Abstract
Background:
The diagnosis of neuromuscular pelvic pain is challenging. Despite the increasing use of magnetic resonance imaging (MRI) in the assessment of pudendal neuralgia, there are limited data describing radiological variations of the nerve. Additionally, the utility of MRI in the assessment of pelvic floor tension myalgia is unknown.
Aims:
To describe the anatomical variations of the pudendal nerve and dimensions of pelvic muscles in patients investigated for neuromuscular pelvic pain using a combined MRI protocol.
Materials and methods:
A retrospective audit of MRI reports for 136 patients referred to a single radiologist was conducted. Reported data included the appearances of the pudendal nerve and its perineural structures, and the maximal thicknesses of pelvic muscles.
Results:
Several anatomical variations were identified in the course of the pudendal nerve at or above the ischial spine. Likely/highly likely nerve compression at this level was present in 34.6% of hemipelves. Variations associated with compression included delayed separation of the sacrospinous and sacrotuberous ligaments (
Conclusions:
This study represents a quantitative and qualitative expansion to the radioanatomical basis of pudendal nerve entrapment on MRI, and highlights the limitations of MRI obtaining reproducible measurements of pelvic muscles. Images from asymptomatic controls and further prospective evaluation of treatment outcomes is required to determine the clinical significance of MRI findings in the assessment of pudendal neuralgia.
Keywords
Introduction
Chronic pelvic and perineal pain (CPPP) is a common condition with wide-ranging impacts on health-related quality of life, healthcare costs and work productivity. 1 CPPP has variable presentations with a multifactorial aetiology. 2 Endometriosis and adenomyosis are common pathologies identified during investigation, however, up to 55% of women have no pathology demonstrated laparoscopically. 3 Pelvic muscle and nerve syndromes such as pelvic floor tension myalgia (PFTM) 4 and pudendal neuralgia 5 are recognised as contributors to CPPP. Diagnosis of these conditions is variable and not consistently reliable; therefore, the development of imaging criteria may facilitate earlier identification and treatment.
PFTM is a poorly understood entity that affects up to 74% of women with CPPP. 6 Rather than a purely somatic pain disorder, PFTM is thought to reflect degrees of peripheral and central sensitisation, viscero-somatic convergence and pelvic organ cross-talk in the context of other pain-generating insults. As a result, the presentation can overlap with other CPPP syndromes including pudendal neuralgia. Diagnosis is best made on digital vaginal examination of the pelvic muscles. 7 Although practical and cost-effective, the reliability of findings is limited by examiner experience 8 with a wide range of inter-rater reliability.9,10 Muscle overactivity appears to be associated with pain2,11 generating the hypothesis that muscles in PFTM may demonstrate shortening or thickening visible on imaging. To date, transperineal/translabial ultrasound studies have not demonstrated differences in pelvic muscle biometry between asymptomatic and CPPP cohorts.12,13 Magnetic resonance imaging (MRI) can produce detailed views of pelvic musculature, 14 however its role in PFTM is unknown.
Pudendal neuralgia is a rare condition characterised by intractable pain affecting the perineum, bladder, vagina and bowel. 5 Symptoms may arise from pudendal nerve pathology from its sacral spinal roots to terminal fibres, or dysfunctional pain processing. The Nantes criteria offer an indirect diagnosis for pudendal neuralgia by entrapment. The criteria require a positive response to a pudendal nerve block, 15 limiting its application due to the invasiveness of pudendal nerve block, subjective definition of a ‘positive’ study (immediate 50% reduction in pain) 16 and possibility of block failure. Peripheral nerve imaging may provide direct evidence of structural nerve entrapment or compression in the clinical assessment of pudendal nerve symptoms. This may supplement the Nantes criteria in guiding the decision to proceed with surgical decompression. MRI is the preferred modality for the small pudendal nerve,17 –19 which negotiates several bony, muscular and ligamentous relations in its course.17,20,21 Pudendal nerve MRI is increasingly incorporated in practice despite limited data for expected findings in affected patients,17,18,22,23 and almost no data for asymptomatic patients. 23 Consequently, the relevance and benefit of MRI in the clinical assessment of pudendal neuralgia remains unsubstantiated.
The aims of this study were to describe the anatomical variations of the pudendal nerve canal and maximal thicknesses of the pelvic muscles in patients with CPPP using a combined MRI protocol.
Materials and methods
This study was approved by the South Eastern Sydney Local Health District Human Research Ethics Committee (2021/ETH11992). This included the specific waiver for patient consent due to the use of retrospective, de-identified data. Retrospective analysis was performed for patients who presented to two gynaecology and pain practices and were investigated by MRI of pelvic nerves and muscles from January 2013 to July 2022. Patients were assessed by gynaecologists experienced in management of CPPP and referred for MRI where pain symptoms were substantive enough to consider a likely diagnosis of pudendal or border neuralgia (neuropathic pain derived from peripheral nerves at the juncture of the abdomen and thigh) based on a clinical diagnosis and not requiring pudendal nerve block as part of that diagnostic criteria. Referrals were addressed to a single radiologist using a dedicated pelvic nerve and muscle MRI protocol.
The key criteria for inclusion in this study was referral and completion of MRI. Potential participants were identified by a search of digital practice software for pudendal nerve block and botulinum toxin injection billing, and consultation notes referencing radiological referral for pudendal nerve MRI. Patient records were reviewed to confirm investigation by MRI and exclude duplicates. MRIs conducted by other radiologists were excluded. Demographic data collected from patient records included age and gender. Where more than one MRI was recorded, the first report was used.
All MRIs were acquired on a GE Medical Systems Signa Architect 3.0T. Patients had a standardised pre procedure bowel prep with clear fluids from 2 pm the day before, and Picoprep 2 sachets at 2 and 7 pm the day before image acquisition. On the day of examination, for 3 h prior, patients were nil by mouth and had a Microlax enema. An AIR coil was utilised with IV Buscopan 10 mg at commencement of image acquisition, a second bolus IV Buscopan 10 mg prior to post contrast series. T2 TSE and Fiesta Dynamic Axial Oblique scan plane planned perpendicular to anal canal were obtained. Dynamic scans were acquired six times at 16 s each alternating sustained Kegel manoeuvre for each scan duration followed by pelvic floor relaxation. Data were reviewed as a cine study. An optional T1 LAVA post contrast in coronal and axial planes was taken in some cases. Specific MRI settings are attached in appendix 1.
Data collected from MRI reports included the appearances of the pelvic nerves and maximal thicknesses of pelvic floor (levator ani), coccygeus, obturator internus and piriformis muscles. There was emphasis on the anatomy of the pudendal nerve and the puendal nerve canal. This included the adjacent structures at or above the level of the ischial spine, such as the sacrospinous and sacrotuberous ligaments and falciform process of the sacrotuberous ligament. Additionally, the reported likelihood of supraspinal perineural canal compression (unlikely, indeterminate, likely, highly likely) at this level and variations in infraspinal regions of the Alcock’s canal and terminal nerve branches were also collected. Figure 1 demonstrates the course of the pudendal nerve. Figure 2 shows MRI views of the pelvic area including the pudendal nerve and associated structures.
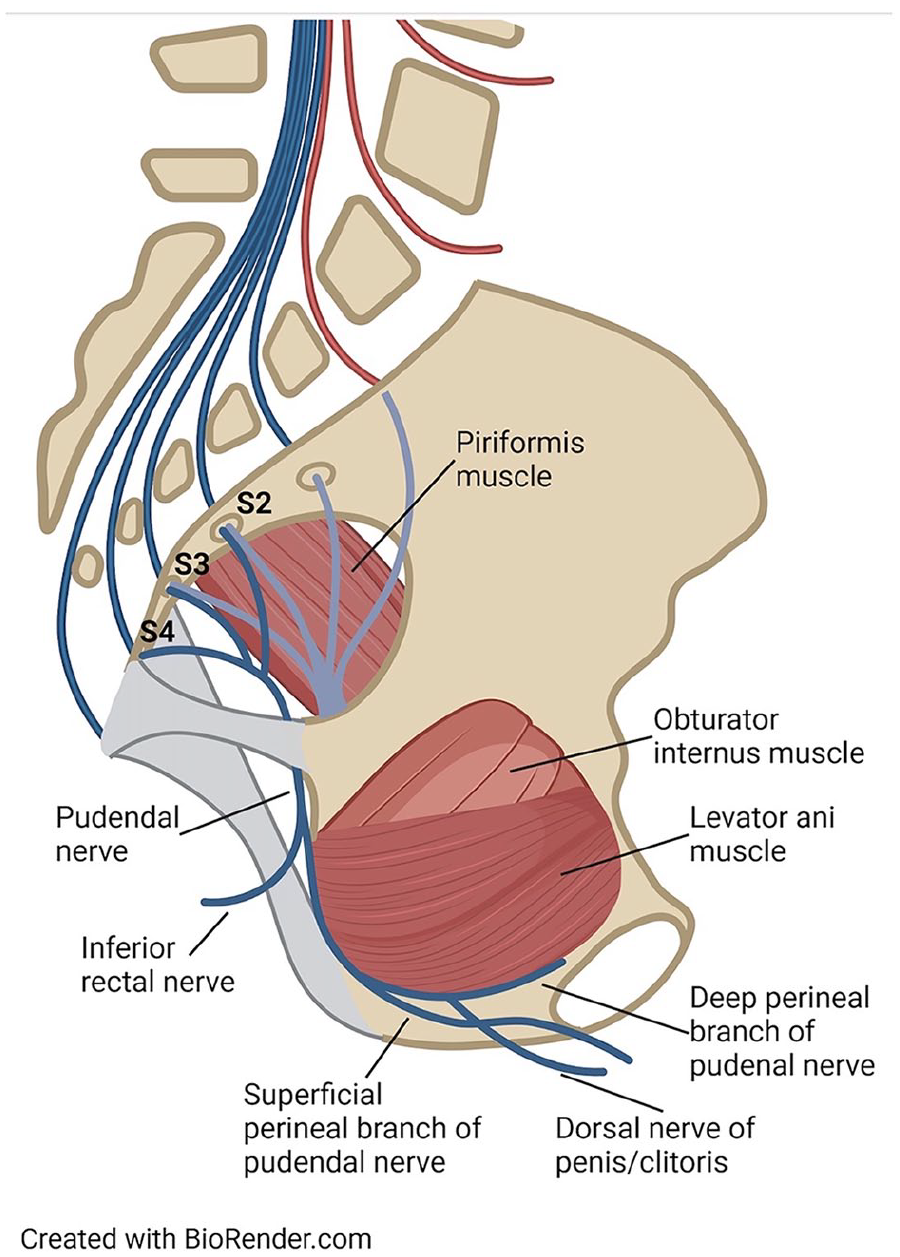
The course of the pudendal nerve.
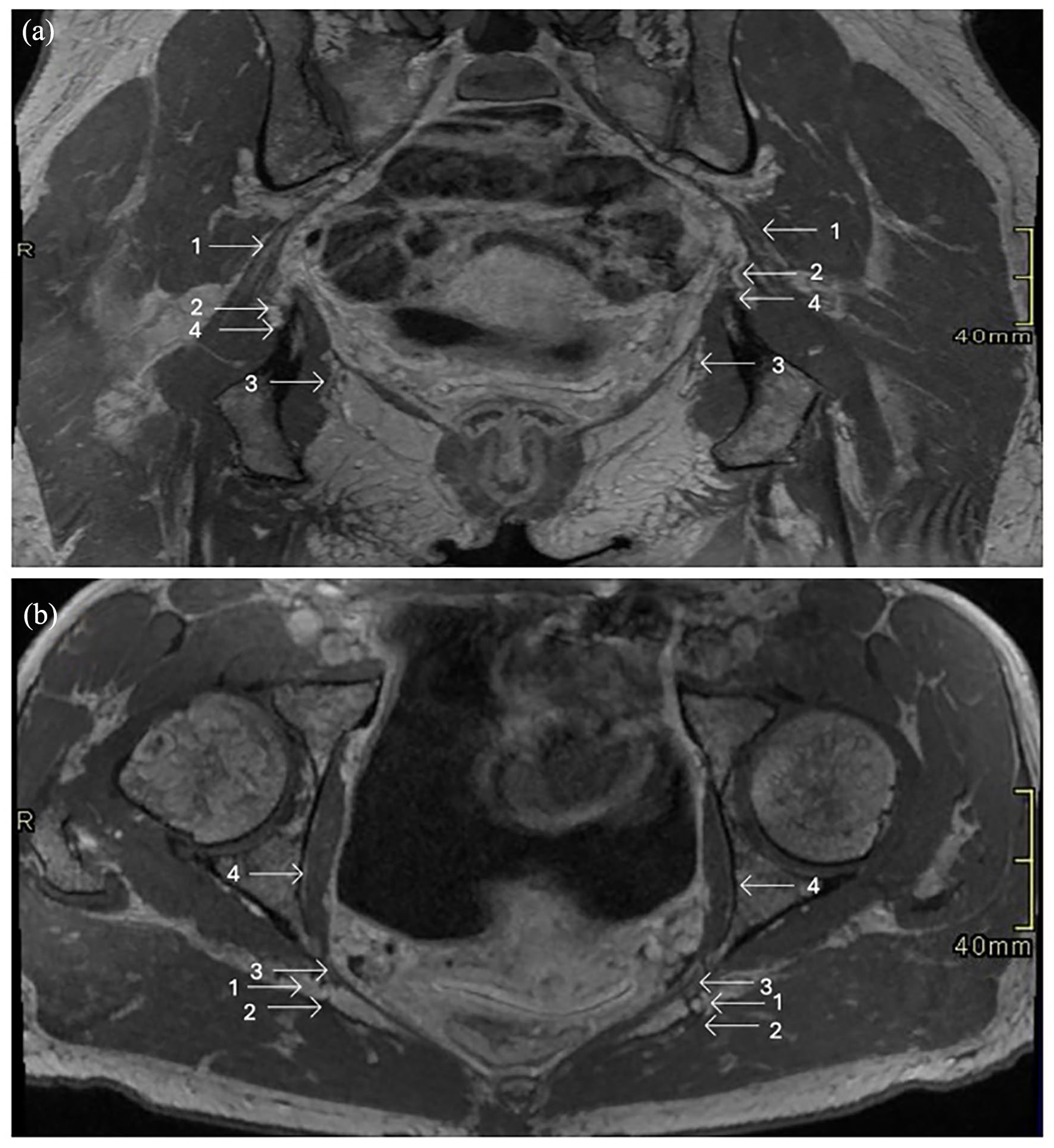
Pudendal nerve anatomy in a female pelvis on magnetic resonance imaging: (a) Coronal oblique T1 weighted MRI. 1 = sciatic nerve, 2 = pudendal nerve above the ischial spine, 3 = pudendal nerve within Alcock’s canal, 4 = ischial spine. (b) Axial T1 weighted MRI pudendal nerves at the ischial spine. 1 = pudendal nerve 2 = sacrotuberous ligament, 3 = sacrospinous ligament proximal to insertion on ischial spine, 4 = obturator internus muscle.
Descriptive statistical analysis was performed using IBM SPSS Statistics for Windows, Version 26.0 (Armonk, NY: IBM Corp). Data are expressed as frequencies, or median and interquartile range. Continuous data were assessed for normality using histogram plots. Categorical data were analysed using Pearson’s Chi-square or Fisher’s exact test. To explore potential associations between anatomical variations and likelihood of perineural compression at or above the ischial spine, the latter was divided into two levels: unlikely/indeterminate and likely/highly likely. A
Results
A total of 136 unique patients had an MRI, including 124 women and 11 men and 1 transwoman. Median age at MRI was 38.5 years (range = 18–72 years). Relevant clinical history was available for 82 patients with high rates of previous intervention including laparoscopy (76.8%), neuropathic pain prescription (75.6%) and pelvic floor physiotherapy (80.5%).
To facilitate analysis, each patient’s left and right hemipelves were considered independently with the sample of hemipelves (
No abnormal variations were observed in the expected courses of the superior and inferior gluteal nerves, sciatic nerve, posterior femero-cutaneous nerve and inferior cluneal nerve in the views obtained. Several anatomical variations of the pudendal nerve and its perineural structures were identified in our neuromuscular CPPP cohort which were predominantly structural findings affecting the space between the sacrospinous and sacrotuberous ligament at or above the level of the ischial spine. The frequency of these variations are outlined in Table 1.
Anatomical variations of the pudendal nerve identified on magnetic resonance imaging.

CM: coccygeus muscle; FP: falciform process; SCJ: sacrococcygeal junction; SSL: sacrospinous ligament; STL: sacrotuberous ligament.
The minimum perineural canal width at or above the ischial spine was reported for 261 hemipelves. Perineural canal widths were not parametrically distributed. Median width across all hemipelves was 4.0 mm (minimum = 1.0 mm; maximum = 11.0 mm; interquartile range = 2.2–5.6 mm). Perineural canal compression was rated as unlikely in 161 hemipelves (59.2%), indeterminate in 17 (6.3%), likely in 65 (23.9%) and highly likely in 29 (10.7%). The probability of association between the observed anatomical variations and likely/highly likely perineural compression are also shown in Table 1.
Interligamentous bands were present in almost two-thirds of hemipelves and were associated with concomitant perineural compression (
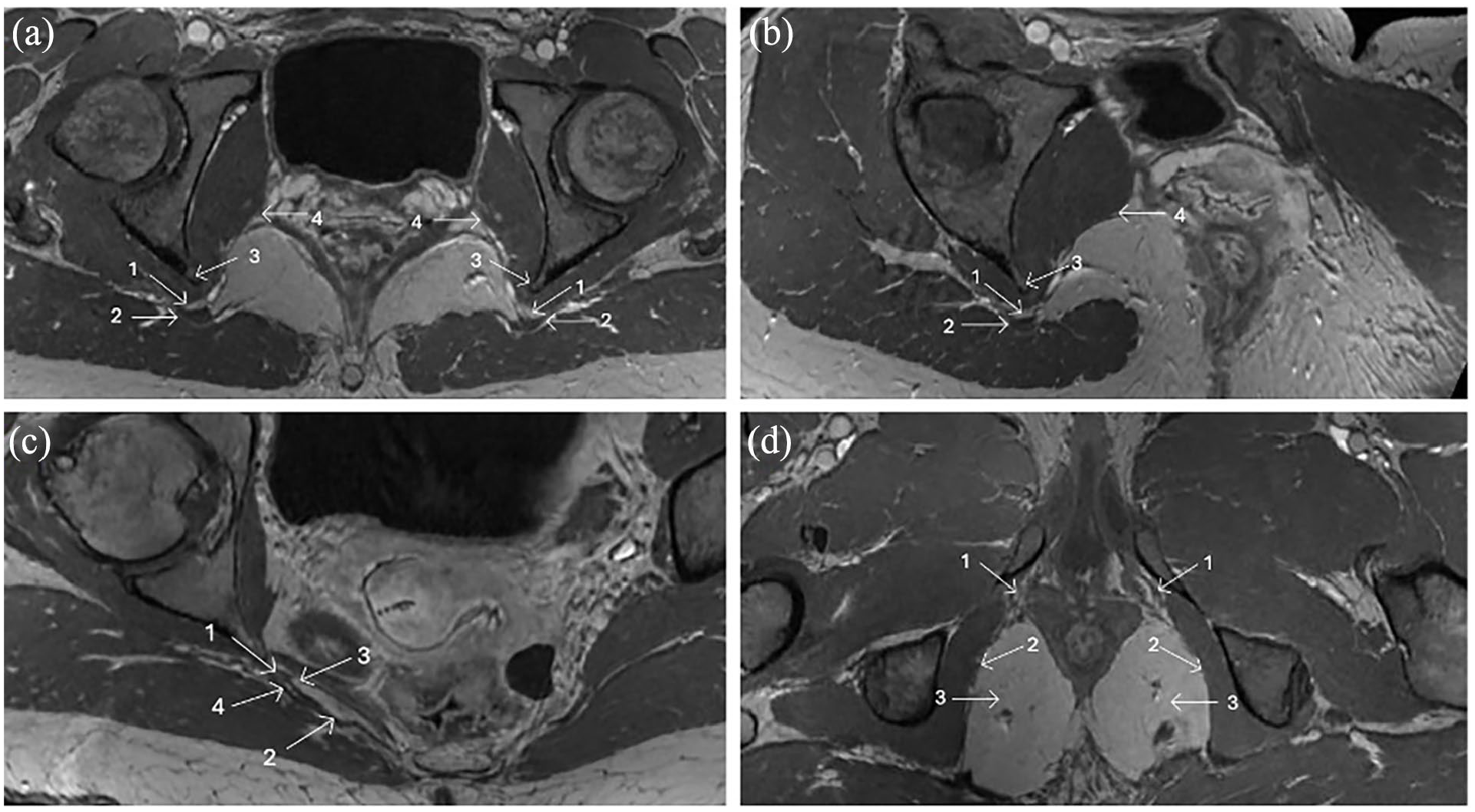
Anatomical variations of the pudendal nerve identified on magnetic resonance imaging: (a) Axial T1 weighted MRI bilateral abnormally thickened falciform process of the sacrotuberous ligament. 1 = pudendal nerve 2 = thickened falciform process 3 = ischial spine 4 = obturator internus muscle. (b) Axial oblique cross-section of A showing the relative narrowing due to the thickened falciform process. 1 = pudendal nerve 2 = thickened falciform process 3 = ischial spine 4 = obturator internus muscle. (c) Axial oblique T1 weighted MRI along the axis of a fused right sacrospinous and sacrotuberous ligament in their medial two thirds. 1 = minimal perineural fat in a narrowed pudendal nerve canal. 2 = thickened fused segment of the sacrospinous and sacrotuberous ligaments, 3 = separated lateral third sacrospinous ligament inserting onto the ischial spine. 4 = separated lateral third sacrotuberous ligament. (d) Axial T1 weighted MRI demonstrating bilateral thickened perineal branches of the distal pudendal nerves due to repetitive microtrauma in a male road cyclist. 1 = thickened distal perineal branches pudendal nerve, 2 = obturator internus muscle, 3 = ischioanal fossa.
The frequency of these variants as unilateral and bilateral findings in the overall patient cohort (
Alcock’s canal variations were demonstrated in 8/263 hemipelves, including prominent perineural vasculature within the canal (4/263), thickened obturator fascia (2/263), a ‘bulky’ obturator internus muscle (1/263) and signal changes indicating potential perineural fibrosis (1/263). Due to their small size, specific findings affecting the terminal branches were not observed. The expected course of terminal branches was evaluated for signal changes, reported for the perineal nerve in 6/250 hemipelves and the inferior rectal nerve in 3/250 hemipelves.
The maximal thicknesses of the pelvic muscles were not parametrically distributed. The median maximal thickness of the obturator internus in reported in 261 hemipelves was 20.7 mm (minimum = 11.0 mm, maximum = 28.4 mm, interquartile range (18.0–23.0)). The median of the pelvic floor in 252 hemipelves was 2.9 mm (minimum = 1.2 mm, maximum = 6.6 mm, interquartile range (2.2–3.5)). The median of the coccygeus in 252 hemipelves was 2.2 mm (minimum = 0.7 mm, maximum = 12.0 mm, interquartile range (1.9–2.7)). The median maximal thickness of the piriformis in 268 hemipelves was 18.7 mm (minimum = 9.6 mm, maximum: 33.6 mm, interquartile range (16.0–21.7)).
Recognising that pudendal neuralgia and PFTM are not exclusively female presentations, available MRI data for male (
Discussion
Pelvic muscle and pudendal nerve dysfunction are known contributors to chronic pelvic and perineal pain5,24 but can be challenging to diagnose. Triggers including prior surgery or procedures, pelvic organ descent, childbirth and pelvic floor dysfunction are all contributory. Imaging is increasingly used to aid diagnosis in a systematic and, reproducible manner. MRI has the potential to provide valuable diagnostic, prognostic and therapeutic information in suspected cases of pudendal neuralgia, including ruling in or excluding structural nerve compression/entrapment, characterising the location and possible mechanism of pathology and identifying concurrent pain-generating pathology.17,18 A clearer diagnostic pathway that demonstrates compression or tethering of the nerve within focal narrowing of the canal may provide more directed treatment such as the potential to decompress the canal by physiotherapy, 25 or interventional means. 26
Data describing MRI for this purpose is heterogenous, without robust quantitative data. A narrative study described MRI findings in 200 patients in a prospective evaluation of novel approaches to surgical decompression, however, no quantitative data were included. 18 A retrospective study of 580 patients reported hyperintensity and distortion of the pudendal nerve pathway in 78% of patients investigated, with concurrent gluteal and sciatic nerve encroachment in 50%. 23 Another retrospective study of imaging-guided nerve block outcomes provided some description of abnormal MRI findings but did quantitate or qualify the 90 MRI studies defined as ‘positive’ for pudendal nerve abnormality. 27 The inability to reliably reproduce data based on imaging studies limits the utility of MRI in this setting, with insufficient evidence to establish radiological criteria for this presentation.
This study describes associated changes in and around the pudendal nerve canal and not the nerve itself. The results highlight the anatomical variability of the sacrospinous and sacrotuberous ligaments that are closely related to the pudendal nerve in its proximal course. Ligamentous variations significantly associated with perineural compression included interligamentous bands that were identified in 63.8% of hemipelves but are not previously reported in MRI studies. Sacrospinous and sacrotuberous thickening has been described previously17,27,28 and were similarly noted in our cohort, and detailed imaging suggests that there are frequent findings of close apposition/fusion and delayed separation of these ligaments. Radiological variations of the falciform process are also a novel finding in this study. These results corroborate with the most common zones of pudendal nerve entrapment reported in previous deceased donor 21 and intra-operative 27 studies, specifically at the ischial spine within the interligamentous space and by the falciform ligament at the entrance to the Alcock’s canal, thereby demonstrating their reproducibility on imaging.
Structures below the ischial spine were poorly defined in this study, representing a limitation of pudendal nerve MRI in visualising infraspinal structures. Findings of obturator fascia thickening, scarring and venous dilation within Alcock’s canal are all reported previously17,18,27 but were rare occurrences in our cohort, despite the Alcock’s canal being another commonly cited entrapment zone. Distal branch findings were similarly rare, with experiences of inconsistent visualisation reported previously.19,29
Other associated MRI findings reported include perineural vessel dilation/congestion and muscular involvement. In this study, prominent supraspinal perineural vasculature was uncommon and not associated with compression. Generally, isolated venous dilation without other features of nerve compression is unlikely to be clinically significant given that dilated, incompetent veins are a common incidental finding in asymptomatic pre-menopausal women. 30 A limited prospective study of 200 patients highlighted entrapment in context of obturator internus and piriformis muscle asymmetry at the entrance of Alcock’s canal and greater sciatic notch. 18 The prevalence and definition of ‘asymmetry’ were not reported, and by comparison the present findings demonstrate only one case of ‘bulky’ obturator internus causing mild narrowing of Alcock’s canal. Overall, these results suggest that vascular and muscular compression are unlikely mechanisms of pudendal nerve entrapment.
There are several proposed risk factors for acquired pudendal nerve injury including obstetric trauma, chronic straining/constipation, repetitive activities such as cycling, and iatrogenic injury from pelvic, colorectal and lower urinary tract surgeries.20,21,31 Congenital or paediatric cases of pudendal neuralgia have not been reported. Accurate documentation of predating injuries was not feasible in this retrospective study, however potential causes of targeted and generalised injuries may be cross-referenced to imaging variations in future studies.
A limitation of this study is that clinical correlation of MRI findings needs to be compared to MRI appearances in asymptomatic controls. Funding limitations prohibited the inclusion of an asymptomatic control group in the current study. Another limitation is the use of investigation by MRI as the main study inclusion criteria. Patients were referred for MRI when gynaecologists experienced in the management of CPPP determined symptoms were suggestive of a possible pudendal nerve contribution to symptoms. This may lead to a selection bias with the anatomical variations noted. However, varied presentations of CPPP are expected and this approach may be more realistic in capturing patients for whom further diagnostic work-up is deemed appropriate. The application and documentation of clinical criteria for pudendal neuralgia would be relevant in a future prospective study comparing pre-intervention MRI findings with response to treatment (i.e. pudendal nerve block) which is currently an essential criterion for diagnosis by the Nantes criteria.
The technical limitations of MRI in obtaining reproducible pelvic muscle measurements, given the multiplanar course and varying morphology of pelvic muscles, suggest that this is unlikely to be helpful in a reproducible method for categorising or diagnosing muscular contributors to CPPP. The widest cross-section of the oblique pelvic muscles, is poorly reproducible, with measurements of the piriformis are likely the most accurate given its largely transverse course. Muscle measurements may be further complicated by other structural abnormalities such as avulsion or prolapse that were not reported in this study. The greater accessibility and ability for real-time capture of functional maneouvres means 3D/4D ultrasound 32 likely remains a superior modality for pelvic floor assessment at this time. MRI may provide useful information for deeper structures that are not assessable on ultrasound, such as the piriformis muscle, hip joints and surrounding musculature, lumbosacral structures and the bony pelvis. Importantly, a thorough clinical assessment remains the first and most important step in diagnosis to assess individual risk factors for PFTM and the contribution of nociplastic mechanisms.
In conclusion, this retrospective study in a neuromuscular CPPP cohort adds anatomical information to a poorly defined evidence base for an increasingly utilised investigation for pudendal neuralgia. Several anatomical variations of the perineural structures of the pudendal nerve were identified and associated with radiological nerve compression, however, MRI appears limited in the visualisation of the infraspinal pudendal nerve and its perineal branches and localisation of widest pelvic muscle cross-sections. The clinical significance of MRI findings and role of MRI in the assessment of pudendal neuralgia require further exploration in a prospective evaluation study.
Supplemental Material
sj-pdf-1-pev-10.1177_22840265241303813 – Supplemental material for Anatomical neuromuscular variations determined by magnetic resonance imaging in chronic pelvic pain
Supplemental material, sj-pdf-1-pev-10.1177_22840265241303813 for Anatomical neuromuscular variations determined by magnetic resonance imaging in chronic pelvic pain by Yuanyuan Peng, Toos Sach, Jason Chow and Jason Abbott in Journal of Endometriosis and Pelvic Pain Disorders
Footnotes
Acknowledgements
We acknowledge the Gynaecological Research and Clinical Evaluation (GRACE) Unit research coordinators Dr Cecilia Ng and Dr Adriana Meneses for their support and guidance in this study.
Author contributions
JA researched literature and conceived the study with input from TS and JC. JA gained ethics approval. All authors were involved in protocol development. YP completed data collection and data analysis and wrote the first draft of the manuscript. TS provided MRI images and interpretation described in the legends. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Data availability
All data generated or analysed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
Ethical approval for this study was obtained from the South Eastern Sydney Local Health District Human Research Ethics Committee on March 7th 2022 (Identifier: 2021/ETH11992).
Consent to participate
Informed consent was not sought nor required for the submitted study due to retrospective de-identified patient data use only. This has been ethics approved.
Consent for publication
Informed consent was not sought nor required for the submitted study due to retrospective de-identified patient data use only. This has been ethics approved.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
