Abstract
Background: Late state colorectal cancer treatments have important side effects that should be avoided in patients where drug effectiveness is not adequate. PrediCTC is a new biomarkers blood test developed to determinate the chemotherapy response in unresectable metastatic colorectal cancer patients that could allow to obviate unnecessary treatments. Aim: To assess from the Spanish Societal Perspective the cost-utility of the test PrediCTC compared to the computed tomography in aim to evaluate chemotherapy treatment response in late stage colorectal cancer patients. Methods: Based on the results of Barbazán et al., a Markov model has been developed, in which the different lines and cycles that the colorectal patient can receive and how they can move between them according to the computed tomography or PrediCTC have been represented. The effectiveness has been expressed in quality adjusted life years (QALYs), avoiding adverse events. Results: Base case analysis shows savings in different types of costs for PrediCTC (per patient): €14.30 in those arise from adverse events, €22,345.73 in chemotherapy costs, €4849.61 in other direct costs, and €306.21 in indirect costs. Although computed tomography 12-week assessed patients gain 0.17 QALYs compared with PrediCTC. Conclusions: From the Spanish Societal Perspective, PrediCTC is not a cost-utility option but allows to identify earlier patients who are not benefiting from first-line chemotherapy avoiding unnecessary side effects and costs.
Introduction
Colorectal cancer is the fourth cause of cancer-related death and the metastasis appearance leads to a significant survival rate reduction. 1 When the disease is unresectable, the therapeutic options are limited to systemic treatments with chemotherapy which are not always well tolerated by the patients. Being able to perform a suitable monitoring treatment response in these patients allows us to avoid administering treatments with notable side effects which are not producing any benefits to the patient. 2
The tumor burden, metastases location, and treatment response are currently assessed with computed tomography (CT) scans according to the RECIST criteria (Response Evaluation Criteria in Solid Tumors). Ramón Domínguez Foundation has developed a specific epithelial and mesenchymal tissue marker panel from peripheral blood (GAPDH, VIL1, CLU, TIMP1, LOXL, ZEB2), to be able to predict with greater accuracy the first-line chemotherapy treatment response in patients with metastatic colorectal (PrediCTC). 2
Tumor cells detection in blood (CTC) via the Cell Search™ System has already been assessed by the Agency for the Evaluation of Health Technologies of Andalusia (AETSA) in its emerging technologies observatory as metastatic breast cancer patients monitoring tool, but the comparative cost-effectiveness analysis with the current clinical practice was not included in the report and other specific colorectal cancer liquid biopsy treatment monitoring tool has not been evaluated in Spain. 3 The main objective of our study is to carry out a cost–utility assessment of PrediCTC compared to CT monitoring the first-line chemotherapy response in patients with unresectable metastatic colorectal cancer from the Spanish Societal Perspective.
Methods
Study design
The model developed for this economic assessment takes as a reference the one published by the National Institute for Health and Care Excellence (NICE) created to evaluate the KRAS gene mutation detection test and the 2014 Metastatic colorectal cancer: ESMO (European Society for Medical Oncology) Clinical Practice Guidelines for the diagnosis, treatment, and follow-up (Figures 1 and 2).4,5
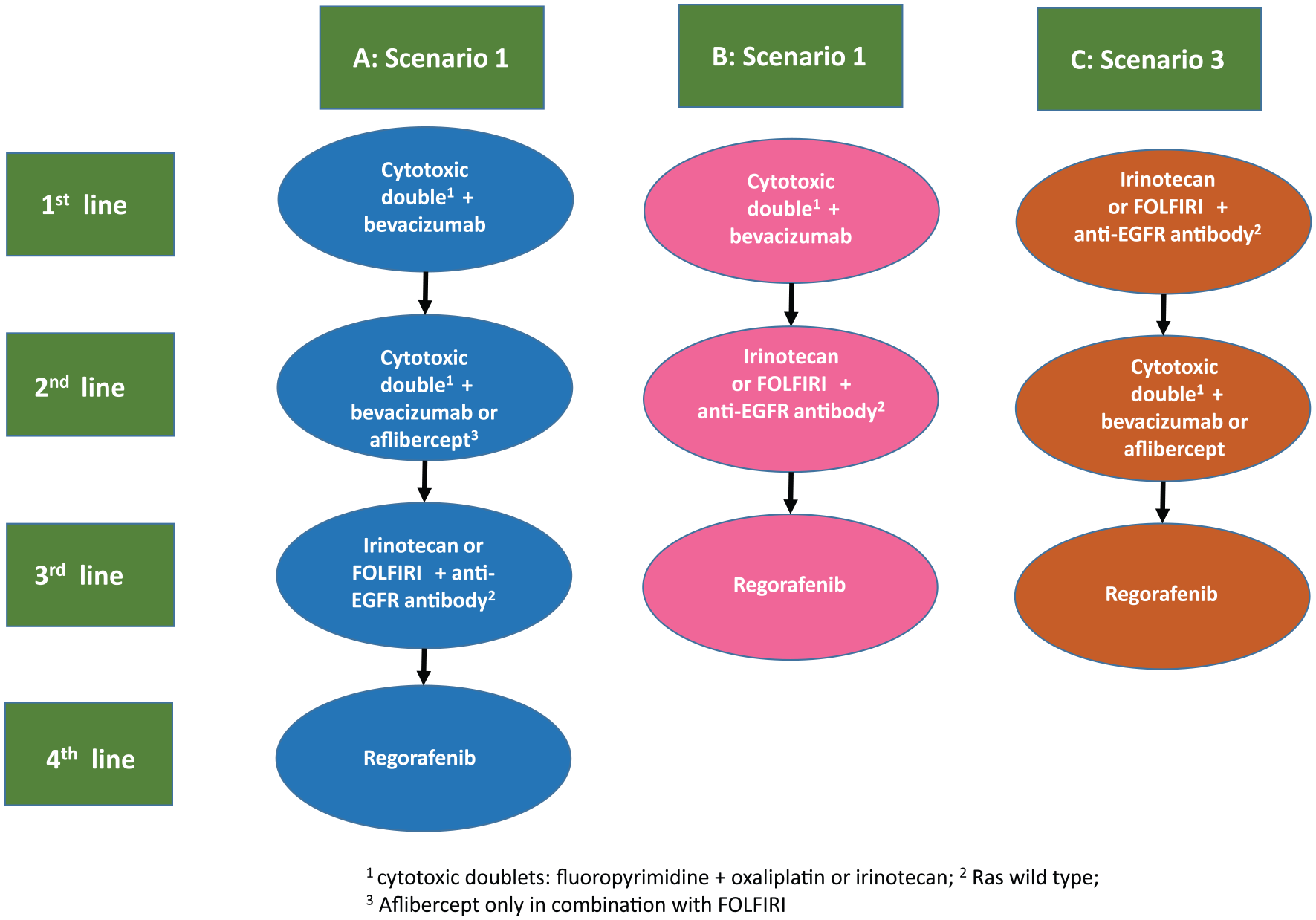
Strategic scenarios in the continuum of care of metastatic colorectal cancer from “2014 Metastatic colorectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow up.” 5
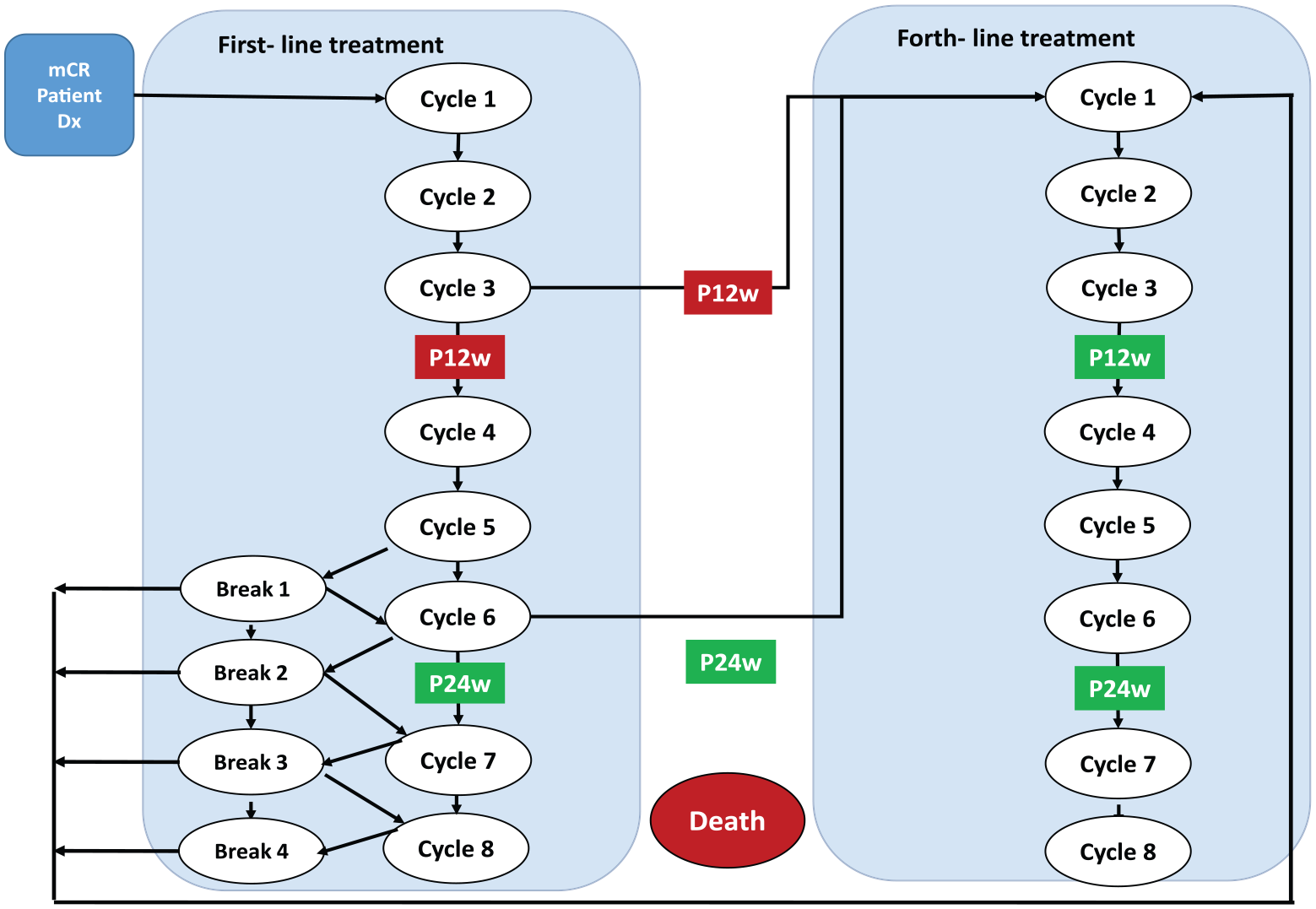
Markov model diagram where CT is used as cancer monitoring chemotherapy response tool every 12 weeks (three cycles) in all the treatment lines and through PrediCTC or CT after the first three chemotherapy cycles in the first treatment line.
With a lifetime horizon (5 years were modeled using 240 cycles) based on the clinical trials survival data and the Barbazán et al. 2 (Tables 1 and 2) and the expert panel consensus. 2 Figure 2 represents the model developed for this study where we have represented the four treatment lines available for these patients. Each treatment line has eight possible health states called “cycles,” each cycle lasting 4 weeks. After cycle 3 and cycle 6 in all the treatment lines, the patients chemotherapy response will be assessed by a CT, except in the first-line treatment in which the patient after cycle 3 has two options: be evaluated through CT or PrediCTC. Also, the first line differs from the other treatment lines because we have the “Break” state, lasting 4 weeks, where patients are in remission without treatment and after which they will continue in the same line. Finally, we have the absorbing state of “Death” where patients cannot move for any other state.
Comparison of therapy response-based patient classification between PrediCTC and first computed tomography evaluation.
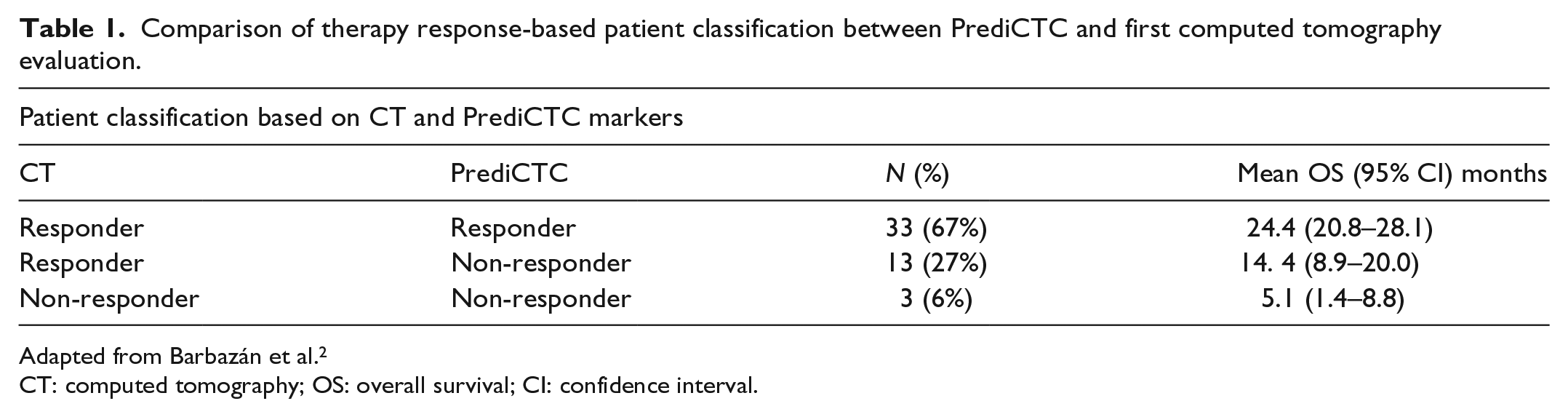
Adapted from Barbazán et al. 2
CT: computed tomography; OS: overall survival; CI: confidence interval.
Therapeutic regimens and OS (overall survival) and PFS (progression-free survival) Kaplan–Meyer curves digitized references.

BST: best support treatment.
Arrows represents possible transition at the end of every 4 weeks during the OS since the diagnostic. For example, first-line treatment possible transitions:
Patients after Cycle 1 can move to Cycle 2 same line or Death state.
After Cycle 2 patients can move to Cycle 3 same line or Death state.
After Cycle 3 patient’s treatment response will be evaluated through CT or PrediCTC moving to Cycle 4 same line if he is a responder, Cycle 1 second treatment line if he is a non-responder or to Death state.
After Cycle 4 patients can move to Cycle 5 same line or Death state.
After Cycle 5 patients can move to Cycle 6, Break 1 or Death state.
After Cycle 6 patients treatment response will be assessed through CT moving to Cycle 7 or Break 2 if he is responding, to Cycle 1 next line if he is not responding or to the Death state.
After Cycle 7 patients can move to Break 3, Cycle 8 or Death state.
After Cycle 8 patients can move to Break 4, next line Cycle 1 or Death state.
Alternatives to compare and efficacy estimation
The alternatives to be compared are as follows:
Treatment assessment response monitored with CT every 12 weeks.
Treatment assessment response monitored with PrediCTC after “Cycle 3” first-line treatment and from that with CT scan every 12 weeks.
Computed tomography transition probabilities used are those drawn from the main clinical trials of the different treatments assigned to the patients. These four lines chemotherapy transition probabilities were obtained with the Plot Digitizer program, digitizing progression-free survival (PFS) and overall survival (OS) Kaplan–Meyer curves of the randomized clinical trial published. All the figures digitized to obtain the data were adjusted with a minimum R2 of 98% and some of them were divided into two to better adjust the final periods. Therapeutic schedules and distribution of the patients were defined by the adjustment of the 2014 ESMO Metastatic Colorectal Cancer Guidelines to the daily practice of the Compostela University Hospital Oncology Service.
The PrediCTC test transition probabilities were drawn from the study carried out at the Translational Medical Oncology Unit from the Health Research Institute, 2 Table 1, which indicates that in week 16, out of 100% of the patients classified as treatment responders using CT, 71.7% (33 from 46) were classified as responders and 28.3% (13 from 26) as non-responders when using the PrediCTC; and 100% patients classified in progression with CT were non-responders with PrediCTC. Under the expert panel consensus, the application week of the transition mediated by the PrediCTC test was change from week 16 to week 12.
The treatment schedules and the references used to obtain the OS, PFS, percentage of drop-outs due to adverse effects, and the grade 3 and 4 side effects (neutropenia, febrile neutropenia, and neurotoxicity, diarrhea and hand–foot syndrome) are shown in Table 2, percentage of patients receiving each treatment defined by the experts in Table 3, and transition probabilities in Table 4.
Therapeutic schedules and percentage of patients treated in each line.
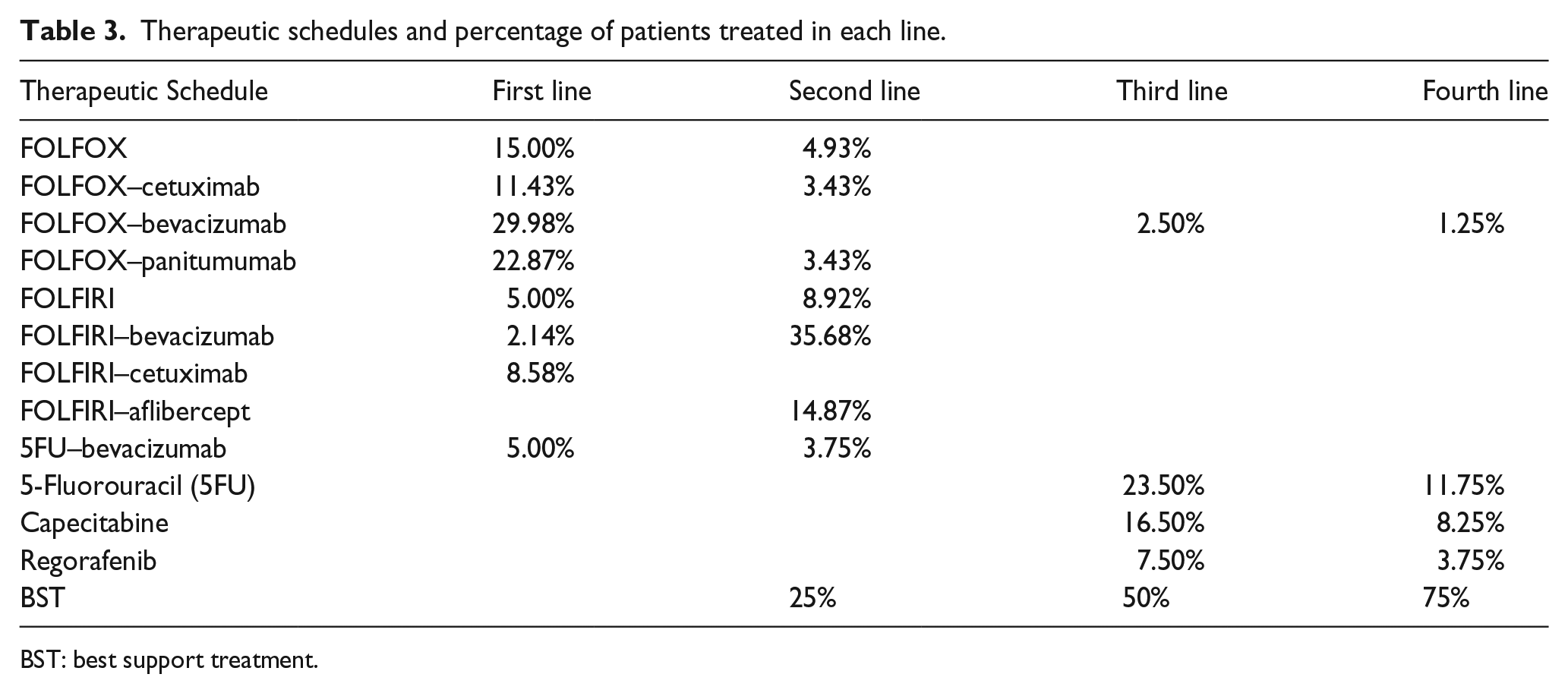
BST: best support treatment.
Model transition probabilities between cycles and treatment lines calculated through the OS and PFS in the studies referred in Table 2.
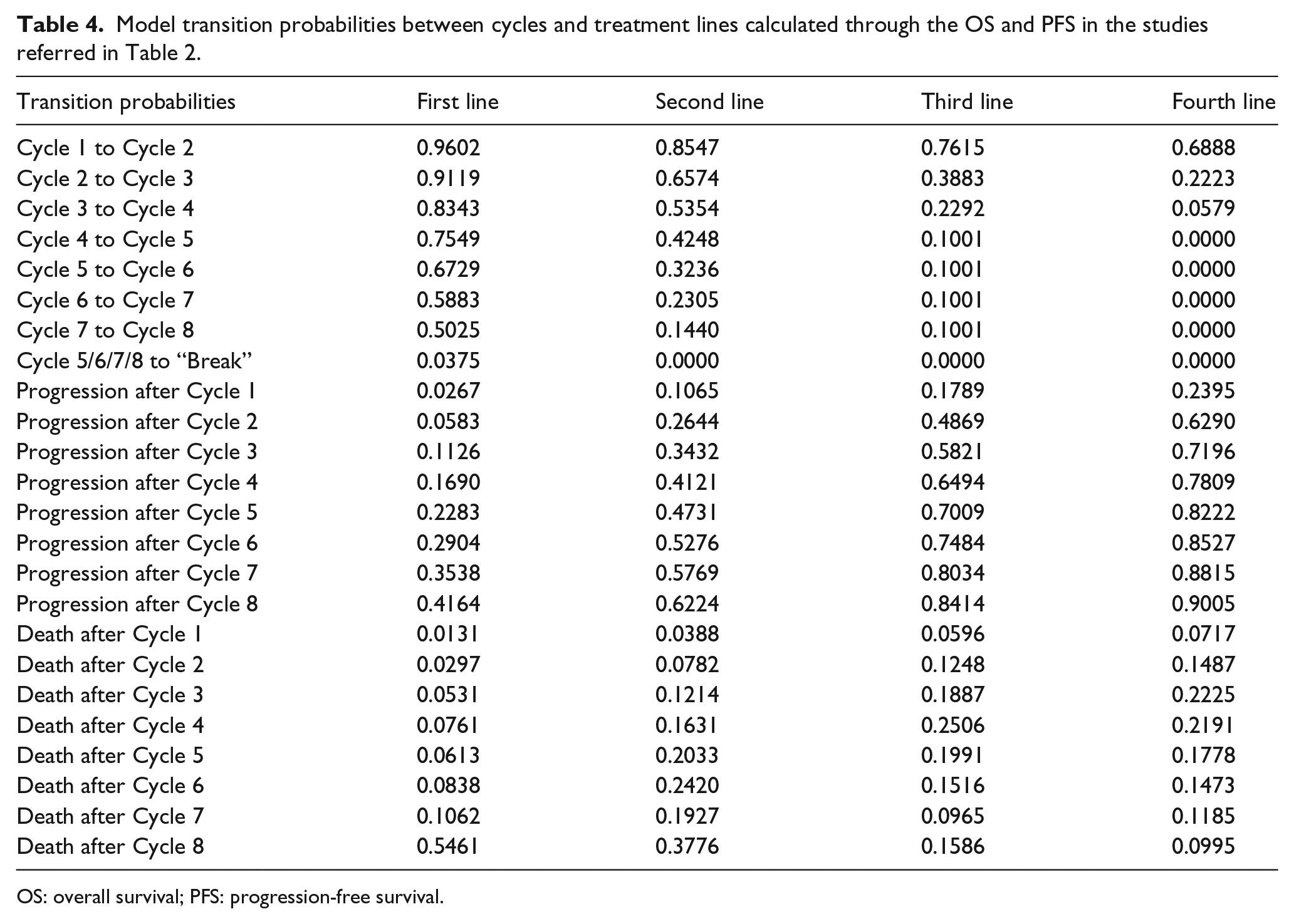
OS: overall survival; PFS: progression-free survival.
To reduce the treatment toxicities, some patients with stable illness, partial or complete response can take a “chemo break” or programmed rest in the treatment and then return once again to the same therapy schedule. The “break” average duration in the first-line treatment described in the OPTIMOX 2 study is 3.9 months. 19 We have include four cycles average of “Break” through which a 15% of first-line patients will transit assuming that these breaks starts from cycle 4 up to cycle 8 continuously until the percentage per line explained above has been reached.
When the chemotherapy outweigh the possible benefits, the patient only receives best supportive care (BSC). The percentage of patients with BSC is of 25% after first-line treatment, 50% after second-line treatment, and 75% after the third line of chemotherapy (Table 3).
The utilities of each chemotherapy schedule and their side effects included in the model are summarized in Table 5; population evaluated in these studies belongs to different countries like USA, UK, and Australia. General mortality data have not been included because OS does not allow to differentiate the mortality due to colorectal cancer from others.
Treatments and their side effects utilities.
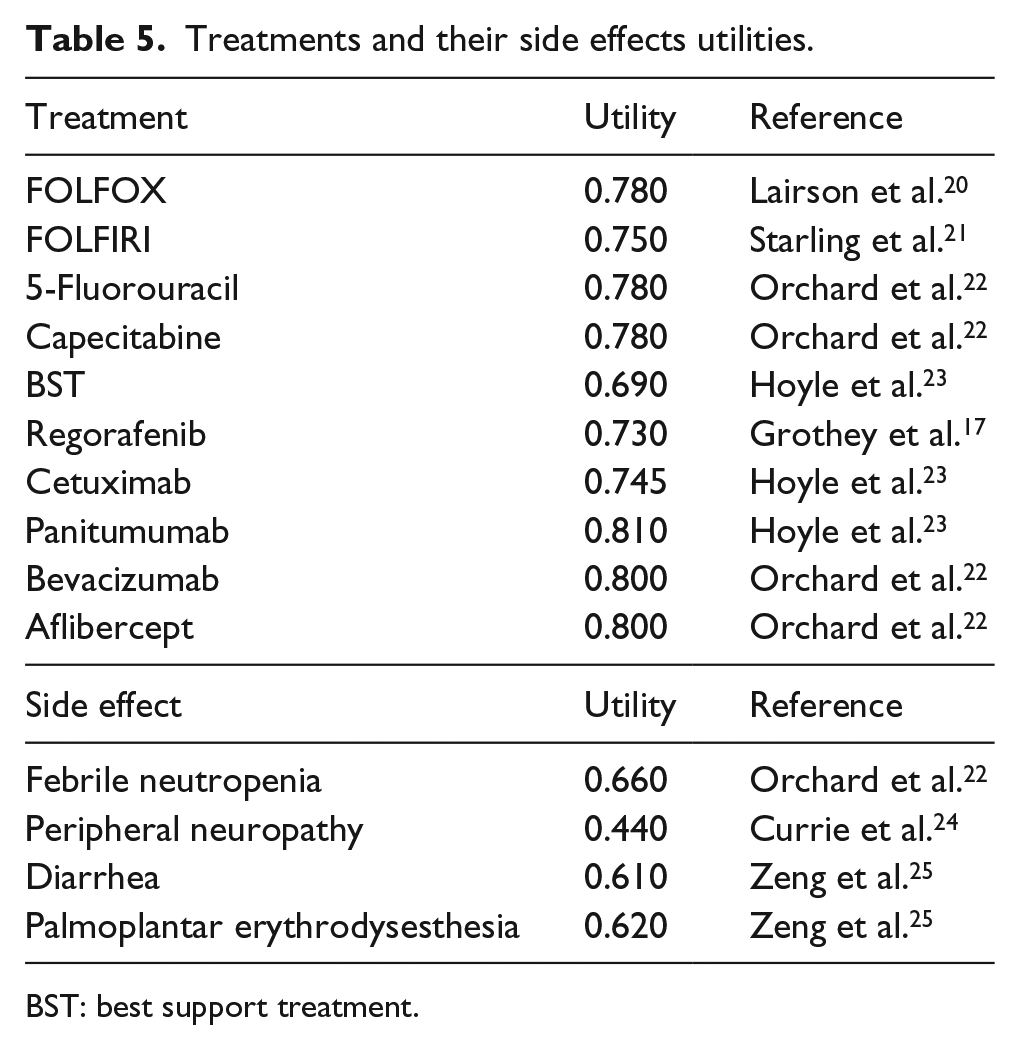
BST: best support treatment.
Cost estimation
The resources, unit cost, and units consumed identification to calculate the healthcare and non-healthcare direct cost and the indirect costs derive from the bibliographical references and the data provided by the panel of experts (Tables 6 to 9). All the costs have been updated to €2015 with the correspondent Consumer Price Index correction factor.
Chemotherapy schedules and best support treatment for each treatment line cycle (4 weeks).

iv: intravenous.
Adverse events grade 3 and 4 treatment cost per cycle (4 weeks).

Disaggregated cost not available. 26
Other direct medical cost included.

Disaggregated cost not available. 26
Parameter distribution function included in the probabilistic sensitivity analysis.

SD: standard deviation.
A discount rate of 3% has been applied to all costs and utilities. 32
Included within the direct healthcare costs are different chemotherapy costs of the cycles and what derives from its side effects, hospitalization costs, outpatient consultations, CT scan cost, PrediCTC test, and other complementary tests cost. For the calculation of the total cost of the different treatments, the dosage data from the technical reports of the medical products have been used, as well as the mean weight and body surface (70.5 kg and 1.7 m2), published by the National Institute of Statistics for individuals (men and women) over 65 years of age. In addition, the price published by the Official Body of Pharmacists has been used, applying a discount of 7.5% and of 4% to the orphan drugs established in articles 8, 9, and 10 of the Royal Decree Law 8/2010 modified by the Royal Decree Law 9/2011 and the costs of the intravenous infusion and day spent at hospital published by the Galician Health Service. 30 The cost of the BSC derives from the composition of the active ingredient, dosage, and distribution published in the study by Pericay et al. 26 The pharmacological treatments included in the BSC are analgesics (delayed-release morphine, metamizole, fentanyl in patches and tablets, tramadol and midazolam), anti-cachetics (megestrol), antiemetics (oral ondansetron), and corticosteroids (dexamethasone and oral prednisone).
All adverse events cost included in the model are summarized in Table 7:
Neutropenia, febrile neutropenia, neuropathy, and diarrhea grade 3 and 4 cost per cycle were estimated from disaggregated resources consume by an expert panel from different Spanish hospitals (Parc Tauli Sanitary Corporation, Catalonia Oncology Institute and Santiago de Compostela Hospital Complex) but the label information is not available in the paper published.
As Hand–Foot syndrome cost is not detailed in Pericay et al., 26 the cost estimation was calculated from the treatment explained in Herrera et al., 27 study and the price published by the Official Body of Pharmacists using the same methodology describe above for the chemo treatment.
The direct healthcare costs not included in the chemotherapy treatment are follow-up treatment test (CT and PrediCTC) and those connected to hospital stays (hospitalization), consultations, emergencies and other direct costs which cover pharmaceutical treatments in outpatient clinics, pathological anatomy, and other complementary test detailed in Table 8. PrediCTC resources consume (material and labor) and their cost was obtained from the Santiago de Compostela Hospital Complex. The sample management cost was based on 1 day (8 average hours) of work for each determination, the assumption that four samples could be evaluated at the same time and a laboratory technician €1300 cost per month. From the public rates for the Galician Health Service, the CT cost and the rest of non-chemotherapy sanitary direct cost from the phase IV colorectal cancer per patient during a 13 months (13.93 cycles) average follow-up taken from the Catalonia Colorectal Cancer Cost Study publish in 2015 (€15,539.05 hospitalizations; €803.56 others €4,228,67). 29
The direct costs not related to healthcare are those such as displacement costs and unofficial care, which are usually paid by the patient or their family. The hourly cost for unofficial care (€8.25) has been calculated using data from the 4th Collective Agreement of the State Framework for services of people and the development of the promotion of personal autonomy, with a total of 40 hours per week. The cost of displacements is based on the cost of non-urgent healthcare transport (€38.52) published in the Official Bulletin of the Autonomous Community of Madrid, multiplied by an average of 2, 1.5, 1, and 1 in first-, second-, third-, and fourth-line treatment, respectively. 31
The indirect costs (productivity potential losses in productivity) have been estimated taking into account the average age of the patients and assuming that 90% are retired and the other 10% are permanently unable to work (assumption approved by the panel of experts). The average cost for permanent disability in Spain is €906.66 and €995.59 as average retirement cost.33,34
Base case analyses
The results of the model have been obtained analyzing the base case and classifying the costs by the following types: test costs, cost of adverse events, cost of chemotherapy, direct costs of the base case. The first-line results have been differentiated from the totals because in the rest of the treatment lines, the drivers of the model for the two alternatives are identical. The effectiveness is expressed in quality adjusted life years (QALYs), adverse effects and costs avoided.
Once the results of the base case were obtained, a univariate analysis was performed by varying the value of all parameters used in the model within a plausible range of ±10% if we did not have the confidence interval or any other statistical dispersion. If CI was available, the upper and lower limits were used. In addition, we performed a probabilistic sensitivity analysis using random values taken from the probability distributions characterized by the average and standard deviation instead of using a fixed value for each parameter used in the base case. The distributions chosen were based on Briggs et al., handbook: “Decision modelling for health economic evaluation” and depicted in Table 9. 35
Results
Base case analysis
Base case results show all cost typologies lower for the PrediCTC alternative (Table 10), mainly due to its higher sensitivity identifying patients who do not respond to first-line treatment, therefore avoiding treatment costs for patients who according to this test, will not respond to it.
Base case results (cost and utilities per patient).
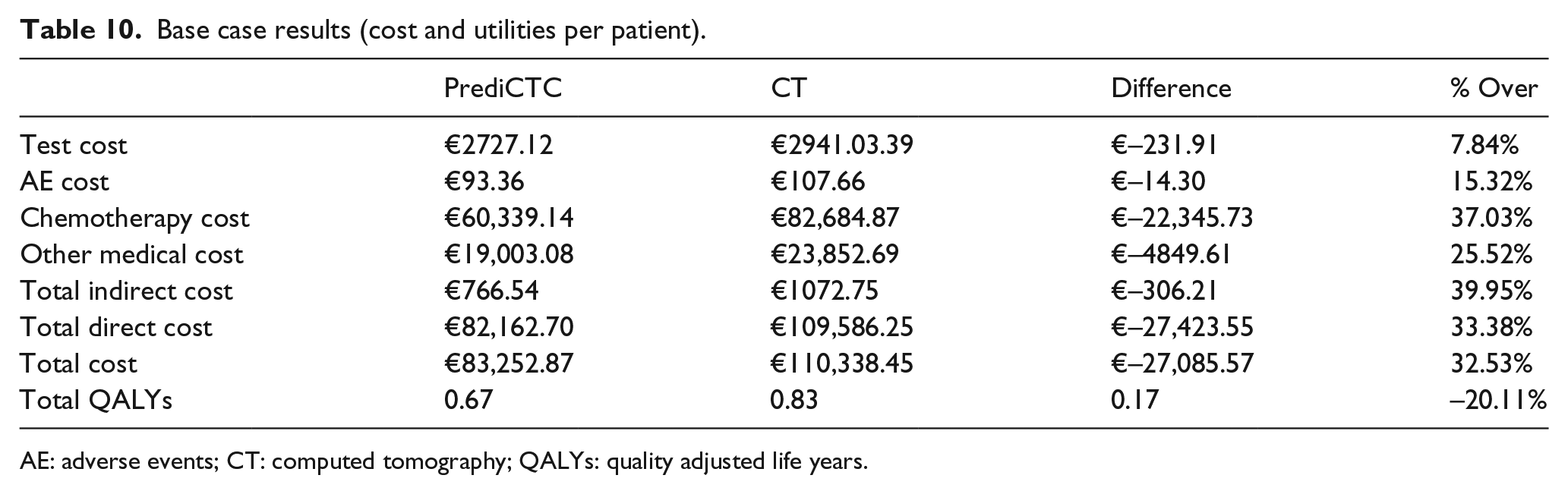
AE: adverse events; CT: computed tomography; QALYs: quality adjusted life years.
The CT alternative achieves greater utility than PrediCTC despite the latter avoiding giving chemotherapy to more patients who will not respond to the treatment scheduled in the first line. This is due to the fact that the model is prepared to give patients less utility as they progress through the treatment lines and with PrediCTC, non-responder patients pass earlier to the second line of treatment. The average utility for the patient in first-, second-, third-, and fourth-line treatment for CT alternative is 0.54, 0.20, 0.06, and 0.02, respectively, and for PrediCTC 0.37, 0.17, 0.05, and 0.05. For this reason, the incremental cost-effectiveness ratio (ICER) is not given as a result of the assessment due to the fact that the alternative is less effective in terms of usefulness (QALYs).
The percentage of patients who survives and moves to the next line treatment using PrediCTC to determinate the treatment response was 33% in the first line, 33% in the second, 22% in the third, and 11% in the fourth. With the CT alternative, this percentage was 41%, 29%, 20%, and 10%, respectively. With regard to adverse events, the PrediCTC alternative produces 0.12 adverse events less, with €27,085 saved per patient.
Univariate sensitivity analysis and probabilistic results
The univariate sensitivity analysis carried out does not show any significant change in the results. And the probabilistic sensitivity analysis provides similar results to the deterministic ones regarding the efficacy and all the cost areas analyzed (Table 11).
Probabilistic analysis results.
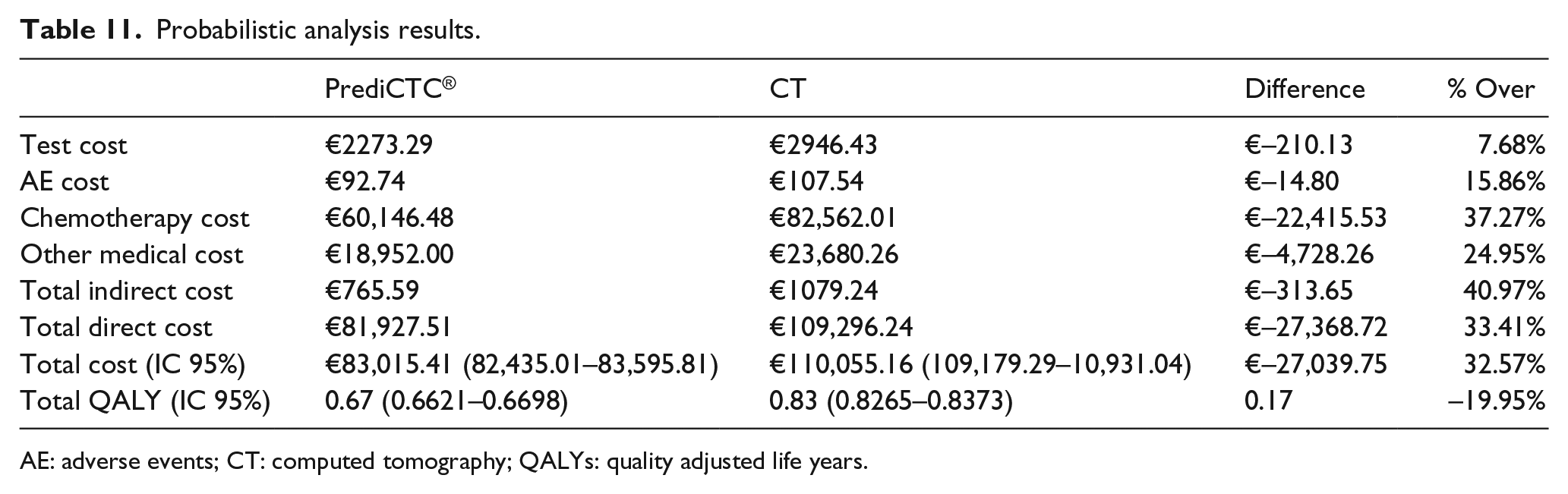
AE: adverse events; CT: computed tomography; QALYs: quality adjusted life years.
On a cost-efficacy level, represented by the 1000 repetitions performed for each alternative, PrediCTC remains the best option in 91.2% of the simulations (Figure 3).
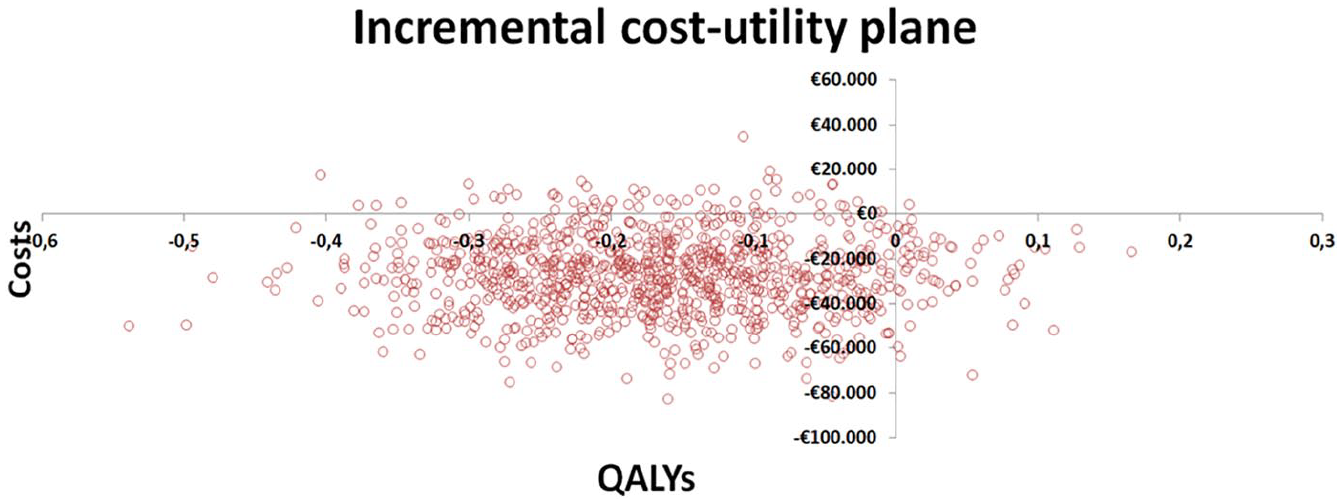
Incremental cost-effective plane.
Discussion
Although PrediCTC has shown a greater sensitivity to detect first-line early non-responder metastatic colorectal patients, compared to the computed tomography (CT) scans, National Oncology experts consider that Barbazán et al. 2 is an exploratory study, with a small sample of 50 patients recruited by the University Hospital of Santiago de Compostela and does not allow to determinate which treatment should be given to the non-responder patients.
The main limits we faced were due to the fact that in the reference study, follow-up is only made on patients in the first line of treatment and no modifications are made to the patient approach given the test results. As a result, we cannot know the real response of the NR patients to the subsequent lines of treatment. Existing possibility is that this subgroup of patients with advanced colorectal cancer who are prematurely classified as NR may have some characteristic in common such as more aggressive tumors or special sensitivity to a particular type of treatment. This would substantially vary the results of effectiveness and usefulness in the subsequent lines and would make them differ from the assumptions made. However, for the moment, there are no data available which allows us to differentiate the results. Therefore, the transition probabilities and usefulness after the first line of treatment are the same for the other alternatives compared.
Most of the new clinical trials published after the selection done by the oncologist from the Santiago de Compostela University Hospital evaluate the efficacy or effectiveness of chemotherapy regimens that do not correspond to the ones used to treat the patients included in the Barbazan study and also most of them include biomarkers to select the treatment to be used.
A National Oncology expert’s focus group was done to analyze the results of the cost–utility evaluation, their main conclusion was that PrediCTC should not replace CT but it could be added to the chemotherapy response assessment when other studies with a large sample prove its accuracy after the first cycle in the first treatment line and in the other clue moments were the treatment response has to be evaluated.
Conclusion
From the Spanish societal perspective, PrediCTC allows to identify patients that are not getting benefits from chemotherapy in unresectable metastatic colorectal cancers and therefore to avoid side effects and cost in non-responder patients, but is not a cost-effectiveness option. Further studies are needed to investigate the real effectiveness of adapted second, third, and fourth chemo schedules for non-responder patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
