Abstract
Background:
Although physical activity is recommended as a therapeutic approach for endometriosis, there is limited evidence on the interaction between endometriosis and skeletal muscle health and function.
Case presentation:
The participant was diagnosed with stage II endometriosis via laparoscopic surgery in 2020. She was undergoing treatment with Dienogest. The study spanned nine consecutive weeks, during which we assessed both functional and structural skeletal muscle parameters, including muscle stiffness, maximal strength, and muscular fatigue outcomes. Saliva samples were collected to analyze estradiol and cortisol concentrations at rest, immediately after, and 30 min following the fatiguing protocol.
Resting estradiol concentrations were within the expected range with a mean value of 2.48 pg/mL (±1.24). estradiol levels did not change significantly following the fatiguing protocol [F (2, 16) = 0.113, p = 0.893, ηp2 = 0.014]. A significant negative relationship between estradiol concentrations and muscle stiffness, both in a relaxed (r = –0.68, p = 0.044) and contracted state (r = –0.763, p = 0.017) was evident.
Mean resting cortisol concentrations were 5.87 nmol/L (±2.18), and significantly decreased after the fatiguing protocol [F (2, 16) = 18.2, p < 0.001, ηp2 = 0.695]. Pearson’s correlation showed a significant positive association between cortisol concentrations and eccentric peak torque (r = 0.723, p = 0.028).
Conclusions:
A diminished hormonal response to physical strain was evident. From these results, we propose the hypothesis that the function of the hypothalamic-pituitary-adrenal (HPA) axis may be impaired following prolonged stress. Muscle stiffness of M. rectus femoris was negatively influenced by estradiol concentrations. Hence, estradiol may modulate muscle stiffness, potentially independent of the underlying pathology.
Introduction
Endometriosis is a chronic inflammatory disease in which uterus-like tissues grow outside the uterus commonly affecting the ovaries, intestines and abdominal cavity. It may affect approximately 10%–15% of women in reproductive age, manifesting in symptoms like chronic pain, painful menstruation (dysmenorrhea), pain during intercourse (dyspareunia), or pain during defecation (dyschezia), and also mental symptoms such as fatigue and depression. 1
A higher number of myofascial trigger points, along with regional allodynia or hyperalgesia—primarily in the abdominal region—was also evident in patients with endometriosis 2 which may in turn lead to higher muscle stiffness and decreased contractility.2,3
Physical activity is often recommended as a therapeutic measure for patients with endometriosis. As a metabolically active tissue, skeletal muscle secretes myokines during contraction which act locally and at distant tissues to establish a systemic anti-inflammatory environment. 4 Thus, physical activity may be one measure to reduce inflammatory processes and endometriosis-associated symptoms. 5
In women with endometriosis only a limited number of antioxidants and higher reactive oxygen species may be present, leading to elevated oxidative stress in the muscle cell compared to healthy controls. 6 Given the detrimental impact of oxidative stress on muscle adaptation and contraction processes, endometriosis-related inflammation may likewise influence the structural and functional integrity of skeletal muscle. 4 However, skeletal muscle function in patients with endometriosis has not been the subject of any studies. Since endometriosis is a multifaceted disease with highly individual symptom profiles, each affected person should be assessed on a case-by-case basis. In this case report, we evaluated structural skeletal muscle properties, including stiffness, and elasticity, as well as functional parameters like maximum strength and fatigue, in a woman with endometriosis. We aimed to gain a deeper understanding of skeletal muscle health and hormonal responses following physical strain in individuals with endometriosis.
Methods
Case description
The subject was a 30-year-old Caucasian woman (172 cm, 61 kg at trial start) who visited the lab between September and November 2023. Diagnosed with stage 2 endometriosis via surgery in March 2020, she has been on daily Dienogest therapy (2 mg) since December 2020. Before treatment, she experienced severe pelvic and body-wide pain, fatigue, and weakness. During the study, she was largely symptom-free except for occasional fatigue. She maintained a weekly training load of ~4 h. The participant engaged in two full-body resistance training sessions per week, utilizing light to moderate weights. No natural menstrual cycle occurred due to hormonal therapy. Further, she was instructed to abstain from physical training 48 h prior to measurements to avoid muscular fatigue.
Diagnostic assessment
The measurements and interventional protocols were conducted once a week over a duration of 9 weeks. All measurements and interventional protocols took place at a standardized time frame between 8 and 10 o’clock in the morning. At every measurement, the same protocol was performed. A baseline assessment (hormonal measurements, body composition, structural skeletal muscle parameters) was followed by an assessment of functional skeletal muscle parameters (maximal strength and fatiguing protocol).
Hormonal measurements
Hormone concentrations of estradiol and cortisol were analyzed by collecting saliva at each appointment at rest (baseline), immediately (post-fatigue) and 30 min (post-fatigue30) after performing the fatigue protocol. The participant was asked to refrain from drinking, eating, and toothbrushing at least one hour before the measurement. Ten minutes after rinsing her mouth, the patient provided a saliva sample via the passive drool technique into a Salivette® without cotton swab. The samples were labeled with the participant’s anonymized code, date and time of the collection and then stored at −20°C. The samples were analyzed by the Dresden Lab Service GmbH.
Body composition
To evaluate body composition, bioelectrical impedance analysis (BIA) was performed using the Nutriguard-MS Body Impedance Analyzer® (Data Input GmbH, Poecking, Germany). The patient laid quietly for at least 5 min before the measurement to ensure the equal distribution of body water. Two electrodes were each attached to the hand and foot of the dominant side of the body. At least two measurements were performed at 5, 50, and 100 kHz. A third measurement was performed if the deviation of the values was higher than 2 Ohm. The conducted values are then entered into the manufacturer’s software Nutriplus© (Data Input GmbH, Poecking, Germany) to calculate body fat, percentage of body fat, total body water, intracellular water and extracellular water.
Functional skeletal muscle parameter
Isokinetic strength measurements were performed on the Isomed 2000 (D. & R. Ferstl GmbH, Regensburg, Germany). The test regimen for concentric and eccentric peak torque of the M. rectus femoris consisted of 1 set of 5 repetitions each, performed at a pre-set angular velocity of 60°s−1. Then, the fatigue protocol was conducted. The fatigue protocol contained 30 consecutive concentric repetitions of m. rectus femoris. Concentric phase was conducted at 120°s−1 angular velocity. The total work and the average work per repetition were recorded in Newton meter. To assess the degree of fatigue, we analyzed the reduction in maximal peak torque, defined as the decrease in Newton meter between the average maximum torque of repetitions 2–6 and that of the final 5 repetitions (26–30). The change score was defined as the decrease in Newton meter between the average of the first and last repetitions. We conducted multiple cross-correlation analyses using SPSS (Version 29.0.0) to examine the relationship between baseline estradiol and cortisol concentrations and various parameters, including total work, average work per repetition (avWork) over 30 repetitions, concentric peak torque (PTcon), and eccentric peak torque (PTecc) across different time lags.
Structural skeletal muscle parameter
To assess muscle stiffness and elasticity, elastography was used. Elastography is a non-invasive technique to evaluate mechanical properties of tissues. 7 Measurements were conducted using an ACUSON Redwood ultrasound (Siemens Healthineers, Erlangen, Germany). A 10L4 linear array probe (15 mm wide) was placed longitudinally on the marked location on the m. rectus femoris. Two circular regions of interest with a diameter of 15 mm were placed directly adjacent to each other beneath the upper fascial sheath of the muscle. Two measurements were taken in a relaxed muscle state, and two measurements were taken during a maximal voluntary contraction. The participant was encouraged verbally to contract the muscle at its maximum. Values of muscle stiffness are presented in meter per second (m·s−1).
Statistical analyses
The statistical tests were performed with SPSS (version 29.0.0). To evaluate changes in hormone concentrations between the average values of the nine measurements at rest and after the fatiguing protocol, a within-subject repeated measures one-way ANOVA was performed. A correlation matrix tested linear relationships between saliva hormone concentrations of estradiol and cortisol at rest and the assessed parameters. Additionally, a cross-correlation analysis between estradiol and cortisol concentrations at rest and the assessed physiological parameters was conducted to explore potential temporal relationships and time-lagged associations. This approach allows for the identification of delayed or anticipatory patterns in hormone–response dynamics, providing insights into how fluctuations in endocrine signals may temporally align with or precede changes in muscle function and stiffness. By evaluating correlations across multiple time lags, this method helps to uncover underlying regulatory mechanisms that may not be apparent through conventional correlation analyses alone. To calculate the t-statistics, p-values, and confidence intervals of the cross-correlation coefficients, a script was generated using R (Version 4.4.0) in RStudio (Version 2024.09.0). The script is provided in the Appendix.
Results
Baseline hormone concentrations at rest
Estradiol concentrations fluctuated considerably over the study period, with a mean value of 2.48 pg·mL−1 (±1.24) peaking at week 4 (5.44 pg·mL−1). Cortisol concentrations also showed substantial variability, with a mean value of 5.87 nmol·L−1 (±2.18), and a maximum of 9.84 nmol·L−1 in week 7. The detailed values are presented in Figure 2. No significant correlation was found between estradiol and cortisol concentrations at rest (r = 0.02, p = 0.959).
Impact of fatigue on hormonal concentrations
Compared to the baseline values at rest, cortisol concentrations decreased immediately subsequent the fatigue protocol and continued to decrease up to 30 min later [F (2,16) = 18.2, p < 0.001, n2p = 0.695]. Post-hoc analysis revealed a significant difference of 1.836 nmol·L−1 (±0.482) between cortisol concentrations at rest and after the fatigue protocol (t (8) = 3.81, p = 0.013). Also, the comparison between concentrations immediately after (post-fatigue) and 30 min after finishing the fatigue protocol (post-fatigue30) showed a significant difference of 0.75 nmol·mL−1 (±0.251), t (8) = 2.99, p = 0.041. Additionally, a significant difference of 2.58 nmol·L−1 (±0.537) between cortisol concentrations at rest and 30 min after the end of the fatiguing protocol was observed (t (8) = 4.81, p = 0.003).
Estradiol concentrations did not change significantly following the fatiguing protocol [F (2,16) = 0.113, p = 0.893, n2p = 0.014]. Figure 1(a) shows hormone concentrations of cortisol and estradiol before and after physical strain.
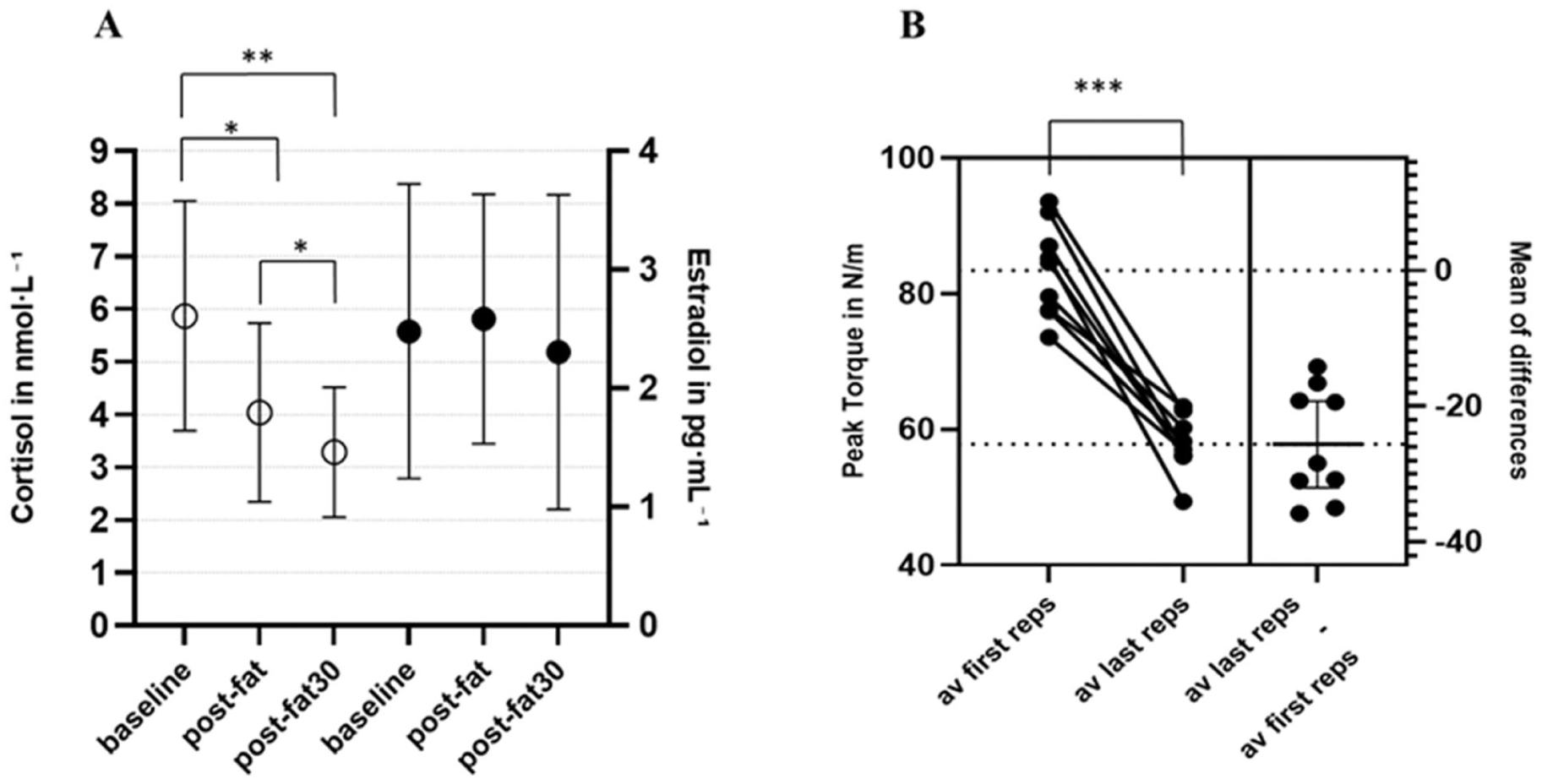
(a) Mean hormonal concentrations of cortisol and estradiol across 9 weeks at different measurement time points, baseline – values at rest, post-fat – values immediately after fatiguing protocol, post-fat30 – values 30 min after fatiguing protocol, *p-value < 0.05, **p-value < 0.01, ***p-value < 0.001. (b) Estimation plot of mean peak torque between first and last repetition. av PT first reps – average Peak Torque of the repetitions 2–6, av last reps – average peak torque of the repetitions 26 to 30, change scores – decrease of average peak torque between repetitions 2–6 and 26–30.
Body composition
The parameters of body composition are presented in Supplemental Table 1 in the Appendix. There was no significant difference observed across the different measurement timepoints in any of the assessed parameters.
Functional skeletal muscle parameter
Estradiol concentrations and total work (Figure 2(a)) as well as the concentric peak torque (Figure 2(b)) follow a similar trend across the time span peaking at week 4.
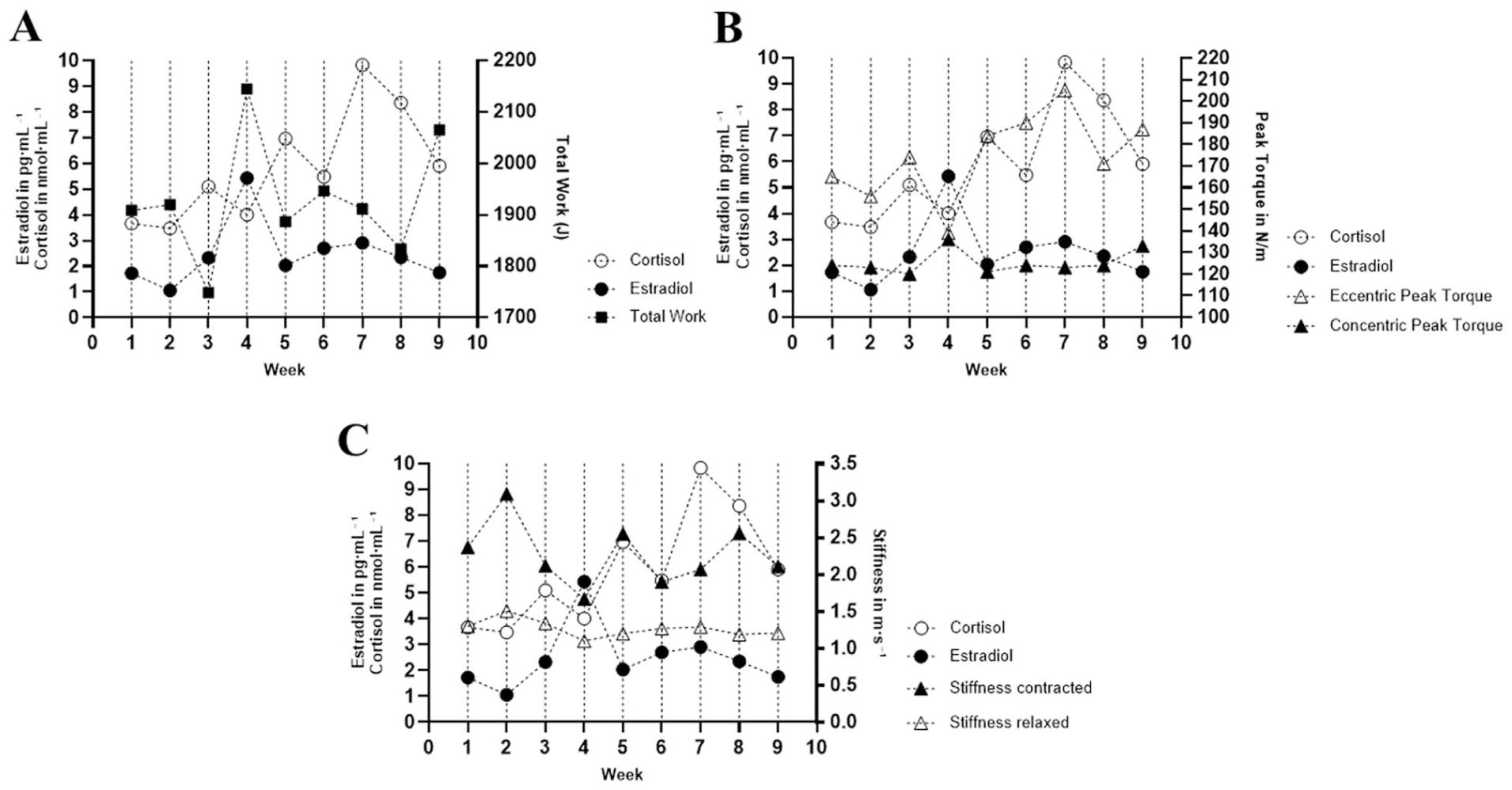
(a) Estradiol and cortisol baseline concentrations and total work across 9 weeks. (b) Baseline cortisol and estradiol concentrations and eccentric and concentric peak torque across 9 weeks; pg·mL−1– picogram per milliliter, J – Joule, nmol·L−1 – nanomole per liter, N/m – Newton meter. (c) Baseline estradiol and cortisol concentrations and muscle stiffness across 9 weeks. pg/mL – picogram per milliliter, nmol/L – nanomole per liter.
Pearson’s correlation analysis did not reveal a significant relationship between estradiol concentrations at rest and total work (r = 0.536, p = 0.137), average work per repetition (r = 0.550, p = 0.125), concentric peak torque (r = 0.591, p = 0.094), or eccentric peak torque (r = −0.346). There was no statistically significant cross-correlation between baseline estradiol concentration and total work, concentric peak torque, eccentric peak torque and average work at any of the examined lags. The table displaying the cross-correlation coefficients of estradiol and the evaluated parameters can be found in the Appendix (Supplemental Table 2 in Supplemental Data).
Results showed a similar trend across the time span of baseline cortisol concentrations and eccentric peak torque. Pearson’s correlation analysis revealed a significant relationship between cortisol and eccentric peak torque (r = 0.723, p = 0.028) and is displayed in Figure 3(b). No significant correlation was found between cortisol concentrations at rest and total work (r = −0.270, p = 0.482), average work per repetition (r = −0.229, p = 0.553), and concentric peak torque (r = −0.251, p = 0.842). The analysis of the cross-correlation and baseline cortisol concentrations and total work, average work per repetition, eccentric peak torque, or concentric peak torque did not reach significance at any of the examined lags (Supplemental Table 3 in Supplemental Data).
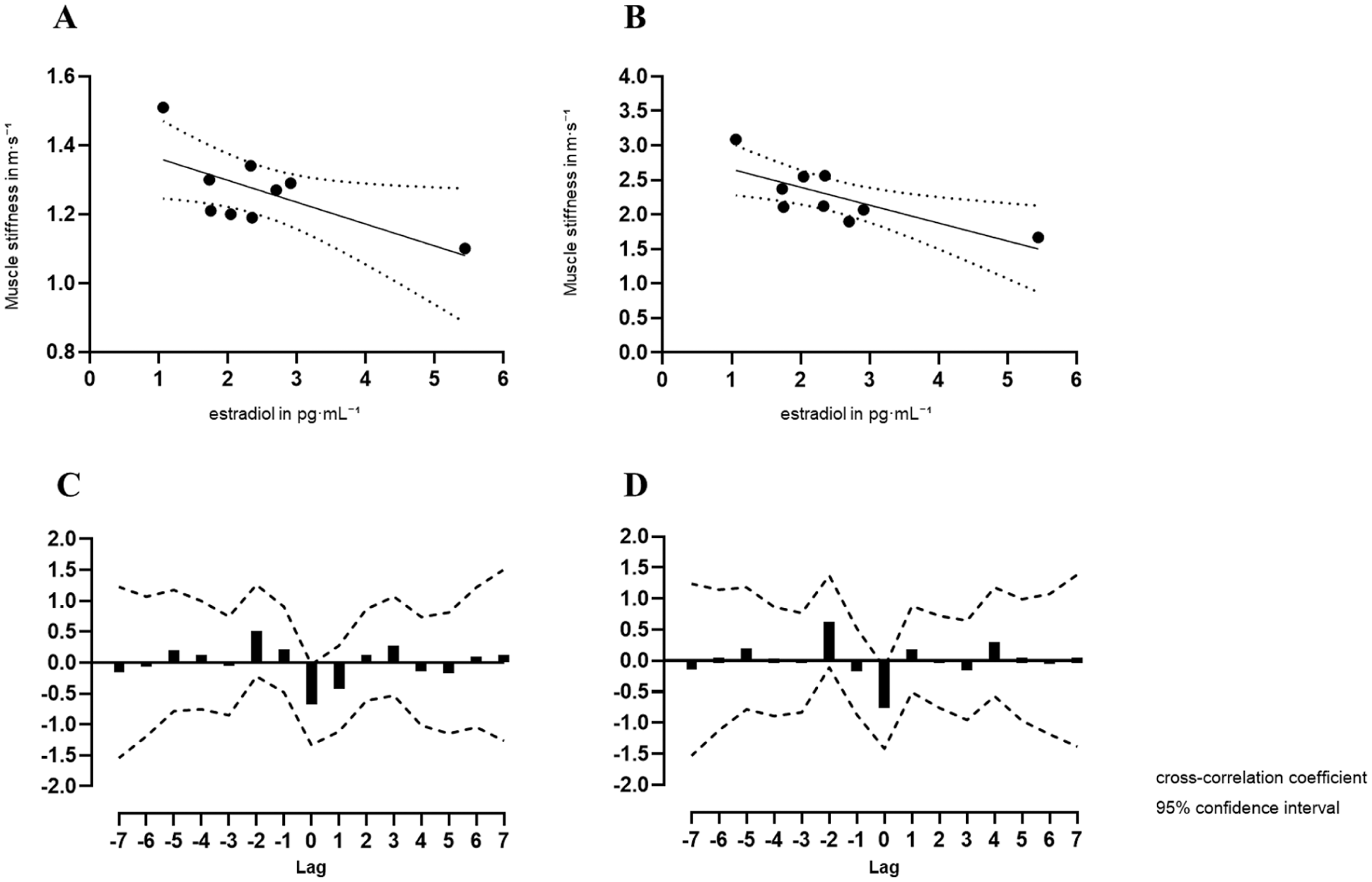
(a) Scatter plot of the linear relationship between baseline estradiol concentrations and muscle stiffness in a relaxed stated, (b) scatter plot of the linear relationship between baseline estradiol concentrations and muscle stiffness in a contracted stated, (c) cross-correlation coefficient of muscle stiffness and baseline estradiol concentrations across 15 lags in a relaxed stated. (d) In a contracted state, pg·mL−1 – picogram per milliliter, m·s−1 – meter per second.
A paired-samples t-test was conducted to compare the average peak torque (avPT) between the first repetitions (av first reps – 2–6) and the last repetitions (av last reps – 26–30) during the fatigue protocol (Figure 1(b)). The results revealed a statistically significant difference between the first and last repetitions, t (8) = 9.29, p < 0.001. The effect size, measured by Cohen’s d, was large (d = 3.10), indicating a substantial difference in peak torque between the first and last repetitions. Pearson’s correlation analysis did not reveal a significant relationship between change scores and baseline estradiol concentrations (r = 0.316, p = 0.408) or cortisol concentrations at rest (r = −0.36, p = 0.928). There was no significant cross-correlation between change scores and baseline estradiol or cortisol concentration at any of the examined lags.
Structural skeletal muscle parameter
Muscle stiffness
Pearson’s correlation analysis showed a significant negative correlation between estradiol concentrations at rest and muscle stiffness in a relaxed state (r = −0.68, p = 0.044) and in a contracted state (r = −0.763, p = 0.017, Figure 3(a)). No significant correlation was found between baseline cortisol concentrations and muscle stiffness relaxed (r = −0.286, p = 0.456) or muscle stiffness contracted (r = −0.087, p = 0.842). The cross-correlation between estradiol concentrations at rest and muscle stiffness in a relaxed or contracted state did not reveal a significant correlation at any of the examined lags (Supplemental Data Table 4).
Discussion
The case report study evaluated skeletal muscle properties and hormone concentrations over a 9-week period in one person with endometriosis who underwent surgery and was receiving Dienogest treatment at the time of data collection. Despite physical activity being commonly recommended as a therapeutic measure, skeletal muscle health and function of persons affected by endometriosis have been largely overlooked.5,8,9 Muscle activity may attenuate inflammatory processes through the release of myokines, which exert anti-inflammatory and immunomodulatory effects. 4 It is consequently assumed that there could be reciprocal effects between skeletal muscle health and endometriosis. 4 Hence, we investigated muscle stiffness, strength parameters and muscular fatigue to improve the understanding of the interaction between endometriosis and muscular function. Further, hormone concentrations were evaluated at rest and after physical strain.
The average value of the baseline cortisol concentrations were in line with previous results indicating lower levels of salivary cortisol in persons with endometriosis. 10 In unaffected individuals, cortisol exerts an adaptive response to physical stress by promoting homeostasis. It modulates energy metabolism through the upregulation of gluconeogenesis, glycogenesis, and glycogenolysis, thereby increasing circulating glucose concentrations and facilitating glucose uptake and utilization in skeletal muscle.10,11 Our findings showed a significant linear correlation between baseline cortisol concentrations and eccentric peak torque values, indicating a positive influence of cortisol on muscular performance by increasing energy availability. However, no clear temporal association was identified between cortisol levels and eccentric peak torque, as cross-correlation analyses yielded non-significant results. Subsequent to the fatiguing protocol, cortisol concentrations decreased, reaching their lowest measured values 30 min after. Previous research has demonstrated that both elite and non-elite athletes, regardless of oral contraceptive use, experience a sharp increase in cortisol levels following competitions and intense training sessions.12–14 Some studies showed mixed cortisol responses to prolonged exhaustive exercise patterns. Some well-trained athletes may display a dampened cortisol response since metabolic strain may not sufficient to promote adapting mechanisms or reaction is blunted due to chronic fatigue and overtraining. 15 However, since our participant is only recreationally active, an influence of training status on cortisol concentrations was not expected. Hence, we speculate that the reduced cortisol response observed in this case may suggest impaired functioning of the hypothalamic-pituitary-adrenal (HPA) axis. This axis is crucial in managing stress, as it triggers the release of the glucocorticoid cortisol during neuroendocrine responses to pain and stress. 16 Symptoms of endometriosis 17 can cause persistent stress, potentially leading to HPA axis dysregulation and, ultimately, hypocortisolism.10,11,16,18 Hypocortisolism indicates an overactive adrenal gland, which can result in adrenal fatigue. 11 Hence, the steep drop of cortisol concentrations points to the possibility that the hormonal answer to physical strain is impaired and HPA axis dysregulation seems to be present even though the participant underwent surgery.18,19 However, as only cortisol concentrations were measured, attributing these findings to HPA axis impairment remains speculative, since key indicators such as corticotropin-releasing hormone (CRH) and adrenocorticotropic-releasing hormone (ACTH) as precursors of cortisol were not assessed. CRH triggers the release of ACTH which subsequently stimulates the adrenal glands to produce cortisol. 14
These findings support the notion that the underlying pathogenetic mechanisms are not fully addressed by surgical intervention alone. 18 Further, since cortisol exerts anti-inflammatory effects, chronically low levels of cortisol and a dampened response to physical strain may compromise regeneration after physical exertion. Hence, patients with low baseline cortisol concentrations seem to be more prone to overtraining symptoms and fatigue, and may display a higher value of perceived exertion.
Further research is necessary to investigate whether the kinetics of biomarkers related to muscle damage and fatigue differ between individuals with endometriosis and such without to tailor exercise programs according to their tolerance of metabolic strain.
Our results demonstrated a significant negative linear relationship between estradiol concentrations at rest and muscle stiffness, both in relaxed and voluntarily contracted muscles. This may indicate that higher estradiol concentrations lead to lower muscle stiffness. Muscle stiffness measured by shear wave elastography may vary significantly between menstrual cycle phases with high and low estradiol concentrations.20,21 Ham et al. (2020) showed that stiffness of M. rectus femoris in healthy females was 12.8 kPa (±2.6) in resting condition and 82.2 kPa (±31.8) at contraction during the early follicular phase and 11.6 kPa (±2.6) rested and 67.3 kPa (±14.8) contracted at ovulation. However, mean values of our participant were substantially lower with 4.81 kPa (±1.04) in the relaxed state and 16.4 kPa (±5.4) at voluntary contraction. One reason for the high deviation between the examined patient and the healthy population could be the suppression of naturally cyclic hormonal concentrations and on average lower hormone concentrations due to the use of hormone therapy. Dienogest as oral progestin dampens the release of gonadotropin releasing hormone and subsequently the production of steroid hormones in the ovaries through PR mediated actions. It has been also shown, that PR signaling leads to a dampen expression of ER receptors in the glands, stroma of endometriotic lesions, and the eutopic endometrium. 22 However, if ER receptor expression is also dampened in muscle cells stays speculative due to a lack of data. A reduction in ER expression could lead to decreased estrogen sensitivity in muscle tissue, which might contribute to higher muscle stiffness in the examined individual. However, this hypothesis is generated based on data from a single person and should be interpreted with caution. In contrast, cohort-based comparisons between oral contraceptive users and non-users have shown that pill users exhibit lower muscle stiffness on average, 23 suggesting that individual responses or specific hormonal formulations such as progestin-only therapy versus combined pills may differentially affect muscle properties. Furthermore, myofascial dysfunction, manifesting as trigger points within skeletal muscle, is a recognized symptom of endometriotic pathology and may contribute to alterations in skeletal muscle stiffness. 24 However, the reduced stiffness values observed in the patient compared to healthy controls do not necessarily indicate a higher prevalence of myofascial trigger point. 24 Since we have not examined the muscle for trigger points, these considerations are speculative.
Nonetheless, our findings provide compelling evidence that estradiol not only influences the elasticity of tendons and ligaments but also exhibits a dose-dependent relationship with skeletal muscle stiffness. 25
Conclusion
This study offers preliminary evidence that supports further research into the potential role of muscle function in endometriosis. Based on observed hormonal dynamics, we hypothesize that HPA axis may be dysregulated, likely linked to chronic stress. However, it remains to be determined whether this observation can be confirmed in a cohort. While the findings suggest a potential link between hormonal modulation and muscle stiffness, the continuous Dienogest treatment in the examined individual complicates the interpretation of estradiol fluctuations, which were evident besides hormonal therapy. Given the inflammatory nature of endometriosis, elevated biomarkers such as ROS, IL-6, and cell-free DNA may impair post-exercise muscle recovery, though this remains speculative in the absence of inflammatory data. 6 Future research should explore these interactions to better understand the role of physical activity in endometriosis management."
Limitations
As a single-case report, the findings lack generalizability. Further research should include a larger cohort and a healthy control group to evaluate differences between individuals with and without endometriosis and to enable more valid conclusions. The 9-week duration may not capture long-term post-surgical developments, and the absence of progesterone and testosterone analyses limits insight into the full hormonal profile. In this study, we only evaluated m. rectus femoris. However, the physical properties of the abdominal and back muscles may be of importance in patients with endometriosis, as pain is often predominantly localized in these areas. Hence, future research should include a broader spectrum of muscles.
While cortisol is known to correlate with perceived exertion and pain, we focused on objective measures and did not assess subjective outcomes. Future studies should include psychological variables, as well-being and quality of life are known to influence endometriosis symptoms. 26
Supplemental Material
sj-docx-1-pev-10.1177_22840265251406153 – Supplemental material for Skeletal muscle function and hormonal dynamics in a woman with endometriosis: A case report following CARE guidelines
Supplemental material, sj-docx-1-pev-10.1177_22840265251406153 for Skeletal muscle function and hormonal dynamics in a woman with endometriosis: A case report following CARE guidelines by Christine Heinrich, Katharina Herzog, Emelie Kneißler, Daniel Niederer and Michael Behringer in Journal of Endometriosis and Pelvic Pain Disorders
Supplemental Material
sj-docx-2-pev-10.1177_22840265251406153 – Supplemental material for Skeletal muscle function and hormonal dynamics in a woman with endometriosis: A case report following CARE guidelines
Supplemental material, sj-docx-2-pev-10.1177_22840265251406153 for Skeletal muscle function and hormonal dynamics in a woman with endometriosis: A case report following CARE guidelines by Christine Heinrich, Katharina Herzog, Emelie Kneißler, Daniel Niederer and Michael Behringer in Journal of Endometriosis and Pelvic Pain Disorders
Supplemental Material
sj-docx-3-pev-10.1177_22840265251406153 – Supplemental material for Skeletal muscle function and hormonal dynamics in a woman with endometriosis: A case report following CARE guidelines
Supplemental material, sj-docx-3-pev-10.1177_22840265251406153 for Skeletal muscle function and hormonal dynamics in a woman with endometriosis: A case report following CARE guidelines by Christine Heinrich, Katharina Herzog, Emelie Kneißler, Daniel Niederer and Michael Behringer in Journal of Endometriosis and Pelvic Pain Disorders
Supplemental Material
sj-docx-4-pev-10.1177_22840265251406153 – Supplemental material for Skeletal muscle function and hormonal dynamics in a woman with endometriosis: A case report following CARE guidelines
Supplemental material, sj-docx-4-pev-10.1177_22840265251406153 for Skeletal muscle function and hormonal dynamics in a woman with endometriosis: A case report following CARE guidelines by Christine Heinrich, Katharina Herzog, Emelie Kneißler, Daniel Niederer and Michael Behringer in Journal of Endometriosis and Pelvic Pain Disorders
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data is available upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
