Abstract
One of the most challenging issues in gynecologic oncology is the high mortality rate of ovarian cancer, largely due to detection of disease in advanced stages. Women with early-stage disease have a significantly improved survival rate and may not require chemotherapy. Thus, the issues to examine are whether there are methods to improve early detection, thereby resulting in a reduction in mortality. Several large, randomized, clinical trials have recently completed evaluating CA 125 and transvaginal sonography as effective strategies to accomplish this goal. These issues and the results of the recent trials will be reviewed in this article.
Keywords
Background
Ovarian cancer is one of the most common gynecologic malignancies and is the fifth leading cause of cancer mortality in women in the USA [1]. The American Cancer Society estimated that there were 21,650 new cases of ovarian cancer and 15,520 women died of the disease in 2008 [1]. With advances in surgery, incorporation of taxanes into first-line therapy, intraperitoneal chemotherapy, introduction of several nonplatinum cytotoxic and targeted therapies, the median survival for patients with advanced-stage disease has improved, depending on the extent of residual disease following initial surgery. From 1975–2004, the overall 5-year survival rate has gone up from 37 to 46% [1]. If diagnosed with disease confined to the ovaries, the 5-year survival rate is 92%. However, less than 20% of ovarian cancers are detected with stage I disease. For women with regional (early-stage) and distant disease (late-stage), 5-year survival rates are 71 and 30%, respectively. The 10-year survival rate for all stages is 38% [1]. These statistics argue for early detection and even more so for new prevention strategies. For a screening test to be effective, detection at an early stage is essential and this test must result in a reduction in mortality.
Risk factors for ovarian cancer are fairly nonspecific and fail to identify a distinct phenotypic prototype. The risk factors are listed in Box 1 [2]. As with other cancers, family history is important, but most women do not have a positive family history for this or other related cancers (breast, colon etc). Pregnancy, long-term use of oral contraceptives, tubal ligation and hysterectomy are protective and reduce the risk of ovarian cancer.
There are no specific symptoms that characterize early-onset ovarian cancer from advanced-stage disease. Many of the symptoms are attributable to benign gastrointestinal, genitourinary, musculoskeletal or gynecologic conditions. Persistent symptoms most commonly associated with ovarian cancer include [3]:
Bloating
Pelvic or abdominal pain
Difficulty eating or early satiety
Urinary urgency or frequency
These symptoms have been reported in the majority of patients with early- and late-stage ovarian cancer [3].
Etiopathogenesis
Understanding the biology of ovarian epithelial tumors is key to developing effective screening strategies and early detection. Ovarian cancer is a spectrum of diseases and not a single disease entity. However, current clinical management of ovarian cancer fails to incorporate this information into the treatment of patients. Given the heterogeneity of this disease, improvements in long-term survival might be achieved by translating recent insights at the molecular and cellular levels into personalized individual treatment strategies. It is widely believed that ovarian epithelial tumors arise in the coelomic epithelium that covers the ovarian surface. Recent histopathologic examination of the distal fallopian tube and molecular genetic analysis of ovarian cancers have begun to question this belief. Emerging data suggest that high-grade serous tumors of the ovary differ from all low-grade serous ovarian carcinomas in terms of their development, prognosis, pathology and underlying molecular genetic alterations. This has led to the classification of ovarian cancers into type 1 tumors, which are low-grade and slowly developing tumors and type 2 tumors, which are rapidly progressing high-grade carcinomas and may originate in the fallopian tube [4–6].
Risk factors for ovarian cancer.
Age: peak incidence in the age group 65–85 years
Infertility or subfertility
Nulliparity
Early menarche
Late menopause
Family history of breast cancer
Family history of ovarian cancer
Family history of hereditary nonpolyposis colon cancer
Highly industrial countries
Data taken from [2].
As noted, extrauterine serous carcinomas are presumed to arise from the ovary, endosalpinx and peritoneal surface [6]. The majority of serous carcinomas in symptomatic women with
Screening
In the USA, the prevalence of ovarian cancer in postmenopausal women (one in 2500) significantly affects the strategies that can be effectively used in the early detection of ovarian cancer. Because of the prevalence, early detection of ovarian cancer requires a testing strategy with high sensitivity (>75% for stage I disease) and very high specificity (>99.6%) to achieve a positive predictive value of 10%. The high specificity is required to avoid alarming a large number of healthy women and triggering a cascade of expensive diagnostic tests and procedures [9]. Screening also presumes that detection of early cancer will improve survival by impacting mortality. However, as noted in the previous section, early-stage disease may be a separate clinical entity to late-stage disease and, therefore, detection at an early stage may not affect the overall survival of patients. Trials evaluating screening strategies will need to demonstrate more than a ‘stage shift’ (i.e., from late-stage to an earlier stage). An improvement in overall survival with long-term follow-up will be needed before such a screening strategy can be adopted for the general population.
Screening strategies include pelvic examinations, serum tumor markers, transvaginal sonography, proteomic approaches or combinations of these tests. Pelvic examination with or without rectal examination has not been demonstrated to be effective in screening for ovarian cancer [10].
Biomarkers
Many studies have evaluated CA 125. CA 125 is produced by mesothelial cells that line the peritoneum. The original CA 125 assay utilized the OC125 antibody that recognizes the CA 125 epitope on a high molecular weight glycoprotein. Monoclonal antibodies have been raised against other epitopes leading to the development of CA 125 II [ii]. Approximately 80% of epithelial ovarian cancers express increased levels of CA 125. However, only 50% of patients with early-stage disease will have an elevated level [12]. There are many benign disorders that are associated with elevated levels of CA 125. There are also other gynecologic cancers (fallopian tube, uterine and peritoneal cancer) and nongynecologic cancers (breast, pancreas and lung cancer) that can cause an elevation in serum CA 125 levels. For these reasons, CA 125 as a single method of screening for ovarian cancer has been disappointing. Its use in large randomized trials in combination with sonography is described later in this review.
Menon and colleagues prospectively evaluated the risk of ovarian cancer (ROC) in a prevalence screen using the ROC algorithm, which interprets serial CA 125 values [13]. Postmenopausal women were assigned to a control group or screen group using the ROC algorithm. If the screen was normal, annual screening was done. If the initial ROC calculation was intermediate, a repeat CA 125 was performed. If the screen was high risk or intermediate on retesting, a transvaginal sonogram was performed. Five ovarian cancers were detected in 144 women at elevated risk. The cancers were detected in stage I/II, one of these was a tumor of low malignant potential. The study reported a specificity of 99.8% and a positive predictive value of 19%. Based on this study, the ROC algorithm was adopted for use in the UK Collaborative Trial of Ovarian Cancer Screening (UKCTOCS) [14].
More than 30 serum biomarkers have been evaluated alone and in combination with CA 125. Some of the more promising include HE4, mesothelin, M-CSF, osteopontin, kalli-kreines, inhibins and soluble EGF receptor [15,16]. However, the report on the use of 24 different biomarkers from an analysis of prediagnostic specimens from the Prostate, Lung, Colorectal, and Ovarian Cancer (PLCO) study concluded that CA 125 had the best performance of these 24 biomarkers [16]. An earlier study also evaluated multiple markers, including leptin, prolactin, osteopontin, IGF-II, macrophage inhibitory factor and CA 125 [17]. Although the initial report suggested a very high specificity and sensitivity for this panel of markers in the early detection of ovarian cancer, this was not validated in the PLCO study. Further analysis of this earlier study revealed inclusion of low malignant potential ovarian cancer and stromal cell tumors in the patient population. Inclusion of these subtypes may have accounted for their favorable results and conclusions.
Recent advances in protein expression profiling (proteomics) may provide novel biomarkers. Protein patterns of tumors can be identified using mass spectrometry. However, plasma and urine proteome profiling for biomarker discovery has been hindered by technical challenges that significantly complicate data interpretation [18]. New fractionation and enrichment strategies are improving the technology; proteomics is a promising technology to aid in the early detection of ovarian cancer.
Transvaginal sonography
The low cost and wide availability of transvaginal ultrasound (TVS) has resulted in its use as a potential screening tool for the early diagnosis of ovarian cancer. Its use has led to an increase in the detection of asymptomatic adnexal masses, which can be found in approximately 10% of premenopausal women [19]. Studies have demonstrated a ‘stage shift’ using this technology as a screening tool. In one study, annual TVS screening was performed in more than 25,000 women who were either high risk for ovarian cancer (family history) or over 50 years of age [20], and demonstrated that annual screening with TVS was associated with a decrease in the stage of disease at detection and with case-specific mortality from ovarian cancer. It was not effective in detecting ovarian cancers in women who had normal ovarian volume. The authors suggest combining TVS with serial serum biomarkers to help identify tumors with normal ovarian volume and morphology [20].
Guidelines on TVS & biomarkers in screening for ovarian cancer
The US Preventive Services Task Force (USPSTF) found fair evidence that screening with serum CA 125 level or transvaginal ultrasound can detect ovarian cancer at an earlier stage than it can be detected in the absence of screening. However, the USPSTF also found fair evidence that earlier detection would likely have a small effect, at best, on the overall mortality from ovarian cancer. Because of the low prevalence of ovarian cancer and the invasive nature of diagnostic testing after a positive screening test, there is fair evidence that screening could likely lead to important harms. The USPSTF concluded that the potential harms of this combination of screening outweigh the potential benefits [10].
The American Cancer Society does not recommend screening for ovarian cancer in the general population [101]. In a Committee Opinion, The American College of Obstetricians and Gynecologists states that currently available screening tests do not appear to be beneficial for screening for ovarian cancer in low-risk asymptomatic women. The opinion recommends that the generalist obstetrician–gynecologists remain vigilant for the signs and symptoms of ovarian cancer, such as abdominal or pelvic pain and unexplained weight loss, and that these symptoms be evaluated by pelvic examination, CA 125, and/or ultrasonography based on the menopausal status of the patient [21]. In view of the recent large randomized screening trials (see below section on ‘Randomized clinical trials’) this committee opinion will be updated.
Given the high risk for ovarian cancer and fallopian tube cancer in women with mutations in
Management of patients at high risk for ovarian cancer
A Gynecologic Oncology Group study recently completed enrollment evaluating the most effective management plan for women at increased genetic risk of ovarian cancer risk [22]. It is a prospective, international, two-cohort, nonrandomized study that allows participants to choose between risk reducing salpingo–oophorectomy (RRSO) or ovarian cancer screening at the time of enrollment. By collecting prospective observational data on these patients, the authors intend to quantify and compare ovarian and breast cancer incidence in the two groups, assess the feasibility and selected performance characteristics of the ROC algorithm screening strategy, evaluate quality of life and nononcologic morbidity related to various interventions in at-risk women, and create a biospecimen repository for subsequent translational research. Accrual of participants was completed in November 2006 and final analysis of the data collected will not be analyzed until the 5-year follow-up period is completed in November 2011.
Given the limitations of current ovarian cancer screening, it has been widely recommended that women with
An American College of Obstetricians and Gynecologists Practice Bulletin recommends that women with hereditary breast and ovarian cancer syndrome be offered RRSO by 40 years of age, or when childbearing is complete [24]. For women with
A recent prospective, observational report from the Nurses' Health Study concluded that bilateral oophorectomy at the time of hysterectomy for benign disease was associated with a decreased risk for breast and ovarian cancer, but an increased risk for all-cause mortality, fatal and nonfatal coronary heart disease and lung cancer [26]. Limitations of the study included that it was observational and ovarian conservation was self-selected. However, the results are consistent with other studies [26]. Thus, RRSO to reduce the incidence and mortality from ovarian cancer can only be recommended for high-risk women with identified genetic mutations or family histories strongly suggestive of one of the genetic syndromes associated with a high risk of ovarian cancer.
Randomized clinical trials
The role of tumor markers in ovarian cancer screening can only be addressed in large-scale studies. Most of these studies seek to define a method by which screening may reduce mortality from ovarian cancer. A variety of screening protocols have been proposed to this end, primarily with TVS and the serum biomarker CA 125. Unfortunately, evidence from even the most recent of these large-scale studies does little to support the routine use of TVS and CA 125 as an ovarian cancer screening modality for the general population.
Buys
Partridge
Menon
The difference between the PLCO and UKCTOCS may be due to the use of CA 125 before referral for ultrasound and improvement in sensitivity and specificity with serial CA 125 testing.
Based on the data in aggregate from these two large studies, screening for ovarian cancer is feasible, but cannot yet be recommended for the general population (Table 1).
Comparison of outcomes of different ovarian screening strategies between the UKCTOCS and PLCO trials.
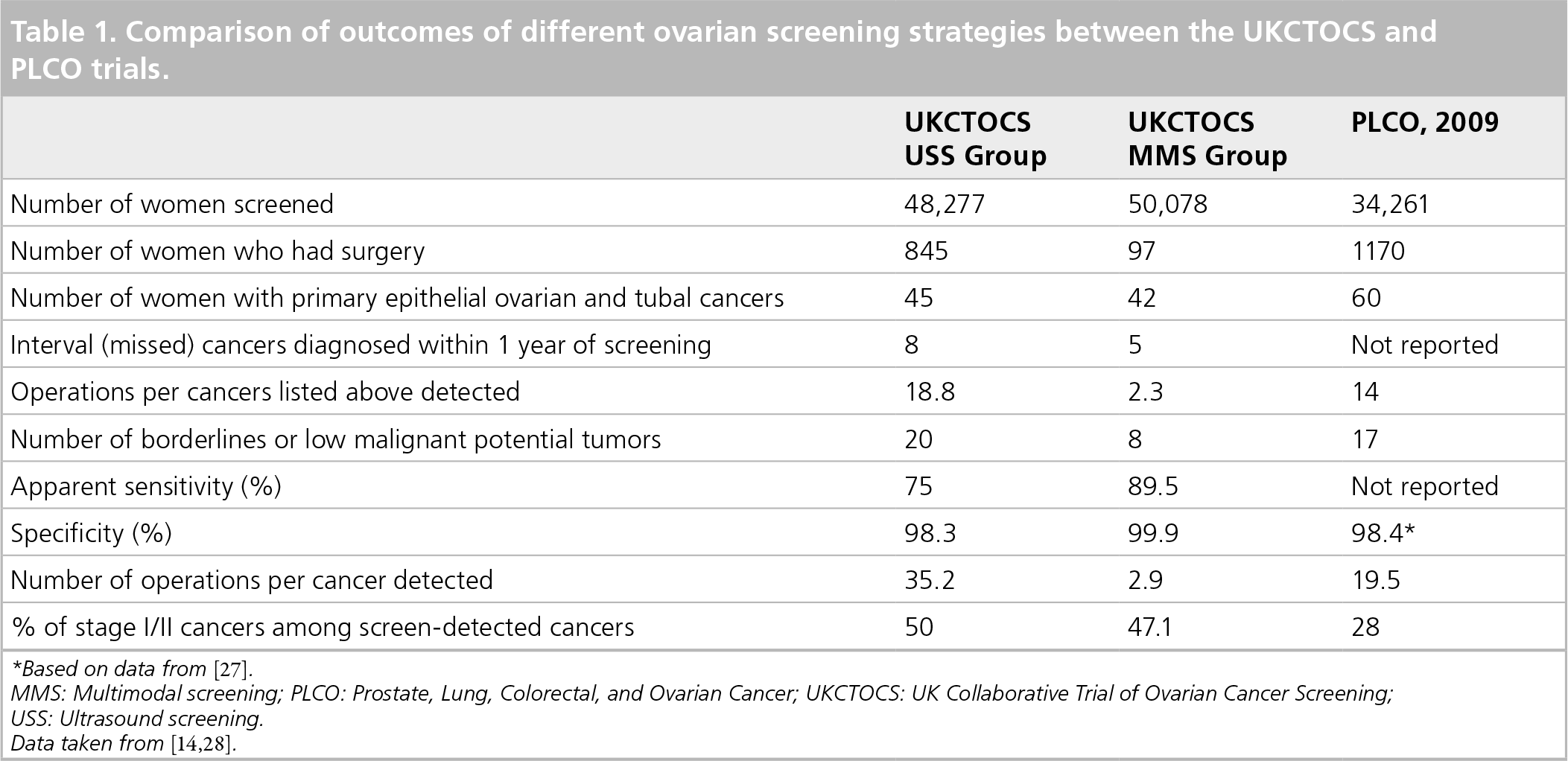
Based on data from [27].
MMS: Multimodal screening; PLCO: Prostate, Lung, Colorectal, and Ovarian Cancer; UKCTOCS: UK Collaborative Trial of Ovarian Cancer Screening
USS: Ultrasound screening.
Future perspective
Understanding the biology of ovarian cancer will greatly enhance our approach to ovarian cancer screening. Improvements in biomarkers, proteomics and other genomic methods may greatly aid in detecting early ovarian cancer.
Executive summary
Understand the etiopathogenesis of ovarian cancer.
Determine if stage I ovarian cancer is biologically related to advanced stage disease.
Develop novel biomarkers through approaches including proteomics, genomics and other technology.
Determine the role of sonography with biomarkers.
Develop new technology employing biologic characteristics of ovarian cancer.
Identify those at highest risk for breast/ovarian cancer to reduce side effects from premature menopause and surgery.
Use new biomarkers and radiographic tests in the general population to screen average risk women when available.
Footnotes
