Abstract
Background:
Energy intake (EI) from daily meals, corresponding to total energy expenditure (TEE), is essential for maintaining good physical condition and performance in athletes. Athletes who continue high-intensity training while consuming insufficient energy from their daily diet are at risk for health problems, such as iron deficiency anemia, endocrine dysfunction, and energy metabolism disorders.
Objectives:
This study aimed to examine the altered health risks in athletes when EI was increased to meet TEE from a daily diet.
Design:
Case series study with dietary intervention.
Methods:
Participants were six female long-distance runners classified as Tier 3. TEE was measured before the nutritional intervention using the doubly labeled water technique to determine the individual target EI and meal content during the intervention. The nutritional intervention was implemented for 6 weeks; sports dietitians provided weekly nutritional advice to each participant on how to eat to meet TEE, in addition to providing appropriate meals (breakfast and dinner) throughout the intervention period. Body composition measurements, resting metabolic rate (RMR) measurements, and blood tests were performed before and after the 6-week nutrition intervention.
Results:
A mean negative energy balance before the intervention in all participants (−548 kcal/day) improved to −31 kcal/day during the intervention. Carbohydrate intake improved to recommended levels (>7 g/kg body weight/day). Five participants were classified as having stage 1 anemia before the intervention, but the condition of four participants improved or was in a recovery process after the intervention. In addition, the suppression of RMR (21.8 and 24.2 kcal/kg fat-free mass/day) in two participants returned to the normal range.
Conclusion:
In this case series, consciously increasing daily EI from meals to meet TEE was associated with a favorable change in health risks, such as improved iron status, recovered from RMR suppression, and decreased cortisol levels. Additionally, practical nutritional advice from sports dietitians appeared to support athletes in overcoming EI challenges.
Registration:
Effect of the diet avoiding relative energy deficiency on physical condition in female long-distance runners. https://center6.umin.ac.jp/cgi-open-bin/ctr/ctr_view.cgi?recptno=R000055221;UMIN000048455
Plain Language Summary
Consciously increasing energy intake to meet total energy expenditure from daily meals is associated with favorable changes for the prevention of anemia and maintenance of athletes’ good physical condition. Practical nutritional advice from sports dietitians helps athletes overcome dietary challenges.
Keywords
Introduction
Performance and training adaptations in athletes are closely linked to adequate and periodized intake of energy and macronutrients. 1 Many athletes, especially female athletes and participants in endurance and aesthetic sports, are chronically energy-deficient, which impairs their performance, growth, and health. 2 To maintain stable body weight (BW), energy intake (EI) must, over time, equal energy expenditure, a state known as energy balance (EB). 3 The energy requirement of an individual is defined as the level of EI from food that balances the total energy expenditure (TEE), consistent with long-term good health. 4 This concept also applies to athletes; athletes require greater EI to maintain EB due to a higher TEE. A meta-analysis of energy deficiency and physical performance demonstrated that a greater negative EB is associated with a decline in physical performance. 5 Therefore, it is important for athletes to maintain their long-term health and physical performance by consuming an energy-balanced diet. In addition, each macronutrient has unique properties that influence health through metabolic and endocrine responses, 6 and all macronutrients are sources of energy. For consecutive days of prolonged endurance running, achieving EB is recommended, alongside the provision of sufficient carbohydrates to meet exercise load demands (i.e., 6–8 g/kg/day for moderate-to-high-intensity exercise of 1–3 h) 7 and consuming sufficient protein to meet daily nitrogen balance (i.e., 1.2–2.0 g/kg/day), to support tissue recovery and adaptations. 8
However, accurately measuring TEE in athletes’ real-life conditions is difficult. The doubly labeled water (DLW) technique is the only tool for accurately assessing energy expenditure under actual living conditions. 9 The DLW method eliminates the need for wearing devices, which reduces psychological pressure on athletes. Motonaga et al. 10 measured the TEE in Japanese male long-distance runners using the DLW method and reported a negative EB of 730 kcal/day. A previous study on elite Kenyan endurance runners also noted negative EB during the period of intense training. 11 A negative EB indicates a low EI relative to the expended energy. Previous studies12,13 using DLW reported that EI in elite female soccer players was lower than TEE and suggested that individualized education and behavior change strategies should focus on “fueling” to optimize health and performance. Energy deficits with concomitant reductions in carbohydrate intake may further increase the risk of iron deficiency owing to greater relative increases in hepcidin levels. 14 In addition, a previous study 15 reported that only 4 weeks of intensified practice reduced energy metabolism-related indices such as resting metabolic rate (RMR) and triiodothyronine (T3) level, and increased cortisol level. Another study 16 observed changes in bone metabolism markers when consuming diets with different energy and carbohydrate availabilities and concluded that nutritional support with adequate energy and carbohydrates appears to reduce unfavorable bone turnover responses to exercise in elite endurance athletes. Previous studies have shown that various health risks in athletes, such as anemia, 17 RMR suppression, 18 and impaired bone health, 19 are caused by energy and carbohydrate deficiencies.
To improve the menstrual status of exercising women, a previous study 20 attempted to increase EI (330 ± 65 kcal/day) over 12-month. Participants were given only energy bars and no intervention to the daily diet. The group that increased EI exhibited improved menstrual function. Nevertheless, the problems that more active athletes face in real life vary widely, and the physical changes caused by consuming adequate energy from their daily diet remain unclear. Fahrenholtz et al. 21 demonstrated a digital intervention for sports nutrition knowledge in female athletes and found strong evidence of improved sports nutrition knowledge, however, did not address the impact of improving actual EI.
This study aimed to determine whether a dietary intervention that increases EI to meet TEE results in altered health risks in athletes. We hypothesized that energy metabolism-related conditions such as RMR suppression, and nutritional conditions such as iron deficiency in female long-distance runners would be improved after the dietary intervention.
Methods
Study design
This case series study implemented a 6-week dietary intervention in a non-randomized, non-blinded setting, following a pre-post design. The research period was from April to July in 2023, schematic overview of the study design is described in Figure 1. All measurements were performed during the regular training season. TEE was measured before nutritional intervention using the DLW technique to determine the individual target EI and meal content during the study. The nutritional intervention was implemented for 6 weeks. All participants lived in the same dormitory, and a team dietitian provided breakfast and dinner during the intervention. The participants were required to consume adequate energy to meet their individual TEE from their daily meals, including lunch and snacks. During the intervention, sports dietitians provided each participant with weekly nutritional advice on how to eat. Body composition, RMR measurements, and blood tests were performed before and after the intervention. The reporting of this study conforms to the CARE guidelines. 22
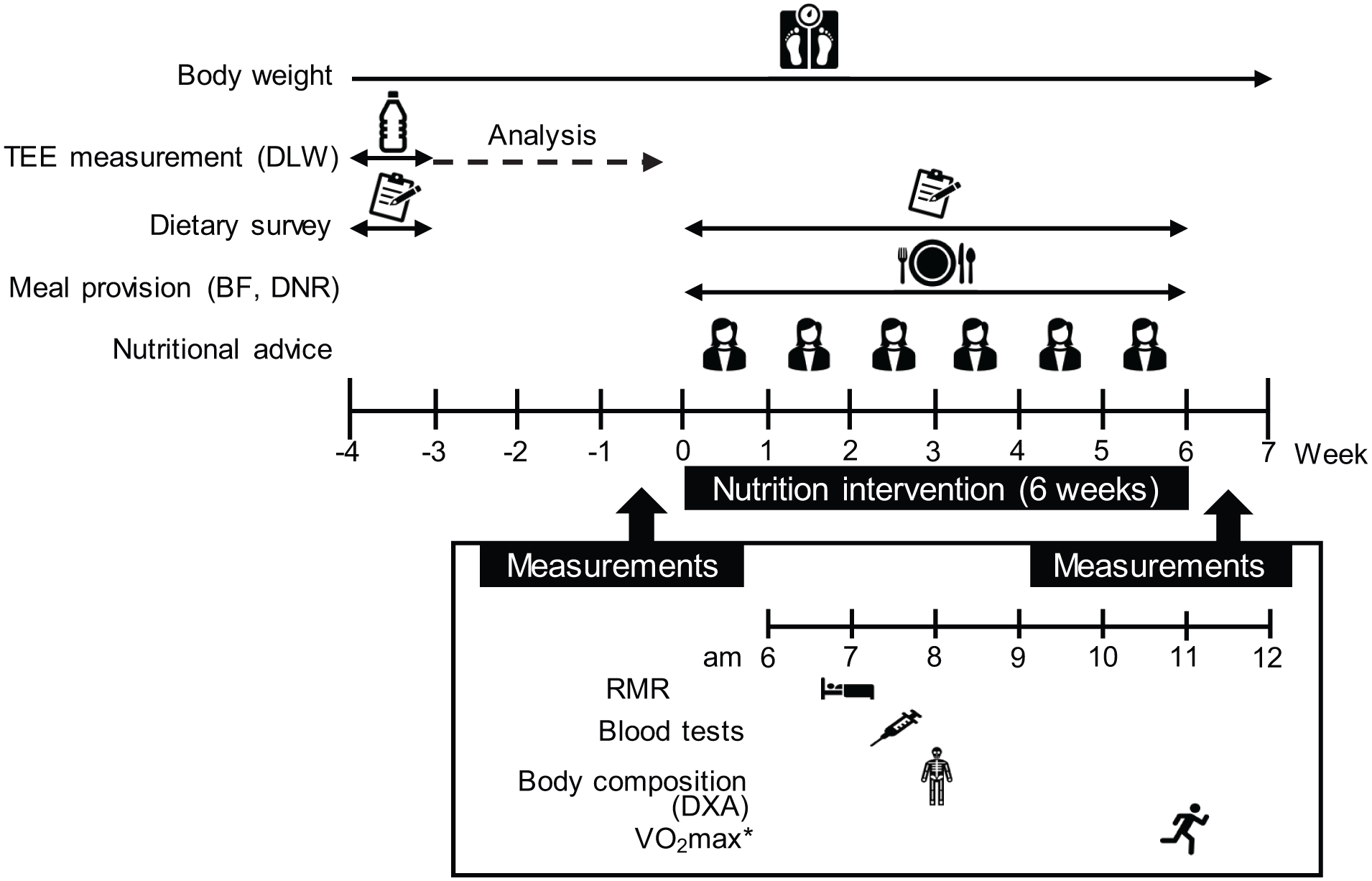
Schematic overview of study design.
Participants
Japan has a unique sports system that allows corporate employees to devote themselves to sports while still maintaining their employee status; female long-distance runners were recruited from a corporate team in Tokyo, Japan. They lived in a dormitory of the company and shared similar lifestyles. The participants worked for the company from 9:00 a.m. to 5:00 p.m. on weekdays and underwent running and physical training every day before and after work. The inclusion criteria were as follows: age 18–30 years, non-smoking, not using thyroid hormone medications, not pregnant, and no eating disorders. Of the 11 team members, 6 runners who met the inclusion criteria were selected as participants for this study. All participants were classified as Tier 3 23 and competed in national and regional competitions. The Eating Attitudes Test-26, Japanese version 24 was used to screen for the risk of eating disorders. Each participant completed the test alone prior to the intervention, and no one scored above 20, which screens for risk of an eating disorder. Although some participants had previously experienced self-reported menstrual dysfunction such as amenorrhea, there were no amenorrheic runners at the beginning of the study. There were no users of oral contraceptives or hormonal intrauterine devices.
All participants received an oral explanation of the study and provided written informed consent before starting it. All contents and procedures of this study were approved by the Ethics Review Committee on Research with Human Subjects of Waseda University (2022-116) and conducted in accordance with the Declaration of Helsinki. The trial was registered on July 25, 2022, at the University Hospital Medical Information Network (ID: UMIN000048455).
Measurement of TEE
TEE was assessed using the DLW method, following the IAEA guidelines 25 and the measurement procedures of the National Institute of Health and Nutrition of Japan. 26 None of the runners was currently losing or gaining BW, and confirmed their BW fluctuation was within 2% from 1 month prior to DLW administration. DLW measurements were performed using a multipoint method. On the evening of the designated day, the runners collected baseline urine sample at the dormitory of the team, then, approximately 0.06 g of 2H2O per kg of BW (99.9 atom%; Sigma Aldrich, MO, USA) and 1.4 g H2 18 O per kg of BW (10.0 atom%; Taiyo Nippon Sanso, Tokyo, Japan) were administered orally to each participant. To ensure a full dose of all isotopes, participants rinsed the cup and their mouths twice with water and swallowed. Urine collection was initiated at least 10 h apart from dosing, as it takes 4–8 h for urine to reach equilibrium. Participants collected their own second void urine samples for the next 8 days at the same predetermined time (±10 min) of the day, and the exact sampling time was recorded. All samples were wrapped with parafilm and stored frozen at −30°C in an airtight container, and then analyzed in the analysis laboratory (Taiyo Nippon Sanso). The isotope enrichment of the urine samples was measured using an isotope ratio mass spectrometer (Sercon Limited, Cheshire, England). Isotope elimination rates were used to calculate CO2 production. TEE analysis was performed using Weir’s equation, 27 based on the CO2 production rate, and the food quotient (FQ) from diet records. The calculation assumed that under conditions of perfect nutrient balance, the FQ equals the respiratory quotient (RQ). The mean FQ in this study was 0.90. Only TEE that passed quality checks according to IAEA guidelines 25 were used.
Measurement of RMR
RMR was measured using the Douglas bag technique early in the morning following an overnight fast, with reference to previous studies.28,29 The participants avoided intense training the day before the measurement and did not eat or drink after dinner, except for water. On the measurement day, participants were transported from the dormitory to the laboratory by car in a resting and fasting state. They rested in the supine position for at least 30 min in a quiet room at room temperature (22°C–24°C) until their heart rate reached the individual’s daily resting state. After the habituation period with the mask, expired gas samples (10 min each) were collected in Douglas bags. The volume of the expired gas was measured using a certified dry gas volume meter (DC-5A; Shinagawa, Tokyo, Japan). The O2 and CO2 concentrations were analyzed using an expiration gas analyzer (AE100i; Minato Medical Science Co., Ltd., Osaka, Japan), and the gas exchange results were converted to RMR (kcal/day) using Weir’s equation. 27 The measurements were repeated until the RMR coefficient of variation was <5%, and the mean value of the two samples was used for the analysis (coefficient of variation 1.9%). RQ was calculated as the ratio of the amount of carbon dioxide produced to the amount of oxygen consumed during the RMR measurement. In a previous study by the authors on 93 Japanese female athletes, the first quartile of the range of RMR per fat-free mass (FFM) was 24.5 kcal/kg FFM/day. 29 In addition, RMR of Japanese athletes with menstrual disorders was at the same level, which was significantly lower than that of the eumenorrheic athletes. 30 In this study, therefore, runners who showed RMR below this value were defined as metabolically suppressed.
Physical activity level was calculated by dividing TEE by RMR measured before intervention.
Measurement of VO2max
At the measurement day before the intervention, the participants completed a maximal graded exercise test to determine VO2max using a treadmill, as an indicator of endurance capacity. After a 3-min rest period, participants started running at 180 m/min, with an increase of 20 m/min per 3 min, with a grade of 3% until the participant reached exhaustion. The expired gases, pulmonary VO2, and VCO2 were analyzed using the breath-by-breath method with an automated gas analyzer (AE310S; Minato Medical Science Co., Ltd.). At the end of each stage, the participants reported a rating of perceived exertion (RPE) using the Borg scale. 31 The endpoints for the maximal exercise test were met if at least two of the following four criteria were achieved: (a) respiratory exchange ratio >1.10, (b) leveling off of VO2 despite an increase in workload, (c) heart rate reaching approximately 95% of the age-predicted maximal heart rate (220 minus age), and (d) RPE>19.
Measurement of body composition and bone status
BW was measured with minimal clothing to the nearest 50 g using an electronic scale (UC-321; A&D Co., Ltd., Tokyo, Japan) in the morning after fasting and voiding. Standing height without shoes was measured to the nearest 0.1 cm using a stadiometer (YG-200; Yagami Inc., Tokyo, Japan). Body mass index was calculated by dividing BW (kg) by the square of height (m). Dual-energy X-ray absorptiometry (Horizon A; version 12.4:3, Fan beam, auto whole-body mode; Hologic Inc., Bedford, MA, USA) was used to measure bone mineral density (BMD) and body fat percentage. All scans and analyses were conducted by an experienced orthopedic surgeon and analyzed using Hologic software (version. 5.6.0; Hologic Inc.). Classic calibration was performed before the measurements, and the mean coefficient of variance of the measurements was <1%. Fat mass was calculated based on BW and body fat percentage, whereas FFM was calculated as BW minus fat mass.
Dietary survey and nutrition intervention
Before the intervention, participants completed a 7-day weighed food record using a food scale, and digital photographs of their meals were taken using their smartphones. Energy and nutrient intakes were calculated using nutrition calculation software based on Japanese standard food tables (Wellness 21; Top Corporation Business System, Okayama, Japan). EB was calculated as EI minus TEE.
The research team presented the team dietitian with menus of breakfast and dinner at the dormitory to ensure that the participants could consume at least two-thirds of the nutrients recommended for long-distance runners as outlined in the International Association of Athletics Federations Consensus Statement 2019: Nutrition for Athletics. 32 During the intervention period, the team dietitian cooked and provided breakfast and dinner while measuring the weight of all foods and seasonings. The participants were required to consume all the provided food and asked to record all their meals, including lunch and snacks, and to document them with photographs. Participants were instructed to consume the individually needed amount of cooked rice for breakfast and dinner; a team dietitian checked whether the participants had any leftover provided foods. They were interviewed weekly throughout the intervention period by sports dietitians, who were the authors of this study, to confirm their daily dietary intake. Simultaneously, sports dietitians provided individualized nutritional advice to ensure that the participants consumed a balanced diet with sufficient energy to meet TEE measured using the DLW method. The research team determined the content of individual dietary guidance based on the energy and nutrient intake status of the previous week. Sports dietitians provided participants with whether the lunch and snack choices they had made were appropriate and confirmed how they would eat for the following week. For example, if there was not enough time and participants just ate something simple for lunch, sports dietitians showed the way when and what to eat for snacks, such as rice balls and fruits. In particular, the guidance focused on ensuring staple food intake, aiming to obtain 7 g/kg/day of carbohydrates 32 since protein intake before the intervention was within the recommended level for athletes (1.3–1.8 g/kg BW/day). 8
Blood tests
Venous blood samples were collected after morning fasting to analyze concentrations of serum T3, growth hormone, insulin, insulin-like growth factor 1 (IGF-1), glucose, triacylglycerol, and iron status (serum ferritin, iron, hemoglobin, and total iron binding capacity (TIBC)), total- and high-density lipoprotein (HDL)- cholesterols, and plasma cortisol. Transferrin saturation (TSAT) was calculated as (serum iron / TIBC) × 100. LDL-cholesterol level was calculated as follows: (total cholesterol minus HDL-cholesterol) minus triacylglycerol/5. The blood analyses were outsourced to the testing company (LSI Medience Corporation, Tokyo, Japan). T3, insulin, ferritin, and cortisol levels were measured using chemiluminescence immunoassays. The growth hormone and IGF-1 levels were measured using an electrochemiluminescence immunoassay. Glucose, triacylglycerol, and HDL-cholesterol were measured using the enzymatic method. Ferritin and TIBC levels were measured using colorimetric methods. Hemoglobin levels were assessed using the sodium lauryl sulfate method. Iron deficiency status was categorized based on a previous study. 33 Stage 1 iron deficiency status was defined as including all of the following: the serum ferritin <35 ng/mL, Hb >11.5 g/mL, and TSAT >16%. “Recovering” (Table 3) was defined as an improvement in any of these three indicators. TSAT below the normal range (<20) was considered to be iron deficiency. Elevated resting cortisol was defined as a level above the normal range (3.7–19.4 μg/L) by the testing company.
Questionnaires
Age at menarche, menstrual status at the start of the study, and past experiences of menstrual abnormalities were determined using a self-administered questionnaire. Amenorrhea was defined as the absence of menstrual cycles for more than 90 days. 34 The participants reported their past and present physical conditions, such as bowel movements, sleep time, injuries, and dietary histories including disordered eating. Daily running distance was recorded by the participants.
Statistical analysis
Since the number of participants was small, the measured data before and after the intervention were shown individually in the tables. SPSS Statistics (version 29; IBM Japan, Tokyo, Japan) was used for statistical analysis. BW between before and after intervention, EB and carbohydrate intake before and during the intervention were compared using the Wilcoxon signed-rank test and statistical significance was set at p < 0.05.
Results
Table 1 presents the baseline characteristics of the participants. The range of TEE was 2299–3014 kcal/day, and the physical activity level was 2.3–2.8. Table 2 summarizes the energy and carbohydrate statuses before and during the intervention. The range of EI before and during intervention was 1794–2403 and 2216–2892 kcal/day, respectively. All participants had a negative EB (−981 to −261 kcal/day) before the intervention, but EB during the intervention improved to −231 to +151 kcal/day (p = 0.028). Carbohydrate intake increased significantly during the intervention (p = 0.026). The weekly weight fluctuation rate during intervention was within 2% for four of the six participants, and within 3% for two, however, a significant difference was not observed between before and after intervention (p = 0.34). As T3, growth hormone, IGF-1, and triacylglycerol levels were within the reference range in all participants both before and after the intervention, the status of other parameters (bone, anemia, metabolic status, and cortisol level) is shown in Table 3. Before the intervention, five of the six participants were in stage 1 iron deficiency status, but all had improved or were in the recovery process after the intervention. The RMR of two participants (#1 and #5) was found to have suppressed before the intervention which was lower than 24.5 kcal/kg FFM/day (21.9 and 19.9 kcal/kg FFM/day, respectively); however, RMR suppression improved after the intervention. RQ changed from 0.76 to 0.87 for #1 and from 0.74 to 0.83 for #5. Cortisol level of participant #1 returned within the normal range following the intervention.
Participant’s characteristics before intervention (baseline).
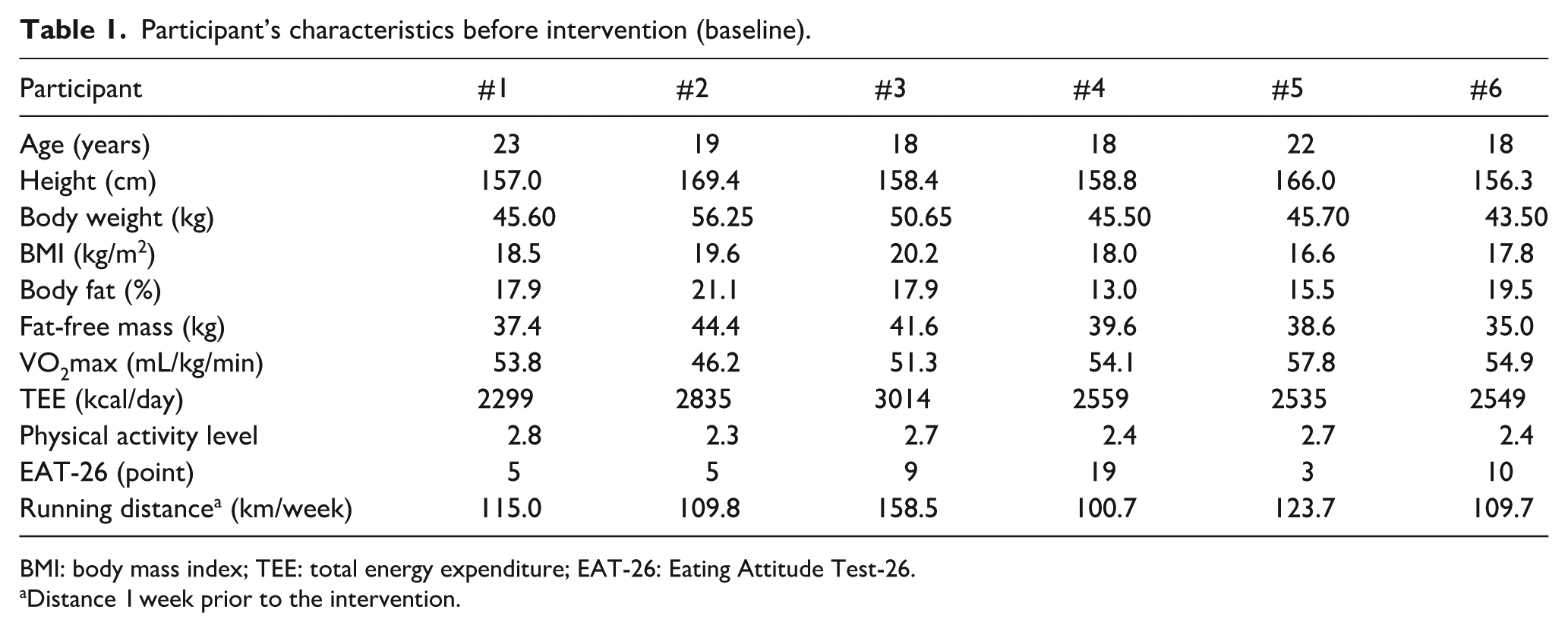
BMI: body mass index; TEE: total energy expenditure; EAT-26: Eating Attitude Test-26.
Distance 1 week prior to the intervention.
Energy and carbohydrate intakes before and during intervention.

TEE: total energy expenditure; EI: energy intake; EB: energy balance; CHO: carbohydrate; BW: body weight. SD: standard deviation.
Mean EI during intervention minus TEE.
Physiological data before and after intervention.

Values outside the reference range are shown in bold. LS: lumbar spine; BMD: bone mineral density; TSAT: transferrin saturation; RMR: resting metabolic rate; FFM: fat-free mass; RQ: respiratory quotient.
Categorized based on Peeling et al. 33 Stage 1 was defined as serum ferritin <35 ng/mL, Hb >11.5 g/mL, and TSAT >16%. Recovering was defined as an improvement in any of the three indicators.
Classified as iron deficiency due to low TSAT value.
Discussion
The study observed that a 6-week intervention to meet TEE involving the provision of meals and nutritional advice improved physiological markers in female long-distance runners, including iron deficiency, metabolic suppression, and elevated cortisol. This study is the first to report that an appropriate diet containing adequate energy can improve several potential outcomes associated with an athlete’s health.
It is difficult to measure the TEE of athletes who perform complex movements daily. Therefore, this study was conducted on runners using the DLW technique, which is the gold standard for measuring TEE. This method is safe and noninvasive, and its notable advantages include objectivity, minimal interference with participants’ daily activities, accuracy, and precision. 35 An energy-balanced status, in which EI matches TEE, is essential to an athlete’s diet; however, the EB of the participants before the intervention was negative. Consequently, in this study, dietary interventions focused on increasing EI to meet TEE. Sports dietitians, the authors of this paper, provided individual nutritional guidance once a week and instructed the participants to eat breakfast and dinner in the dormitory, ensuring no leftovers. The necessary amount of staple food (rice) per meal was presented individually. In addition, we provided participants with instructions on how to choose their own lunch and supplementary snacks. As a result, mean EB improved significantly from −548 kcal to −31 kcal/day (p = 0.028), and carbohydrate intake increased to the recommended level of >7 g/kg BW/day (p = 0.026). Although it is possible that adding energy may increase BW and body fat, BW and fat mass did not change over the 6 weeks, and FFM was maintained in this study. Maintenance of BW and body composition is an indication of being in a state of EB where EI equals TEE. 36 In addition to daily high-intensity long-distance running, the participants also performed high-intensity and intermittent training 37 two or three times per week. The measured TEE included energy expenditure of all training regimens and NEAT due to commuting or working of the participants. Therefore, the nutritional intervention method used to obtain appropriate energy to meet TEE in this study is meaningful.
Impaired bone health is a potential health outcome of REDs, 38 however, a change in the delayed onset of menarche is impossible, and low BMD z-scores cannot be improved in a short duration by a 6-week dietary intervention. At the beginning of this study, two participants had a lumbar spine BMD z-score of < −1 (low BMD), and one had a z-score of < −2 (osteoporosis). 34 Indeed, bone status of the participants remained unchanged after the intervention. Thus, the present study focused on changes in indicators of energy deficiency in athletes, such as hematological status and energy metabolism (Table 3).
Iron deficiency status was classified based on a previous study of athletes, 33 and five participants were in stage one of iron deficiency before the intervention. The mean iron intake before the intervention was approximately 14 mg (6.6 mg/1000 kcal), which was greater than that of elite female athletes in a previous study. 39 However, intense exercise elevates cytokine levels, which, in turn, boost hepcidin production in the liver. 40 A recent study 41 suggests that an energy-deficient diet or low carbohydrate availability may increase hepcidin levels without causing inflammation. The upregulation of hepcidin negatively affects iron transport and absorption channels, which explains the potential mechanism underlying iron deficiency in athletes. 42 Although we did not measure hepcidin levels, it is possible that negative EB prior to the intervention affected iron metabolism. After the intervention, iron deficiency improved in four participants, including two in the process of recovery. The reason iron deficiency status did not completely improve in 6 weeks in participant #2 may be that the degree of improvement in EB was less than that of the other participants (Table 2). By contrast, participant #6, whose status did not improve in this study, had ferritin levels <35 ng/mL before the intervention. Serum iron level and TSAT decreased even after the intervention. Her secondary amenorrhea resolved 3 months prior to the start of this study, which may have increased her iron needs.
A previous study 15 reported that athletes undertook a 4-week intensified endurance training intervention without any apparent increase in EI led to reduced RMR. An 8% reduction in RMR has been reported in elite male endurance athletes with low EI compared to those with adequate EI. 43 Similar results in RMR have been reported in athletes with amenorrhea compared with eumenorrheic athletes.44,45 These studies suggest an energy-conserving mechanism for the maintenance of BW and bodily functions under negative EB status. Participants #1 and #5 had menstrual bleeding when they were enrolled in this study; however, both experienced amenorrhea during their careers at university before joining the team. Therefore, low BMD z-scores of these runners may reflect past energy deficiencies. Thyroid hormones help regulate many metabolic functions in the body, and it has been reported that T3 contributes to RMR in athletes. 46 However, the T3 levels of participants #1 and #5 were within the reference range. One previous study 30 reported that female athletes with menstrual disorders had lower T3 levels. Participants #1 and #5 did not have amenorrhea and eating disorders; Suppression of RMR before intervention may reflect previous endocrine status rather than current T3 levels. Thus, this study suggests that adequate EI may help avoid the suppression of RMR. The RQ of #1 and #5 with RMR suppression returned to normal, consistent with studies in healthy Japanese adults who ate normal meals.47,48 This indicates that an increased intake of carbohydrates raises the oxidation rate of carbohydrates as an energy substrate. Participant #5 showed the most observable improvement in EB in this study, and considerable progress may have eliminated the effects of past energy deficiencies and improved RMR suppression and iron deficiency status. In participant #1, cortisol, a regulatory hormone released in response to stress, was elevated above the reference range before the intervention. Because she was a small eater and had gastrointestinal problems, she had previously been in a state of energy deficiency. Thus, many problems exist, including low BMD, iron deficiency, RMR suppression, and elevated cortisol levels. To increase her EI, we advised consuming multiple meals, including supplemental snacks, rather than merely increasing one meal. Except for BMD, her multiple health risks have been improved by increasing EI in this study; therefore, improving daily energy supplementation by using smaller, frequent meals and snacks to address dietary issues is worthwhile.
Strengths and limitations
In this study, the DLW method, the gold standard for TEE measurement, was used to set individualized EI targets, which has the greatest advantage of preventing underestimation of the target EI of female athletes. Using the DLW method eliminates the psychological burden on participants and allows interventions to focus on promoting adequate EI. In addition, the research team, which included multiple certified sports dietitians, facilitated the assessment of the provided meals and the participants’ energy and nutrient intakes without delay throughout the dietary intervention.
However, several limitations should be noted. First, this study was conducted in a small sample size and a limited number of sporting events. Future research with more robust designs (larger sample size, randomized controlled) is warranted to confirm these conclusions. Second, this study was designed to examine the altered health risks in athletes under real-life conditions and did not consider the menstrual cycle when measuring RMR. Third, the underestimation of EI by dietary survey is also listed as a limitation. It has been reported that dietary surveys of athletes tend to underestimate by 10%–45%. 49 However, in this study, breakfast and dinner were prepared and served while measuring the amount of all ingredients. Participants were asked to take photographs of all lunches and snacks during the intervention period, and detailed interviews by sports dietitians were also conducted weekly. Therefore, the rate of underestimation is thought to be smaller than when a dietary survey is conducted on all meals eaten in the day.
Conclusion
In this case series, consciously increasing daily EI from meals to meet TEE was associated with a favorable change in health risks, such as improved iron status, recovered from RMR suppression, and decreased cortisol levels. Future research with more robust designs is warranted to confirm these conclusions. Additionally, practical nutritional advice from sports dietitians appeared to support athletes in overcoming EI challenges.
Footnotes
Acknowledgements
The authors would like to thank the coach for his assistance and participants in this study. We also thank Ms. Madoka Mukai, the team dietitian.
Ethical considerations
The study was approved by the Ethics Review Committee on Research with Human Subjects of Waseda University (2022-116) and conducted in accordance with the Declaration of Helsinki. The trial was registered on July 25, 2022, at the University Hospital Medical Information Network (ID: UMIN000048455).
Consent to participate
All participants received an oral explanation of the study and provided written informed consent before starting it.
Consent for publication
The authors have received consent for publication from the participants.
Author contributions
Motoko Taguchi: Conceptualization; Project administration; Supervision; Formal analysis; Investigation; Methodology; Visualization; Writing—original draft; Writing—review and editing.
Tatsuya Ishizu: Conceptualization; Investigation; Methodology; Data curation; Writing—review and editing.
Mika Goshozono: Funding acquisition; Conceptualization; Investigation; Methodology; Data curation; Writing—review and editing.
Kuniko Moto: Investigation; Data curation; Writing—review and editing.
Nozomi Miura: Investigation; Data curation; Writing—review and editing.
Sumire Endo: Investigation; Data curation; Writing—review and editing.
Eriko Yasuda: Investigation; Data curation; Writing—review and editing.
Suguru Torii: Investigation; Data curation; Writing—review and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Japan Keirin Autorace Foundation (JKA) and its promotion funds from KEIRIN RACE, grant number 2023P-385.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data used to research conclusions in this study are not publicly available due to ethical reasons. Further inquiries can be directed to the corresponding author.
