Abstract
Introduction:
Chronic pelvic pain (CPP) affects up to 40% of reproductive-age women and often involves myofascial pelvic pain syndrome (MPPS), a musculoskeletal condition associated with pelvic floor dysfunction. Effective treatment options are limited, and pudendal nerve blocks have been proposed as an adjunct to pelvic floor physical therapy (PFPT). This study aimed to evaluate the impact of adding pudendal nerve blocks to PFPT on pain reduction and quality of life in patients with MPPS.
Methods:
This prospective, randomized, double-blind trial enrolled women with MPPS referred for PFPT. Participants were randomized to receive either pudendal nerve blocks with local anesthetic and steroid or placebo saline injections weekly for 6 weeks. The primary outcome was pain measured using a visual analog scale (VAS) at 8 weeks. Secondary outcomes included muscle tenderness, pelvic floor strength, and quality-of-life metrics (PFDI, PFIQ, SF-36, and FSFI). Statistical analyses included paired
Results:
Both groups experienced significant improvements in VAS scores (
Conclusions:
While pudendal blocks did not significantly enhance outcomes over placebo injections, PFPT alone likely drove the observed improvements in pain and quality of life. Future studies with larger sample sizes and additional comparison groups are needed to explore the potential role of adjunctive therapies in managing MPPS.
Keywords
Introduction
Chronic pelvic pain (CPP) is not uncommon and represents a challenging clinical problem that affects 4%–40% of reproductive age women.1–3 It is defined as non-menstrual or non-cyclic pain of at least 6 months duration with significant intensity such that it causes functional disability and requires medical or surgical treatment. CPP is not a disease, but a symptom caused by a wide variety of conditions, making the differential diagnosis and identification of the underlying cause challenging. 4 Symptoms may be caused by urologic, gynecologic, gastrointestinal, and other pelvic floor disorders. Initiation of treatment is often delayed or nonspecific until a specific diagnosis is made. Myofascial Pelvic Pain Syndrome (MPPS)—formerly known as pelvic floor tension myalgia—is increasingly identified in patients with CPP. This increase has been associated with an increase in awareness of MPPS rather than an increase in actual cases. 5 As many as 85% of patients with CPP have dysfunction of the musculoskeletal system, ranging from lumbar lordosis, knee hyperextension and pelvic anteriorization as well as pelvic floor muscle spasm. 1
MPPS can be a primary or secondary cause of CPP. Shortening of the pelvic floor musculature with development of trigger points can develop secondary to underlying pelvic diseases such as endometriosis, painful bladder syndrome, history of pelvic inflammatory disease, gastrointestinal disease, or after pelvic trauma such as surgery, radiation, or physical/sexual trauma.1,6 Animal studies have shown a connection between inflammation and injury in one pelvic structure/area resulting in effects in other pelvic structures, described as neurogenic inflammation.7–9 Neurogenic inflammation results from noxious stimuli, causing both orthodromic (toward the spinal cord) and antidromic (toward the periphery) neuronal transmission. Even if the underlying cause of the noxious stimulus is treated, without addressing the MPPS, the pelvic pain often persists. Furthermore, treatment of the underlying MPPS can relieve bladder urgency, frequency and pain symptoms.10–12
Pelvic floor physical therapy (PFPT) focuses on the musculature of the pelvis and the spine, with the goal of improving muscular, urinary, defecatory and sexual function. PFPT alone has been extensively studied as a promising, efficacious treatment modality of CPP. Many studies to date, including randomized controlled trials10,13–15 and prospective studies,16–20 have found significant improvements in muscle resting tone and reported pain, but rarely found significant differences in quality of life or sexual function and dyspareunia, as further reported in a recent systematic review of the literature. 21
To date, few studies have been conducted to address specific therapies for MPPS outside of PFPT. Weiss studied a group of patients with either painful bladder or urgency-frequency syndrome and showed that 83% of patients with urgency-frequency and 70% of patients with painful bladder syndrome had marked improvement or complete resolution with manual physical therapy. 22 Case reports as well as small case series have shown improvement in dyspareunia and pelvic pain with injection of Botox into hypertonic pelvic floor muscles.23,24 A randomized controlled double-blinded trial of 60 patients in which Botox versus saline injection was used, where an improvement in pelvic pain and dyspareunia was seen for the Botox group with improvement in dyspareunia only in the saline group; however, more recent data in a randomized controlled trial suggest no differences in those receiving onabotulinumtoxin A injections for myofascial pelvic pain compared to placebo group.25,26 In another study of patients with CPP and levator ani trigger points, 13 of 18 patients had greater than 50% improvement at a mean follow up of 3 months after a single trigger point injection with a mixture of steroid and local anesthetic. 27 Two separate randomized control trials of intra-vaginal diazepam suppositories showed no difference in resting pelvic tone or as a treatement for pelvic hypertonicity, respectively, when compared with placebo.28,29
The current study was designed to further investigate injection therapy to treat CPP and MPPS, specifically in combination with PFPT. Patients with these conditions can have refractory courses that may not respond to treatment, or have a protracted response time to physical therapy. Pudendal blocks have been used for decades in fields including but not limited to obstetrics, urology and colorectal surgery with minimal morbidity.30–32 Pudendal blocks with local anesthetic or local anesthetic with steroid have been used as an adjunctive treatment to physical therapy for patients with MPPS. In patients with MPPS, pudendal blocks provide anesthesia, making manual vaginal physical therapy more tolerable. 10 These blocks also decrease nerve input to the pelvic floor, potentially decreasing pelvic spasm and pain. 33 Such therapy has been utilized by practitioners in treating MPPS, however, there have been no prospective clinical studies published evaluating this intervention.10,33 The goal of this study was to evaluate whether the addition of pudendal nerve blocks, using a combination of steroid and local anesthetic, could enhance the therapeutic effects of pelvic floor physical therapy (PFPT) in women with myofascial pelvic pain syndrome (MPPS). Specifically, the study aimed to assess improvements in pain reduction, muscle tenderness, pelvic floor strength, and quality of life metrics, comparing outcomes between patients receiving pudendal blocks and those receiving placebo injections over an 8-week period.
Methods
Trial design
This was a prospective, randomized, double-blind, placebo-controlled trial with a 2:1 allocation ratio for active versus placebo treatments. Participants were randomized to receive pudendal nerve blocks (PNBs) with either an active intervention or a placebo injection in addition to pelvic floor physical therapy (PFPT). There were no changes to the trial design after commencement.
Participants
Eligibility criteria included non-pregnant women aged 18 years or older diagnosed with myofascial pelvic pain syndrome (MPPS), naïve to PFPT, able to provide informed consent, and willing to accept randomization. Exclusion criteria included prior PFPT, allergy to any injection components, inability to complete study questionnaires, or inability to attend follow-up visits. Participants were recruited from the Division of Urogynecology and Reconstructive Pelvic Surgery (URPS) at the University of California, Irvine Medical Center. IRB approval was obtained (study HS#2009-6784), and all participants provided written informed consent before enrollment.
Interventions
A standard baseline exam was conducted by the physical therapist using validated instruments to assess the location of pelvic pain and tension. Subjective pelvic floor pain was graded using visual analog scale from 1 to 10. Pelvic floor strength was graded using the modified oxford scale. Myalgia with palpation of the pelvic floor musculature was graded on a scale of 1–4 with 1 denoting no pain with digital palpation and 2, 3, and 4 denotes mild, moderate, and severe pain with palpation respectively. The areas of maximal discomfort were identified and noted. Baseline vaginal electromyography (EMG) readings were measured at rest and with maximal pelvic floor contraction to assess pelvic floor hypertonicity. The participants completed baseline questionnaires including the pelvic floor impact questionnaire (PFIQ), pelvic floor distress inventory (PFDI), and female sexual function index (FSFI) and the Short Form 36 Health Survey (SF-36).
All participants received standardized PFPT weekly for 8 weeks. During the treatment period, standard physical therapy techniques were utilized including internal manual palpation and trigger point release, muscle stretching, biofeedback, and abdominal scar release. Participants performed a self-assessment of their pain using a visual analog scale (VAS) weekly. A myalgia score, modified oxford score and point of maximal tenderness were noted by the treating physical therapist at each visit. Participants in the active group received weekly PNBs comprising 8 mL of 0.5% bupivacaine, 1 mL of 10 mg/mL triamcinolone, and 1 mL of 8.4% sodium bicarbonate (total 10 mL, 5 mL per site). Placebo injections consisted of 5 mL saline per site. Injections were performed under ultrasound guidance by two board-certified URPS physicians over 6 weeks at the initiation of each PFPT session. Patients were observed for 1 h post-injection during their first visit.
Outcomes
The primary outcome was pain reduction, measured using the Visual Analog Scale (VAS) at baseline and 8 weeks post-treatment. Secondary outcomes included pelvic floor strength (Modified Oxford Scale), myalgia scores (graded 1–4), and quality-of-life metrics assessed using the Pelvic Floor Distress Inventory (PFDI), Pelvic Floor Impact Questionnaire (PFIQ), Female Sexual Function Index (FSFI), and Short Form Health Survey (SF-36). Vaginal electromyography (EMG) was used to assess hypertonicity at rest and during maximal contraction. There were no changes to outcome measures after the trial commenced.
Sample size
Power analysis indicated that 32 participants were required to achieve 80% power to detect a 50% reduction in VAS scores between groups at a 5% significance level, accounting for potential dropouts.
Randomization and blinding
Randomization was conducted in a 2:1 ratio using sealed, consecutively numbered envelopes containing computer-generated allocations. The decision was made to have more patients in the treatment versus control groups due to the high published drop out/retention rates of placebo studies for pain. The allocation sequence was implemented by a study coordinator not involved in patient care. Both participants and treating physical therapists were blinded to treatment assignments. Active and placebo injections were visually indistinguishable to ensure blinding integrity.
Statistical methods
Statistical analysis followed intention-to-treat principles. Paired
Results
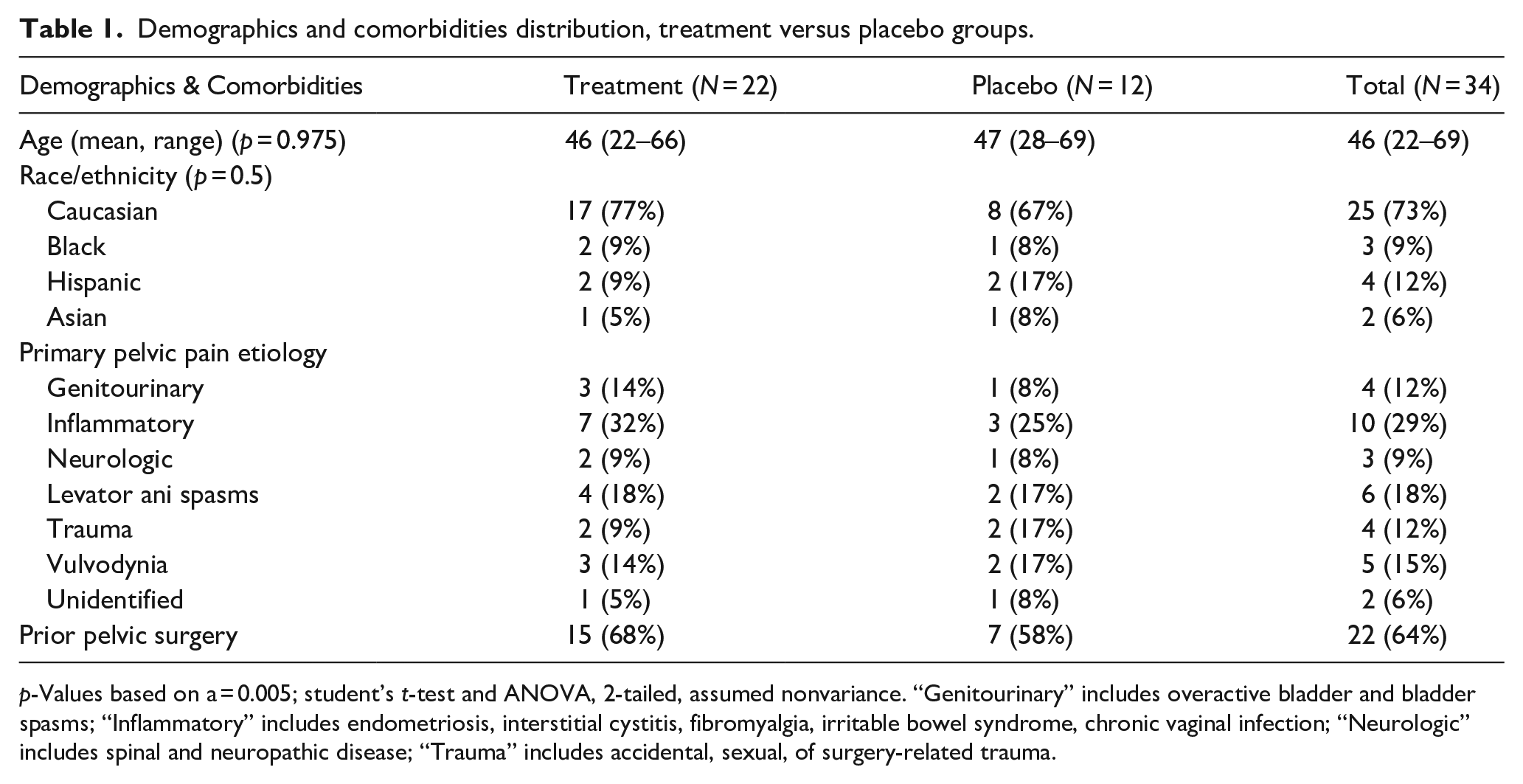
A total of 122 patients were screened throughout the study period from 2009 to 2014. The most common reason for exclusion was refusal to accept randomization to placebo. Forty patients were enrolled, six patients dropped out following the baseline internal pelvic floor assessment. Thirty-four patients were randomized in a 2:1 fashion to pudendal or placebo injections (Figure 1). The majority of the patients were Caucasian women (74% Caucasian, 12% Hispanic, 9% African American, and 6% Asian) with a median age of 46 years (range 22–69 years,
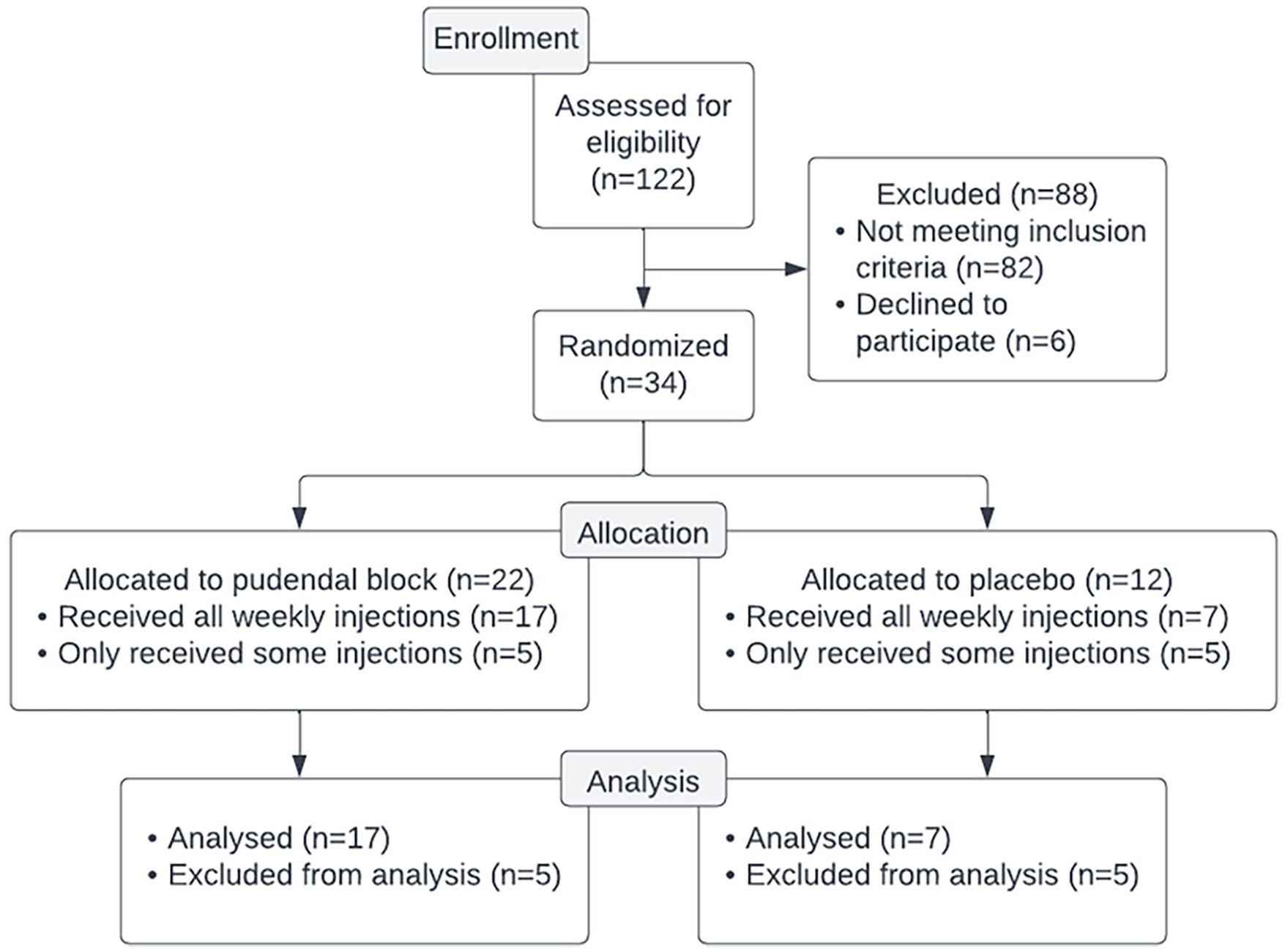
CONSORT flow diagram.
Demographics and comorbidities distribution, treatment versus placebo groups.
The primary outcome measure pain by VAS was 7.1 ± 0.6 in the pudendal group and 6.4 ± 0.8 in the placebo group at baseline, and 3.7 ± 0.8 and 3.6 ± 1.1 at the 8 week follow up. Overtime both groups had significant reductions in pain (
Pain as measured by visual analog scale.
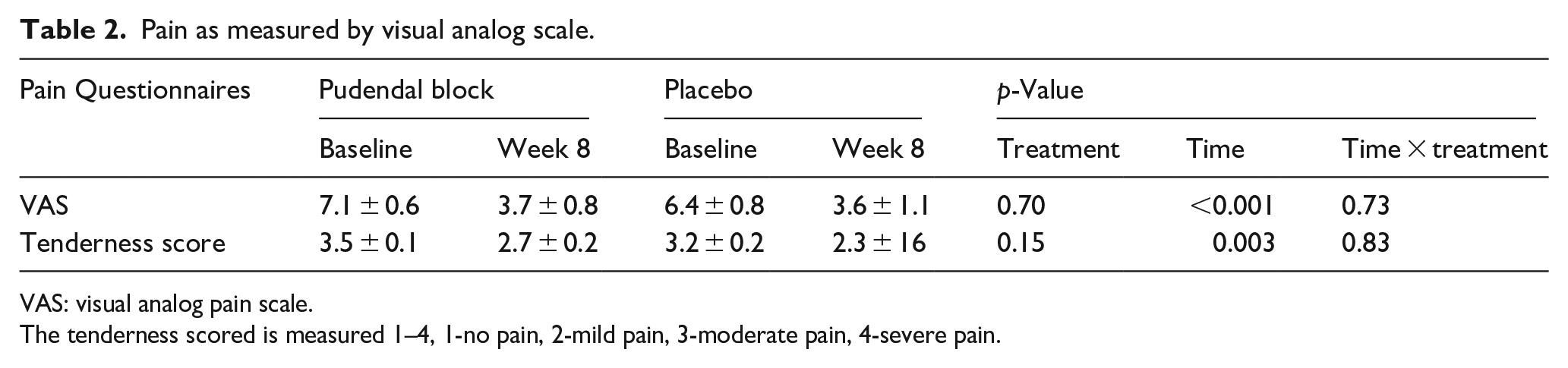
VAS: visual analog pain scale.
The tenderness scored is measured 1–4, 1-no pain, 2-mild pain, 3-moderate pain, 4-severe pain.
Vaginal EMG measured in microvolts.
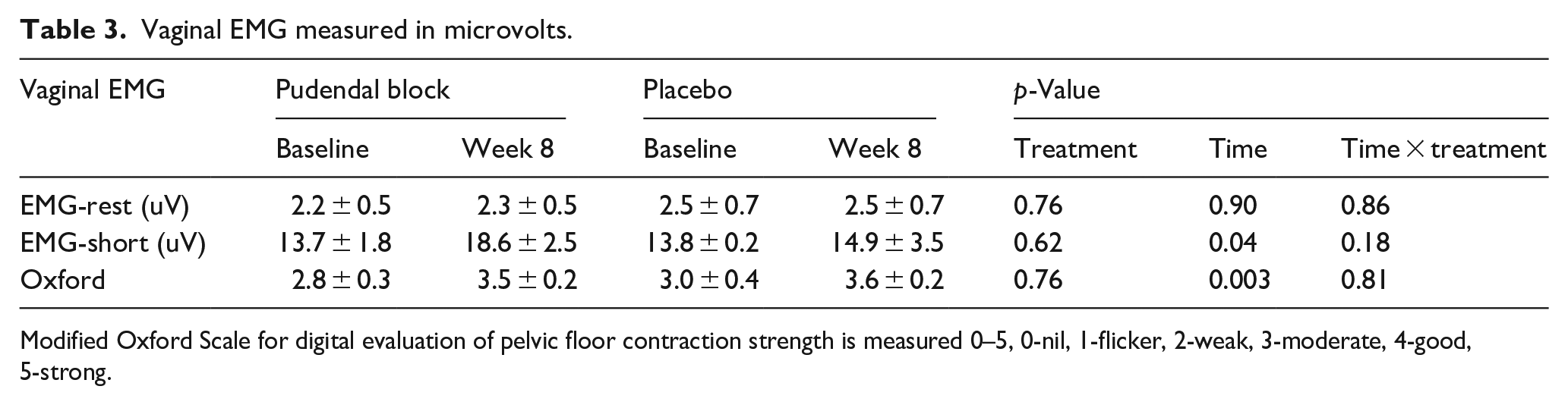
Modified Oxford Scale for digital evaluation of pelvic floor contraction strength is measured 0–5, 0-nil, 1-flicker, 2-weak, 3-moderate, 4-good, 5-strong.
The PFDI and PFIQ showed significant improvements at 8 week follow up (
Quality of life measures.
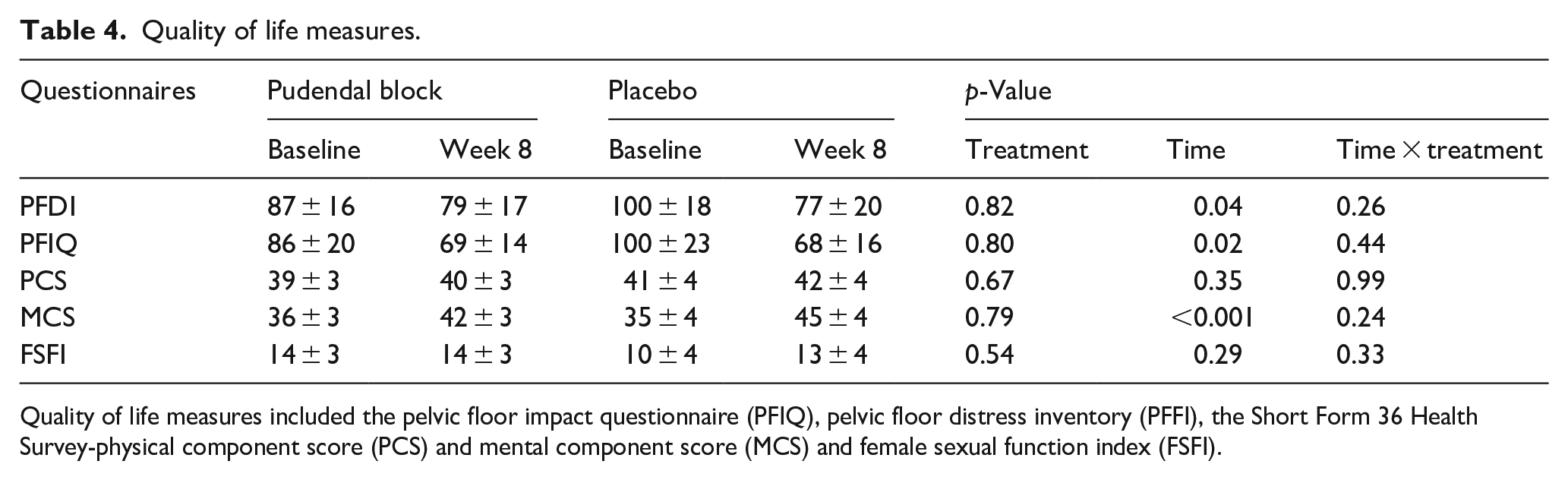
Quality of life measures included the pelvic floor impact questionnaire (PFIQ), pelvic floor distress inventory (PFFI), the Short Form 36 Health Survey-physical component score (PCS) and mental component score (MCS) and female sexual function index (FSFI).
Eight Dindo grade 1 complications (minor postoperative issue that requires no invasive treatment and resolves with standard care) occurred during the study period. Four in the pudendal group and four in the placebo group. Two patients in the pudendal group experienced transient leg/toe numbness and two experienced moderate vaginal bleeding at the injection site, all resolved spontaneously. All four patients in the placebo group experienced vaginal bleeding, three resolved with pressure and one required vaginal packing prior to resolution.
Among the 12 patients initially treated with placebo, 8 participants elected to cross over to physical therapy with pudendal block following the 8 week follow-up visit. Four (18%) of the pudendal group and five (42%) of the placebo group completed the 6-month follow-up evaluations and questionnaires after the initial 8 weeks of treatment. Consequently, no conclusions were drawn regarding the durability of the treatment outcomes in either group.
Discussion
CPP and MPPS are complex conditions that pose diagnostic and treatment challenges. This study aimed to investigate the effectiveness of pudendal blocks in combination with physical therapy for the management of MPPS and CPP. While the findings revealed a significant reduction in pain scores over time in both the pudendal and placebo groups, the improvements in pain scores were similar between the placebo and the pudendal block groups. This suggests that the observed improvements may be attributed to the overall effect of pelvic floor therapy alone rather than the specific intervention of pudendal blocks. Notably, the study did not include a group receiving pelvic floor therapy alone, limiting the ability to draw definitive conclusions regarding the individual contributions of pudendal blocks, as well as the effect of injection alone on pain control.
Similar findings have been observed in prior studies investigating interventions for CPP. For instance, a randomized trial by FitzGerald et al. 14 demonstrated that myofascial physical therapy significantly improved symptoms in patients with urologic pelvic pain syndromes, although the addition of targeted trigger point injections yielded mixed results. Another study by Weiss 22 suggested that while pudendal nerve blocks may offer some benefit, the majority of symptom relief was attributed to comprehensive physical therapy programs.
Muscle tenderness scores also decreased significantly in both groups, indicating a reduction in pelvic floor muscle tension. The inverse correlation observed between pain scores and pelvic muscle strength suggests that as pain decreases, pelvic muscle strength increases. This may be indicative of the beneficial impact of physical therapy on muscle function and pain reduction.
In terms of quality of life, both groups reported less distress and improvements in mental health. However, no significant differences were observed in female sexual function between the groups. It is worth noting that the study had a low number of sexually active participants, which may have influenced the results. Further investigation with a larger sample size and a focus on sexual function is warranted to gain a better understanding of the relationship between CPP, treatment interventions, and sexual health.
It is important to acknowledge the limitations of this study. The lack of longterm follow-up limits the ability to establish whether more time is necessary to see the full potential of the intervention. Additionally, a substantial number of participants lost to follow-up hindered the ability to assess the durability of pain reduction and treatment outcomes. The study was also powered to detect a 50% difference in pain between groups, potentially overlooking smaller yet clinically significant differences that could exist.
Furthermore, a lack of a no-intervention group restricted our ability to draw conclusions on the effects of dry/wet needling. While some case series may suggest a potential benefit from needling in the treatment of pelvic pain, 34 there have been no prospective studies looking into the effects of dry needling of the pelvis. However, both wet and dry needling have shown promising results in the treatment of pain in other body locations.35,36 Unfortunately, no such conclusions can be drawn from this study and further research comparing PFPT with and without dry/wet needling will be necessary to explore its potential benefits.
The observed improvements in pain and muscle tenderness in both the pudendal and placebo groups also raises questions regarding the placebo effect. The literature exploring the benefits of the placebo effect on pain control is vast, and suggests upwards of 35% pain improvement attributable to placebo alone.37–40 Some studies have shown that placebo-controlled trials in chronic prostatitis/chronic pelvic pain syndrome often reveal substantial placebo responses, which suggests that patient expectations and psychological factors play a crucial role in the perception and management of pain. 41 Expectations of pain relief can lead to actual pain reduction, emphasizing the need for further investigation into the specific mechanisms of action and the most effective treatment strategies for CPP.
In conclusion, this study suggests that pudendal blocks may not offer a significant benefit compared to saline injection when combined with PFPT; a PFPT group alone was not included, the effect of dry/wet needling alone could not be analyzed, and the effect of placebo was not controlled for. On the other hand, significant improvement in pain scores and quality of life measurements were seen in both groups, further strengthening the known benefits of PFPT alone in the treatment of CPP. The results highlight the importance of larger studies with longer follow-up periods to determine the durability of pain reduction, and to explore other potential treatment modalities in order to optimize and individualize patient care with this challenging condition.
Footnotes
Author’s participation
J Ramstein: Manuscript writing, Data analysis.
D Markle Price: Project development, Data Collection, Data analysis, Manuscript writing.
A Al Saleh: Manuscript writing.
F Lane: Project development, Data Collection, Data analysis, Manuscript writing.
Data availability statement
Research data is available upon request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AUGS grant, PFD Research Foundation Grant.
IRB Approval
IRB STUDY, HS# 2009-6784; University of California, Irvine Medical Center.
Trail Registration
ClinicalTrials.gov Identifier: NCT0092856.
