Abstract
The approval of new non-insulin treatments has broadened the therapeutic arsenal, but it has also increased the complexity of choice for the treatment of type 2 diabetes mellitus (DM2). The objective of this study was to systematically review the literature on economic evaluations associated with non-insulin antidiabetic drugs (NIADs) for DM2. We searched in Medline, IBECS, Doyma and SciELO databases for full economic evaluations of NIADs in adults with DM2 applied after the failure of the first line of pharmacological treatment, published between 2010 and 2017, focusing on studies that incorporated quality-adjusted life years (QALYs). The review included a total of 57 studies, in which 134 comparisons were made between NIADs. Under an acceptability threshold of 25,000 euros per QALY gained, iSLGT-2 were preferable to iDPP-4 and sulfonylureas in terms of incremental cost-utility. By contrast, there were no conclusive comparative results for the other two new NIAD groups (GLP-1 and iDPP-4). The heterogeneity of the studies’ methodologies and results hindered our ability to determine under what specific clinical assumptions some NIADs would be more cost-effective than others. Economic evaluations of healthcare should be used as part of the decision-making process, so multifactorial therapeutic management strategies should be established based on the patients’ clinical characteristics and preferences as principal criteria.
Introduction
Diabetes mellitus type 2 (DM2) is one of the chronic diseases with the greatest impact on public health in developed countries, due to its high prevalence and associated morbidity and mortality. 1 It is estimated that diabetes accounts for 11.6% of total healthcare expenditure worldwide and for 8.2% of public health expenditure in Spain.2,3
The objectives of the treatment for DM2 are to reduce blood glucose levels to values close to normal, to prevent complications and finally to prolong survival, so adequate control of the disease is crucial. 4 That is why the clinical practice guidelines recommend starting to control the glucose levels of the newly diagnosed patient with physical exercise, changes in diet and therapeutical education, unless the patients fulfil the criteria for immediate insulinisation. If after 3 months, the disease cannot be controlled, and a pharmacological treatment should be started.5,6
In general, the starting drug of choice is metformin. 5 In cases of intolerance or contraindication, other drugs should be considered. 7 When glycaemic control is not adequate in a monotherapy regimen, in general, a dual therapy would be used, combining the pharmacological treatment of two non-insulin antidiabetic drugs (NIADs), or with one insulin, assessing also different aspects of the patient and the medication in order to decide the best therapeutic option. 6 The choice of the second drug should be made taking into account different aspects such as efficacy, risk of hypoglycaemia, effects on weight and other adverse effects, comorbidity, life expectancy and patient preferences, as well as the cost. Similarly, if adequate glycaemic control is not achieved under double therapy, it is recommended to start a triple therapy of NIADs in those patients who cannot or do not agree to receive insulinisation. Evaluating the different factors of the available therapeutical options is recommended to choose the most appropriate one in each case. 6
The drugs to be added may be sulphonylureas, glitazones, inhibitors of dipeptidyl peptidase-4 (iDPP-4), analogues of the glucagon-like peptide 1 (aGLP-1) receptor or inhibitors of the sodium-glucose cotransporter type 2 (iSGLT-2). The most recently marketed NIADs have been the iDPP-4, aGLP-1 (since 2007) and the iSGLT-2 (since 2012). All of them achieve glycaemic control similar to that of the classic drugs, but with the additional benefit of having a lower risk of hypoglycaemia and a significant loss of weight, which often results in an improvement in patient’s quality of life and a decrease in the total costs associated with the disease.8,9 However, it is necessary to analyse whether these additional clinical benefits compensate for the relatively high price of these drugs.
The introduction of new NIADs has allowed the available therapeutical arsenal to be expanded, but at the same time it has increased the complexity of choice of treatment, so it has become more difficult to know which is the optimal pharmacological intensification.10,11 Organisations such as the NICE or the American Diabetes Association have glycaemic control algorithms that try to facilitate these decisions.12,13
In the context of limited budgetary resources, prioritising the use of efficient healthcare interventions is essential for the rational use of those resources and, therefore, for the sustainability of the system. The economic evaluation is a fundamental tool for making rational decisions, which allow one to determine whether the interventions are cost-effective and whether they are worth (in terms of health) what they cost (in financial terms). Thus, with the appearance of new treatments, the economic evaluations must also be updated.
In 2009, a systematic review of economic evaluations of glucose-controlling drugs marketed in Spain was published. It concluded that all the treatments available at that time were cost-effective compared to placebo for a willingness to pay of €30,000 per year of quality-adjusted life-years (QALY) gained, with metformin being the most cost-effective treatment, so it was concluded that the second-generation oral antidiabetics should be used as a complement, and not as an alternative, to metformin. 14
Just as the clinical practice guidelines need to be updated to adapt to the availability of new evidence and the development of new treatments, it is also advisable to update the mentioned report, incorporating the economic evaluations carried out recently in the field of DM2.
The main objective of this work is thus to evaluate the efficiency of NIADs in DM2, through a systematic review of the published literature about the subject. After identifying the published articles which meet the specified inclusion criteria, we compare the results obtained by the reviewed articles and assess the quality of the evidence provided. In the discussion section, the results obtained are contextualised and some considerations of interest are detailed, as well as the potential limitations of the work. Detailed information about the studies found in the review is provided in the supplementary material.
Methods
Design
To respond to the objective of the study, a systematic review of the literature was carried out in the following stages, recommended in the ‘CRD’s guidance for undertaking reviews in health care’ of the University of York: 15 (1) search of the literature, (2) selection of studies, (3) evaluation of the quality of the studies, (4) data extraction, (5) synthesis and analysis of the data, (6) preparation of the preliminary report, and (7) preparation of the final report.
Search strategy
The search strategy took into account the following terms, in both free text and controlled language (MESH terms): ‘diabetes mellitus’, ‘DM2’, ‘type 2 diabetes’, ‘glycaemic control’, ‘HbA1c’, ‘economic evaluation’, ‘cost-effectiveness’, ‘cost-utility’, ‘cost-benefit’, ‘cost minimisation’, ‘costs’, ‘effectiveness’, ‘economics’, ‘cost-analysis’ and ‘QALY’, associating these terms with each of the names of the active principles under study (see Annex 1 in Supplemental material). The search strategy was conducted in January 2018.
The search strategy was launched in the following databases: Medline (through PubMed), SciELO Spain, Índice Bibliográfico Español en Ciencias de la Salud (IBECS), Doyma. The scientific evidence published in the indicated databases between January 2010 and December 2017 was reviewed, and no additional filter was applied.
Inclusion criteria
The inclusion criteria that were applied in the review of the literature are indicated in Table 1.
Criteria for inclusion of the systematic review of the literature.

DM2: diabetes mellitus type 2; DPP: dipeptidyl peptidase; SGLT: sodium-glucose cotransporter type; QALY: quality-adjusted life years; GLP: glucagon-like peptide 1.
Selection and synthesis
The studies were initially selected by two researchers, applying the criteria for inclusion and exclusion. Two researchers carried out, in parallel, the extraction of data about the effectiveness and costs of the selected studies, entering the information into a database specifically designed for that purpose. A synthesis of the most relevant variables was carried out by a descriptive analysis which summarises the relevant information. In order to facilitate the direct comparison of the results, a conversion of the cost components was performed to express the results in euros of the year 2017, applying the official exchange rate of the year in question and the variation in the harmonised consumer price index of Spain.16,17 A maximum cost-effectiveness threshold of €25,000/QALY gained was considered, based on the implicit threshold defined for Spain. 18
Evaluation of the quality of the studies
Simultaneously with the extraction of data, an evaluation of the quality of the selected studies was carried out, applying the methodology proposed by the University of York, 15 and following the quality scale of Drummond, which consists of 36 items. 19 Each question was evaluated, answering ‘yes’, ‘no’, ‘partly’, ‘impossible to judge’ to each of them as appropriate.
Results
Search results
The search of the literature identified a total of 601 studies. After eliminating the duplicates, the titles and abstracts of the 553 resulting articles were reviewed, from which 155 studies of potential interest were selected. After a review of their entire texts, 98 papers were excluded, for different reasons. The number of final studies included in the review amounted to 57 (Figure 1).
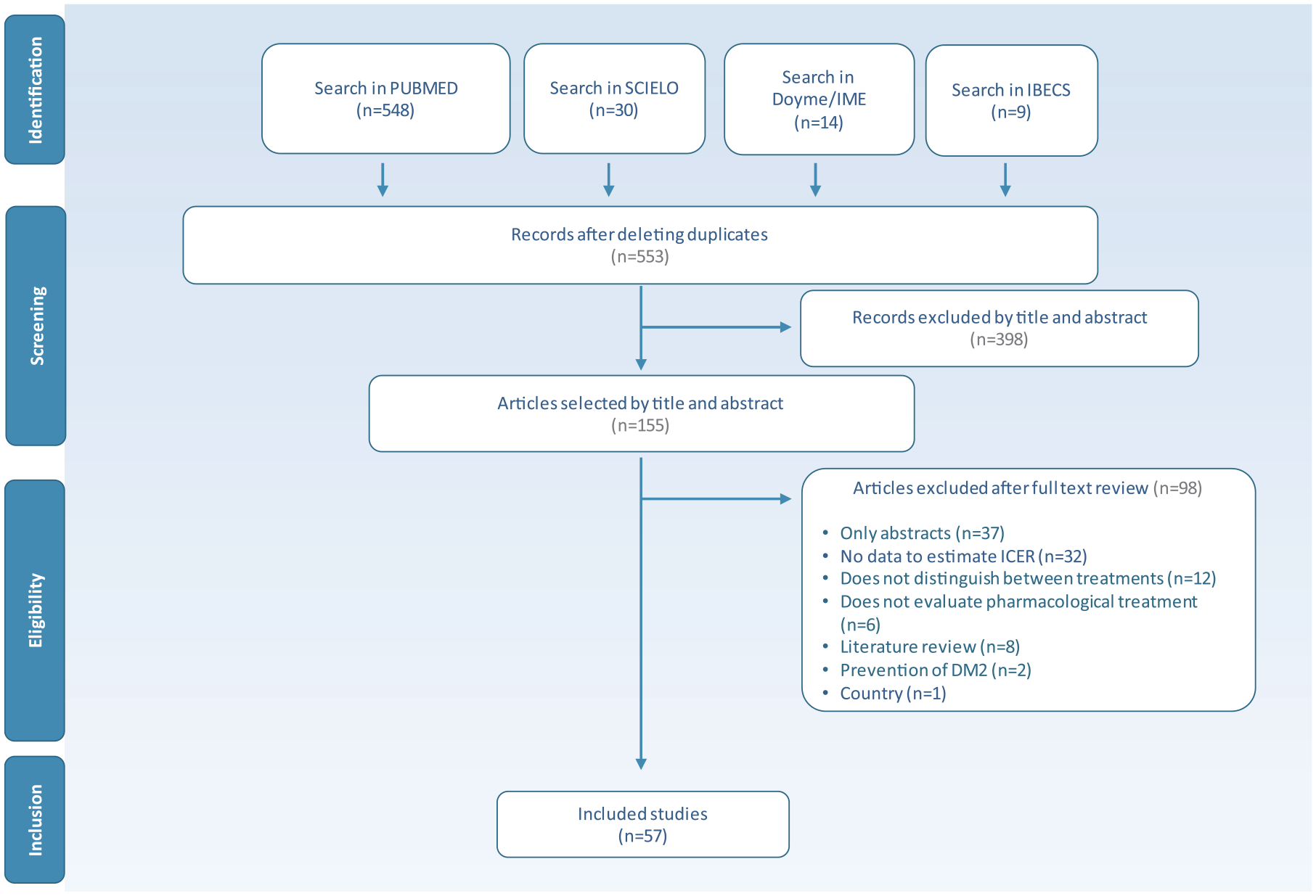
Flow diagram of PRISMA about the process of bibliographic search and selection of studies.
Description of the economic evaluations included
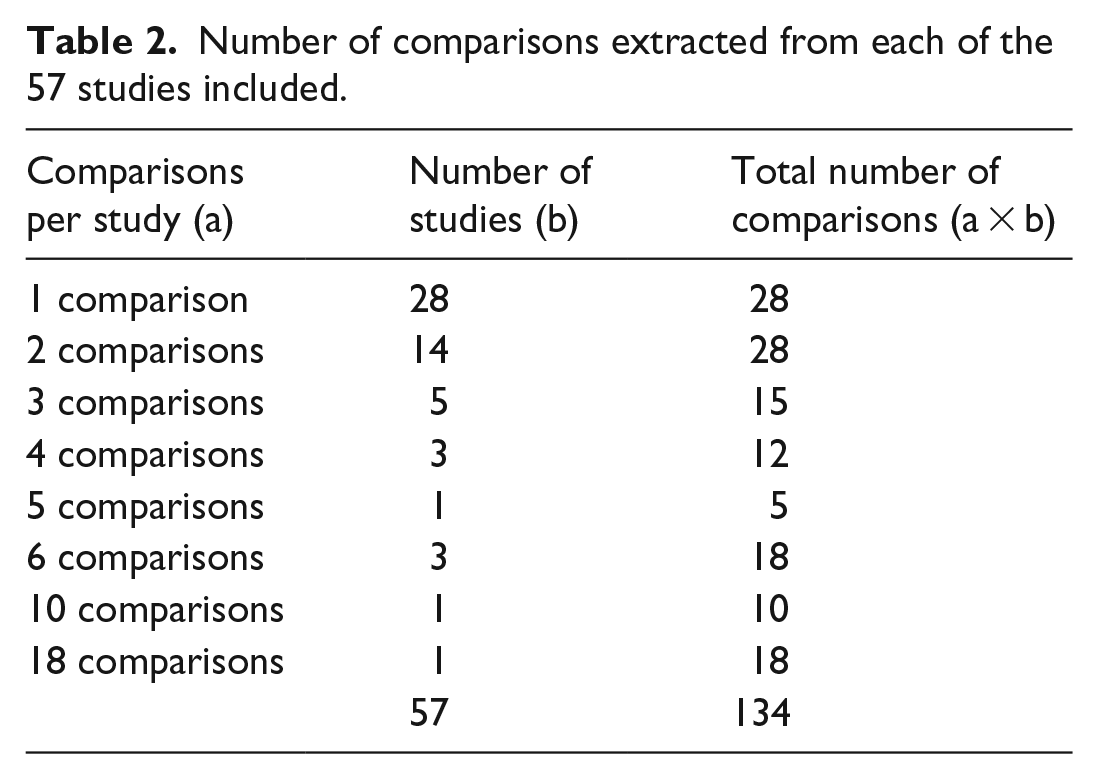
From the 57 studies found, 134 drug comparisons in which one of the NIADs under review was involved were extracted. Only 28 of the 57 studies required a single comparison between the two drugs. From the rest, 2 (n = 14), 3 (n = 5), 4 (n = 3), 5 (n = 1), 6 (n = 3), 10 (n = 1) and 18 (n = 1) comparisons per study were extracted (Table 2). The number of comparisons extracted per study depended on the number of active substances compared, but also on other variables, such as the dose applied, the country (if results are provided for three countries, three comparisons were extracted), the time horizon, the added therapy, or the type of costs included. The comparisons can be considered from the perspective of the treatment or of the comparator. Thus, there will be 134 comparisons in one way and another 134 comparisons in the opposite way. The comparisons can relate to two of the types of NIADs included or to one of them compared to another antidiabetic drug, not a subject of this review, such as acarbose, insulin or placebo. Since only those comparisons of two NIADs that were subject to this study’s review were considered twice, we finally analysed a total of 223 comparisons (Figure 2).
Number of comparisons extracted from each of the 57 studies included.
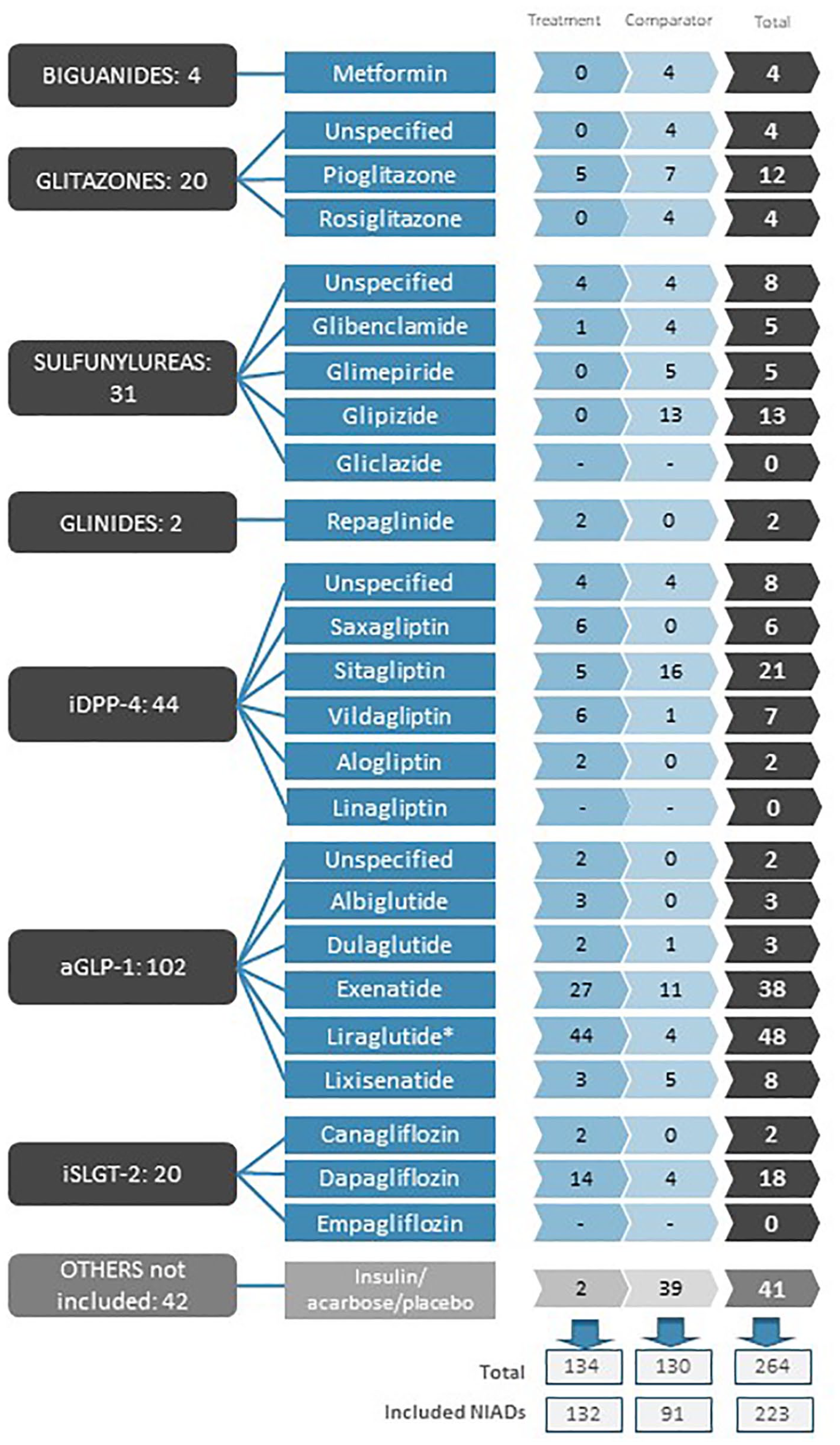
Drugs participating in the 134 comparisons drawn from the 57 studies.
In these 134 comparisons, 22 NIADs (or NIAD groups) that were the subject of this review participated. No study included gliclazide, linagliptin or empagliflozin. The aGLP-1 liraglutide was the most frequently evaluated active substance (48 times), followed by the aGLP-1 exenatide (38 times) and the iDPP4 sitagliptin (21 times). Insulin, sitagliptin and glipizide were the most commonly used drugs as comparators (37, 16 and 13 times, respectively) (Figure 2). The most frequent comparison was exenatide versus insulin (18 times), followed by liraglutide versus sitagliptin and liraglutide versus exenatide (nine times each) (Figure 3).
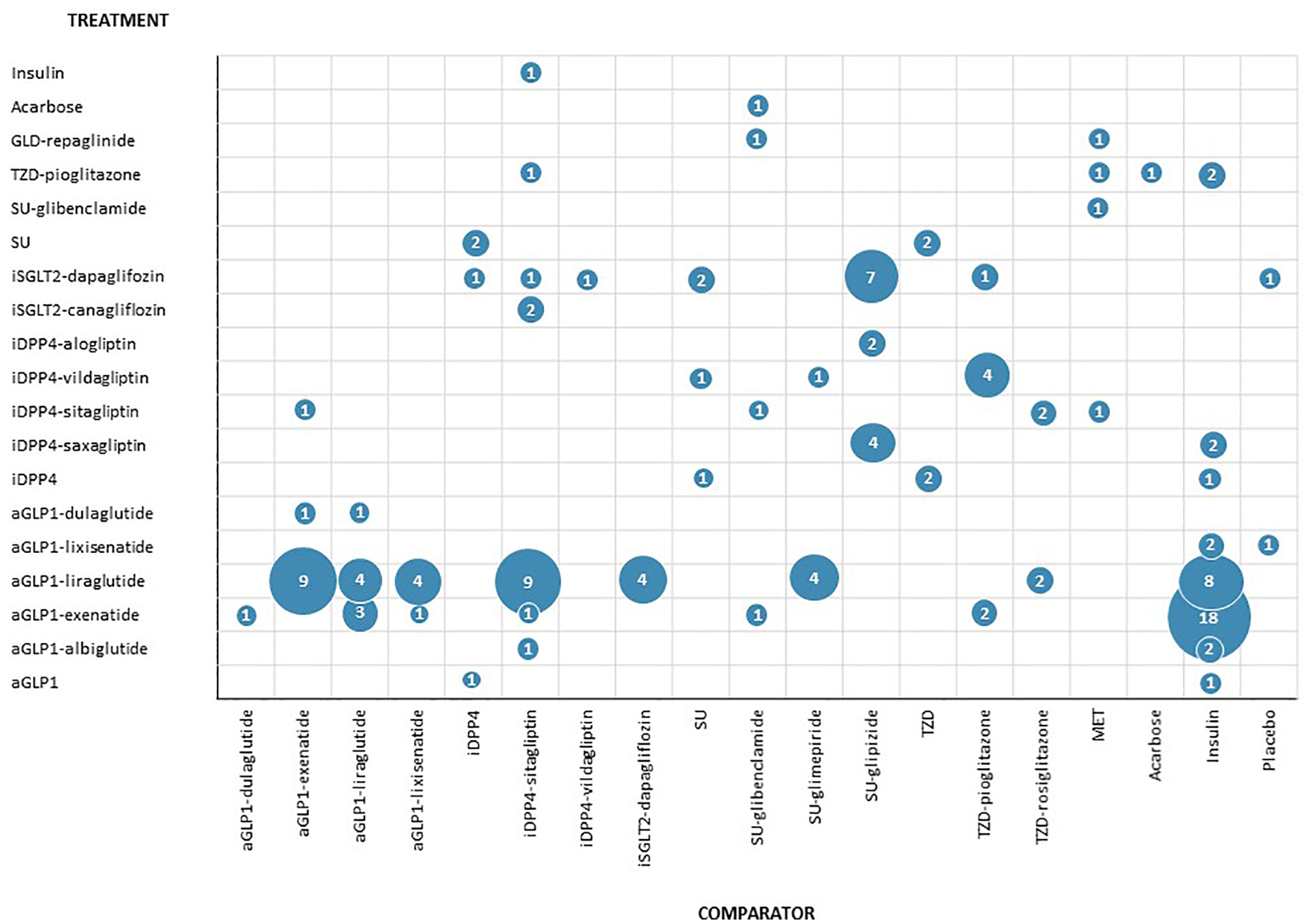
Diagram of the comparisons extracted from the 57 studies included depending on whether the drug acts as a treatment or as a comparator.
With regard to the main characteristics of the 57 studies included in the review, the following aspects should be noted (Table 3). 77% of the studies were carried out in European countries, although the United States was the predominant country of reference, with 10 studies. The perspective of the analysis most frequently used was that of the healthcare financer (in 53 studies), while four of them considered the social perspective, that is, they included both direct and indirect costs.20–23 In 88% of cases, baseline data about patients and treatment efficacy came from randomised clinical trials: 35 studies used data from a unique clinical trial (17 different trials), 4 studies were anchored on various trials (n = 4) and 11 studies used information arising from a literature review or meta-analysis (n = 11). The remaining 12% of cases came from observational studies (n = 4) or from patients’ databases (n = 3).
General characteristics of the 57 studies included in the review.
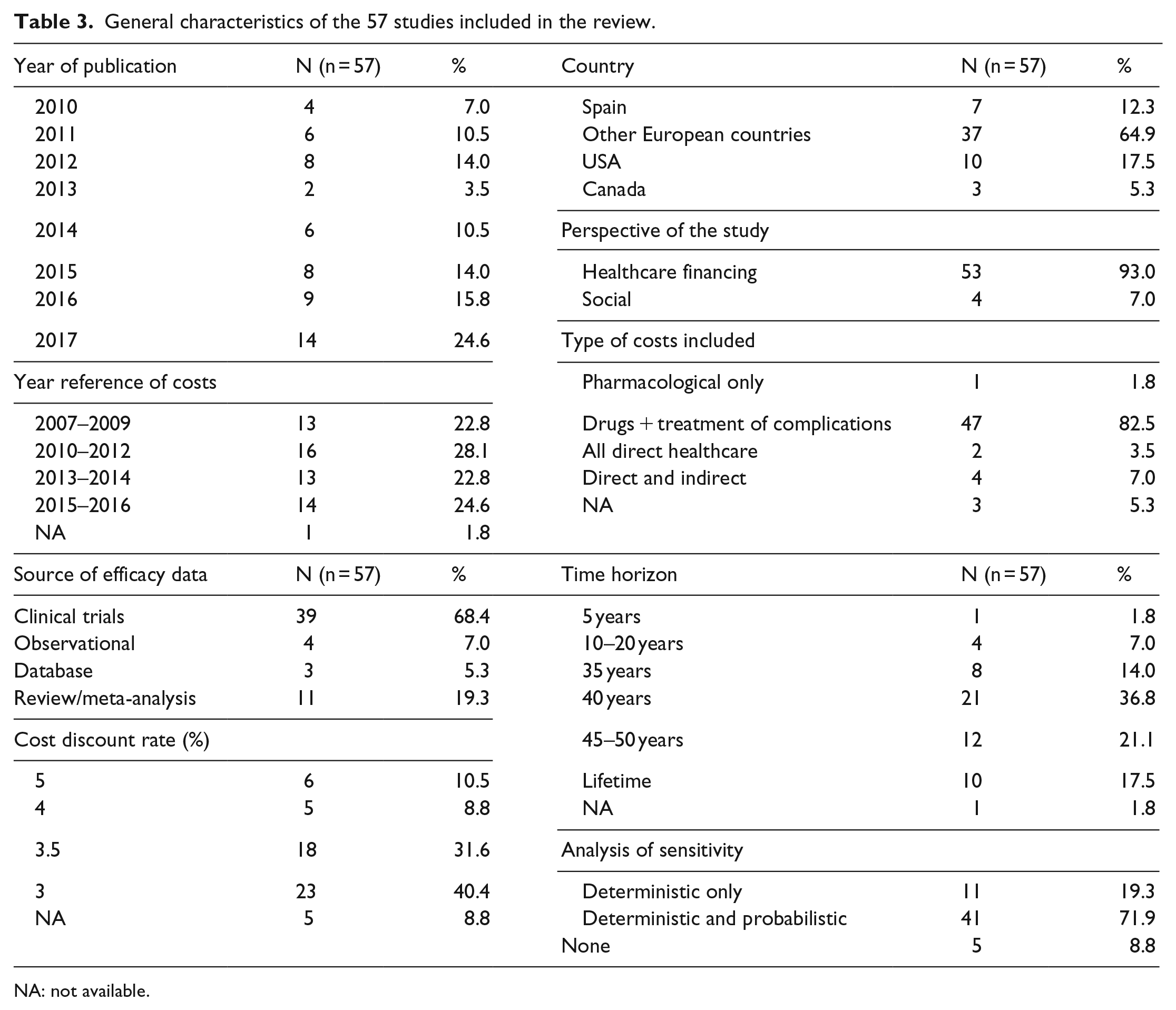
NA: not available.
The most frequently used time horizon was 40 years (n = 21), followed by between 45–50 years (n = 11) and 35 years (n = 8). The most used simulation models were the CORE Diabetes Model (n = 25) and the Cardiff Diabetes Model (n = 16). 52 of the 57 studies discounted the results, and only two of them applied a discount rate to the costs that were different from that applied to the clinical benefits.24,25 The most common discount rate was 3% (n = 23), followed by 3.5% (n = 18). 91% of the studies performed a sensitivity analysis on the results (n = 52). In 11 studies, the analysis was only deterministic, and in the other 41 studies, it was both deterministic and probabilistic.
Regarding the characteristics of the clinical trials used as a source of efficacy data, and the baseline characteristics of the patients included, the following points should be noted (Table 4). The duration of the trials ranged between 18 weeks and 2 years, the most common duration among the comparisons was 26 weeks (6 months) (n = 40). The sample size of the clinical trials varied considerably. The average age of the subjects included in the trials was 57.2 years, and most of the comparisons (n = 94) were based on trials whose subjects had an average age between 55 and 60 years. The average duration of diabetes type II was 6–7 years (n = 52). Diabetes was newly diagnosed only in three of the combinations extracted, while in 17 combinations the patients had had the disease for more than 10 years. 9% of the studies were based on trials conducted on a sample of overweight patients, while in 77% of the comparisons made, the sample of patients had an average body mass index (BMI) of more than 30 (obesity). Sadly, the high heterogeneity of the studies, as well as the limitations in the information offered, prevented us from offering a quantitative synthesis of the studies or a global summary measure.
Duration and sample size of clinical trials.
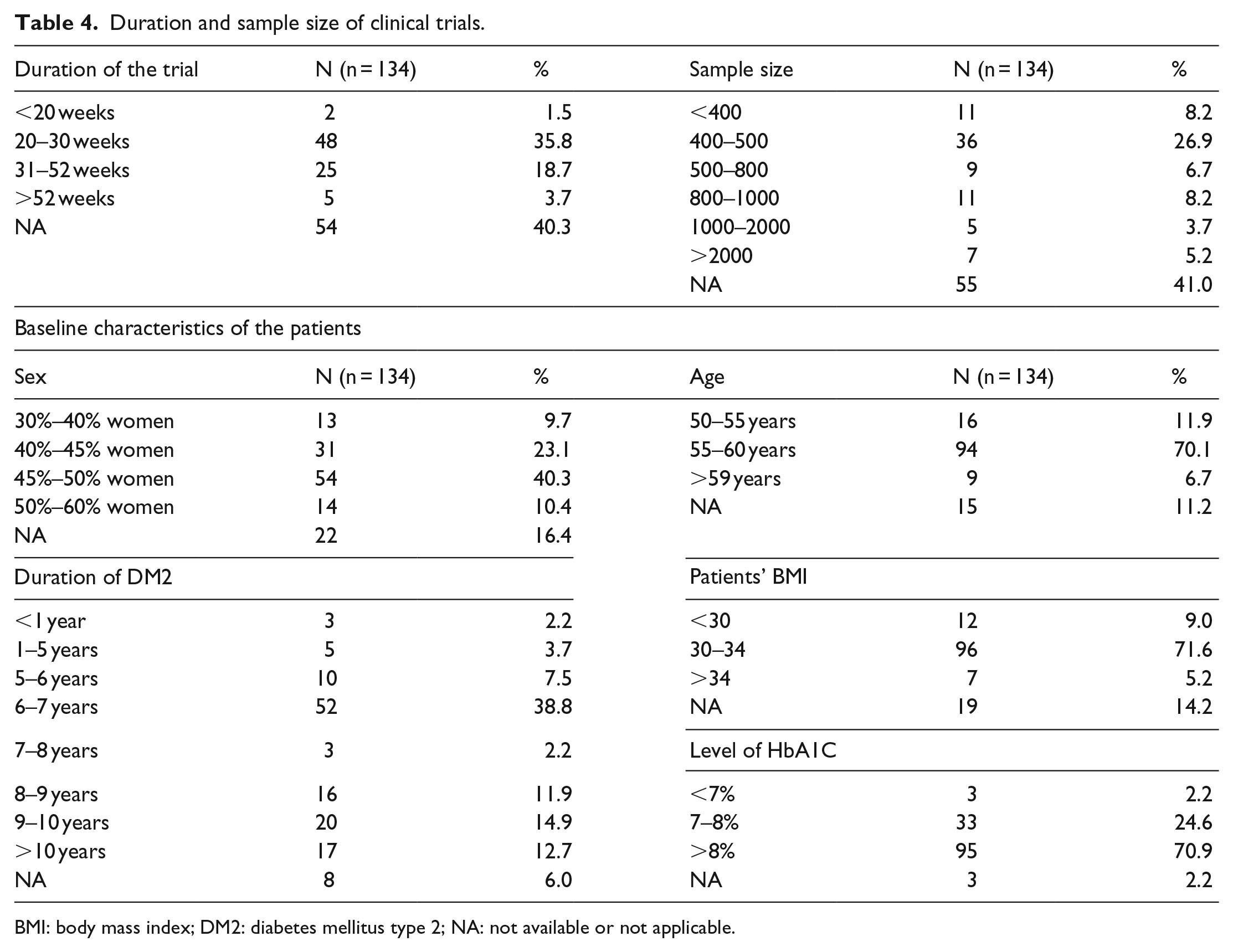
BMI: body mass index; DM2: diabetes mellitus type 2; NA: not available or not applicable.
Quality of the studies
The quality of the economic evaluations found, which was evaluated using the Drummond checklist, 19 was acceptable (Figure 4). The aspects most appropriately collected in the studies were the research question (P1.), the answer to the question of the study (P33.) and the derivation of conclusions (P34.), while the most common problems found, were those related to the details of the statistical tests and confidence intervals (P26.), the justification of the choice of the discount rate (P24.), the questions of generalisation (P36.) and the justification of the model used and its key parameters (P21.).
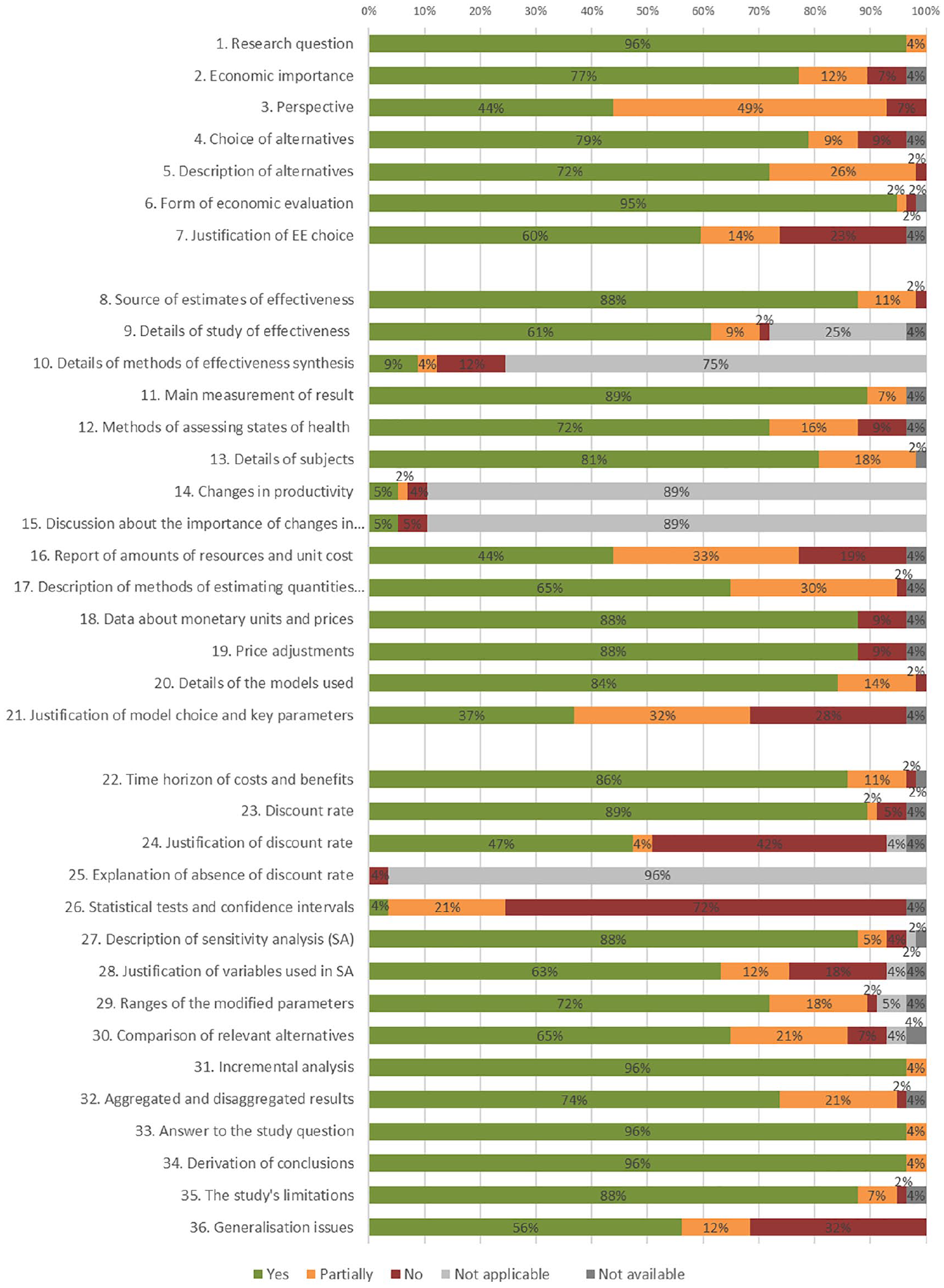
Results of the evaluation of the methodological quality applied in the studies included in the review.
Efficiency of non-insulin antidiabetic treatments
Table 5 summarises the efficiency results obtained for each comparison included in this review, considering a cost-effectiveness threshold of 25,000 euros per additional QALY gained. The detailed information contained in each study can be found in Table 6.
Summary of the results of the economic evaluations included in terms of incremental cost per QALY gained for the NIADs (threshold €25,000/QALY).

QALY: quality-adjusted life years; NIADs: non-insulin antidiabetic drugs; SGLT: sodium-glucose cotransporter type; DPP: dipeptidyl peptidase; GLP-1: glucagon-like peptide 1; TZD: thiazolidinedione; NPH: neutral protamine Hagedorn.
Results of the economic evaluations found in the NIAD under review.
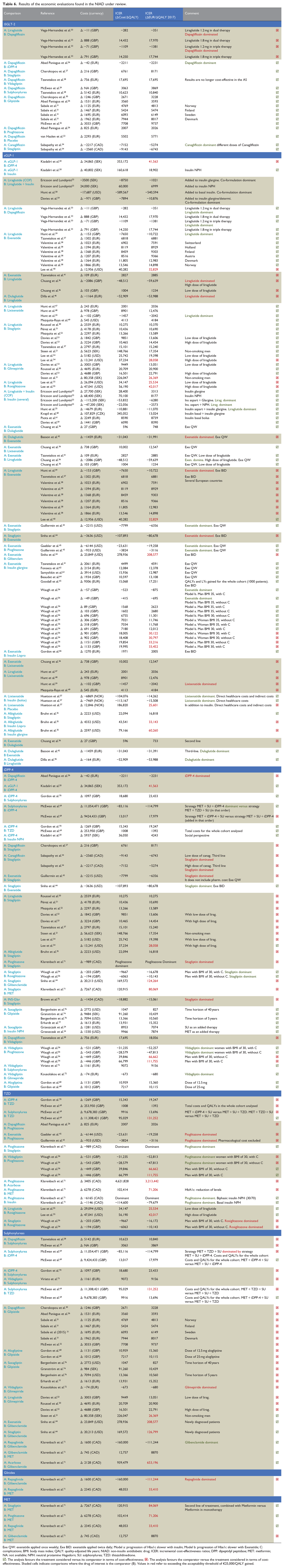
Exe QW: exenatide applied once weekly; Exe BID: exenatide applied twice daily; Model a: progression of Hba1c slower with insulin; Model b: progression of Hba1c slower with Exenatide; C: complications; BMI: body mass index; QALY: quality-adjusted life years; NIAD: non-insulin antidiabetic drug; ICER: incremental cost-effectiveness ratios; DPP: dipeptidyl peptidase; MET: metformin; NA: not available; NPH: neutral protamine Hagedorn; SU: sulphonylurea; TZD: thiazolidinedione.
☑: The analysis favours the treatment considered versus its comparator in terms of cost-effectiveness; ☒: The analysis favours the comparator versus the treatment considered in terms of cost-effectiveness. Shaded cells indicate comparisons where the drug of interest is the comparator (B). Values in red refer to exceeding the acceptability threshold of €25,000/QALY gained.
Based on the results found, the inhibitors of SGLT-2 (dapagliflozin and canagliflozin) were preferable, in terms of cost-effectiveness, to the iDPP-4, the sulphonylureas and pioglitazone.22,26–34 However, the same cannot be said about their superiority over the aGLP-1 (Table 5, Figure 5). Specifically, the iSGLT-2 participated in 20 of 223 comparisons included (18 for dapagliflozin and 2 for canagliflozin), which were extracted from 10 economic evaluations. No comparison was found between these two iSGLT-2, nor any for empagliflozin. Dapagliflozin was a more efficient option than the iDPP-4: it was dominant (less expensive and more effective) versus the group of iDPP-4 in general; 26 and cost-effective versus sitagliptin 27 and vildagliptin, 28 with incremental cost-effectiveness ratios (ICERs) of €8000 and €17,700 updated to 2017 per QALY gained, respectively. Dapagliflozin was also cost-effective versus the sulphonylureas, both at the group level28,29 and versus glipizide26,30–32 and versus pioglitazone (TZD) (ICER of €2.000/QALY). 26 However, in the four comparisons with liraglutide, dapagliflozin was a dominant or non-cost-effective option. 33 In addition, in the only two comparisons found for canagliflozin, from the same study in which different doses of this NIAD were evaluated versus sitagliptin in the third line of treatment, canagliflozin was a dominant treatment option when compared with this iDPP-4. 34
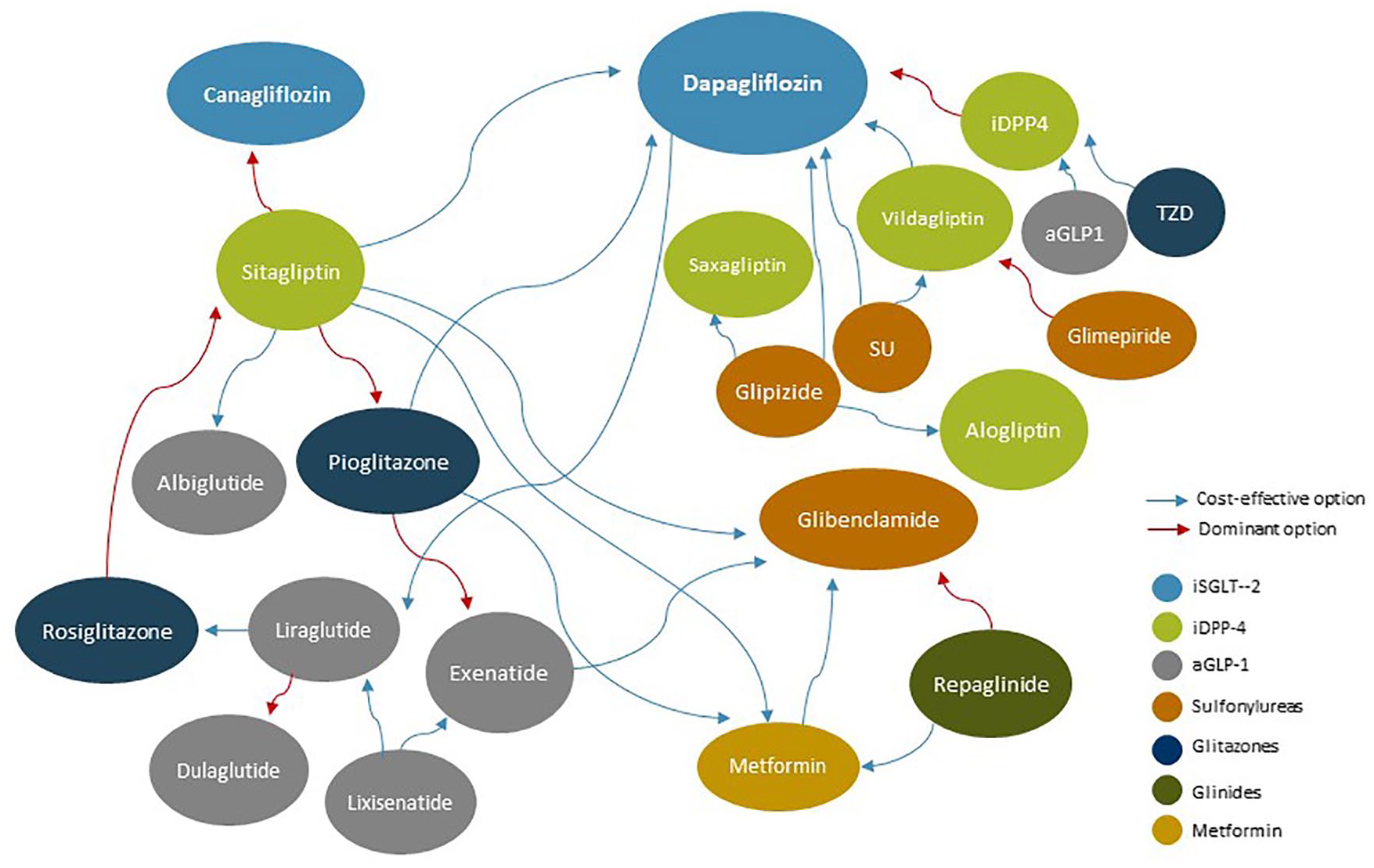
Summary of cost-effectiveness results found in the systematic review of literature 2010–2017, under the threshold of €25,000/QALY gained, by NIAD groups.
The results were not conclusive for the aGLP-1.20,21,23,25,33,35–66 Albiglutide appeared to be a more cost-effective option than sitagliptin, 35 but the results were the opposite when comparing the general subgroups to which these NIADs belonged (aGLP-1 versus iDPP-4). 20 Exenatide was a more cost-effective treatment than another analogue such as lixisenatide (ICER of €12,600/QALY), 36 but no conclusive results were obtained in comparison with other analogues such as liraglutide,36–41 or dulaglutide.36,42 Nor were the results conclusive for sitagliptin, where converse results were obtained.43,44 Exenatide was a dominant option over pioglitazone in the two studies that analysed this comparison,43,45 but it was not cost-effective compared with a sulphonylurea such as glibenclamide. 44 In terms of cost-effectiveness, the results for liraglutide were favourable versus dapagliflozin, 33 but the results compared with other aGLP-1 were inconclusive: it was a dominated option versus dulaglutide 46 and cost-effective or dominant versus lixisenatide,47–49,67 but with divergent results versus exenatide.36–41 Liraglutide was cost-effective versus sitagliptin (iDPP-4) in 8 of the 9 comparisons found,21,37,50–54 and was a cost-effective option versus glimepiride in 3 of the 4 comparisons,21,50,53 but not versus rosiglitazone. 55 Lixisenatide did not appear to be a more cost-effective option than other analogues such as exenatide 36 or liraglutide.39,47–49 Dulaglutide was dominant over liraglutide, 46 but contradictory results were obtained versus exenatide.36,42
For the iDPP-4, no conclusive results were obtained,20,21,24,26–28,34,35,37,43,44,50–54,56,68–77 except when they were compared with the iSGLT–2. In this case, their results were favourable in all comparisons made at group and individual level.26,28,30,34 At the group level, the iDPP-4 were more cost-effective than the aGLP1 20 and glitazones.69,70 However, a specific iDPP-4 such as sitagliptin was a non-cost-effective option compared with an aGLP1 such as albiglutide 35 and there were contradictory results for the glitazones, which dominated rosiglitazone, 56 but were dominated by pioglitazone.35,71 When these NIADs were compared with the sulphonylureas, there were again no clear results: at the group level, the results were inconclusive;69,70 saxagliptin and alogliptin were more cost-effective than glipizide,68,78 and vildagliptin was cost-effective versus glimepiride 72 and versus sulphonylureas in general; 73 but sitagliptin did not appear to be cost-effective compared with a sulphonylurea such as glibenclamide used in the second line. 44 The only iDPP-4 which was compared with an aGLP-1 was sitagliptin, but no conclusive results in only one way were obtained: it was not cost-effective versus albiglutide 35 and there were converse results versus exenatide43,44 and liraglutide.21,37,50–54
Nor were the results conclusive for the glitazones.26,43,45,55,56,69–71 At the group level, glitazones were a non-cost-effective option compared with the group of iDPP-4,69,70 and rosiglitazone was an option dominated by sitagliptin, 56 but pioglitazone was dominant over sitagliptin, 71 and there were conflicting results in the comparison with vildagliptin. 56 Results were not conclusive in the comparison with the sulphonylureas group, results being obtained in both ways.69,70 When compared with the aGLP-1, pioglitazone had unfavourable results compared with exenatide,43,45 whereas rosiglitazone was a cost-effective option compared with liraglutide under the threshold considered. 55 In the only comparison found with an iSGLT-2, dapagliflozin was more cost-effective than pioglitazone. 26
Seen in the opposite way, it was not possible, either, to establish a clear option for sulphonylureas based on their efficiency,21,26,28–32,44,50,53,68–74,76,77 with the exception of dapagliflozin, which was more cost-effective than sulphonylureas in all the comparisons found.28,29 In contrast, the results were not conclusive either versus the group of iDPP-470,73 or versus the group of glitazones. 69 Glibenclamide was a cost-effective option compared with sitagliptin, 44 but glipizide was non-cost-effective compared with other iDPP-4 such as saxagliptin and alogliptin,68,74,78 and glimepiride was dominated by vildagliptina. 72 Regarding the aGLP1, glibenclamide was cost-effective compared with exenatide, 44 but there were no conclusive results for glimepiride versus liraglutide,21,50,53 regardless of the dose applied. Glibenclamide was a dominant option over repaglinide and more cost-effective than metformin. 71
Discussion
Economic evaluation is a tool that facilitates complex decision-making. In the field of DM2, with the introduction of new therapeutic alternatives, often safer and more effective, but also more expensive than the previous ones, it has become more difficult to know which is the optimal pharmacological intensification. In this sense, this systematic review of the literature aims to help determine which NIADs are the most efficient in each case. To do so, we updated an earlier review, carried out in 2009, with the new evidence available, compiling the most recent economic evaluations and comparing the results obtained.
The description and analysis of the results were carried out by making comparisons among drugs, and not only among studies. To facilitate comparability, the incremental cost-utility ratios were updated to euros for 2017, and the implicit threshold of acceptability most recently published for Spain was used. 18 The search of the literature yielded a total of 57 economic evaluations published in the last 8 years, from which it was possible to extract 134 comparisons for the included non-insulin antidiabetics. The results show the growing interest in economically evaluating the different NIADs. It seems that the debate about the first line of pharmacological treatment has already been overtaken, and the focus has shifted to an evaluation of the different NIADs among themselves in the second and third lines.
In this review, the only NIADs for which conclusive favourable results were obtained in terms of incremental cost-utility seem to be the iSGLT-2 versus the iDPP4, sulphonylureas and glitazones. In this regard, the literature has shown the usefulness of this type of NIADs among patients with long-term diabetes.26,34 In addition, the most recent NICE guidelines recommend, as the first treatment intensification, the addition of an iDPP-4, pioglitazone or a sulphonylurea to the metformin, opening the possibility of prescribing an iSGLT-2 in certain circumstances. 79 In particular, the British agency states that iSGLT-2 can also be considered an alternative treatment in patients with inadequate glycaemic control, especially if they are overweight because they are associated with a loss of weight. Nevertheless, they should be prescribed with caution in patients with impaired renal function and propensity to suffer urinary tract infections.80–82 In triple therapy, the NICE places at the same level of recommendation as insulinisation, the adding of pioglitazone or an iDPP-4 to metformin-sulphonylurea, and keeps the aGLP-1 as a second option, while considering the use of iSGLT-2 as a third-line treatment option, either in combination with metformin, a sulphonylurea or a glitazone.81–83
When interpreting and contextualising the results of this review, certain considerations should be taken into account. First, the inclusion criteria are limited to studies published from 2010 onwards, so the conclusions apply specifically to those studies, which have been mainly performed with the most recently marketed drugs. This means that there is no general view of the cost-effectiveness of all available drugs; therefore, it is difficult to derive generic recommendations for use.
Second, the interpretation of the results obtained in terms of efficiency depends directly on the threshold of acceptability considered. Some countries have an explicit threshold, but this is not the case in Spain, so the maximum implicit threshold of acceptability published most recently for our country has been used: between €21,000 and €24,000/QALY gained.18,84 Consequently, the conclusions obtained under this threshold will differ from the results of each study, which will take into account the threshold that applies to each area. Likewise, our conclusions could vary if the results obtained were considered under the prism of a substantially different alternative threshold.
Third, there is a high degree of heterogeneity in the methodology used in studies that evaluate the efficiency of these drugs, which makes it difficult to compare results and extrapolate conclusions. The studies use different sources of information, modelling of the disease, types of costs, discount rates, time horizons, baseline characteristics of patients, treatment intensification thresholds, sensitivity analyses and so on. The high degree of heterogeneity of the studies, together with the limitations in the information offered, prevented the formal estimation of an overall summary measurement through a meta-analysis.
Fourth, economic evaluations suffer from certain limitations. Data relating to effectiveness are often lacking, and efficacy data are only available from old clinical trials conducted in a given context and group of patients. 85 Models, risk equations and assumed utilities were often developed for a different context and time, without necessarily being validated within the scope of the study. Likewise, there is uncertainty about the efficacy data and the costs of treatment in the medium/long term. 67 The lack of information is alleviated by different assumptions, extrapolations and indirect evidence.27,57
Fifth, it is necessary to pay attention to the potential publication bias in the studies analysed. Publication bias appears because the studies published are usually those which are in favour of experimental treatment instead of control treatment.86,87 This and other biases (selection, implementation, detection, attrition and/or notification, 88 quantifiable by different techniques, often come from the clinical trials on which efficacy and safety results are based, and which subsequently inherit the economic evaluations that are based on them. It should be mentioned that the good methodological practice of clinical trials and economic evaluations is the way to contain and manage the appearance of biases.
Finally, our study is not without limitations either. In systematic reviews, there is the possibility that the strategy has not been sufficiently sensitive when identifying relevant studies to answer the research question. However, a structured search approach has been followed so that the results are replicable. In addition, the cost-effectiveness ratios of the comparisons made, do not always appear in the studies but have sometimes been derived from the disaggregated results. In addition, the heterogeneity of the studies and the potential publication bias limit the external validity of the results, which in turn directly depend on the cost-effectiveness threshold considered. Finally, there are some NIADs for which no new evidence was found, as in the cases of gliclazide, empagliflozin, and linagliptin.
In conclusion, under an acceptability threshold of €25,000/QALY, the only NIADs for which conclusive favourable results seem to be obtained in terms of efficiency appear to be the ones most recently marketed, namely, the iSGLT-2 versus iDPP4, sulphonylureas and glitazones. However, these conclusions should be viewed with caution, since the heterogeneity between studies and results makes it difficult to draw unambiguous conclusions about the cost-effectiveness of the various NIADs, or to determine under what specific clinical conditions some non-insulin antidiabetics would be more effective than others. Also, the number of economic evaluations published about the iSGLT-2 is still short, and there is uncertainty about their safety and effectiveness in the medium and long term, so it does not seem appropriate to extrapolate the results to a generic recommendation of use. Some agencies warn about the increased risk of diabetic ketoacidosis, fractures, amputations and genitourinary infections79,89–91 associated with iSGLT-2.
This study aims to be one additional supportive tool in healthcare decision-making, but we must not lose sight of the fact that clinical criteria should always be the basis for deciding the most appropriated individualised treatment for each patient at each moment, based on their clinical characteristics and their preferences, in order to optimise the effectiveness of the treatment, but also the costs associated with the disease throughout the patient’s life. 5
In the future, it would be desirable to carry out more clinical trials and economic evaluations adapted to current clinical practice, as well as to limit to the highest extent the appearance of possible biases which may compromise the internal and external validity of the results.
Supplemental Material
Supplemental material for A systematic review of economic evaluations in non-insulin antidiabetic treatments for patients with type 2 diabetes mellitus
Supplemental material, Supplemental_Material for A systematic review of economic evaluations in non-insulin antidiabetic treatments for patients with type 2 diabetes mellitus by Néboa Zozaya, Margarita Capel, Susana Simón and Alfonso Soto-González in Global & Regional Health Technology Assessment
Footnotes
Acknowledgements
The authors want to thank Jhon Galindo (Weber consultant) for the technical help performed with the figures and tables included in the article.
Author contributions
N.Z. developed the design and implementation of the review, the analysis of the results and the writing of the manuscript; A.S.G. provided a clinical review of the work. A.S.G., M.C. and S.S. helped to interpret the data and contributed to the final version of the manuscript. All authors approved the final version to be published.
Availability of data and materials
The datasets used during the current study are available from the corresponding author on a reasonable request.
Ethical approval
As a systematic review, this work did not require any Ethics Committee approval.
Human rights
This work did not involve human subjects.
Informed consent
As a systematic review, this work did not require any informed consent. No data of patients are shown.
Declaration of conflicting interests
M.C. and S.S. are employers of AstraZeneca. A.S.G. received funding for the clinical review of this article. N.Z. is employee of Weber, a consultancy firm which received research funding from AstraZeneca to conduct the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by AstraZeneca Spain.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
