Abstract
Introduction:
When peritoneal endometriosis is clinically suspected, diagnostic and therapeutic laparoscopy may be offered after failed or unsuitable medical therapy. Our objective was to determine how often endometriosis is verified in these laparoscopies.
Methods:
In our registry-based historical cohort we identified 296 women aged 15–49 years undergoing laparoscopy due to chronic pelvic pain, between 2009 and 2020 at Turku University Hospital. Women without preoperative ultrasound findings and no history of endometriosis were included in the study.
Results:
Macroscopic endometriosis was detected in 52.4% of laparoscopies, and the rate was similar throughout the study period. Peritoneal endometriosis was found in 52% of participants, deep endometriosis in 4.7%, and a small endometrioma in 2%. Women with and without endometriosis reported similar preoperative rates of acyclic abdominal pain (91.8% vs 93.9%, p = 0.64) and dyspareunia (84.6% vs 78.9%, p = 0.32), while dyschezia was significantly more common among women with endometriosis (44.4% vs 26.9%, p = 0.006). The prevalence of dysmenorrhea is not reported, as 62.5% of patients used hormonal therapy commonly aiming to amenorrhea. The median age at the onset of symptoms was significantly lower in women with endometriosis (14 vs 19, p = 0.048).
Conclusions:
Endometriosis was confirmed in 52.4% of the women undergoing laparoscopy due to chronic pelvic pain, and in most women, peritoneal endometriosis was the only finding. However, the other half of the women did not have endometriosis despite having similar pain symptoms. It remains unknown whether patients benefit long-term from undergoing diagnostic and therapeutic laparoscopy.
Introduction
Diagnostic laparoscopy with positive histology has previously been the gold standard diagnostic method for endometriosis. The latest European Society of Human Reproduction and Embryology (ESHRE) guidelines challenge this idea and suggest that laparoscopy should be offered only to women who have no signs of endometriosis in ultrasound (ultrasound negative) or MRI imaging and when empirical treatment has failed or is not suitable. 1
Limited data exist on the prevalence of endometriosis among ultrasound negative women with chronic pelvic pain (CPP). According to the latest systematic review, visible endometriosis is detected via laparoscopy in 64% of adolescents with pelvic pain. 2 In 42% of adult women with CPP, endometriosis occurs. 3 The prevalence of endometriosis is reported to be 21% in adult women undergoing laparoscopic hysterectomy due to CPP. 4
Turku University Hospital, Department of Obstetrics and Gynecology, is a tertiary referral center, and during our study period (2009–2020) diagnostic laparoscopies were performed following current recommendations by the ESHRE. Laparoscopy was performed if a woman had pain symptoms that decreased her quality of life despite hormonal treatment or if hormonal treatment was inappropriate because of pregnancy seeking, side effects, or contraindications. When performing a laparoscopy there was an intention to treat endometriosis if it was found.
Considering this background, the primary aim of our study was to determine how often endometriosis is detected in our diagnostic laparoscopies. In addition, our objective was to determine the annual number of these procedures and to explore the characteristics of these women.
Methods
This retrospective registry study was conducted at the Department of Obstetrics and Gynecology, at Turku University Hospital. Our intention was to identify women who had undergone diagnostic laparoscopy due to suspicion of endometriosis between years 2009 and 2020. The formation of the study cohort is illustrated in detail in Figure 1.
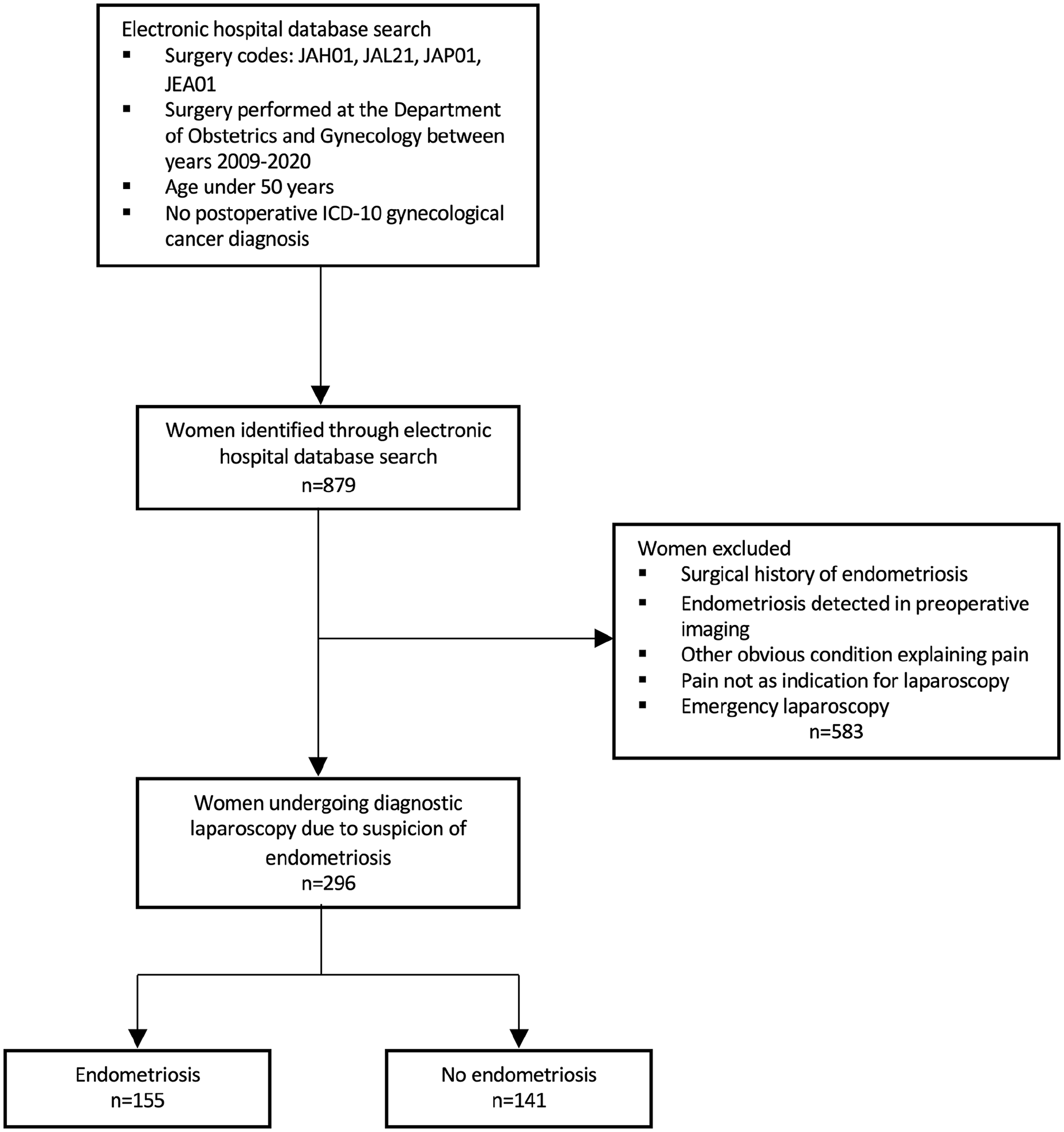
Flow chart of the study population.
We searched for eligible women under the age of 50 from the electronic hospital database by using the Finnish Classification of Surgical Procedures, 5 which is based on the Nordic Classification of Surgical Procedures (NCPS). The following procedures were searched: (1) diagnostic laparoscopy (JAH01) as the main or subsidiary procedure, (2) laparoscopic excision or destruction of peritoneal lesions (JAL21), (3) laparoscopic adhesiolysis in the peritoneal cavity (JAP01), or (4) laparoscopic appendectomy (JEA01) as the main procedure. Laparoscopies performed due to gynecological cancer were excluded from the search by ruling out postoperative International Classification of Diseases, Tenth Revision (ICD-10) diagnosis codes C50-58.
There were 879 women found in the primary data search. All these women had undergone gynecological examination and transvaginal ultrasound prior to laparoscopic surgery. These investigations were performed at the Gynecological Outpatient Clinic by a specialist or a trainee in Obstetrics and Gynecology, who are consistently trained to perform ultrasound imaging.
The women’s medical history was systemically screened by using a general questionnaire that included data on dysmenorrhea and dyspareunia (Supplemental File 1). This questionnaire was routinely used for all the patient groups visiting the Gynecological Outpatient Unit at Turku University Hospital. Patients were also asked about other endometriosis-related pain symptoms (yes/no). A visual analog scale was not systemically used. The women’s medical history as well as clinical examination and ultrasound findings were documented in the electronic patient files by the gynecologist. We retrospectively examined the preoperative and surgical notes from the patient files and excluded women with (1) a previous visual or histological diagnosis of endometriosis via laparoscopy or laparotomy, (2) deep or ovarian endometriosis detected via preoperative ultrasound, (3) other obvious preoperative findings than endometriosis explaining pain symptoms, (4) emergency laparoscopy due to acute pain, and (5) the indication for surgery solely other than pain or suspicion of endometriosis.
After these exclusions, our final cohort consisted of 296 women. These women were referred to the Gynecology Outpatient Clinic for the most part by private gynecologists (37.9%), public health care general practitioners (34.4%), or other discipline units within the hospital (15.1%). After careful medical history taking and clinical evaluation, laparoscopy was considered indicated if a woman had pain symptoms decreasing her quality of life despite hormonal treatment or if hormonal treatment was inappropriate because of pregnancy seeking, side effects, or contraindications. The waiting time for laparoscopy was less than 6 months. Most operations, that is, 223/296 (75.3%), were performed or assisted by a gynecological surgeon of the endometriosis team, operating advanced endometriosis in a weekly basis. In addition, 33/296 (11.1%) patients were operated or assisted by a specialist having a subspeciality in urogynecology and gynecological surgery, 33/296 cases (11.1%) were done by a specialist in obstetrics and gynecology working in the gynecological surgery department, and 7 (2%) by a trainee undergoing gynecological surgery training period.
The preoperative characteristics and intraoperative findings were collected from the patient files to a uniform Excel spreadsheet. Positive cases for endometriosis were identified based on documentation of the presence of macroscopic endometriosis lesions (superficial, deep, or ovarian) in the electronic operation notes. Histological samples were not systematically collected if peritoneal endometriosis was visually detected, and peritoneal sampling was not routinely performed in cases with negative laparoscopy. Thus, we report visually detected endometriosis as a positive laparoscopy. The surgical prevalence of endometriosis was the primary outcome. Second, we wanted to explore preoperative pain symptoms.
Statistical analyses
Statistical analyses were performed using JMP pro16.2.0. The data were pseudoanonymized prior to analysis. The basic characteristics are presented as the mean and standard deviation (SD) for normally distributed continuous variables, median, and interquartile range (IQR) for nonnormally distributed continuous variables and number and percentage for categorical variables.
Basic characteristics were compared using two sample t-test for normally distributed variables, Wilcoxon rank sum test for nonnormally distributed variables, and Fisher’s exact test for nominal variables. Fisher’s exact test was also used when comparing different age groups.
Ethical approval
According to Finnish Medical Research Act (488/1999) patient’s informed consent or ethical approval is not required in a registry-based study. This study was approved by Southwest Finland Hospital District in April 2021 (approval number: T07/009/21). Participant confidentiality was protected, and the data was pseudoanonymized prior to analysis.
Results
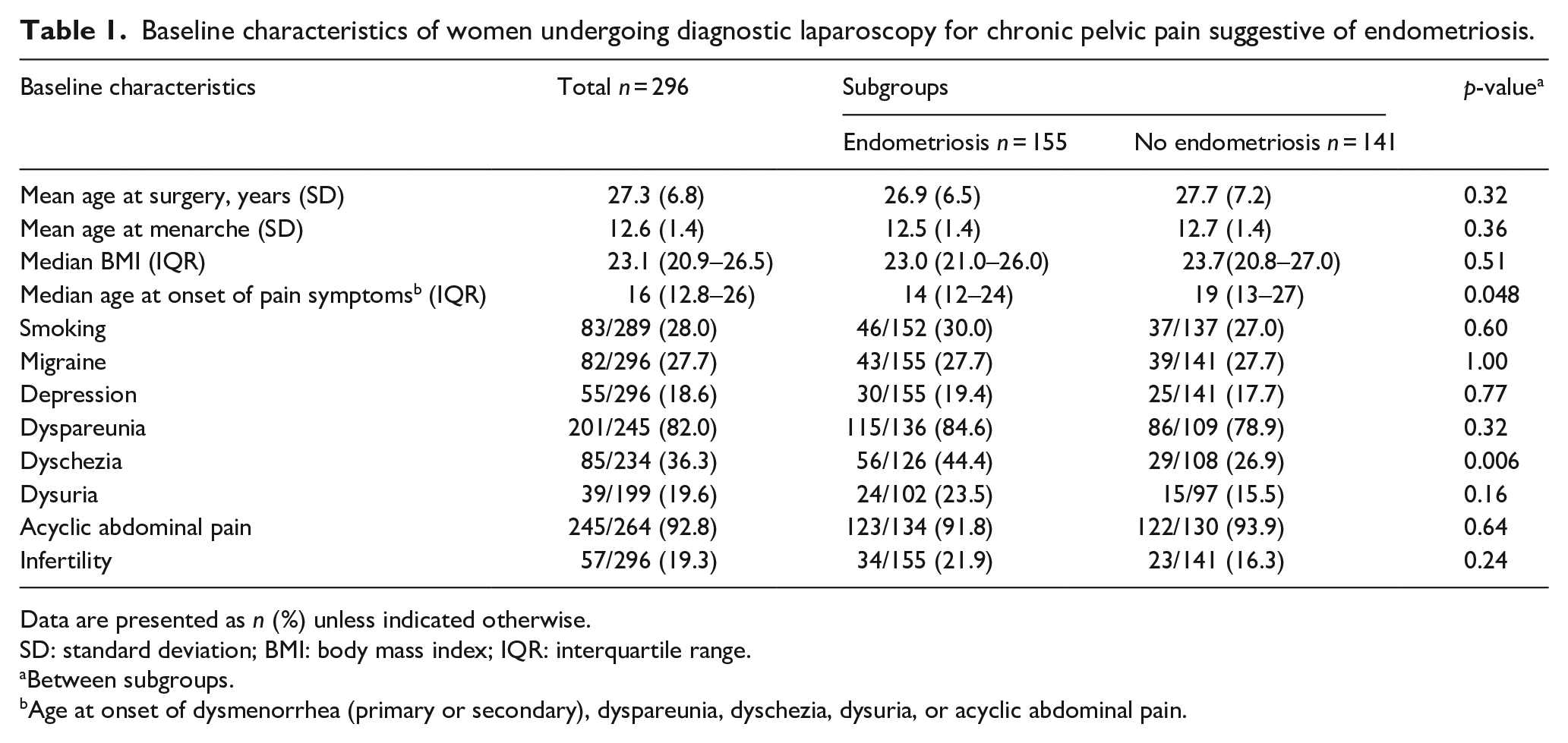
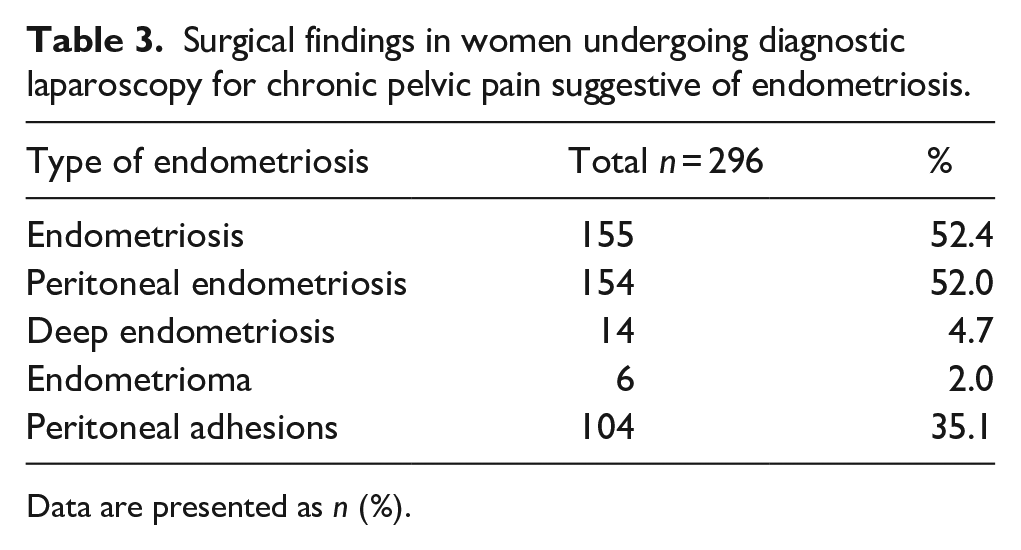
Between 2009 and 2020, laparoscopy was performed on 296 women with pain symptoms suggestive of endometriosis. The annual number of these procedures varied between 13 and 39 (mean = 24.6 and SD = 7.1). The mean age of the women was 27.3 years (SD = 6.8). The baseline characteristics of the study population are presented in Tables 1 and 2. Women with and without endometriosis were comparable by age, BMI, and age at menarche. Macroscopic endometriosis was found in 155 (52.4%) laparoscopies (Table 3), and the most common finding was peritoneal endometriosis.
Baseline characteristics of women undergoing diagnostic laparoscopy for chronic pelvic pain suggestive of endometriosis.
Data are presented as n (%) unless indicated otherwise.
SD: standard deviation; BMI: body mass index; IQR: interquartile range.
Between subgroups.
Age at onset of dysmenorrhea (primary or secondary), dyspareunia, dyschezia, dysuria, or acyclic abdominal pain.
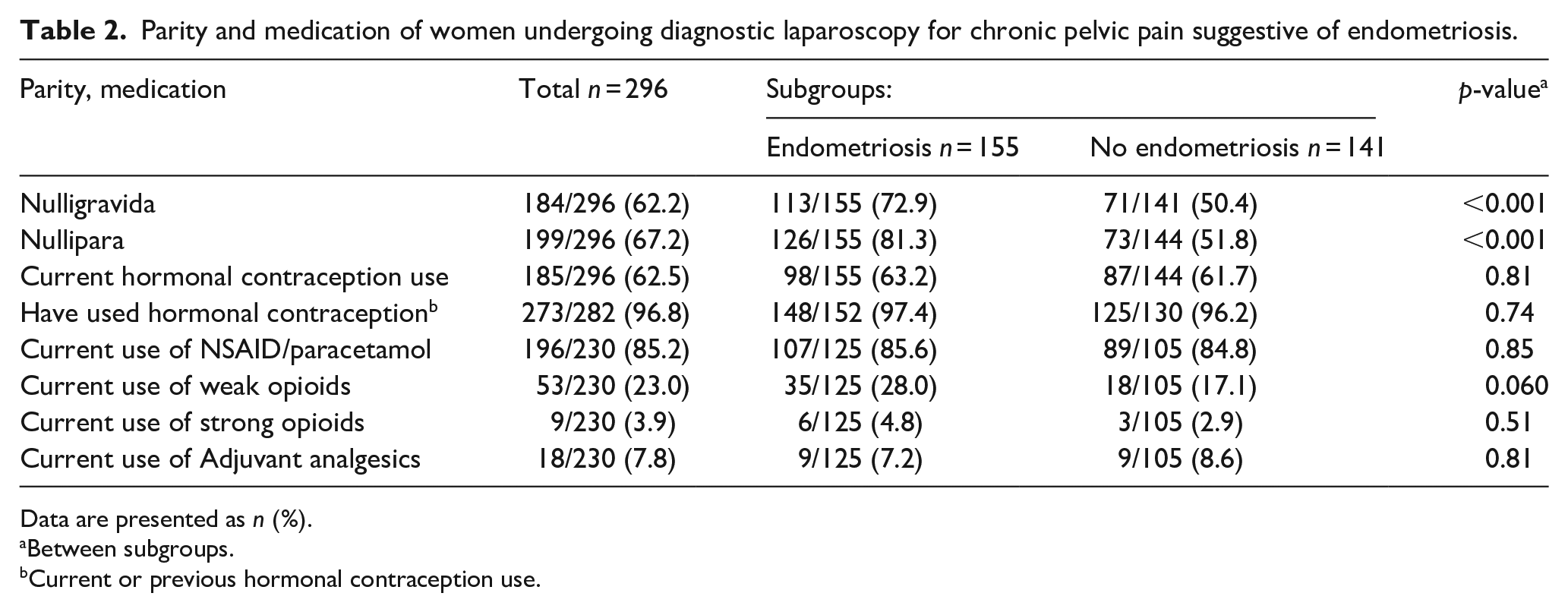
Parity and medication of women undergoing diagnostic laparoscopy for chronic pelvic pain suggestive of endometriosis.
Data are presented as n (%).
Between subgroups.
Current or previous hormonal contraception use.
Surgical findings in women undergoing diagnostic laparoscopy for chronic pelvic pain suggestive of endometriosis.
Data are presented as n (%).
Peritoneal endometriosis was most frequently located on the uterosacral ligaments (n = 75/154, 48.7%), on the ligamentum latum (n = 46, 29.9%), anterior to the uterus (n = 46, 29.9%), and on the surface of the ovaries (n = 29, 18.8%). Other common sites reported were nearby the uterosacral ligaments (n = 23, 14.9%), posterior surface of the cervix (n = 16, 10.4%), and posterior surface of the uterus (n = 10, 6.5%). Three women had endometriosis on the fallopian tubes (1.9%) and three women had diaphragmatic endometriosis. In addition, in 49 (31.8%) cases peritoneal endometriosis was detected elsewhere including different locations of the pelvic sidewall, on top of the ureters, under the ovaries, on the ovarian ligaments, on the mesenterial fat, and/or on the surface of the intestine.
The most common preoperative symptoms other than dysmenorrhea were acyclic abdominal pain (92.8%) and dyspareunia (82.0%; Table 1). There was no statistically significant difference in the prevalence of these symptoms between women with and without endometriosis.
In total 85/234 (36.3%) women reported dyschezia. Women with endometriosis reported dyschezia more often than women with no endometriosis, 44.4% and 26.9%, respectively (p = 0.006). The prevalence of current dysmenorrhea is not reported, as many patients used continuous hormonal therapy aimed to induce amenorrhea. Preoperative hormonal and analgetic medical therapy are described in Table 2. At the end of laparoscopy an intrauterine device was inserted for 68 (23.0%) women.
In this study, no laparoscopies were performed solely because of infertility, but 57 (19.3%) women had infertility with coexisting pelvic pain (Table 1). There was no statistically significant difference in the prevalence of infertility between women with (21.9%) and without (16.3%) endometriosis (p = 0.24). Interestingly, compared with women without visual endometriosis in laparoscopy, women with endometriosis were more often nulligravida (72.6% vs 50.4%, p < 0.001) and nulliparous (81.3% vs 51.8%, p < 0.001).
The age distribution of our cohort is presented in Figure 2. To evaluate whether there were any age-related differences in the prevalence of endometriosis, we divided women into five subgroups: under 20, 20–24, 25–29, 30–34, and over 34 (34–49) years old. The prevalence was highest in the age groups of 20–24 (56.8%) and 25–29 (56.2%), and lowest among women older than 34 years old (38.6%). There was no statistically significant difference in the prevalence of endometriosis between the age groups (p = 0.34).
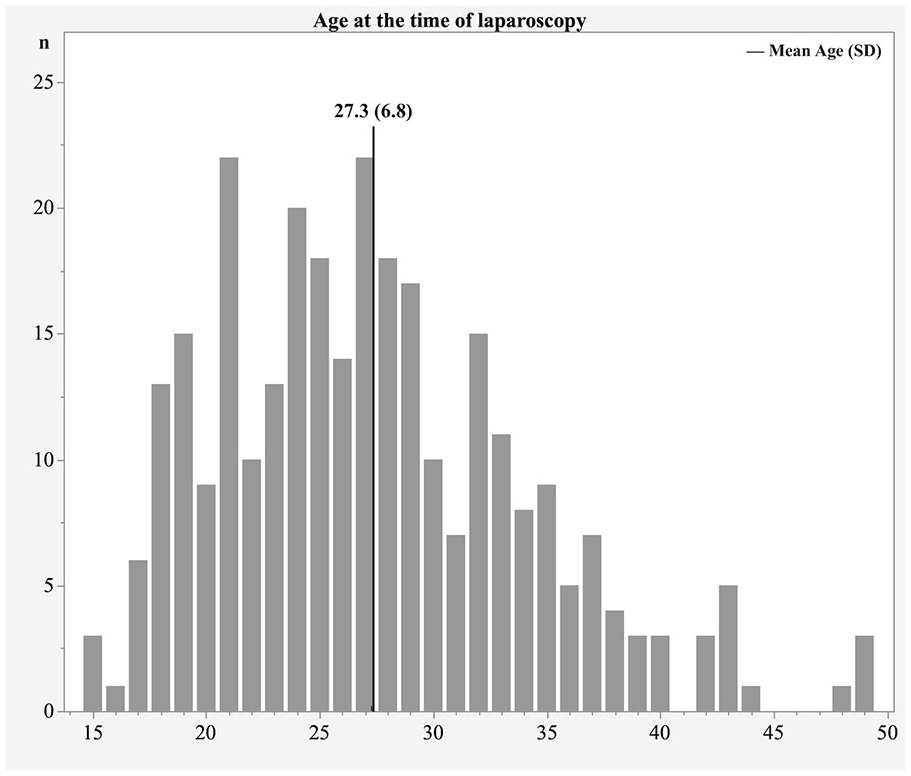
Age distribution of women undergoing diagnostic laparoscopy for chronic pelvic pain suggestive of endometriosis.
Table 4 shows data on preoperative imaging, laboratory testing, and endoscopies performed due to pain symptoms. Investigations were mostly performed by primary health care doctors or specialists in other disciplines prior to referral to the Gynecological Outpatient Clinic.
Frequency of preoperative investigations among the study population.
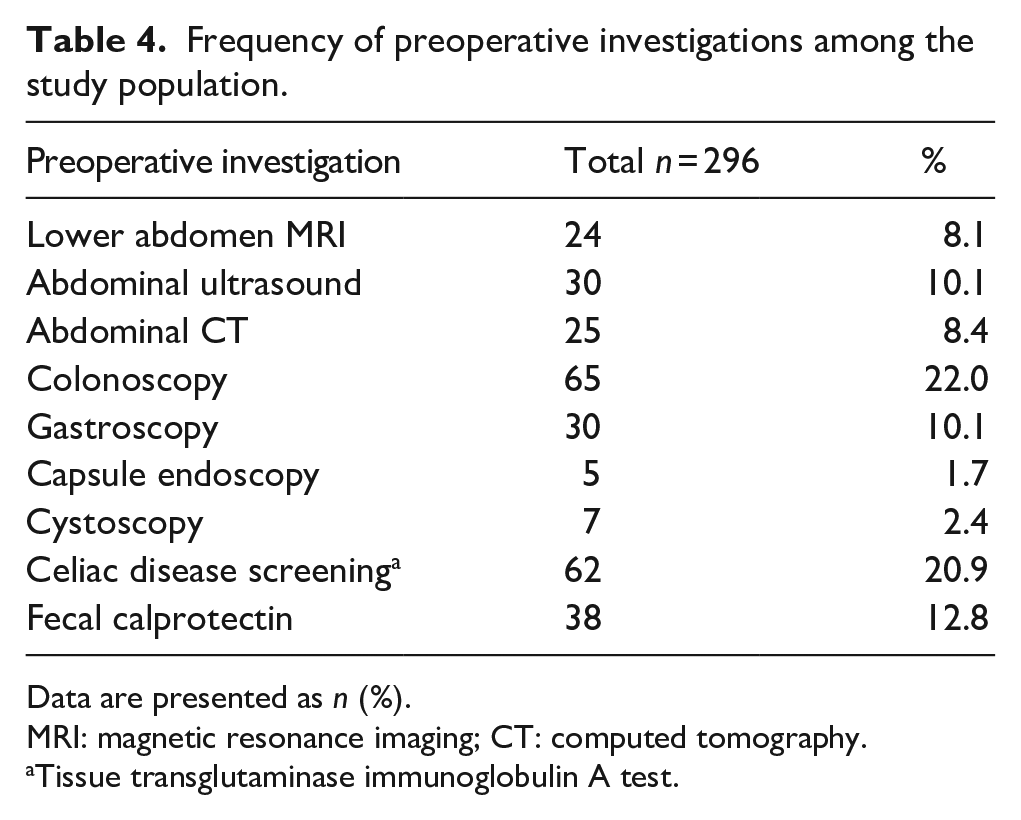
Data are presented as n (%).
MRI: magnetic resonance imaging; CT: computed tomography.
Tissue transglutaminase immunoglobulin A test.
The total complication rate was 7.7% (23/296), including 14 cases in the endometriosis group and 9 cases in healthy women. Altogether five intraoperative complications occurred and one of them was considered as serious. This patient had small intestine perforation, that was diagnosed 6 days after primary operation and treated in laparotomy with small bowel resection. Four uterine perforations were reported during laparoscopy, but the recoveries were uneventful.
One woman complained hypoesthesia of thigh after the laparoscopy, three women received antibiotics due to postoperative endometritis, eleven wound infections were treated, and three other infections were reported. One of these patients underwent re-laparoscopy due to peritonitis, but no organ injury was detected.
Discussion
In our historical cohort study, more than half of the women (52.4%) had endometriosis in diagnostic laparoscopy performed due to CPP. Our study included only women of reproductive age (15–49 years), and surgery was performed with the intention to treat endometriosis if detected. According to previous meta-analyses, visible endometriosis is found via laparoscopy in 64% of adolescents and in 42% of adult women with CPP.2,3 Thus, our results are in line with previous findings.
To our knowledge, our study exploring the prevalence of endometriosis among fertile aged women with CPP is the largest performed in recent years. During last decades, ultrasound imaging of endometriosis and knowledge about the appearance of peritoneal endometriosis in laparoscopy as well as surgical equipment have notably developed, making it difficult to compare our results with those of previous studies.
Many previous studies have included postmenopausal women, for example Moradi et al. 3 in their meta-analysis of adult women with CPP. In addition, a cohort consisting of 3068 diagnostic laparoscopies, performed on women aged 15–66 years due to CPP between 1994 and 2005 reported a prevalence of 60.2% of endometriosis. 6 The most recent study consisting of 150 women aged 16–30 years, with a similar study design to ours, reported surgically verified endometriosis in only 20% of laparoscopies. 7 These women suffered from CPP and were reported to be ultrasound negative and to have a normal pelvis in clinical examination preoperatively.
In our study, dyschezia was the only preoperative symptom that differed significantly in prevalence between women with and without endometriosis (44.4% vs 26.9%, p = 0.006). Similar to our findings, in previous studies the prevalence of dyschezia among women with laparoscopic endometriosis diagnosis has been reported to be 32.2%–44.2%.8,9 In addition, dyschezia during menstruation has been shown to have a strong correlation with the risk of endometriosis. 10
The mean age at the time of diagnostic laparoscopy was similar in women with and without endometriosis, 26.9 and 27.7 years, respectively (p = 0.32). However, women with endometriosis had significantly earlier onset of pain than women without endometriosis (14 vs 19 years, respectively, p = 0.048). This is a major difference clinically and for the individual suffering from pain symptoms. According to the literature, in two-thirds of patients with endometriosis, symptoms start before the age of 20.11,12 In one recent study of women with surgically confirmed endometriosis, the median age at the onset of endometriosis symptoms was 14 years old. 13
Laparoscopy should no longer be considered as the gold standard diagnostic tool for endometriosis, but rather an option when endometriosis cannot be detected by noninvasive imaging and empirical treatment is unsuccessful. 1 Recently this transition in the diagnostic process has been widely discussed.14,15 Furthermore, it is suggested that the definition of endometriosis should not be based on histology but rather on clinical symptoms and findings. This modification to the definition could help change the diagnostic approach globally away from laparoscopy. 16
The ESHRE guidelines state that there is no comprehensive evidence that endometriosis can be predicted by women’s symptoms, nevertheless having multiple symptoms increases the likelihood of endometriosis. 1 In our study, it would have been impossible to discriminate women with and without endometriosis based on preoperative symptoms only, and almost half of the women (47.6%) undergoing laparoscopy did not have visible endometriosis. Nongynecological pelvic and nonpelvic conditions such as interstitial cystitis, irritable bowel syndrome, fibromyalgia, and migraine often contribute to pain symptoms in women with CPP pain. 17 It is estimated that even in 80% of CPP the origin of the pain is not gynecological. 18
Importantly, there is no clear evidence if laparoscopic treatment of peritoneal endometriosis reduces pain symptoms in the long term. A recent systematic review on laparoscopic surgery for endometriosis concluded, that it remains uncertain whether surgery is effective for pain symptoms compared to diagnostic laparoscopy alone. 19 Presently, two ongoing blinded randomized controlled trials intend to provide information on whether laparoscopic excision of peritoneal endometriosis is helpful or harmful.20,21 Importantly, it has been suggested that laparoscopy may enhance pelvic pain through several different mechanisms, such as increased sensory hypersensitivity or the development of neuropathic pain. 21
It has long been a high priority to reduce the diagnostic delay of endometriosis and establish a non-invasive diagnostic tool. Transvaginal ultrasound is recommended when endometriosis is suspected, and ovarian endometrioma is easily detected. Even deep endometriosis can be accurately diagnosed with imaging, but it is highly operator-dependent, and also dependent on the location of the lesions. 22 However, superficial endometriosis is the most common disease type with estimated prevalence of 80% among patients, and to date it has been impossible to rule it out without laparoscopy. 23 Nevertheless, in expert hands peritoneal lesions may be seen in the pouch of Douglas. 23
Nnoaham et al. 10 concluded that a symptom-based diagnostic model predicts any-stage endometriosis relatively poorly with area under curve (AUC) of 68.3. When including ultrasound to the model, the AUC is improved (80.0), but the sensitivity remains low (58%). In other words, ruling out disease with ultrasound in symptomatic women is impossible. Our data similarly indicates that CPP in ultrasound negative women poorly predicts endometriosis.
In our study, half of the patients underwent unnecessary surgery with negative findings and were exposed to complications. From monetary or the society’s perspective, this finding in a government funded hospital among closely selected women suggests, that the present practice is far from cost-effective. Importantly, we should take notice that women without findings are typically very disappointed when surgery does not give a reason for their suffering. So how could we improve current diagnostic path?
Recently, two new promising non-invasive diagnostic methods to detect all types of endometriosis have become commercially available: a saliva test composed of 109 microRNAs (Endotest®) and an electroviscerogram measuring the activity of the stomach and viscera (Endosure®). MicroRNAs have been widely investigated as a new potential biomarker for endometriosis. 24 Bendifallah et al. 25 have developed a saliva-based microRNA signature for the diagnostics. Their results suggest that salivary microRNA test could predict endometriosis with a high sensitivity, specificity, and AUC of 96.9%, 100%, and 98.3%, respectively. 26 Yet, the saliva microRNA test is fairly expensive. A cost-effectiveness evaluation was conducted comparing different diagnostic strategies in a French setting. They concluded that the saliva test performed after negative ultrasonography appeared to be most relevant both clinically and economically. 27 Another novel non-invasive diagnostic technique is the gastrointestinal myoelectrical activity (GIMA) unique to endometriosis, and this could be an easily available low-cost option. 28 According to Noar et al., 28 GIMA biomarker threshold scoring showed 95%/91% sensitivity and positive predictive value and 96/95% specificity and negative predictive value, respectively. Larger validation studies are needed to find out the usefulness and the optimal timing of these new tools in clinical practice.
The strengths of our study include the fact, that most laparoscopies were performed or assisted by an experienced surgeon familiar with endometriosis. Therefore, the visual diagnosis of endometriosis is considered reliable. Another strength is that the indications for laparoscopy remained predominantly the same during our study period, and laparoscopy was not the first-line treatment option nor solely a diagnostic method. In addition, the study cohort was moderately sized.
The most important limitation of our study is the retrospective study design. All preoperative symptoms were not collected via a uniform questionnaire; thus, the data may be incomplete. In addition, several gynecologists performed preoperative visits. This increases the heterogeneity of the medical record notes eventually collected for this study. We consider the sensitivity of preoperative ultrasound imaging acceptable as deep endometriosis was found in 4.7% and endometrioma in 2.0% of participants considered ultrasound negative. Another limitation was that no histological samples were routinely collected.
Conclusions
Endometriosis was confirmed in approximately in one half of the women undergoing laparoscopy due to CPP. In most women peritoneal endometriosis was the only finding suggesting that endometrioma and deep endometriosis are detected preoperatively in most cases. Importantly, the other half of the women did not have endometriosis despite having similar pain symptoms prior to surgery, and these laparoscopies can be thought as unnecessary invasive procedures. Irrespective of laparoscopy findings, women with CPP should be offered comprehensive and multimodal treatment of chronic pain to improve their quality of life.
To date, it remains unknown whether patients benefit long-term from undergoing diagnostic and therapeutic laparoscopy or surgical treatment for peritoneal endometriosis. Hopefully, ongoing randomized clinical trials will provide us with answers to this well-recognized question.
Supplemental Material
sj-tiff-1-pev-10.1177_22840265251363707 – Supplemental material for Prevalence of endometriosis in diagnostic and therapeutic laparoscopy among women with chronic pelvic pain
Supplemental material, sj-tiff-1-pev-10.1177_22840265251363707 for Prevalence of endometriosis in diagnostic and therapeutic laparoscopy among women with chronic pelvic pain by Saima Siivonen and Pia A Suvitie in Journal of Endometriosis and Pelvic Pain Disorders
Footnotes
Acknowledgements
Not applicable.
Author contributions
PS and SS planned and designed the study. SS collected the data with input from PS. SS performed the statistical analysis. SS drafted the first manuscript, which was further thoroughly edited by PS. Both authors contributed to all faces of this study and approved the final version.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Saima Siivonen has stated that there are no conflicts of interest. Pia A. Suvitie has received consultancy and speaker’s fees from Gedeon Richter Nordics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
This study was approved by Southwest Finland Hospital District in April 2021 (approval number: T07/009/21).
Consent to participate
According to Finnish Medical Research Act (488/1999) patient’s informed consent or ethical approval is not required in a registry-based study. Participant confidentiality was protected, and the data was pseudoanonymized prior to analysis.
Consent for publication
Not applicable.
Trial registration
Not applicable.
Data availability statement
The datasets generated and analyzed during the current study are not publicly available due to privacy restrictions, but anonymized data are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
