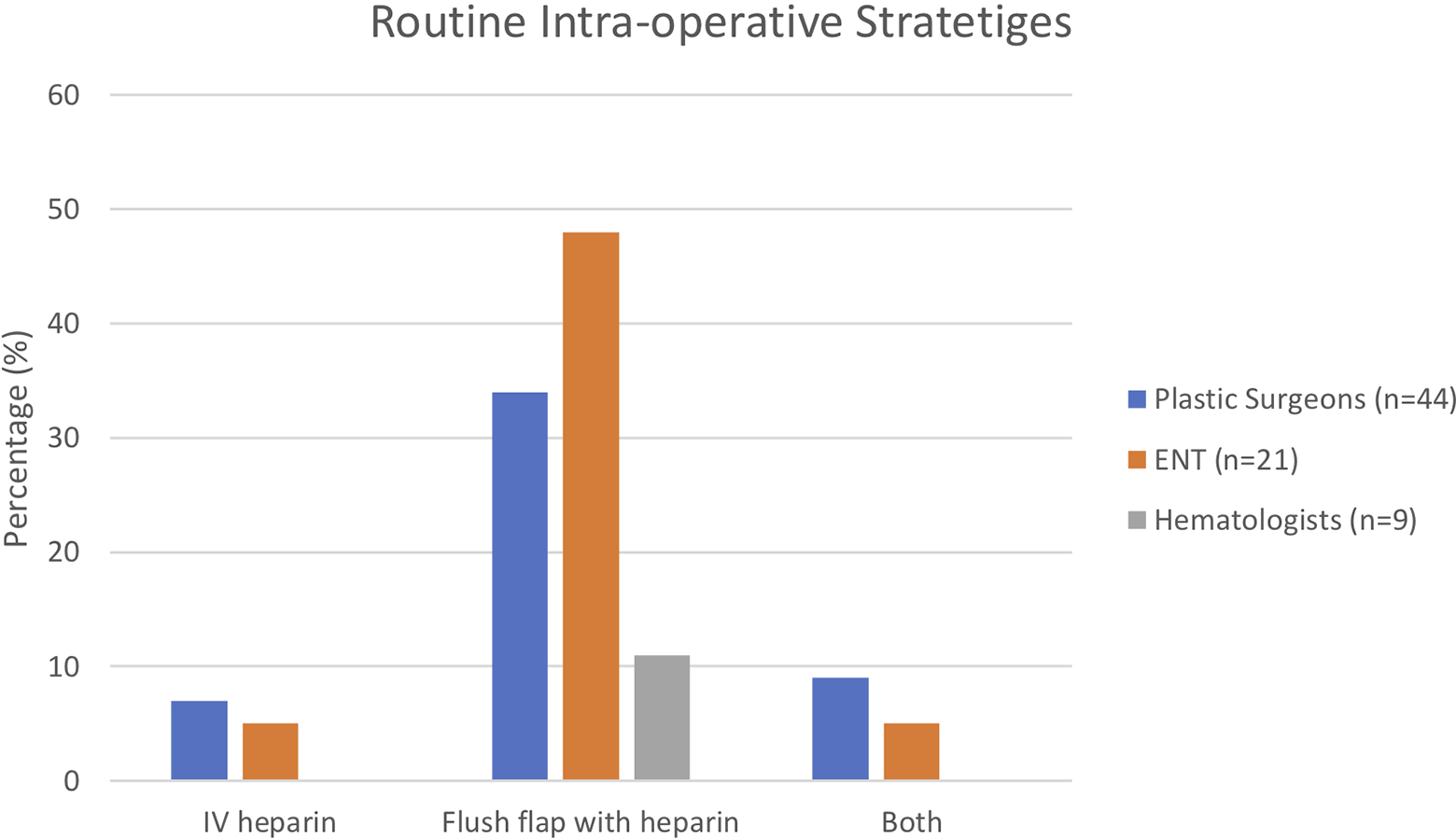
Routine intra-operative strategies.
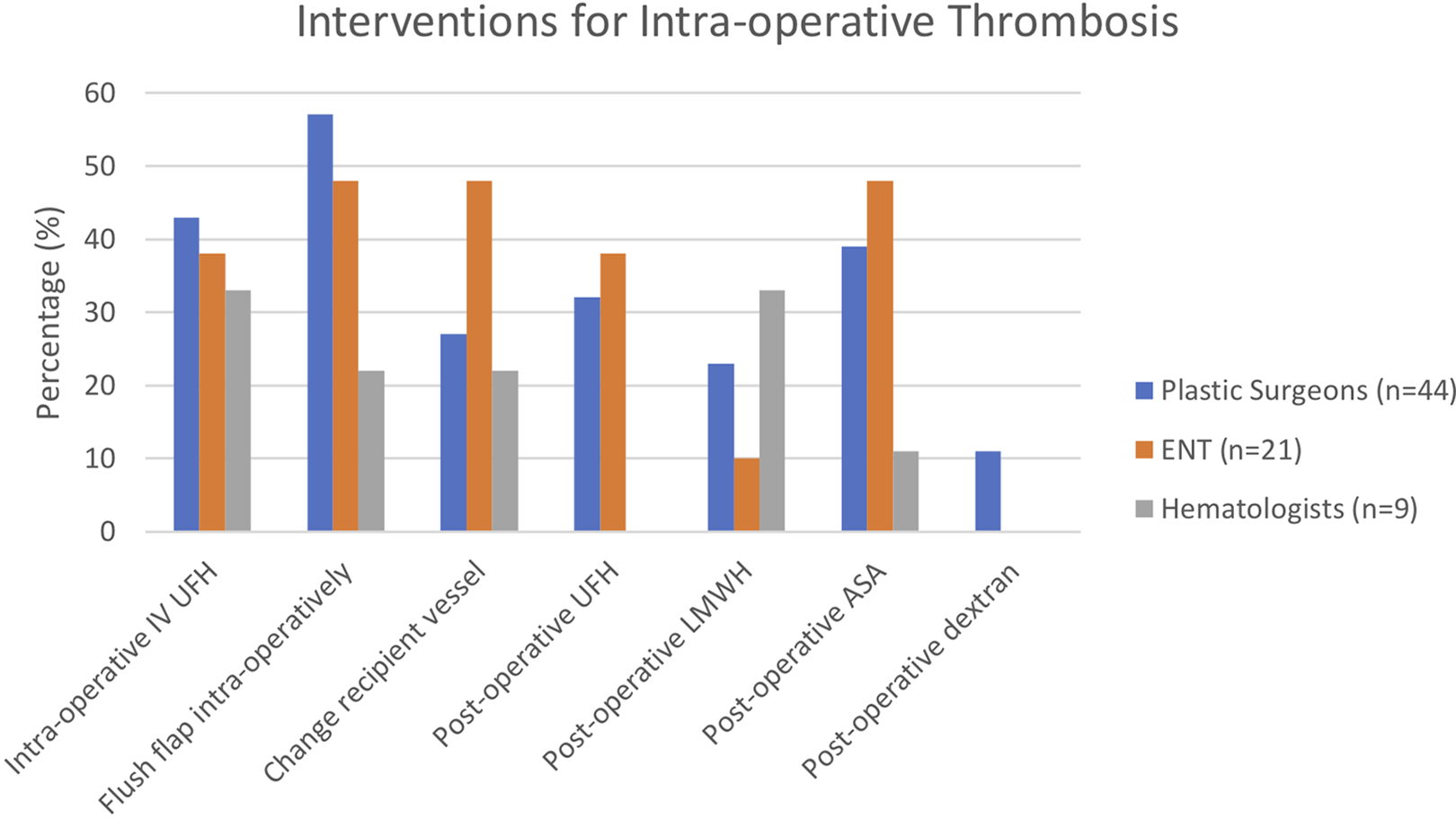
Interventions for intra-operative thrombosis. LMWH indicates low-molecular-weight heparin; UFH, unfractionated heparin.
Surgeons reported the use of a range of post-operative agents and combinations. Single agent strategies were used by 36% of respondents (UFH, LMWH, or ASA were used by 12% [PS = 6; ENT = 2], 11% [PS = 3; ENT = 4], and 14% [PS = 4; ENT = 5] of surgeons, respectively). Dual agent therapy was used by 20% of respondents (ASA was combined with UFH or LMWH 12% [PS = 4; ENT = 4] and 8% [PS = 2; ENT = 3] of the time, respectively). Other combinations of UFH, LMWH, ASA, and dextran were used 8% (PS = 5; ENT = 0) of the time. Hematologists who were involved in post-operative management almost exclusively used LMWH (56%) alone or in combination with UFH (11%; Figure 5).
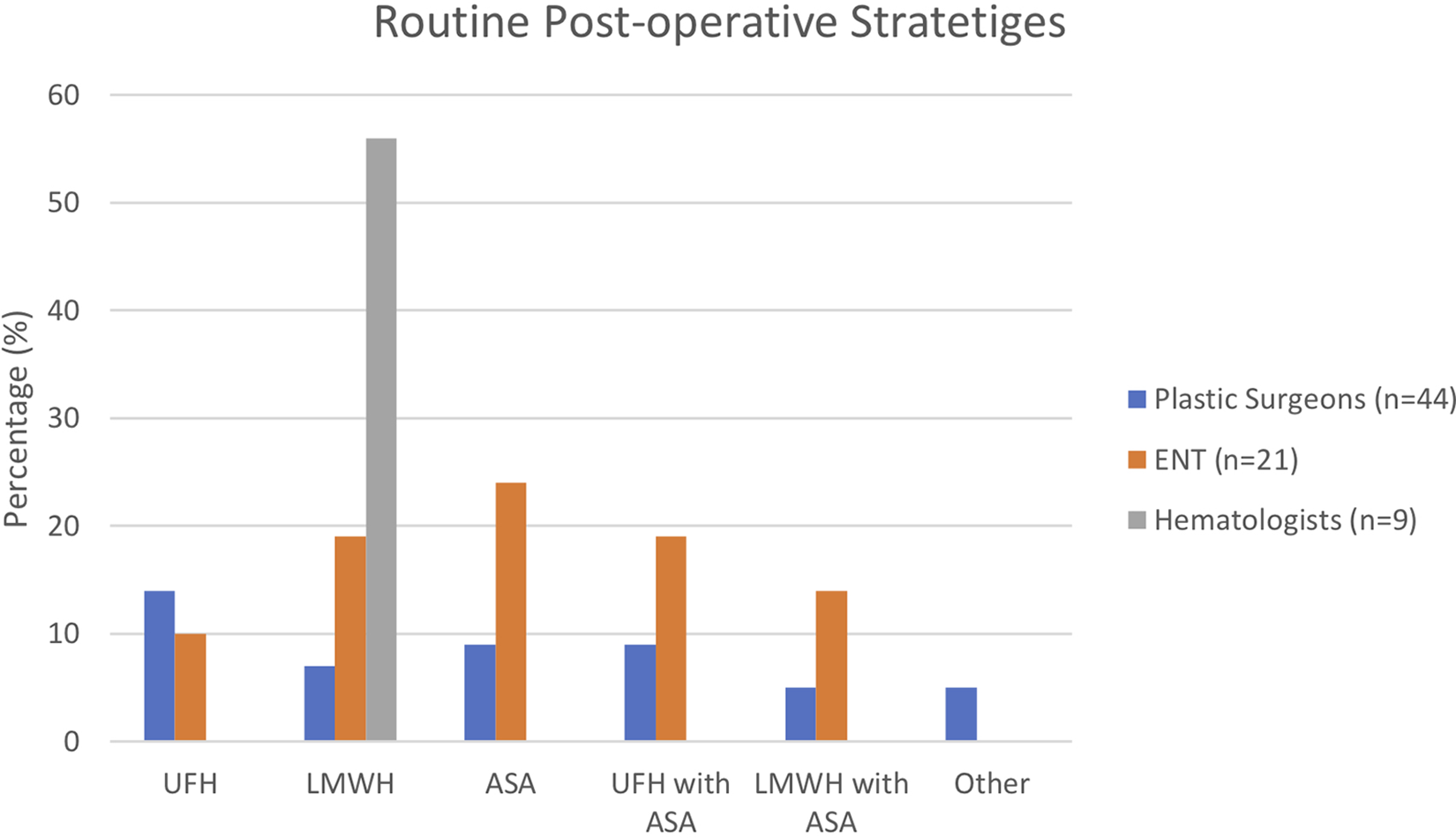
Routine post-operative strategies. LMWH indicates low-molecular-weight heparin; UFH, unfractionated heparin.
Patients who had thrombosis in the early post-operative course were more likely to receive anti-thrombotic agents if technical factors were not identified by surgeons (58%) or hematologists (33%). If a technical factor was found and corrected, surgeons (6%) and hematologists (11%) were less likely to add an anti-thrombotic agent intra- or post-operatively.
Discussion
There was significant variation in reported prophylactic and therapeutic anti-thrombotic regimens for free flap surgery. There also appeared to be a discordance between surgeons and hematologists with respect to perceived risk factors for flap thrombosis. These differences in perceived risk factors affected whether or not anti-thrombotic agents were administered.
Surveys of anti-thrombotic practice in microvascular reconstruction have been previously performed; however, these are largely historic or evaluated as a secondary outcome. In 1982, Davies 16 performed a survey on anticoagulation practices by microsurgeons. Since then, only Salemark, 17 in 1991, and Pannucci et al, 18 in 2010, have documented anticoagulation practices in microsurgery as secondary outcomes. The analysis performed by Davies and Salemark showed heavy use of dextran and fibrinolytics, which is a stark contrast to contemporary regimens. Furthermore, the study by Pannucci et al lacked stringent documentation of the anticoagulation practices, with only UFH, LMWH, and ASA use being recorded. Other agents, such as dextran or fibrinolytics, were not reported. No other recent study has examined the practice patterns of pharmacological flap management or the perspectives of our hematology colleagues.
Risk Factors
The literature concerning free flap reconstruction in the setting of thrombophilia is limited. Several studies assert an association between a hypercoagulable state and flap loss. 19 -25 Other studies, meanwhile, refute these claims. 13,26,27 Both Wang et al 22 and Mirzabeigi et al 23 performed a retrospective review of 2032 and 2260 free flaps, respectively. Patients with thrombophilia were identified and shown to have poorer outcomes, salvage rates, and flap survival compared to patients without thrombophilia. Historically, hypercoagulable states were routinely screened due to concerns of venous thromboembolism (VTE) risk. The current literature suggests that the increased risk for VTE events is only clinically appreciable in patients with a prior thrombotic event. 28,29 In fact, the American Society of Hematology recommends against regular screening of thrombophilia as it often does not change management. 30,31 These studies, however, were not meant to guide prevention of venous thrombosis in free tissue transfer.
There is a paucity in the literature for the association between preoperative chemotherapy and radiation on flap survival. Many studies include the 2 variables as patient demographic information, but only as a secondary outcome of their studies. Thus, there is no definitive conclusion if preoperative chemotherapy and radiation are risk factors for flap viability. 12,14,32 -40 Surgeons may have rated radiation and chemotherapy as less of a risk factor than hematologists because they routinely use flaps in oncologic patients receiving adjuvant therapy.
Lastly, surgeons did not perceive diabetes to be as important of a risk factor when compared with hematologists. Uncontrolled diabetes can lead to vascular issues that will ultimately damage the blood vessels. Similar to the other risk factors, the literature is equivocal if patients with diabetes are truly at an increased risk of flap failure. 13,14,23,41 -45 Surgeons may have rated diabetes as less of an important risk factor for flap failure due to the lack of evidence between diabetes and flap failure or being outside their scope of practice. Hematologists may have emphasized diabetes as a risk factor due to the vascular consequences of diabetes in general, not solely in the context of flap viability. In addition, several studies have proposed that patients with diabetes are at a higher risk for VTEs than the general population due to increased endothelial and coagulation cascade dysfunction resulting in a hypercoagulable state. 46,47 However, recent systematic reviews and meta-analyses 48,49 fail to discern an association between diabetes as an independent risk factor for VTEs. Therefore, the role of diabetes as a risk factor for flap failure, and VTEs, remains equivocal and no strong recommendations can be given.
Preoperative Management
The majority of surgeons (54%; PS = 18; ENT = 17) and hematologists (55%) routinely add an anti-coagulant with no discernible preference between UFH and LMWH. Surgeons (64%) explicitly stated that preoperative anti-thrombotics were specifically used for deep vein thrombosis (DVT) prophylaxis with improved flap survival as a possible secondary benefit. The choice between UFH and LMWH is ultimately due to personal preference for patients undergoing microsurgery.
The benefits of UFH and LMWH remain to be properly studied in relation to flaps, but there does not seem to be increased complications with preoperative anti-coagulation. 50,51 However, preoperative thromboprophylaxis may provide ancillary benefits such as reducing the rates of VTEs after microsurgery. Bertolaccini et al 52 investigated the pharmacodynamics of fixed-dosed intra-operative UFH (500 U/h) and discovered that it did not reach therapeutic levels to prevent VTEs. For patients who are not being appropriately weight dosed with intra-operative or post-operative anti-thrombotic agents, preoperative agents may provide a crucial role of DVT prophylaxis. Thus, recommendations on the use of preoperative anti-coagulation for flap viability remain equivocal, but it may provide ancillary benefits.
Intra-Operative Management
Intra-operative prophylactic anti-thrombotic strategies are diverse, with the literature illustrating the variation in expert opinion. Current practice patterns in our study support this diversity. Moreover, if a thrombosis occurs intra-operatively, treatment strategies are equally as varied. Intra-operative interventions ranged from flushing the flap to introducing post-operative dextran or using a combination of anti-thrombotic agents.
The benefit of intra-operative prophylactic or therapeutic anti-thrombotic agents has yet to be determined. However, our study discovered that agents such as dextran, ketorolac, and Tissue plasminogen activator (TPA) are still being used as intra- and post-operative interventions despite a lack of data supporting their effectiveness or their risk for severe complications. 53 -59
Post-Operative Management
Diverse pharmaceutical regimens were also employed for post-operative management. Although dosing and choice of anti-thrombotic varied greatly, most employed UFH, LMWH, or ASA. If a post-operative thrombosis occurred, the overwhelming majority would initiate an anti-platelet, anti-coagulant, or both. Hematologists preferred LMWH to UFH in post-operative management.
Several studies 53,56,60,61 have demonstrated the increased risk of complications with post-operative agents. Current evidence does not show increased flap viability with routine use of post-operative agents compared to no anti-thrombotic agents. 56,60 -64 Without data supporting its use, the common practice of using post-operative anti-thrombotic agents may be a futile endeavor to improve flap survival. Although clear recommendations cannot be made for which anti-thrombotic strategy is superior, agents such as dextran are generally to be avoided in light of the current literature.
Historically, dextran was a popular option for microsurgeons due to its anti-thrombotic effects. Complications were uncommon but could be severe such as anaphylaxis, volume overload, pulmonary edema, cerebral edema, platelet dysfunction, and renal failure. 15,37 Disa et al 53 performed a randomized prospective study comparing the use of post-operative dextran and ASA. Patients receiving dextran developed significantly more systemic complications (29%-51% depending on duration of dextran administration) compared to ASA alone (relative risk ratio [RRR]: 3.9-7.2). Despite this, a recent survey demonstrated dextran use remains quite prevalent. 65 Over 45% of microsurgeons in the United Kingdom routinely used dextran for free tissue transfers and digital replants without significant reported complications. Due to the severity of the complications and dubious benefits, however, current studies on dextran recommend against routine use. 53 -59
The current practice recommendations by Thrombosis Canada 66,67 for routine thromboprophylaxis in non-orthopedic surgery are LMWH (ie, enoxaparin 40 mg subcutaneously once daily, dalteparin 5000 units subcutaneously once daily, etc) or a direct oral anti-coagulant (ie, rivaroxaban 10 mg oral once daily, etc). Patients should start their first dose of anti-coagulant at least 0 to 2 hours before surgery and are typically continued until discharge. Anti-platelet recommendations are more diverse (ie, discontinue before minor surgery if possible, risk stratify patients with coronary artery disease, etc). For patients who present with a VTE, strategies for therapy range from using a single agent, such as a DOAC, or initiating therapy with LMWH followed by warfarin or transitioning to a DOAC. Therapeutic dosing (ie, rivaroxaban 15 mg twice daily, apixaban 10 mg twice daily, etc) is initially started and the duration of therapy is tailored to each patient. The choice of duration is balanced by the risk of having a recurrent VTE, such as patients with an unprovoked VTE, and risk of bleeding with long-term anti-coagulation. 66,67 No specific recommendations, however, have been made by Thrombosis Canada in relation to flaps.
Limitations
Our study focused on practice variations in Canada and may not be generalized to other countries due to differences in resource availability and personal preferences for anti-thrombotic regimes. Although there was a limited response rate (18%), this was within the typical response range of most surveys. Moreover, we suspect this was in keeping with the expected number of physicians who are involved in the management of flaps. 68 Lastly, limitations due to the nature of the surveys include close-ended questions, the use of an unvalidated 7-point Likert scale to understand the perceived risk factors for flap thrombosis. Reported protocols simply indicate current practice patterns and may not reflect “best practices” based on available evidence.
Ultimately, free flap failure is an uncommon event and, as a result, large numbers are required to generate sufficient evidence to guide practice. Further collaboration with thrombosis experts may be beneficial to better understand the mechanisms of thrombosis in free flaps and to understand the potential role of anti-thrombotic agents in mitigating these complications. This is especially important given the rapid development and introduction of novel agents. The large practice variation that exists highlights the need for the development of evidence-based consensus and guidelines.
Conclusion
There is a diverse number of anti-thrombotic agents available with no consensus on their efficacy for free tissue transfers. It has been over 30 years since the international survey by Davies 16 was published, and there has been little progress toward understanding the use anti-thrombotic agents to prevent flap thrombosis. Given the paucity of existing evidence, our study confirms the heterogeneous practice in the management of free flaps. Physicians should individualize their approach for each patient given the paucity of evidence for specific anti-thrombotic strategies and algorithms. Care should be taken with the routine use of dextran and aspirin post-operatively; higher rates of complications have been demonstrated in the absence of improved flap outcomes. 56 Future efforts should be concentrated on developing strategies based on evidence-based medicine and standardizing indications and dosing regimens for specific agents based on pharmacokinetics and risk.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
