Abstract
Objective:
The purpose of the study is to estimate the financial impact on the Italian National Health Service of the use of BD Micro-Fine™ 4 mm 32G needles for insulin pens and of the BD Educational Starter Kit programme as compared to the use of longer pen needles, without the support of any educational programme.
Methods:
A budget impact model was developed to estimate the direct medical costs up to 12 months.
The resource utilisation (insulin and complications associated with diabetes) was derived from literature. Only direct medical costs were considered. Ex-factory prices and National Tariffs were considered to estimate the costs of insulin and medical resource used to manage the complications associated with diabetes, respectively. The analysis was restricted to insulinised patients diagnosed with type 2 diabetes. Key variables were tested in the sensitivity analysis.
Results:
The cost per patient treated with BD needles presents a reduction of €32.08 of average annual cost (€690.02) compared to that associated with the patient treated with needles of other brands (€722.11). Given an increase in the percentage of use of BD needles that, compared to the base case, could involve 20%, 50% or 100% of patients, there would be a significant reduction in the expense incurred by the Italian National Health Service in the range of €2,536,710–€12,683,551.
Conclusion:
The use of the BD needles and the BD educational programme, as an alternative to the use of longer pen needles without the support of any educational programme, could represent an effective option to a low cost for the Italian National Health Service.
Keywords
Introduction
The control of glycated haemoglobin levels and the consequent corrections to insulin therapy are the main issues investigated during a diabetology visit; less time, however, is spent investigating the insulin injection technique used by patients. 1 Yet, the latter is fundamental in guaranteeing that the drug acts properly, and therefore, it should be considered as significant as the type of insulin selected or the administration dosage. 1 In addition to injection site rotation and the procedure for inserting the needle into the skin, a correct injection technique also involves selecting the proper needle length. 1 Many studies have investigated this last issue demonstrating patient’s preference, effectiveness and safety of 4 mm 32G needles for insulin pens.2–8 These needles provide the same level of blood glucose control as longer needles (12.7, 8, 6 and 5 mm), while reducing the risks and pain associated with intramuscular injections.2–8
The results of a recent analysis conducted in the North of Italy by Grassi et al. 1 demonstrated the importance of a proper injection technique. The adoption of 4 mm needles for insulin pens (BD Micro-Fine™ 4 mm 32G) and of the joint educational programme on injection technique (BD Educational Starter Kit), in the 3-month observation period of the study, resulted in a significant reduction of glycated haemoglobin (−0.58%; 95% confidence interval (CI): 0.50%–0.66%) in insulinised patients (p < 0.05) as compared to the use of longer needles (5, 6, 8 and 12.7 mm). 1 In addition to the improved blood glucose control, the study demonstrated how a proper injection technique allows the consumption of insulin to be significantly reduced (−2 IU; 95% CI: 1.4–2.5 IU).
Based on these clinical results, 1 it seemed appropriate to also perform a budget impact analysis (BIA) to estimate the financial impact on the Italian National Health Service (NHS) of the use of BD Micro-Fine 4 mm 32G needles for insulin pens and of the BD Educational Starter Kit programme for the proper administration of insulin, as compared to the use of longer pen needles (5, 6, 8 and 12.7 mm) without the support of any educational programme.
Methods
To evaluate the economic impact of BD needles and of the BD educational programme, a budget impact model (BIM) was developed in Excel®. The healthcare expenditures considered here intend to describe, for insulinised patients, the cost of needles, of pharmacological treatment (insulin) and of managing the complications associated with diabetes. The analysis was only restricted to insulinised patients (without any other anti-diabetics drugs) diagnosed with type 2 diabetes. The BIA covers a period of 1 year. Figure 1 schematically shows the structure of the BIM. The analysis was conducted in accordance with the Guidelines of the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) for conducting budget impact analyses.9,10
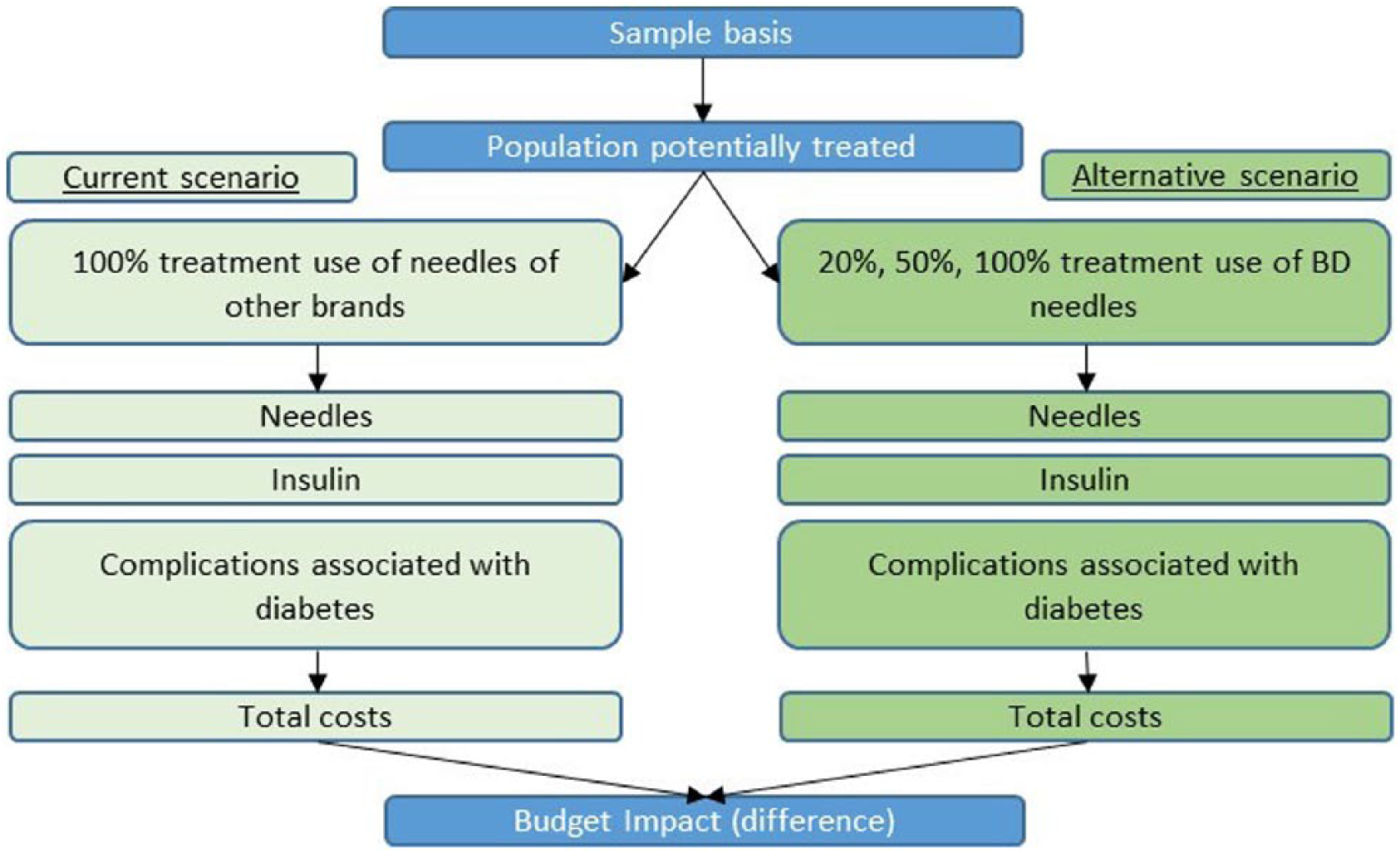
Structure of the budget impact model.a
Assumptions of the BIM
The number of patients subject to the BIA was estimated based on the resident population in Italy on 1 January 2017 (sample basis). 11 The population with diabetes was calculated applying a prevalence rate of 6.2%. 12 It was then possible to stratify the population in order to determine the number of patients with type 2 diabetes in treatment with insulin alone.12,13 In line with the methodology adopted here, the population of patients with type 1 diabetes, which is, according to the most recent estimations, 13 around 5.4% of the total population, was excluded from the analysis (see section ‘Discussion’).
In the base case, 100% of patients use needles of other brands without the adoption of any educational programme. In addition to this case, the BIA also evaluated three other scenarios in which an increased level of adoption of the BD needles and of the joint educational programme was hypothesised (20%, 50% and 100%).
Input data for the BIM
Population
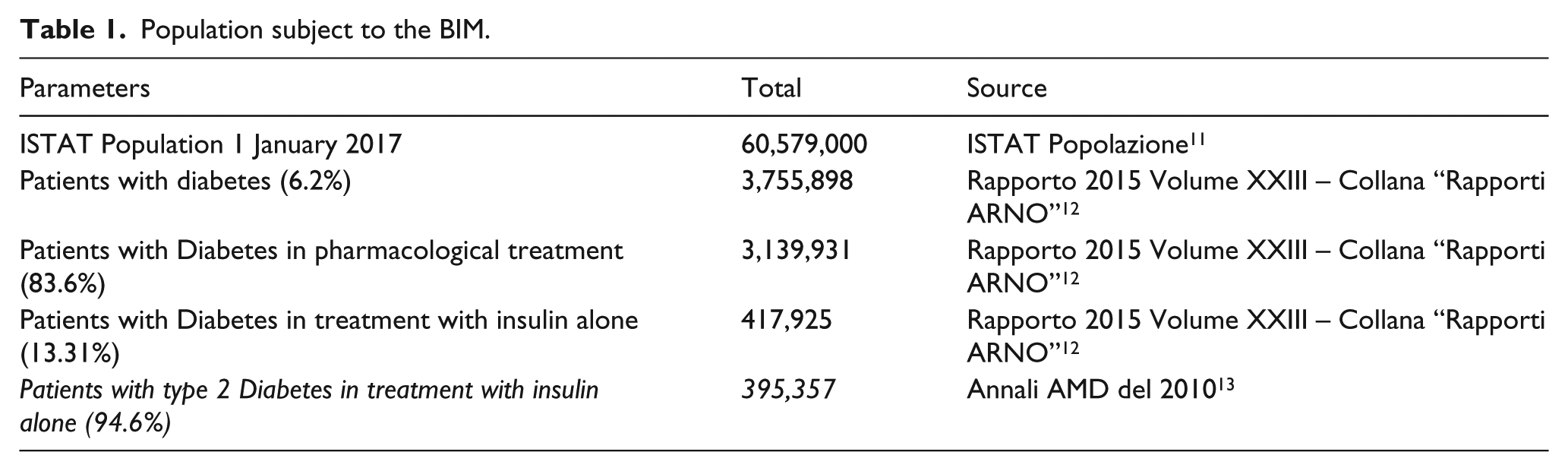
The reference population is composed of patients with type 2 diabetes in treatment with insulin only. Table 1 reports in detail the estimation process that, from the sample basis (resident population in Italy on 1 January 2017), leads to the identification of the number of patients subject to this BIA, around 400,000.11–13
Population subject to the BIM.
Treatment groups
Following the methodological approach adopted in the clinical study by Grassi et al., 1 two treatment groups were compared in the BIM:
Patients who use the BD 4 mm 32G needles and the BD educational programme (henceforth indicated by ‘BD needles’).
Patients who use other needles of greater length (5, 6, 8 and 12.7 mm) without the support of an educational programme (henceforth indicated by ‘needles of other brands’).
Clinical data
The study by Grassi et al. 1 provided the main clinical data, as well as the data for the consumption of necessary healthcare resources, for estimating the economic impact on the Italian NHS resulting from the use of BD needles or of needles of other brands. Overall, between February and April 2012, 346 patients were considered (age ≥ 12 years, diagnosed with diabetes and in insulin therapy for at least 4 years) pertaining to 18 clinics located in the North of Italy. No patient was enrolled who used 4 mm needles (5 mm: 33.3%; 6 mm: 41.7%; 8 mm: 23.7%; and 12.7 mm: 1.2%). Upon enrolment, the nurse always examined the injection sites of the patient to evaluate the presence of abnormalities such as lipohypertrophy. Subsequently, each patient received a first general education session on injection technique together with the BD Educational Starter Kit. This first phase was then followed by a personalised training session during which, through a special questionnaire administered to the patient by the nurse, ways to correct the adopted injection technique were demonstrated. All patients were then asked to properly rotate injection sites to avoid lipohypertrophy and asked to switch to BD Micro-Fine 4 mm 32G needles to avoid intramuscular injections. Finally, they were instructed to not reuse needles. At 3 months of follow-up, a reduction was recorded: (1) of HbA1c of 0.58 (95% CI: 0.50%–0.66%), (2) of the average daily dose of insulin of 2 IU (95% CI: 1.4–2.5 IU) and (3) of fasting glycaemia of 14 mg/dL (95% CI: 10.2–17.8 mg/dL; Table 2). The study did not provide separate analysis on patients with type 1 or 2 diabetes.
Characteristics of patients and results from the analysis conducted in the North of Italy. 1
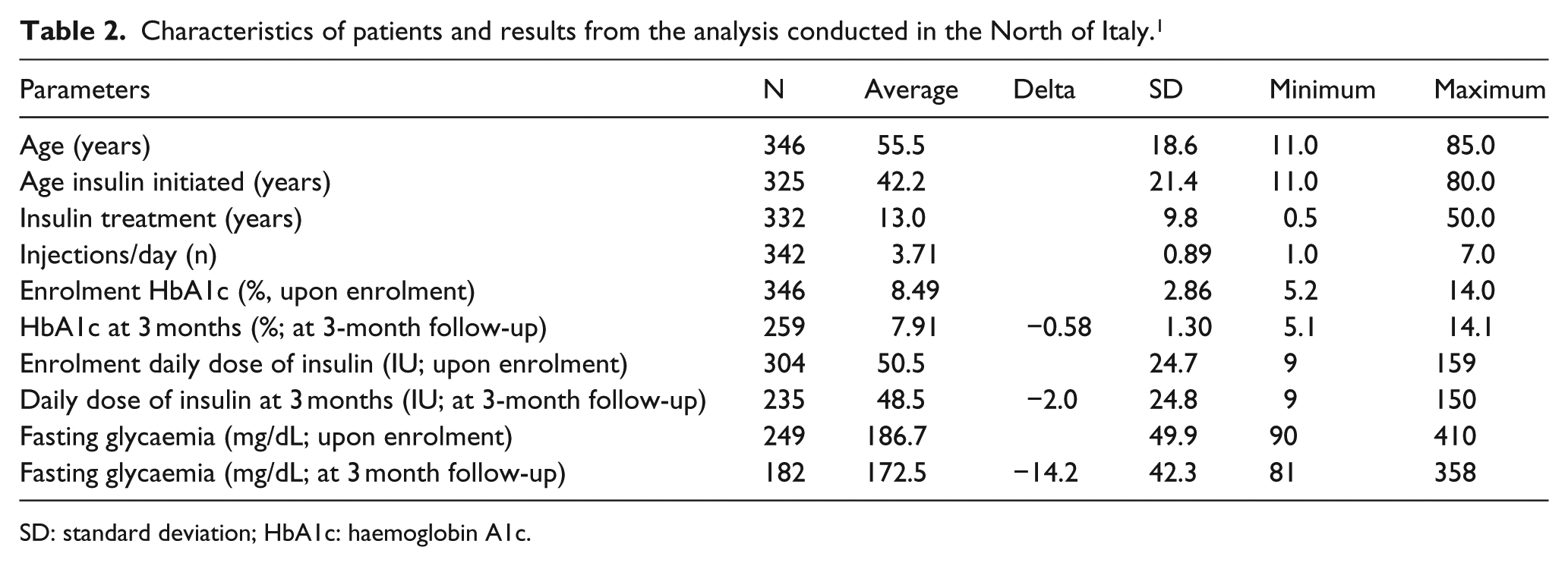
SD: standard deviation; HbA1c: haemoglobin A1c.
Cost of needles
On the hypothesis that needle selection does not influence the average number of daily injections per patient and that therefore this value remains constant before and after enrolment, in this analysis, it was decided not to value the consumption of needles. The respective reimbursement price adopted by regions does not result in any differentiation based on length (4, 5, 6, 8 or 12.7 mm), and this would thus not result in any difference in cost between the two alternatives under consideration.
Cost of insulin
An average price of €0.0203 per international unit (IU) of insulin was entered into the BIM. This value reflects the net ex-factory price after the discounts required by law for fast- and long-acting insulin (pens) knowing that these represent 56% and 38% of the market, respectively. 14 Based on this cost and on the consumption levels indicated in Table 2, the average daily cost of insulin treatment was calculated given the use of BD needles and given the use of other brands.
Management and cost of diabetes complications
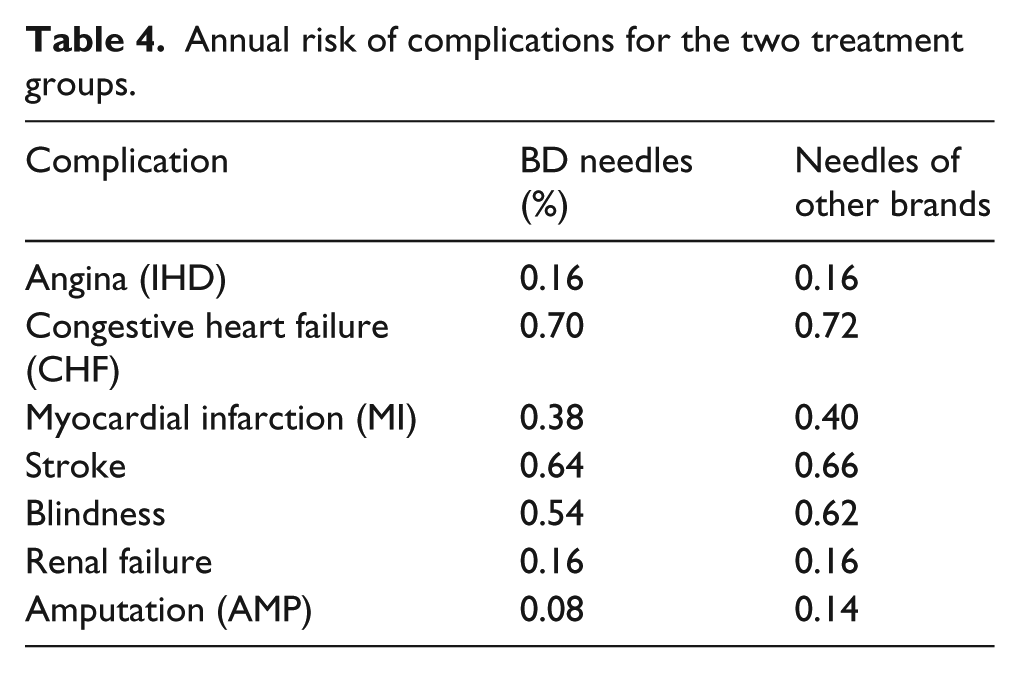
The BIM also estimated the economic impact relating to the risk of complications associated with diabetes, with a distinction by treatment group. This estimation required a complex process described briefly below. Table 3 shows the probability (in percentage) that a patient with type 2 diabetes could have one of the following complications: angina, congestive heart failure, myocardial infarction, stroke, blindness, renal failure and amputation. 15 In addition to the percentage figure, Table 3 also reports the cost data used to determine the value of the healthcare costs associated with the management of each individual event. 15 These data, percentage figures and costs, were extrapolated from the results of a recent study conducted nationally that evaluated the cost-effectiveness of a long-term programme for improving the quality of care provided to the diabetic patient. 15 Subsequently, based on the equations for forecasting the occurrence of diabetes-related complications derived from the United Kingdom Prospective Diabetes Study (UKPDS) 16 and in accordance with the glycated haemoglobin value (enrolment vs follow-up) determined from the study by Grassi et al., 1 the annual risk of complications was estimated for the two treatment groups (Table 4). Finally, by multiplying these risk values by the annual costs associated with individual events (Table 3), the average annual cost per patient for the management of complications related to type 2 diabetes was calculated.
Diabetic complications and treatment costs. 15
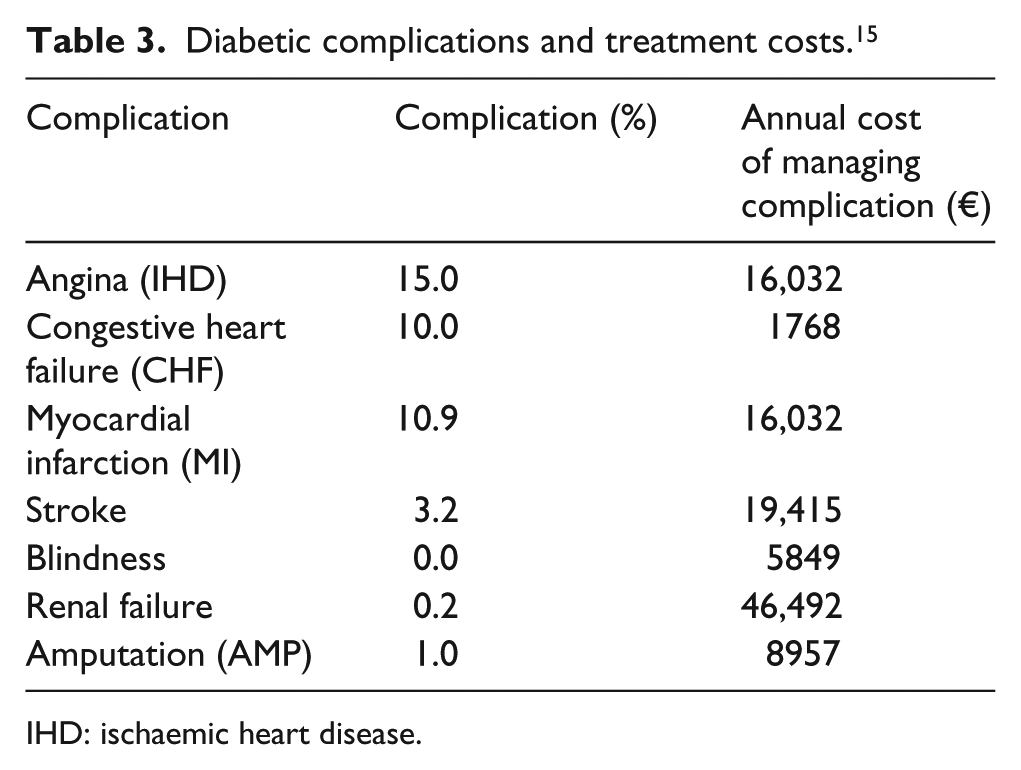
IHD: ischaemic heart disease.
Annual risk of complications for the two treatment groups.
Output data from the BIM
Budget impact
The BIA, based on the eligible population and on the estimated average annual cost per patient in therapy with BD needles or with needles of other brands, was performed over 1 year.
The choice of this time period was carried out with the aim of making the economic impact as consistent as possible with the observation period adopted in the study by Grassi et al., without extrapolating clinical data beyond the year.
The difference in the expense between the base case (other needles: 100%; BD needles: 0%) and the three alternative scenarios in which an increased percentage of use of BD needles was hypothesised (20%, 50% and 100%). The decision to present different scenarios, as compared to the base, involving the increased use of BD needles, was made with the intent of reproducing a similar effect as would be obtained by varying the percentages of use of BD needles along a greater time period (e.g. 3, 4 or 5 years).
Sensitivity analysis
A deterministic analysis (one way) was conducted to estimate the sensitivity of the results estimated from the model to variations in insulin price, cost of managing complications, complications annual risk and HbA1c reduction.
Results
Cost per patient treated
The cost per patient treated with BD needles presents a reduction of €32.08 of average annual cost (€690.02) compared to that associated with the patient treated with needles of other brands (€722.11; Table 5). This difference, to the advantage of BD needles, resulted from the reduced use of insulin (−€14.59) and the reduced need to treat any possible complications caused by diabetes (−€17.50).
Average annual cost of treatment per patient.

Expense incurred by the Italian NHS
The expense incurred by the Italian NHS associated with the base case, calculated from the number of patients in treatment with needles of other brands (n = 395,357), came out to €285,489,784; €139,892,567 (49.0%) of this is for the management of complications, and €145,597,217 (51.0%) of it is for the consumption of insulin (Figure 2).
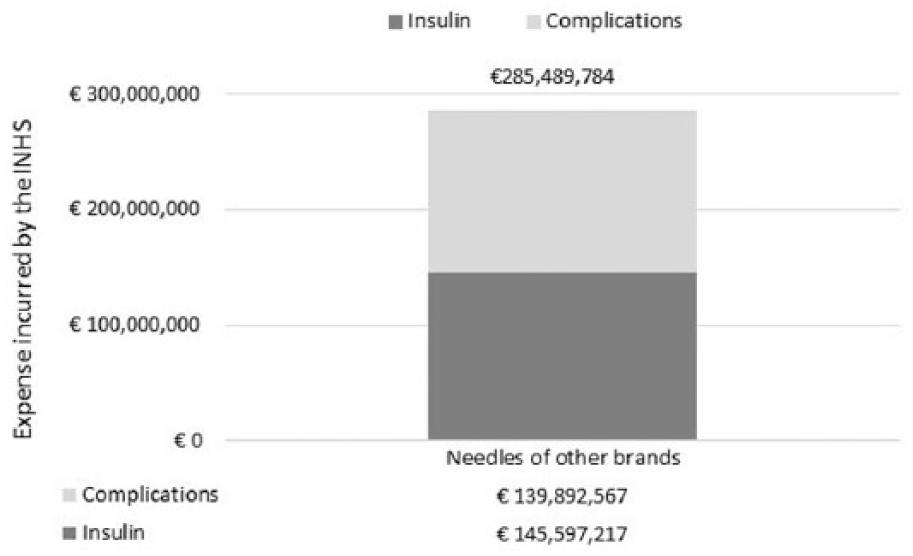
Annual expense incurred by the Italian NHS: base case.
Given an increase in the percentage of use of BD needles that, compared to the base case, could involve 20%, 50% or 100% of patients, there would be a significant reduction in the expense incurred by the Italian NHS in the range of €2,536,710–€12,683,551 (20% scenario: −€2,536,710; 50% scenario: −€6,341,775; and 100% scenario: −€12,683,551; Figure 3).
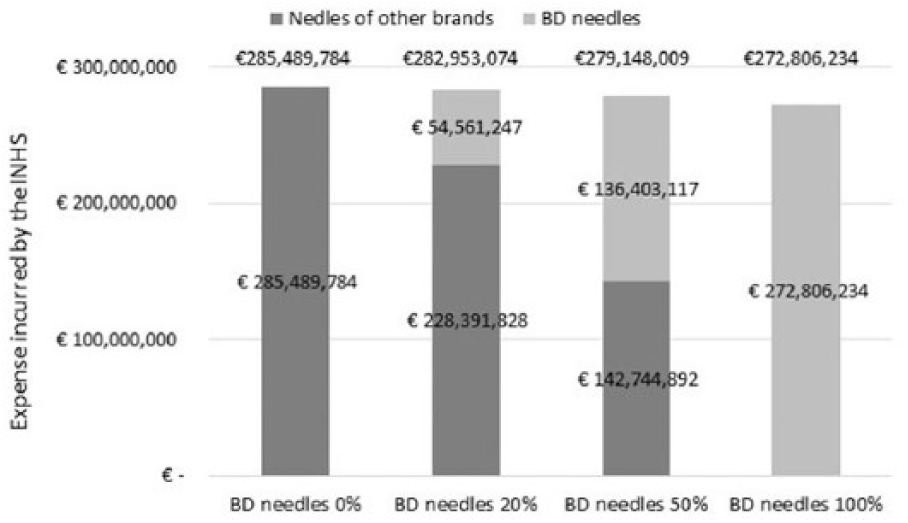
Expense incurred by the Italian NHS: base case versus variation in the percentage of use of BD needles.
Sensitivity analysis
Table 6 reports the results of the sensitivity analysis. Even if there is a variation in the annual risk of complications or there are significant reductions in the price of insulin or in the cost of managing complications or of the HbA1c reductions at follow-up, BD needles together with the special educational programme would represent the option with the lowest average costs per patient treated under the Italian NHS. Table 7 shows the variations in the financial impact of the use of BD needles as compared to the needles of other brands in the light of variations in the price of insulin or in managing complications. In all proposed scenarios, the adoption of the BD needles results in a reduction of the expense incurred by the Italian NHS.
Sensitivity analysis: average treatment cost.
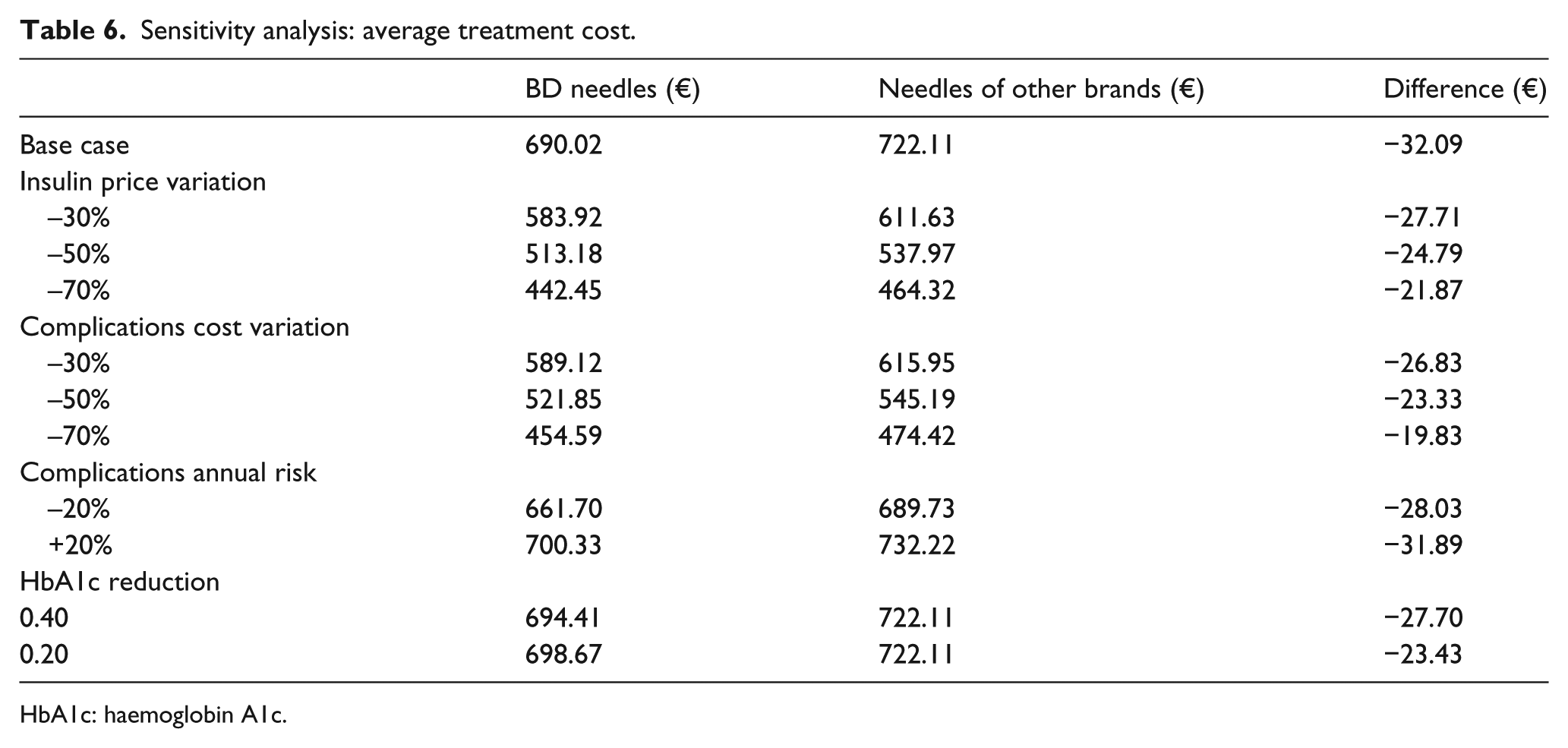
HbA1c: haemoglobin A1c.
Sensitivity analysis: Budget Impact.
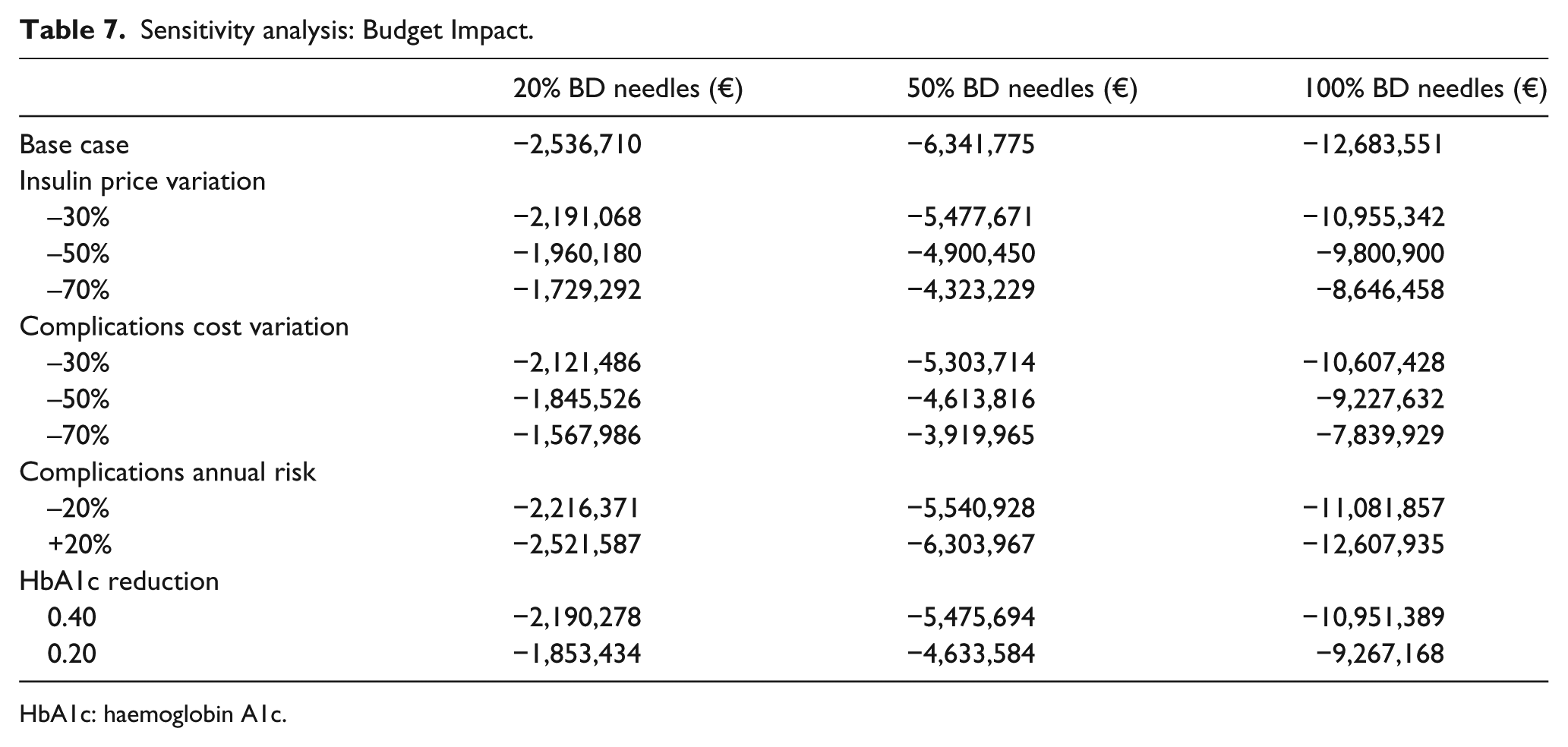
HbA1c: haemoglobin A1c.
Discussion
Due to a greater reduction of HbA1c and a lower daily consumption of insulin with the BD needles as compared to the needles of other brands, 1 the evaluation of the economic impact on the Italian NHS was deemed opportune. Therefore, the BIA produced a financial estimation of the costs associated with the consumption of insulin and with the management of complications related to diabetes in patients with type 2 diabetes in treatment with insulin only, with the aim of providing an indication of the expense of the Italian NHS would incur for the management of patients in treatment with BD needles or with needles of other brands. As Regions offer a single reimbursement price that does not differ with regard to needle length, the BIA did not consider the costs associated with the use of needles. According to the evaluation provided by the BIM, the greater use of BD needles compared to the use of needles of other brands would reduce the healthcare expense incurred by the Italian NHS in the range of €2,536,710 (20% scenario)–€12,683,551 (100% scenario).
Unlike the study by Grassi et al., 1 in this analysis, only patients with type 2 diabetes were considered, assuming that clinical results are the same for both subgroups (type 1 and type 2) as patients treated with insulin alone have been considered. This choice (considering only patients with type 2 diabetes) is a direct result of the methodology used to determine the rates of risk associated with diabetes-related complications. The rates of risk were indeed calculated based on equations derived from the UKPDS study that refer strictly to a population affected by type 2 diabetes. 16 Although the most recent estimates indicate that the population with type 1 diabetes represents only 5.4% of the total population, 13 it is likely reasonable to assume that the exclusion of these patients has somewhat reduced the cost savings potential of the BD needles as compared to the alternative option. An additional element that may have reduced the potential advantage of BD needles is represented by the choice, in line with the study by Grassi et al., to have considered patients in treatment with insulin alone. The effectiveness of BD needles would likely also be demonstrated in patients treated with insulin and oral drugs. Currently, however, there is no data to support these hypotheses.
In the absence of similar trials conducted nationally or internationally, it was not possible to compare these results with those of other studies. It was possible, however, to compare the results of the clinical study by Grassi et al. 1 with those of a recent trial conducted by Smith et al. 17 in the United Kingdom. In 18 hospitals, an educational programme on injection technique was implemented in order to evaluate the impact of this lipohypertrophy and other clinical variables such as glycated haemoglobin and the daily dosage of insulin; 75 patients in treatment with insulin were followed for 3–6 months. 17 At the end of the observation period, the presence of lipohypertrophy at injection sites had disappeared or was reduced by more than 75%. Upon enrolment only one-third of patients (38%) used a 4 mm needle, while at the end of the observation period, almost all patients (96%) were in treatment with the 4 mm needle. 17 At the end of the observation period, a significant reduction (p < 0.01) was also observed in the level of HbA1c (−4.1 mmol; 70.0 vs 65.9 mmol/mol) and in the average daily dosage of insulin (−5.6 IU; 71.6 vs 66.0 IU). 17 Finally, an unexpected but significant reduction in hypoglycaemic events was observed. In this study as well, the adoption of the 4 mm needle and of an educational programme on injection technique resulted in an improvement in the clinical conditions of the insulinised patient, reducing the amount of glycated haemoglobin and of insulin consumption. These results confirm the robustness of the clinical data used in this BIA. 1
The study by Grassi et al., as opposed to that conducted by Smith et al., 17 did not provide any estimation regarding the reduction/increase in hypoglycaemic events during follow-up. As a result, it was not possible to estimate the potential financial impact resulting from this event. It is presumable, however (given the findings in the study by Smith et al. 17 ), that if this aspect had been evaluated, as a result of the greater effectiveness of the BD needles, there may have been recorded a reduction in hypoglycaemic events with a consequent decrease in treatment costs.
Although here the economic impact associated with the management of lipohypertrophy was not evaluated, according to the results of a Spanish study, the adoption of a proper site-rotation technique would be fundamental in reducing it. 18 Also, according to the results of the Spanish study, lipohypertrophy would increase both the risk of a hypoglycaemic event and the variability of blood sugar levels. 18 Another important result from this study is the correlation between the daily dose of insulin and lipohypertrophy: patients with lipohypertrophy present a significantly higher daily consumption of insulin. 18 The increase in hypoglycaemic risk, the greater variability of blood sugar levels and the increase in insulin consumption constitute factors liable to compound the cost of the insulinised diabetic patient under the Italian NHS’s responsibility.
As occurs, whenever it is necessary to use a simulation model, it looks appropriate reading the results in the light of some considerations. The most important of these is represented by having extended for 1 year the results of the Grassi et al. 1 study, which indeed only referred to a time period of 3 months. While the extrapolation to a medium- to long-term time period of clinical data referring to analyses of a brief period is now a well-established practice in pharmacoeconomics, in justification of the choice adopted here, it is noted that as a precautionary measure, the clinical and resource-consumption data estimated for 3 months were actually kept constant through the year, to avoid extrapolating across the year and thus increasing the clinical effect associated with the use of the BD needles and programme.
The model provides, over a 1-year time period, the difference in expense generated from the base case (100% needles of other brands) compared to three different alternative scenarios in which an increase in the percentage of use of the BD needles and programme (20%, 50% and 100%) is hypothesised. All of this is done assuming that the population subject to the analysis is static and not dynamic during the observation year, that is to say, a possible reduction in the size of the population due to mortality was not considered. It is believed that given the brevity of the observation period, this effect would be insignificant.
The univariate sensitivity analysis was conducted in order to determine how much the selection of criteria for determining the value of the consumption of healthcare resources could influence the results of the BIM. Even significantly reducing the reimbursement price for insulin or the costs used to determine the value of complication management, the patient who uses BD needles still represents the lowest cost of treatment and a significant cost savings for the Italian NHS.
Conclusion
While the reservations mentioned above should be taken into account, this analysis described a reliable economic scenario deriving from a greater use of the BD needles and the joint educational programme and how such a course of treatment could result in a lower cost incurred by the Italian NHS for the management of insulinised patients with type 2 diabetes. As this analysis represents one of the first attempts, it is hoped that in the near future, this result can be checked against what is demonstrated by clinical practice. Possible data obtained from Italian records could in fact definitively enhance the analyses coming from BIMs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This analysis has been supported by an unconditional research grant from Becton Dickinson S.p.A.
