Abstract
Background:
Although significant advancements have been made in the diagnostic and therapeutic methods of gynecological oncology, ovarian cancer continues to pose a significant challenge. The measurement of CA-125 levels is commonly considered the preferred approach for assessing the necessity of surgical intervention, hence guaranteeing a conclusive diagnosis. The objective of this study is to conduct a comparative analysis of existing literature about the effectiveness of serum CA-125 levels in distinguishing between endometriosis-associated ovarian cancer (EAOC) from ovarian endometrioma.
Material and methods:
The researchers conducted separate searches in the PubMed and Web of Science databases to identify peer-reviewed papers that examined the serum or plasma CA-125 marker. The search was conducted from the start of both databases until April 4th, 2024.
Results:
After conducting a literature search, a total of 114 studies were identified for review. Out of these, three studies satisfied the inclusion criteria and were consequently included. The results of the pooled analysis indicate that there is no statistically significant difference between the groups (336.3 ± 667.6 vs 530.6 ± 548.9, respectively; standard deviation = 0.51; 95% confidence interval: −0.63 to 1.64; p = 0.39).
Conclusion:
Unfortunately, the biomarker CA-125, which is commonly selected, has exhibited restricted effectiveness in distinguishing between EAOC and endometriomas. The existing body of literature on this topic indicates a significant dearth of research regarding the use of CA-125 as a means of differentiating between EAOC and endometriomas. Enhanced diagnostic techniques are necessary for the detection and characterization of ovarian tumors, encompassing those originating from endometriosis.
Introduction
Despite notable progress in the diagnostic and therapeutic approaches in the field of gynecological oncology, ovarian cancer remains a severe obstacle, especially endometriosis-associated ovarian cancer (EAOC). An array of biomarkers and clinical diagnostic indicators have been explored. At present, the utilisation of ultrasound examination in accordance with the International Ovarian Tumour Analysis (IOTA) criteria, 1 in conjunction with the evaluation of serum Ovarian cancer-related tumor marker CA-125 levels, is widely regarded as the preferred method for determining the need for surgical intervention, hence ensuring a definitive diagnosis.2,3 Nevertheless, the diagnostic procedures suggested have not been successful in mitigating the mortality related to ovarian cancer. CA-125, a Mucin-16 glycoprotein, often shows elevated levels in patients with ovarian cancer, with a rise observed in around 80% of patients, thus providing evidence for diagnosis.3–5 However, CA-125 levels may also be elevated in benign conditions, such as endometriosis, which is a chronic and progressive ailment that impacts approximately 15% of women, what make it less useful in patients with both conditions.
In certain instances, the development of ovarian cancer may arise from the presence of preexisting endometriosis on the ovaries, a condition known as endometrioma. The clinical overlap between EAOC and endometriomas lacks specificity. Due to the generally asymptomatic progression of ovarian cancer, lesions are frequently diagnosed at an advanced stage, contributing to a higher mortality rate. 3 Moreover, previous findings indicate that neither biomarker screening nor routine ultrasound correlates with improved survival rates in ovarian cancer, and the coexistence of endometriomas complicates both screening and diagnosis. 2 Consequently, akin to endometriomas, EAOC is commonly detected during the advanced phases of the condition, posing difficulties in its management. 5 Distinguishing between advanced EAOC on top of preexisting endometriomas using CA-125 and ultrasound data is scarce about this differentiation.
The objective of this study is to conduct a comparative analysis of existing literature about the effectiveness of serum CA-125 levels in distinguishing between endometriomas and endometriosis-related ovarian neoplasms.
Material and methods
The preferred reporting items for systematic reviews and meta-analysis (PRISMA) statement guidelines were followed when performing this study. 6 The authors independently searched the PubMed and Web of Science databases for peer-reviewed studies reporting on serum or plasma CA-125 marker from databases inception to 4-th April 2024. The keyword terms were: ‘CA-125’ OR ‘Cancer Antigen-125’ AND ‘endometriosis-related ovarian neoplasms’ OR ‘ovarian endometrioma’ OR ‘ovarian cancer’ OR ‘endometriosis’ OR ‘endometrioma’. We have imposed English language restriction. References of the previously published meta-analyses or reviews were also searched for eligible trials. The quality assessment of included studies was done using the Newcastle–Ottawa Scale (NOS).
We assessed all pertinent study designs for inclusion. This review encompassed studies that consisted of women diagnosed with endometriosis-associated ovarian cancer, as well as a control group of patients diagnosed with ovarian endometrioma. Including the assessment of the CA-125 level in these patients was an essential requirement for their inclusion.
All types of media outlets were excluded, as well as expert opinions, letters to the editor, case reports, case series, and conference papers that were not published in their entirety. Additionally, the absence or inappropriate description of a control group was an exclusion criterion.
Pooled analysis was done in RevMan software (Review Manager (RevMan). Version 5.4. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, Denmark). The standardized mean difference (SMD) and 95% CIs are reported and were generated by using a random effects model according to the Der-Simonian Laird model. Statistical significance was considered in p-values < 0.05.
Results
The literature search indicated 114 studies for review, three of which met the inclusion criteria and were, therefore, included, as shown in the study flow chart (Figure 1).7–9 All included studies were at low risk of bias, as assessed by the NOS tool.
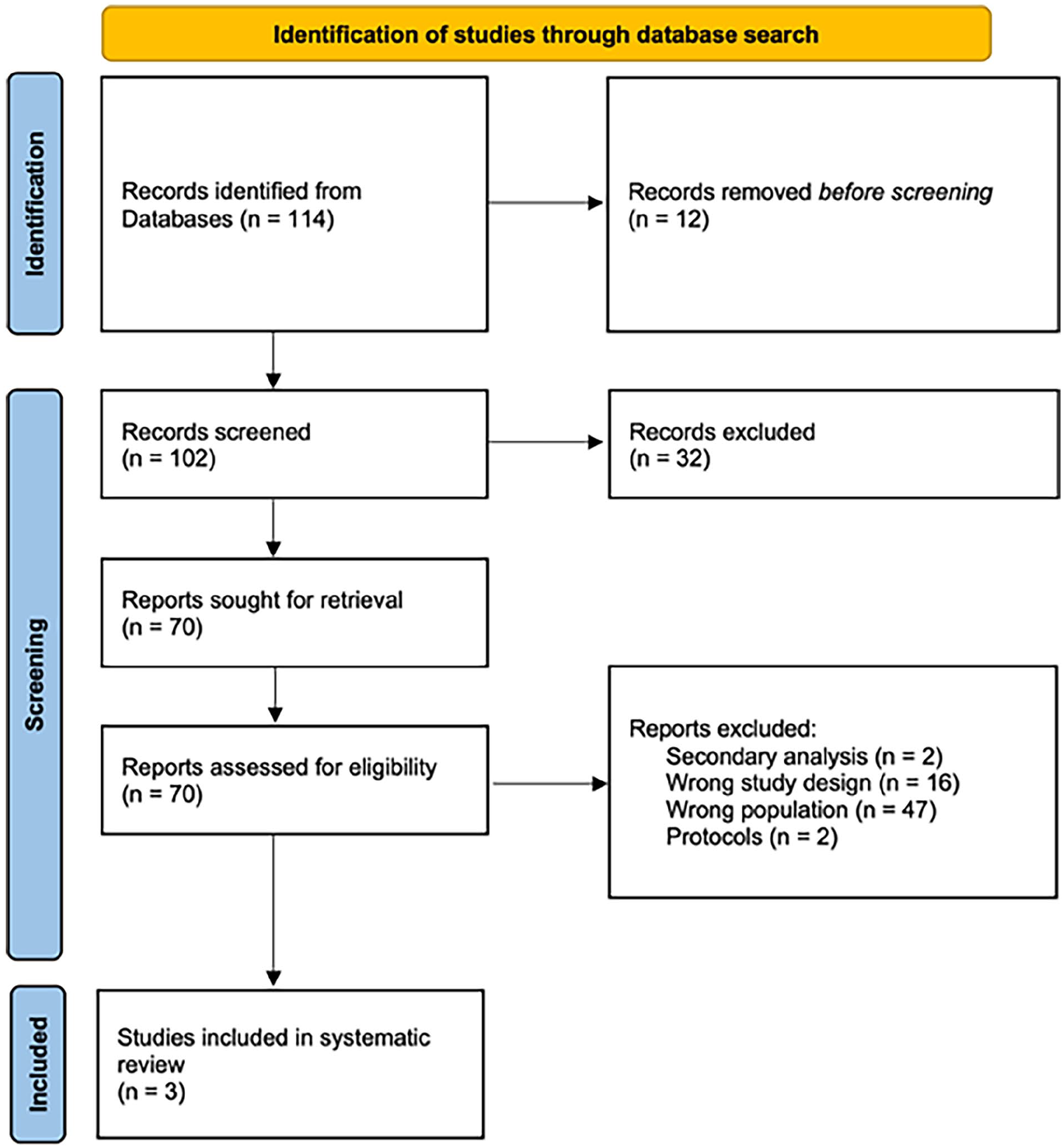
PRISMA flow diagram.
Three eligible trials with a total of 814 participants (endometriosis-associated = 357; non-endometriosis-associated primary ovarian cancer = 263) reported the CA-125 levels. Pooled analysis shows a non-significant statistical difference between those groups (336.3 ± 667.6 vs 530.6 ± 548.9, respectively; SMD = 0.51; 95% CI: −0.63 to 1.64; p = 0.39; Figure 2).
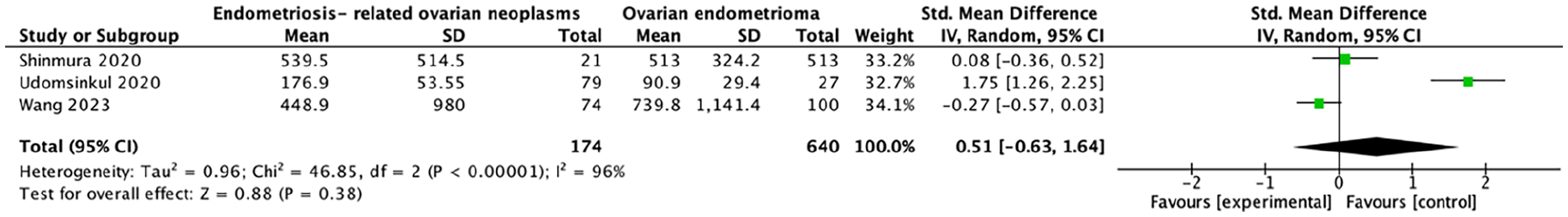
Metanalysis plot chart of the included studies.
Discussion
Our analysis indicates that CA-125 is not a reliable marker for differentiating endometriomas from EAOC.7–9 Several investigations, such as those conducted by Taniguchi et al. 10 and Kadan et al., 11 have examined the levels of serum CA-125, yet have yielded inconclusive findings. Although the aforementioned studies did not observe any statistically significant disparities in CA-125 levels between individuals diagnosed with EAOC and those diagnosed with ovarian endometriomas. 7 The study of Gentry-Maharaj’s et al. 12 suggested that patients with endometriosis-associated ovarian cancer generally exhibited lower CA-125 levels in comparison to those with typical epithelial ovarian cancer.
CA-125 is synthesised by both malignant cells and non-cancerous tissues in the pelvic and abdominal regions, including the endometrium, fallopian tubes, ovaries, pleura, and peritoneum. Endometriosis can stimulate an inflammatory response and irritation, which can result in an elevation of CA-125 production, causing an increase in its concentration in the bloodstream. Due to its association with both malignant and non-malignant illnesses, CA-125 can be detected at higher levels in several benign diseases. The absence of specificity diminishes the dependability of CA-125 as an indicator for differentiating between ovarian cancer and non-malignant diseases such as endometriosis.
However, when ovarian cancer coexists with preexisting endometriosis, there can be noticeable variations in CA-125 levels. 13 This is because the growth rates of cancerous cells differ from those of endometrial cells. The lack of substantial variations in CA-125 levels between ovarian endometriosis and EAOC in our study may be attributed to the diverse characteristics of the group under investigation. CA-125 levels exhibit variability based on the age of women, with levels being influenced by hormonal fluctuations, menopause status, and other age-related ailments.14–16 The comparison may be confounded by potential differences in baseline CA-125 levels between younger and older women. In addition, larger endometriomas and more advanced stages of endometriosis may exhibit elevated CA-125 values. CA-125 is produced not only by endometrial tissue but also by other tissues in response to inflammation or chronic disorders such as heart failure or pelvic inflammatory disease.17–19 Consequently, elevated levels of CA-125 can be attributed to these conditions as well. Furthermore, the levels of CA-125 can be influenced by the phase of the menstrual cycle and the menopausal status.20,21 During menstruation and in premenopausal individuals, higher levels of CA-125 are typically detected. The levels of CA-125 can also be influenced by hormonal medications, including as oral contraceptives and hormone replacement therapy.22–24 Differences in the usage of these medicines between women with endometriomas and those with EAOC could potentially complicate the interpretation of the data.
Although serum CA-125 is commonly used alongside transvaginal ultrasonography to distinguish adnexal masses, its effectiveness in diagnosing ovarian malignancies linked with endometriosis is still restricted. However, the IOTA research highlights the efficacy of transvaginal ultrasonography in detecting epithelial ovarian cancer before surgery in cases with a high malignancy risk. 1 The present methodological framework facilitates the precise evaluation of the malignancy potential of ovarian tumors by physicians through the analysis of ultrasound features, hence providing valuable information for surgical decision-making.25,26 Nevertheless, the identification and treatment of ovarian cancer are significantly dependent on the diligent and specialized oncological vigilance of healthcare practitioners. Experienced healthcare professionals play a pivotal role in the interpretation of diagnostic findings and the identification of subtle indicators that may suggest the existence of ovarian cancer. This, in turn, enhances the rates of early detection, which are of utmost importance for patient prognosis. However, advanced ovarian endometriomas can hinder ultrasound evaluation, potentially leading to an underestimation of ovarian cancer risk. 3
Within the preoperative setting, Udomsinkul et al. 8 conducted a study that revealed ovarian tumours as frequently difficult to categorise as either benign or malignant. In this study, prior to surgical intervention, a limited number of cases of EAOC were identified as ovarian cancer. A precise diagnosis is crucial for the development of an efficient management plan, which may entail counselling, a collaborative approach, consultations with an oncologist, and efforts to preserve fertility. It is essential to identify patients who are at high risk for tumour rupture during ovarian cystectomy efforts. This is because spilling during the surgery can alter the patient’s disease stage and increase the requirement for additional chemotherapy. Moreover, the absence of a diagnosis for ovarian cancer heightens the likelihood of subsequent surgical procedures for re-staging.
Wang et al., in their study, demonstrate that there were no significant differences in the epidemiological or pathological characteristics and prognosis between ovarian tumours associated with endometriosis and those not associated with endometriosis, as determined by the Sampson and Scott criteria.9,27,28 The results of this study provide evidence in favour of the dualistic interpretation of ovarian cancer, indicating that the development of both endometrioid and clear cell carcinomas may be attributed to the advancement of endometriosis. This observation leads to a reevaluation of the rigorous standards for malignant transformation in endometriosis, highlighting the necessity for updated diagnostic methods to account for the intricacies of this condition.
Crucial preoperative factors for distinguishing between endometriomas and EAOC include age, tumor size, and the presence of mural nodules. Interestingly, symptomatic women with endometriosis often undergo more frequent ultrasounds, facilitating the early-stage detection of over 60% of endometriotic cyst-associated ovarian cancers.7–9 However, the prognostic significance of this early detection remains unclear and warrants further study. Differentiation between ovarian alterations and ovarian cancer has been achieved by the utilization of alternative biomarkers or a combination of clinical of several biomarkers for example ROMA algorithm, using despite CA-125 the HE-4 and menopausal status of the risk patients.3,29,30 In addition, the domain of gynecological oncology is currently experiencing the advent of novel diagnostic methodologies, including the use of Bionized NanoFerrite particles (BNF), genetical testing, and endometrial tissue analysis.31,32 Presently, these tests are predominantly employed for the identification of endometriosis, however they possess the capacity for wider-ranging applications. As scientific investigations advance and yield novel understandings about the molecular and genetic indicators of ovarian cancer, it is plausible to modify these assays to facilitate the timely identification and distinction of ovarian neoplasms. The dynamic nature of diagnostics has the potential to greatly expand our capacity to diagnose and treat ovarian cancer with more efficacy, ultimately leading to improved patient outcomes. Nevertheless, the dearth of scholarly investigations in this domain is a significant challenge, and there exists a dearth of distinction in terms of non-invasive methodologies, as the majority of conclusions are drawn solely from histopathological outcomes. Numerous biomarkers have been investigated and distinct glycosylation patterns observed in cancer cases offer a diagnostic potential.30,33 Nevertheless, the available information about their efficacy remains constrained and further research is needed. 34
Nevertheless, our analysis has several limitations, including a limited number of included studies and a restricted population size. Our study primarily focused on analyzing ovarian cancer that emerges from endometriosis, rather than examining the simultaneous existence of endometriosis and ovarian cancer. There is a need to enhance research on the correlation between endometriosis and ovarian cancer and to perform thorough investigations in this area since it remains a relatively untouched subject. These limitations may arise from challenges in categorizing patients, as endometriosis can vary considerably in its stage and is prone to errors due to the possible coexistence of endometriosis and ovarian cancer, as well as ovarian cancer developing as a consequence of endometriosis. The complex character of this event highlights the need for more research to explore the likely connections.
Conclusion
Regrettably, the biomarker CA-125, which is widely chosen, has demonstrated limited efficacy in differentiating between EAOC and endometriomas. The literature study on this subject reveals a notable scarcity of studies pertaining to the application of CA-125 for distinguishing between EAOC and endometriomas. Continuing research on the correlation between endometriosis and ovarian cancer is crucial in order to identify novel biomarkers to improve predictive assessments, hence enhancing patient care and practical implementation.
Footnotes
Data availability statement
Research data are shared on request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
Conceptualization: SF and MP; Methodology: MP and LS; Formal analysis: MC; Investigation: SF and MP; Resources: MC; Writing – original draft preparation: SF, MP and MB; Writing – review and editing: LS, KS, and MC; Supervision: LS; Project administration: LS. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
