Abstract
Background:
Abdominal myomectomy is a commonly performed surgical procedure for removing fibroids. Various techniques and interventions have been explored to minimize blood loss during abdominal myomectomy. This study aims to assess and compare the efficacy of perioperative vaginal misoprostol and intraoperative pericervical hemostatic tourniquet in minimizing blood loss during abdominal myomectomy.
Method:
Eligible studies were identified using several databases. Analysis was conducted using RevMan version 5.3. This systematic review and meta-analysis were registered in PROSPERO on June 20, 2023, with registration number CRD42023434191. We included three studies with 88 participants.
Results:
Of the 73 records identified initially, 3 met the inclusion criteria—sample sizes of the included studies involving 88 participants. There was a significant mean difference in intraoperative blood loss (p = 0.022 (95% CI, 0.0706–0.901) and operative time (p = 0.015, 95% CI, 0.116–1.066) between groups. There was no significant mean difference in postoperative hematocrit (p = 0.053 (95% CI, −0.734 to 0.00411)) and intraoperative blood transfusion (OR = 1.60 (95% CI, 0.587–4.405), p = 0.355)) between group.
Conclusion:
Our studies indicate that using tourniquets lowered intraoperative bleeding and shortened operating time among the participants. Intraoperative vaginal misoprostol has no significant advantage over the tourniquet group in either intraoperative blood transfusion needs or postoperative hematocrit.
Introduction
Uterine leiomyomas represent the prevailing pelvic tumor pathology observed in the female reproductive age population. 1 The prevalence and frequency of leiomyomas increase gradually as women age during their reproductive years. Notably, a greater incidence of leiomyomas is observed among African women. Studies indicate that approximately 70% of white women and more than 80% of women of African ancestry receive a diagnosis of leiomyomas at some point in their lifetime.2,3
Patients experiencing symptomatic fibroids commonly express various symptoms and exhibit concerns regarding the impact of fibroids on their quality of life. 4 Hysterectomy is recommended as the decisive surgical choice for a patient experiencing abnormal uterine bleeding and bulk symptoms linked to uterine leiomyoma, who either do not plan on future childbearing or prefer not to keep their uterus. 5 Conversely, myomectomy is preferable for women who desire future fertility or preservation of the uterus. 6
Abdominal myomectomy is a commonly performed surgical procedure for removing uterine fibroids.7,8 Lap-aroscopic myomectomy is the gold standard management in carefully selected women but abdominal myomectomy is still required for women with large and multiple fibroids. 9 One of the major concerns during the former procedure is blood loss, which can lead to anemia, the need for blood transfusions, and other postoperative complications.6,10,11 Factors contributing to a higher risk of excessive blood loss during myomectomy include the presence of large fibroids, multiple fibroids, and fibroids located in the lower pelvis. 12
Various techniques and interventions have been explored to minimize blood loss during abdominal myomectomy, including pharmacological agents and mechanical devices. Vaginal misoprostol is a prostaglandin analog used in gynecological procedures to induce cervical ripening and uterine contractions, which may help reduce bleeding. 13 Additionally, during myomectomy procedures, a mechanical device called the pericervical hemostatic tourniquet can be placed around the cervix to occlude the uterine blood supply and reduce blood loss.14,15 This study aims to assess and compare the efficacy of perioperative vaginal misoprostol and intraoperative pericervical hemostatic tourniquet in minimizing blood loss during abdominal myomectomy.
Methods
Information sources
We conducted an extensive electronic search across various databases, including PubMed, EBSCO, and ProQuest, up to June 12, 2023. Our search method incorporated MeSH terms, keywords, and different variations of terms associated with “tourniquet,” “myomectomy,” and “misoprostol.” Additionally, we reviewed the reference lists of pertinent articles and reviews to identify any supplementary studies.
Search strategy
To conduct a systematic review and meta-analysis of studies investigating the comparison between perioperative vaginal misoprostol and pericervical hemostatic tourniquet for reducing blood loss during abdominal myomectomy, we employed the following search terms: “tourniquet” or “hemostatic tourniquet” or “pericervical tourniquet”; “uterine myomectomy” or “myomectomy” or “abdominal myo-mectomy”; and “misoprostol.”
Eligibility criteria
The literature search was conducted with a focus on clinical studies published in English. To meet the inclusion criteria, studies were required to satisfy the following conditions: (1) the participants must have received a diagnosis of uterine leiomyoma, (2) the patients must have undergone abdominal myomectomy, and (3) the patients must have received 400 μg of misoprostol intravaginally at least 1 h before the procedure or a pericervical tourniquet during the procedure. Our review excluded single case reports/series, editorials, commentaries, letters, and nonfull texts. The exclusion criteria encompassed conditions such as pregnancy, adenomyosis, bleeding disorders, anemia, hypertension, chronic diseases, previous surgical history, preoperative hormonal therapy, and oral contraceptive pills. The primary outcomes of interest were the estimated amount of blood loss during the surgery, the need for blood transfusion, and the postoperative hematocrit levels. The secondary outcome of interest was the duration of the surgical procedure.
Data Extraction
Our study adhered to the reporting guidelines provided by the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement guidelines and the Cochrane Risk of Bias Tool (Cochrane ROB-2).16,17 All abstracts were assessed independently by two authors. Any discrepancies regarding the relevance of a particular study were resolved through discussion with a third author (CNRS). In cases where multiple studies had the same participant group and addressed identical outcomes, we selected the study with the most comprehensive information while excluding the others to prevent duplication. Two authors (NA and GM) independently evaluated the articles’ quality in this systematic review and meta-analysis.
Statistical analysis
Meta-analysis of proportions was used to combine all the data; logit transformations were conducted before meta-analysis, and the reported pooled proportions of depression were calculated using a random-effects model. All meta-analyses were performed using MedCalc, 18 version 19.5.1, and Review Manager (RevMan), 19 version 5.3. The p-value for the overall effect, p < 0.05 with two-tailed, was considered statistically significant. I2 was used to assess the heterogeneity of all the detailed studies. When it was lower than 50%, studies with acceptable heterogeneity were considered, and the fixed-effects model was used; otherwise, a random-effects model with the DerSimonian and Laird method was adopted.
Egger’s linear regression test assessed Publication bias for each pooled study group. Begg’s rank correlation was also applied to assess potential publication bias; when p was >0.05, there was no publication bias in the study. This systematic review and meta-analysis were registered in PROSPERO on June 20, 2023, with registration number CRD42023434191.
Results
Study selection and data extraction
The electronic database search initially yielded 73 studies. After screening titles and abstracts, seven articles were identified for further evaluation. Eventually, three studies were included in this meta-analysis. The search process and selection criteria used in this study are shown in Figure 1.
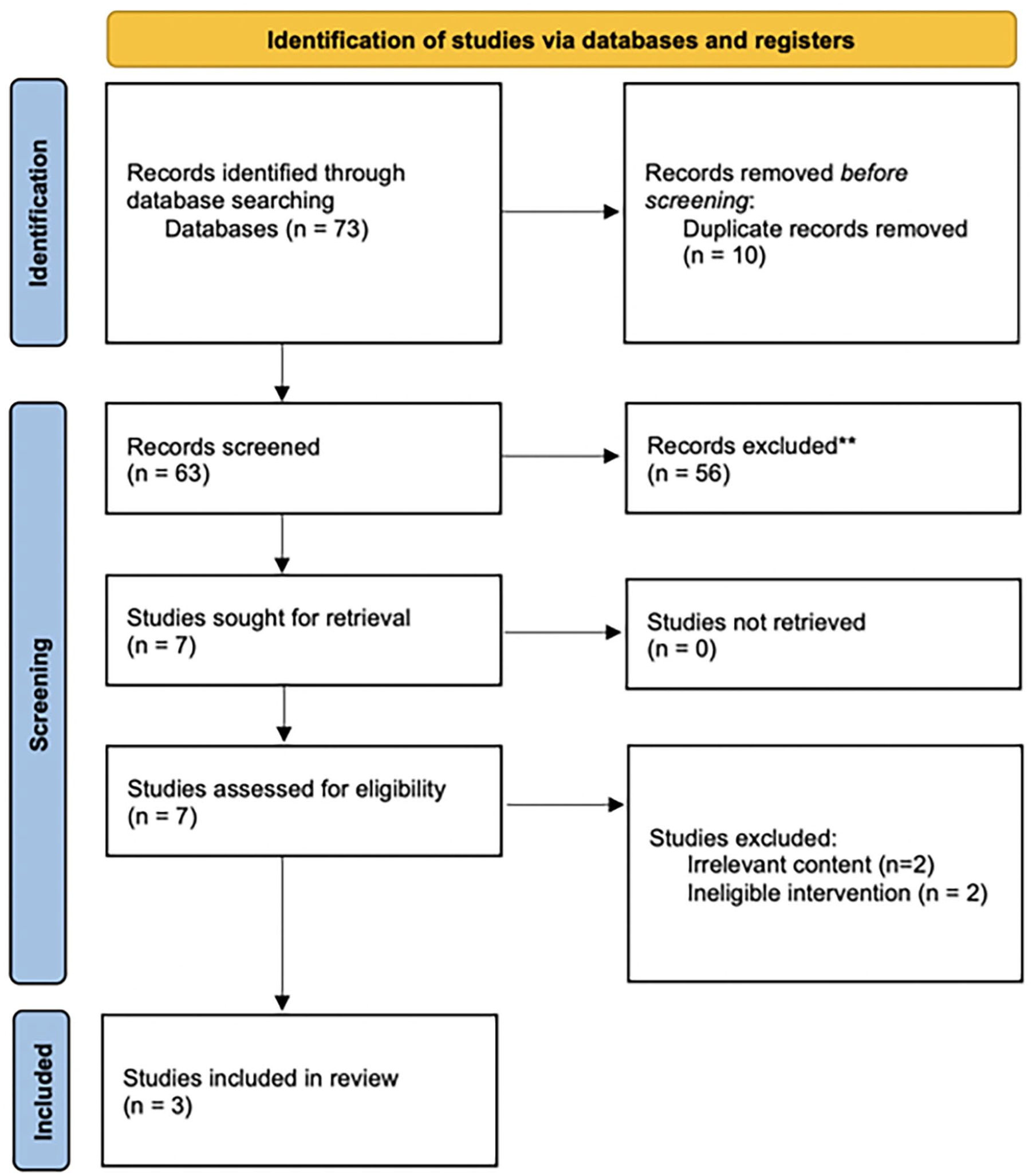
The preferred reporting items for systematic reviews and meta-analysis flow diagram. The diagram summarizes the search strategy and selection process to include articles eligible for this meta-analysis. 16
Study Characteristics
This systematic review and meta-analysis incorporated three primary studies (Table 1) conducted from 2019 to 2022, involving 88 participants. All of the included studies were randomized controlled trials. One study employed an infant feeding tube as the pericervical tourniquet, while the remaining used a Foley catheter. In all studies, 400 µg of misoprostol was administered intravaginally at least 1 h before the surgical procedure. The geographical scope of this review encompassed studies conducted in Africa. The average maternal age ranges from 30 to 37 years.
Study characteristic.
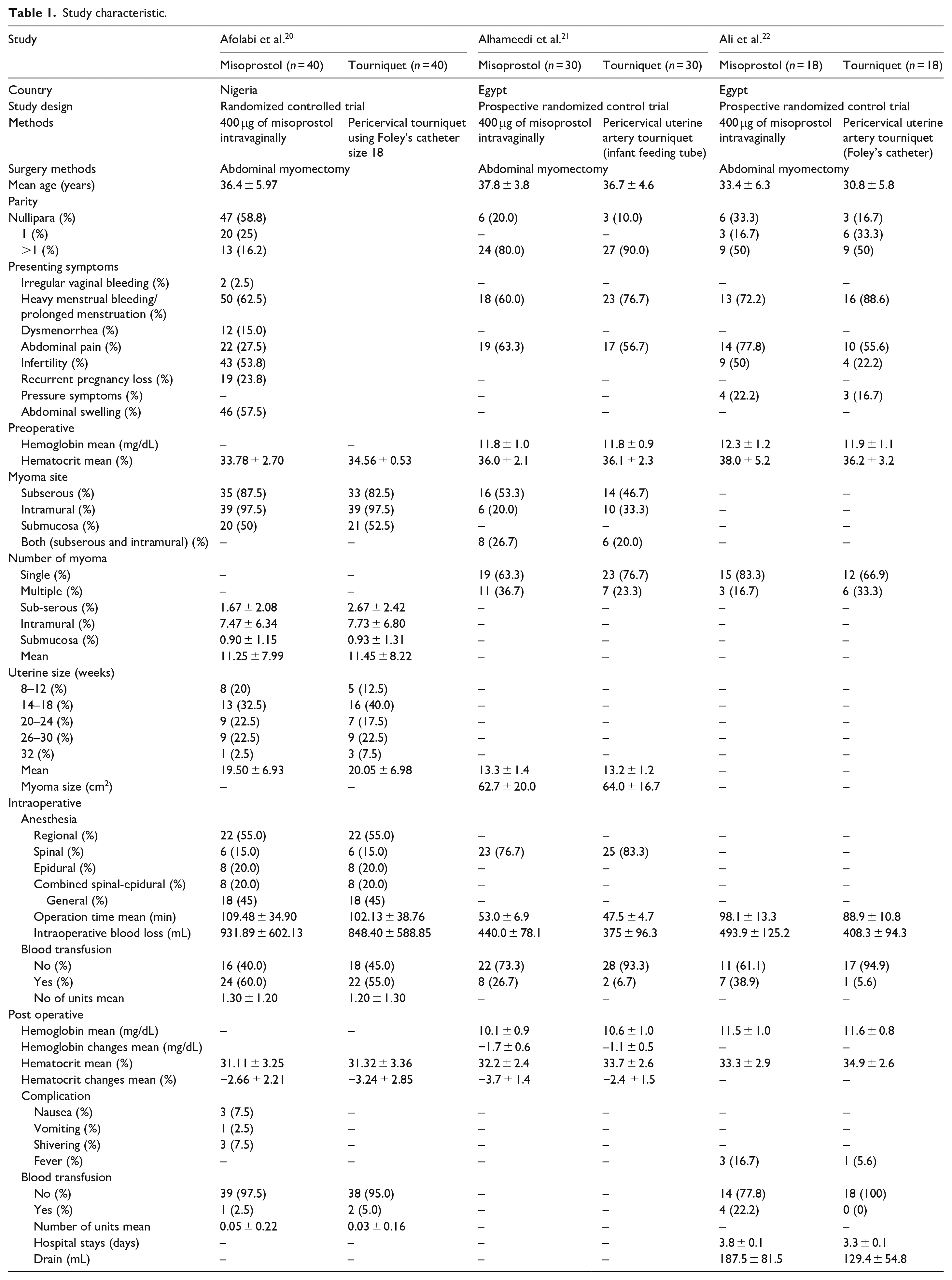
The most commonly observed symptoms across all included studies were predominantly characterized by excessive menstrual bleeding or prolonged menstruation accompanied by abdominal pain. Other reported symptoms included irregular vaginal bleeding, painful me-nstruation, difficulty conceiving or infertility, repeated pregnancy loss, pressure sensations, and abdominal sw-elling.
The Cochrane Risk of Bias Tool (Cochrane ROB-2) was utilized to assess the quality of the included studies. 17 All of the studies exhibited a low level of bias in relation to deviations from the intended intervention, missing outcome data, and outcome measurement. However, in one study, the randomization method was not specified, raising suspicions that participants and enrolling investigators were aware of the assigned intervention. Consequently, this study was deemed to have a high risk of bias. Additionally, another study raised concerns regarding the selection of the reported result, as it lacked a pre-specified analysis plan, making it impossible to compare the published report with the planned measurements. The evaluation of study quality is visually depicted in Figure 2.

Methodological quality. 17
Intraoperative blood loss
The pooled effect estimate showed a significant difference between misoprostol and tourniquet groups (p = 0.022 (95% CI, 0.0706–0.901). The absence of publication bias in this meta-analysis was confirmed by applying statistical tests, namely Begg’s test and Egger’s test of intercept. Additionally, no significant heterogeneity was observed between the included studies (Q = 3.6585, p = 0.1605, I2 = 45.33). The forest plot of the data is shown in Figure 3.
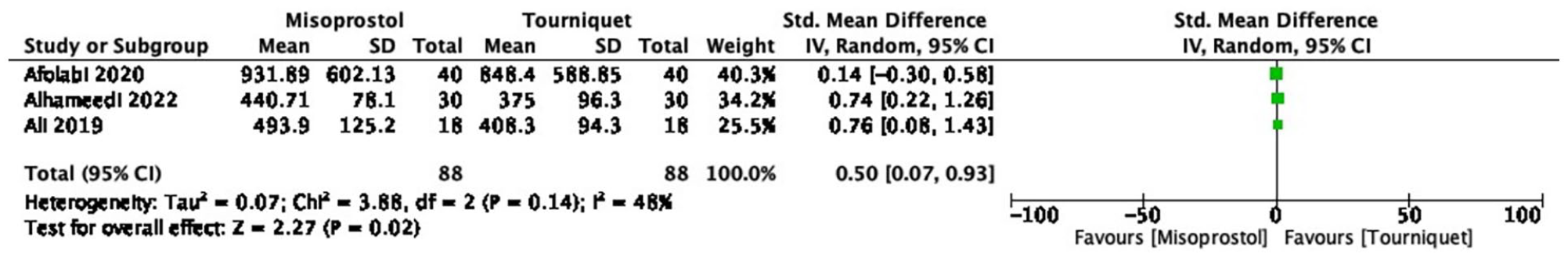
Show the result of perioperative vaginal misoprostol versus tourniquet mean difference in intraoperative blood loss.
Post-operative hematocrit
The pooled mean difference showed no statistically significant difference between the misoprostol group postoperative hematocrit and the tourniquet group (p = 0.053 (95% CI, −0.734 to 0.00411)). The pooled studies were heterogeneous (Q = 2.9679, p = 0.2267, I2 = 32.61). The forest plot of the data is shown in Figure 4.

Show the result of perioperative vaginal misoprostol versus tourniquet mean difference in post-operative hematocrit.
Intraoperative blood transfusion
The pooled risk ratio showed no statistically significant difference in intraoperative blood transfusion (OR = 1.60 (95% CI, 0.587–4.405), p = 0.355)). Pooled results were homogeneous (Q = 3.7206, p = 0.155, I2 = 46.25). The forest plot of the data is shown in Figure 5.

Show the result of perioperative vaginal misoprostol versus tourniquet mean difference in intraoperative blood transfusion.
Operative time
The pooled showed a statistically significant difference in operation time (p = 0.015, 95% CI, 0.116–1.066). The absence of publication bias in this meta-analysis was confirmed by applying statistical tests, namely Begg’s test and Egger’s test of intercept. Additionally, no significant heterogeneity was observed between the included studies (Q = 4.6940, p = 0.0957, I2 = 57.39). The forest plot of the data is shown in Figure 6.
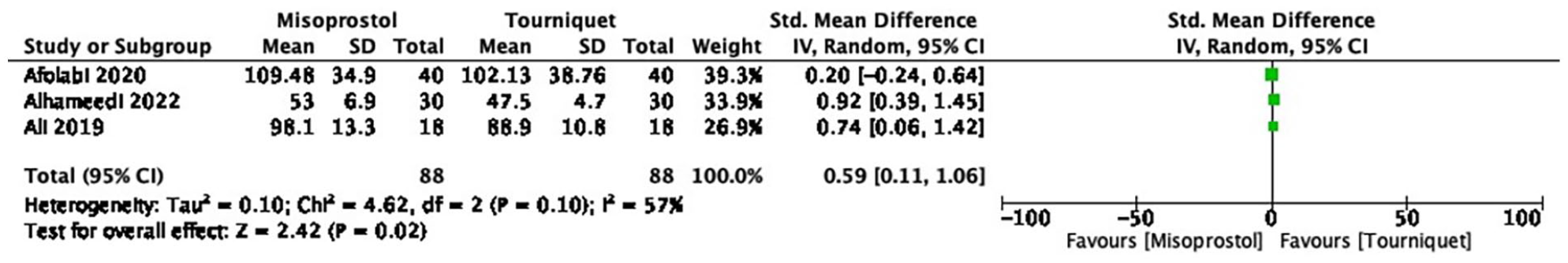
Show the result of perioperative vaginal misoprostol versus tourniquet mean difference in operative time.
Discussion
Our study’s outcomes indicate that the utilization of tourniquets lowered intraoperative bleeding among the participants. Intraoperative vaginal misoprostol has no significant advantage over the tourniquet group in either intraoperative blood transfusion needs or post-operative hematocrit. However, there are statistically significant differences in the mean values of intraoperative blood loss and operating time between the group that received misoprostol and the group that employed a pericervical tourniquet.
Abdominal myomectomy is an option for the management of fibroids in women who do not wish to have a hysterectomy particularly if they have large and multiple fibroids. However, this surgical procedure has undergone various modifications over the years to enhance its safety profile, as it is associated with significant intraoperative hemorrhage, posing a life-threatening situation and potentially requiring blood transfusion. 3 Using a pericervical tourniquet during an open abdominal myomectomy to minimize blood loss has become increasingly popular and is considered a standard practice.23,24 However, it is important to note that despite this practice, abdominal myomectomy is still associated with a substantial risk of intraoperative bleeding, leading to anemia, and the requirement for blood transfusion. 25
Various interventions have been investigated to minimize blood loss during abdominal myomectomy, including pharmacological agents and mechanical devices. Vaginal misoprostol is a prostaglandin analog used in gynecological procedures to induce cervical ripening and uterine contractions, which may help to subside bleeding. 11 Misoprostol has been reported to be effective in reducing blood loss in myomectomy compared to placebo. It stimulates consistent uterine contractions within 1–2 h after vaginal administration, which persist for up to 6 h. Hence, the utilization of misoprostol can effectively prevent postoperative blood loss resulting from uterine relaxation for a period of up to 6 h.26,27
In this study, no statistically significant difference was observed in the requirement for intraoperative blood transfusion, indicating comparable efficacy between the two approaches in decreasing blood loss. Nevertheless, it is worth stating that the misoprostol group revealed more cases needing a blood transfusion, which aligns with the findings reported in the included studies.20 –22 Contrarily, another study reported no instances of blood transfusion requirements. 28
The average postoperative hematocrit levels did not show statistical significance. These results demonstrate that both groups effectively reduced blood loss during the procedure. Most studies included analyzed the tourniquet group showed more minor changes in hematocrit levels.21,22 However, one of the included studies reported lower changes in hematocrit levels in the misoprostol group. 20 This finding aligns with another study that observed higher hemoglobin levels and a lower prevalence of anemia among patients in the tourniquet group. 29
Regarding intraoperative blood loss, the tourniquet group demonstrated significantly lower blood loss than the misoprostol group. This observation is in accordance with a study that demonstrated the effectiveness of employing a tourniquet with intermittent release to ligate the main arterial supply to the uterus, resulting in a noteworthy reduction in intraoperative blood loss.30,31 The decreased blood loss observed in the group using a tourniquet may account for the reduced necessity of intraoperative transfusions among patients.
There was a statistically significant disparity in the duration of the surgical procedure between the groups, with the tourniquet group displaying a notably shorter duration. This outcome corresponds with all the studies included in the analysis.20 –22 Additionally, one study reported a decreased operation duration in the tourniquet group compared to the control group, even after considering the time necessary for the placement and release of the tourniquet. 32
The participants in our study reported mild and self-limiting adverse effects associated with misoprostol, such as nausea, vomiting, diarrhea, shivering, and fever.20,22 These findings align with a previous report, indicating that the adverse effects were considered self-limiting and did not necessitate intervention. 28
Strengths and limitations of the study
The strength of this research lies in the novelty of it being the first systematic review and meta-analysis and in the rigorous methodological and statistical approaches that we have adhered to. Several limitations were identified in our review. Firstly, the small sample size and limited number of studies restricted our findings’ generalizability. Additionally, the studies included in our review were conducted in a limited study area, resulting in potential regional variations in results. Moreover, the heterogeneity in the number and size of myomas among the included studies introduced variability in the outcomes. Another limitation was the absence of a standardized cut-off for intraoperative transfusion, which may have impacted the interpretation of transfusion-related data.
Conclusion
The utilization of a pericervical tourniquet during abdominal myomectomy suggested a substantial reduction in intraoperative blood loss when contrasted with the use of perioperative vaginal misoprostol. Additionally, the tourniquet method was linked to a shorter duration of the surgical procedure. However, we advise the use of perioperative vaginal misoprostol as an alternative when the application of a tourniquet is challenged by notable pelvic adhesions or leiomyoma at the intended site. Moreover, the cost-effectiveness, ready availability, and wide-ranging benefits of misoprostol make it a valuable tool, offering sustained effects even post-surgery.
To further validate these findings, conducting a larger sample size requires multi-center randomized trials, and assessing the impact of the pericervical tourniquet and misoprostol in relation to uterine size and number of large fibroid nodules.
Supplemental Material
sj-pdf-1-pev-10.1177_22840265231212706 – Supplemental material for Perioperative vaginal misoprostol versus intraoperative pericervical hemostatic tourniquet outcome during abdominal myomectomy: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-pev-10.1177_22840265231212706 for Perioperative vaginal misoprostol versus intraoperative pericervical hemostatic tourniquet outcome during abdominal myomectomy: A systematic review and meta-analysis by Nicholas Adrianto, Ghea Mangkuliguna and Candra Novi Ricardo Sibarani in Journal of Endometriosis and Pelvic Pain Disorders
Footnotes
Author contributions
NA, GM, and CRS: developed the protocol, and contributed to the study design, literature review, and selection of studies, quality assessment, data extraction, statistical analysis, interpretation of the data, and read and approved the final manuscript.
Availability of data and material
All datasets have been presented within the manuscript. The datasets supporting the conclusions of this article is available from the corresponding authors on request via (
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
