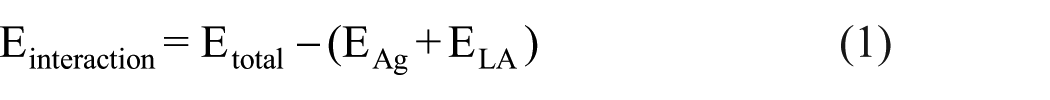Abstract
Silver nanoparticles (AgNPs) are increasingly explored as alternatives to conventional antibiotics in wound care due to their broad-spectrum antimicrobial activity. This study evaluates the antibacterial performance of α-lipoic acid-functionalized silver nanoparticles (AgNPs-LA) using an integrated experimental-computational approach combining molecular dynamics simulations and experimental validation. AgNPs-LA were synthesized via a controlled hydrothermal method employing biogenic reduction of silver ions to minimize cytotoxicity. Structural and compositional analyses confirmed successful surface functionalization with α-lipoic acid and D-glucose. Morphological characterization revealed predominantly spherical nanoparticles with diameters ranging from 37 to 90 nm. Antimicrobial agar disk diffusion assays demonstrated concentration-dependent inhibition of Staphylococcus aureus, Pseudomonas aeruginosa, and Escherichia coli. Specifically, AgNPs-LA at 40 μg/mL produced inhibition zones statistically comparable to gentamicin against S. aureus and P. aeruginosa (p > 0.05). Molecular dynamics simulations performed under isothermal-isochoric (constant number of particles, volume, and temperature; NVT) canonical ensemble indicated that the disulfide moiety of lipoic acid serves as the primary anchoring group to the silver surface, conferring stability through the formation of a structured hydrophobic surface layer. This interfacial configuration enhances nanoparticle stability and may improve pharmacokinetic clearance, thereby reducing the risk of systemic toxicity. Collectively, these findings highlight the potential of AgNPs-LA as a safe and effective antimicrobial platform for wound management applications.
Introduction
The skin, as the body’s largest organ, serves as the primary barrier against physical injury and various forms of trauma, including scratches and burns. Significant disruption of epithelial and connective tissue integrity resulting from such injuries compromises this protective barrier, markedly reducing the body’s ability to defend against external environmental factors.1,2 While normal wound healing typically progresses through well-defined phases without complications, numerous factors, both extrinsic and intrinsic, can disrupt this orderly process. These include necrotic tissue, retained foreign bodies, microbial contamination, and underlying comorbidities, all of which can substantially impede tissue repair and regeneration.1,3–6
Microbial colonization represents the predominant challenge in wound closure and management.7–10 The early stages of wound healing are commonly affected by Staphylococcus aureus (S. aureus) and methicillin-resistant S. aureus (MRSA), whereas deeper tissue layers in chronic wounds frequently harbor Escherichia coli (E. coli) and Pseudomonas aeruginosa (P. aeruginosa).11,12 Furthermore, synergistic interactions among bacterial species often result in polymicrobial biofilm formation, contributing to the chronicity and therapeutic resistance of wound infections.13–16 The need for targeted and effective interventions has driven the evolution of wound dressings beyond passive barrier functions to active therapeutic delivery systems. Contemporary advanced wound dressings incorporate broad and narrow-spectrum antibiotics and various antimicrobial agents to address the complexity of polymicrobial wound environments. 17 However, the escalating prevalence of antimicrobial resistance poses significant challenges to both topical and systemic antibiotic therapies. Consequently, there has been renewed interest in novel non-antibiotic antimicrobial approaches.
Metallic nanoparticles such as silver and copper nanoparticles, have emerged as promising candidates in regenerative medicine and infection control due to their unique physicochemical properties, including nanoscale dimensions and high surface area-to-volume ratios.18–21 Particularly, silver nanoparticles (AgNPs) account for approximately 55.4% of commercially available nanotechnology-based products, with their antimicrobial efficacy attributed to broad-spectrum activity, oligodynamic properties, and lower propensity for inducing microbial resistance mechanisms.22–26 Conventional AgNP synthesis methods, although effective, are often limited by high energy consumption, operational expenditures, and the use of potentially hazardous chemical reagents. 27 The physicochemical characteristics of AgNPs, specifically their particle size, morphology, and density, facilitate cellular penetration and site-specific accumulation. 28 Their surface chemistry enables free radical generation, potentially inducing oxidative stress. 29
Dermatological studies have reported correlations between cutaneous AgNPs exposure and tissue accumulation, with temporal and dose dependent effects characterized by epidermal and papillary layer thinning, inflammatory marker elevation, and Langerhans cell proliferation. 28 Moreover, cytotoxicity evaluations of commercially available nanocrystalline silver-containing wound dressings (e.g., Acticoat, Absorbent, and PolyMem Ag) have demonstrated detrimental effects on diabetic fibroblast viability, collagen synthesis, and re-epithelialization processes.29,30
Addressing these limitations is essential to fully realize the therapeutic potential of AgNPs in wound dressing applications. The wound-healing performance of AgNPs-based systems depends on their physicochemical properties, optimized synthesis strategies, mitigation of toxicity and stability concerns, and careful evaluation of therapeutic efficacy. Among the emerging approaches, α-lipoic acid-functionalized silver nanoparticles (AgNPs-LA) have shown notable promise in wound management. These functionalized nanoparticles exhibit robust antibacterial efficacy against various pathogens while showing reduced cytotoxicity and hemolytic effects compared to uncoated AgNPs, thereby achieving a critical balance between therapeutic potency and biocompatibility essential for clinical applications.31,32 Nonetheless, several challenges impede their clinical implementation, including the potential for bacterial resistance (albeit less frequent than with conventional antibiotics), colloidal instability due to agglomeration, and the complex relationship between surface charge and antimicrobial performance, which requires precise control during synthesis.32–34
Despite these promising attributes, a comprehensive assessment of AgNPs-LA must also address concerns related to their long-term effects on human health and the environment. The potential release of nanoparticles from medical devices represent an emerging risk that warrants thorough evaluation using standardized protocols.31,33–35 Future research should focus on clarifying the molecular mechanisms associated with bacterial adaption or resistance to AgNPs, developing advanced functionalization strategies to enhance nanoparticle stability without compromising antimicrobial activity, standardizing cytotoxicity assessments across relevant cell types, and implementing sustainable production and disposal practices. Integrated experimental and computational investigations will be critical for translating these functionalized nanomaterials into clinically viable wound care technologies that maximizes therapeutic benefits while minimizing potential risks.
In this study, we investigated the antibacterial properties of α-lipoic acid-functionalized silver nanoparticles synthesized (AgNPs-LA) via a green hydrothermal route using D-glucose as a reducing agent. An integrated experimental-computational approach was employed, combining physicochemical characterization, qualitative antibacterial evaluation against representative Gram-positive and Gram-negative bacteria, and molecular dynamics simulations to examine nanoparticle-ligand and nanoparticle–biomolecule interactions. While the present work emphasizes structure-function relationships and an initial evaluation of antibacterial activity, the study highlights mechanistic understanding and initial antibacterial behavior, thereby providing a foundation for future quantitative validation, expanded biological assessment, and translational development.
Materials and methods
Materials and equipment
All reagents were of analytic grade and used without further purification. Silver nitrate (AgNO3) was used as the precursor salt, D-glucose (dextrose anhydrous) served as the green reducing agent, and α-lipoic acid (≥99%, titration, Sigma-Aldrich) was employed as the stabilizing agent. Solutions were freshly prepared using distilled water.
The bacterial strains Staphylococcus aureus UPCC 1143, Pseudomonas aeruginosa UPCC 1244, and Escherichia coli UPCC 1195, were obtained from the culture collection of the University of the Philippines-Natural Sciences Research Institute (NSRI), Diliman, Quezon City, Philippines.
Green synthesis of α-lipoic acid functionalized AgNPs
The synthesis of α-lipoic acid capped silver nanoparticles (AgNPs-LA) was carried out using a hydrothermal green chemistry approach, as shown in Figure 1. Initially, 0.083 g of α-lipoic acid was dissolved in 50 mL of distilled water. Subsequently, 10 mL of 0.4 M AgNO3 was added, followed by 40 mL of 0.1 M D-glucose. The resulting mixture was transferred to a Teflon-lined stainless-steel autoclave, sealed, and heated at 160°C for 1 h. After completion of the reaction, the autoclave was allowed to cool naturally to room temperature. The synthesized AgNPs-LA were purified by repeated centrifugation at 4500 rpm for 70 min and washed with distilled water to remove residual silver ions and unreacted species. The purified nanoparticles were finally redispersed in distilled water for subsequent characterization and analyses.

Schematic diagram of the green hydrothermal synthesis of AgNPs-LA.
Experimental design
This study investigated two primary variables: (1) the effect of reaction time (1, 2, and 3 h) on the physicochemical properties of green-synthesized AgNPs-LA and (2) the antibacterial efficacy of AgNPs-LA synthesized at 160°C for 1 h at three concentrations (20 µg/mL, 30 µg/mL, and 40 µg/mL) against Staphylococcus aureus, Pseudomonas aeruginosa, and Escherichia coli. Antibacterial activity was evaluated by measuring zones of inhibition (mm) with gentamicin and ethanol serving as positive and negative controls, respectively. All experiments were performed in triplicate, and statistical significance was evaluated at p < 0.05.
Characterization techniques
Fourier transform infrared (FTIR) spectroscopy
Samples were finely ground with potassium bromide (KBr) using a mortar and pestle and then compressed into a clear disc via manual press. FTIR spectra were recorded using a Nicolet iS50 FTIR Spectrometer over the range of 400 to 4000 cm−1.
Field-emission scanning electron microscopy with energy dispersive X-ray spectroscopy (FESEM-EDX)
Nanoparticle samples were deposited onto carbon tape, and loosely bound particles were removed by gentle aspiration. Imaging and elemental analysis were performed at two distinct spots using a Hitachi SU8230 Field-Emission Scanning Electron Microscope equipped with a Horiba xMax-N 50-mm window X-ray detector.
Ultraviolet-visible (UV-Vis) spectroscopy
The formation of AgNPs-LA was confirmed by optical absorption measurements using a Hitachi UH5300 UV-Vis spectrophotometer. Samples were dispersed in distilled water, sonicated, and transferred to 1.0 mL quartz cuvettes (1 cm optical path length). Absorption spectra were recorded at room temperature from 200 to 600 nm with a spectral resolution of 2 nm, using distilled water as the blank reference.
Antibacterial assay
The agar disk diffusion method was used as a screening-level comparative tool to evaluate the antimicrobial activity of the synthesized AgNPs-LA relative to a clinically established antibiotic (gentamicin) under standardized conditions against one Gram-positive bacterial strain (S. aureus) and two Gram-negative bacterial strains (P. aeruginosa and E. coli). Fresh 18-24-h bacterial cultures were used to prepare microbial suspensions in 0.1% peptone water. The microbial suspensions were inoculated into the pre-poured Mueller Hinton Agar (MHA), about 3 mm thick, by uniformly swabbing the agar surface. Sterile cotton swabs were immersed in the microbial suspension, rotated several times, and pressed firmly against the inner wall of the tube above the fluid level to remove excess inoculum before streaking the agar surface. The plates were rotated by 60° and the swabbing procedure was repeated twice to ensure even bacterial distribution. Using a 10 mm diameter cork borer, three equidistant wells were created on each agar plate, corresponding to individual trials. Each well was filled with 200 µL of AgNPs-LA solution at concentrations of 20, 30, or 40 µg/mL, while absolute ethanol served as the negative control. Gentamicin (10 µg) was used as the positive control.
Each concentration was tested in triplicate. The MHA plates were incubated at 35°C for 24 h, after which the diameters of the inhibition zones were subsequently measured in millimeters.
Statistical analysis
One-Way Analysis of Variance (ANOVA) was used to determine the statistically significant differences (p < 0.05) among the zones of inhibition obtained for different concentrations of AgNPs-LA. When significant differences were observed, the Games-Howell post hoc test was applied. All statistical analyses were performed SPSS version 20.
Computational modeling of AgNPs-LA
Molecular dynamics (MD) simulations were conducted using Materials Studio Software (Accelrys Inc.) to investigate the interaction between silver nanoparticles (AgNPs) with α-lipoic acid. 36 All simulations were performed using fine quality setting. Silver (unit cell lattice parameters of u = v = 2.89Å and ϴ = 90°) was constructed, and the (2 0 0) crystallographic plane was cleaved to serve as the interaction surface for contact with the α-lipoic acid (LA). Only the topmost atomic layers of silver were allowed to move during the calculations, while the coordinates of other silver atoms were constrained.
Force field calculations were performed using the Forcite module, employing the condensed-phase optimized molecular potentials for atomistic simulation studies (COMPASS) force field. Simulations were conducted under the isothermal-isochoric (NVT) canonical ensemble at 298.15 K and 1 atm, with a time step of 1 fs and total simulation durations ranging from 0 to 200 ps. The interaction energy (Einteraction) between the α-lipoic acid and the silver surface was calculated according to equation (1). wherein Etotal represents the total energy of Ag and LA in contact at equilibrium (AgNPs-LA), and EAg and ELA correspond to the total energies of isolated Ag and LA, respectively, separated in vacuum at equilibrium.
Results
Infrared spectroscopic analysis
The hydrothermal synthesis of silver nanoparticles (AgNPS) functionalized with α-lipoic acid (AgNPs-LA) represents a simple approach to nanoparticle preparation, initiated by introducing the D-glucose to the α-lipoic acid (LA)/AgNO₃ solution. The α-lipoic acid serves as the primary anchoring ligand, while the D-glucose functions primarily as a reducing agent during nanoparticle formation and as a secondary stabilizing component. The reaction progression was visually indicated by a gradual color change from pale yellow to black, reflecting the complex molecular interactions associated with nanoparticle nucleation and growth.
Figure 2 shows the comparative Fourier transform infrared (FTIR) spectra of AgNPs-LA, pristine α-lipoic acid (LA), and D-glucose, providing insights into the molecular composition and surface chemistry of the synthesized nanoparticles. A broad absorption band at 3340 cm−1 revealed O–H stretching vibrations from hydrogen-bonded hydroxyl groups, predominantly originating from D-glucose incorporation.37–39 The pronounced peak at 1393 cm−1, observed to both AgNPs-LA and D-glucose spectra, was attributed to O–H bending vibrations, further substantiating the critical role of D-glucose in surface functionalization. 40

FTIR spectra of synthesized AgNPs-LA, lactic acid (LA), and D-glucose.
Additional spectral features include aliphatic C–H stretching vibration at 2921 cm−1,41 observed in both LA and D-glucose, and a characteristic C=O stretching peak at 1694 cm−1,42,43 attributed to carboxylic acid groups, as evident in the LA spectrum. Notably, the S–S stretching vibration at 732 cm−1, originating from the disulfide linkage in α-lipoic acid, was significantly attenuated in the AgNPs-LA spectrum relative to pristine LA. 44
In general, α-lipoic acid forms strong coordination interactions via its disulfide/thiolate moieties and the silver surface, thereby ensuring persistent surface attachment. In contrast, D-glucose contributes hydrophilic hydroxyl groups that promote steric stabilization and enhance dispersion in aqueous environments. This complementary ligand functionality enables effective surface anchoring by α-lipoic acid while simultaneously improving colloidal stability through glucose-mediated hydration and steric effects.
Surface morphology
Figure 3 shows the globular morphology of AgNPs-LA, with size distributions varying as a function of synthesis duration. Nanoparticles synthesized after 1 h exhibited globular shapes with diameters ranging from 37 to 90 nm, whereas extending the synthesis to 2 h resulted in larger particles with sizes between 85 and 236 nm. After 3 h, the particle sizes ranged from 66 to 220 nm. This time-dependent increase in particle size is consistent with the observations of Darroudi et al. 45 for green-synthesized silver nanoparticles. The formation of larger particles may be attributed to the agglomeration of smaller nanoparticles during the drying process. 46

SEM micrograph of AgNPs-LA synthesized at: (a) 1 h, (b) 2 h, (c) 3 h, and (d) the elemental mapping of sample (a).
Energy-dispersive x-ray spectroscopy (EDX) spectra revealed prominent silver atom signals at 3 keV, characteristic of silver nanocrystallites, confirming the presence of AgNPs. Elemental mapping further detected sulfur, likely attributed to α-lipoic acid, while carbon and oxygen signals were associated with both LA and D-glucose.
UV-Vis spectroscopy
The surface plasmon resonance (SPR) properties of the synthesized AgNPs-LA were investigated using UV-vis spectroscopy to confirm formation and monitor particle size evolution as a function of aging time. As shown in Figure 4, all samples exhibited a characteristic SPR absorption band in the range of approximately 380 to 450 nm, consistent with spherical silver nanoparticles and previous reports.47,48 The presence of a single dominant SPR band further supports a predominantly spherical morphology, as anisotropic AgNPs, such as nanorods, typically exhibit two or more distinct resonance bands. 49

UV-Vis spectra of AgNPs-LA prepared at: (a) different aging time and optical band gap based on Tauc plot for the samples synthesized at: (b) 1 h, (c) 2 h, and (d) 3 h.
The SPR peak wavelength exhibits a size-dependent shift, with smaller AgNPs showing plasmon bands at shorter wavelengths, while increasing particle size leads to a red shift of the SPR peak.50–52 In this study, the sample synthesized for 1 h displayed a relatively blue-shifted and broadened SPR peak, indicative of smaller particle sizes with a wider size distribution and incomplete stabilization. The appearance of a shoulder at longer wavelengths suggests a fraction of larger AgNPs, consistent with findings from previous spectroscopic studies. 53 As synthesis duration increased to 3 h, a red-shift in the SPR peak was observed, confirming particle growth and an increase in average particle size. Optical band gap values estimated from Tauc plots (Figure 4(b)–(d)) decreased with increasing reaction time, reflecting reduced quantum confinement effects in larger particles.
Antibacterial activity
The disk diffusion method was employed as an initial qualitative screening tool to assess the antibacterial activity of AgNPs-LA relative to a clinically established antibiotic. This qualitative, concentration-specific approach provided comparative insights only, rather than definitive bacteriostatic threshold, and was not indicative of equivalent minimum inhibitory concentration (MIC) potency. Gentamicin and absolute ethanol served as positive and negative controls, respectively. This screening-based methodology was designed to evaluate the relative antibacterial performance of the nanoparticle system rather than establish definitive bactericidal or bacteriostatic thresholds.
Figure 5 illustrates the mean inhibition zones for AgNPs-LA at concentrations of 20, 30, and 40 µg/mL, gentamicin, and absolute ethanol against Staphylococcus aureus, Pseudomonas aeruginosa, and Escherichia coli. The selected concentrations were based on prior studies indicating that 40 µg/mL remains non-toxic to human gingival fibroblasts. 54 The observed inhibition trends, including comparatively smaller zones for E. coli, are consistent with previous reports.55–59 Figure 6 presents the images of the inhibition zones from all tested sample plates. Non-functionalized AgNPs and free α-lipoic acid were not included as separate control groups, which we acknowledge as a limitation of the present study. The primary objective was to evaluate the antibacterial performance of the integrated AgNPs-lipoic acid functionalized system as a unified platform, rather than to dissect the individual antimicrobial contributions of each component.
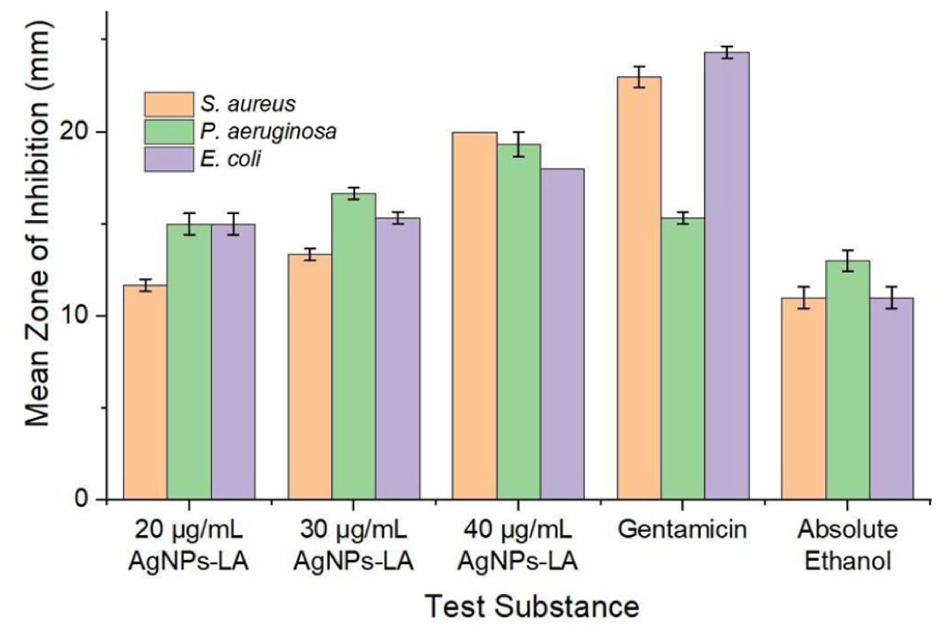
Mean zones of inhibition of AgNPs-LA and reference substances against different bacterial strain.

Images showing zones of inhibition for: (a) 20 μg/mL, (b) 30 μg/mL, and (c) 40 μg/mL of AgNPs-LA, and (d) absolute ethanol (left), and gentamicin (right), tested against three different bacterial strains.
Statistical analysis using one-way ANOVA revealed significant differences among inhibition zones (p < 0.05), with post hoc Games-Howell testing indicating enhanced antibacterial efficacy at higher AgNPs-LA concentrations. At 40 µg/mL, AgNPs-LA exhibited antibacterial activity comparable to gentamicin against S. aureus and P. aeruginosa. However, at 20 µg/mL, antibacterial activity of AgNPs-LA was not significantly different from that of absolute ethanol, indicating limited efficacy at lower concentration. The optimal concentration for significant antibacterial activity was identified as 30 µg/mL. The smaller inhibition zones observed for E. coli relative to S. aureus and P. aeruginosa may result from electrostatic repulsion between the negatively charged AgNPs-LA and the negatively charged E. coli cell membrane. 60
While this qualitative approach is appropriate for preliminary comparison, it is acknowledged that quantitative assays such as minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) determination, broth microdilution assays, and optical density-based bacterial growth kinetics would provide higher resolution and deeper mechanistic insight into antibacterial efficacy. The absence of these analyses represents a limitation of the present study and is explicitly identified as an important direction for future investigations. Furthermore, non-functionalized AgNPs and free α-lipoic acid were not included as separate control groups. This was a deliberate design choice, as the present work focused on evaluating the antibacterial performance of the integrated AgNP-LA system as a functional nanomaterial platform intended for wound-dressing applications, where synergistic surface functionality and overall biological performance are of primary relevance. Nevertheless, future studies will incorporate comparative antibacterial assessments of non-functionalized AgNPs and free α-lipoic acid to more clearly delineate their individual and synergistic contributions to antimicrobial activity.
Molecular dynamics study
Molecular dynamics (MD) simulations were performed using the Forcite module in Materials Studio to investigate the interaction energetics between α-lipoic acid (LA) and the AgNP surface. In this classical framework, atoms are treated as classical particles governed by Newton’s equations of motion, and their interactions are described using parameterized interatomic potentials. Electronic effects are incorporated implicitly through the force field; however, electrons are not explicitly modeled. The system evolves through iterative calculation of atomic positions and velocities, allowing relaxation toward energetically favorable configurations.
The simulations primarily describe adsorption stability, molecular orientation, and interfacial energetic trends rather than quantum-level chemisorption or charge transfer phenomena. Thus, the MD results provide insight into structural adsorption behavior and surface stabilization mechanisms at the Ag-LA interface.
Figures 7(a) to (f) illustrate the behavior of LA on the AgNP surface at various time points (i.e., 0, 5, 10, 15, 100, and 200 ps) where images of time progression show relaxation of the LA onto the Ag surface indicating effective anchoring and adsorption. It is observed that the S–S group of LA serves as the anchoring group for adsorption on the Ag surface while its terminal carboxyl functional group is freely exposed for subsequent interactions. Figure 7(g) shows the interaction energy between AgNPs-LA becomes increasingly negative over time confirming the enhanced interfacial stabilization of LA adsorption onto the Ag surface with time. 61 The Einteraction vs. time plot also suggests that the interfacial stabilization is not first order adsorption since MD calculations neglect the effect of electron sharing and/or exchange, especially when chemisorption is likely involved.

Molecular dynamics simulation of the AgNPs-LA formation at: (a) 0 ps, (b) 5 ps, (c) 10 ps, (d) 15 ps, (e) 100 ps, (f) 200 ps, and the (g) interaction energy versus time of AgNPs-LA from 5 to 200 ps.
Figure 8 presents the molecular dynamics results of LA adsorption onto AgNPs in the presence of water molecules at different time points. The S–S group remains closely bound to the AgNP surface, acting as the primary anchoring site. LA forms a structural layer around the AgNPs, shielding them from water molecules and preventing self-aggregation in the aqueous environment.

Molecular dynamics simulation of AgNPs-LA formation in the presence of water molecules at: (a) 0 ps, (b) 5 ps, (c) 10 ps, (d) 15 ps, (e) 100 ps, and (f) 200 ps.
Additional simulations examined interactions between bare AgNPs and LA-functionalized AgNPs with representative biomolecules of the skin lipid layer, including stearic acid, palmitic acid, and glucose (Figure 9). For bare AgNPs (Figure 9(a)), decreasing intermolecular distances over time indicate attractive interactions, with stearic acid exhibiting the strongest affinity. In contrast, functionalization with LA alters these interactions (Figure 9(b)), generally increasing the separation distance between the nanoparticle surface and the biomolecules. This behavior can be attributed to the hydrophobic backbone of LA, which modulates interactions with lipid molecules, while the exposed hydrophilic carboxyl groups partially mitigate repulsion effects, particularly for more polar biomolecules such as glucose.
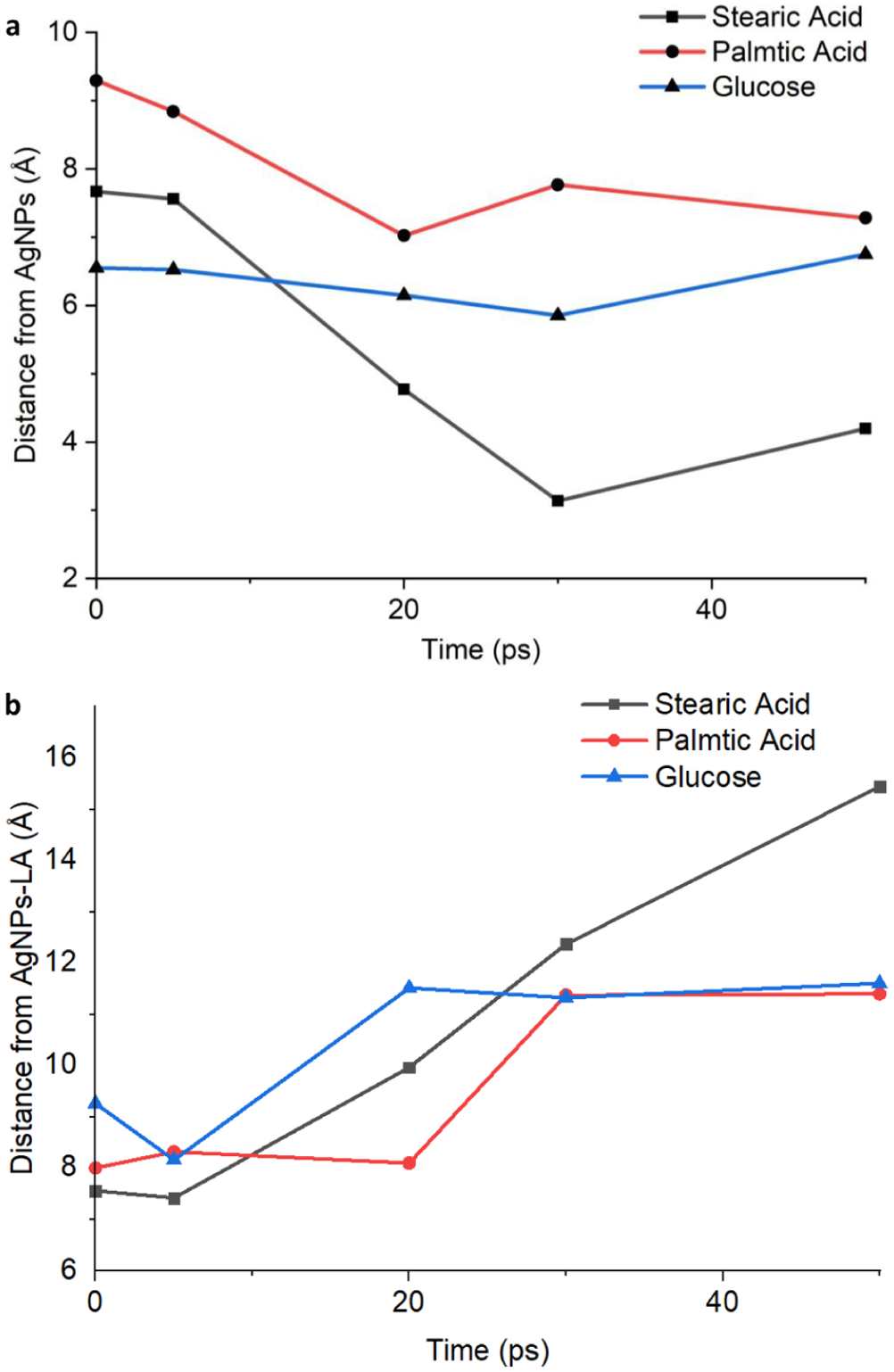
Distances between: (a) AgNPs and (b) AgNPs-LA with the representative biomolecules of the lipid layer of the skin from 0 to 50 ps.
The primary objective of the computational analysis was to investigate the adsorption behavior and structural stabilization of the AgNP-LA interface. The molecular dynamics (MD) simulations employed in this study primarily capture physisorption processes and interfacial stabilization trends rather than electronic-level chemisorption mechanisms. Nevertheless, complementary experimental evidence and established silver–thiol chemistry suggest that chemisorption involving Ag–S coordination is likely present in the AgNPs-LA system. In particular, the attenuation of the S–S stretching band observed in the FTIR spectra indicates possible disulfide bond cleavage and formation of thiolate species capable of interacting strongly with the silver surface. This interpretation is consistent with well-documented silver–thiol interactions reported in the literature and with the stable adsorption orientation observed for lipoic acid molecules in the MD simulations.
Capturing the electronic structure associated with Ag–S bond formation and charge transfer would require computational approaches that explicitly account for electron density, such as density functional theory (DFT) or reactive force field simulations (e.g., ReaxFF). These methods provide a more detailed description of chemisorption phenomena but are considerably more computationally intensive and were therefore beyond the scope of the present work. Consequently, the MD simulations presented here should be interpreted as providing structural and thermodynamic insight into ligand adsorption and interfacial stabilization rather than a complete electronic description of chemisorption. Future computational studies incorporating electronic-structure-based methods will be valuable for further elucidating the detailed bonding characteristics of the Ag–S interaction in the AgNPs-LA system.
Discussion
Surface chemistry and functionalization mechanism
The FTIR analysis provided compelling evidence for a dual-ligand functionalization mechanism involving both α-lipoic acid and D-glucose. The hydroxyl group vibrations at 3340 cm−1 and 1393 cm−1 confirmed the incorporation of glucose-derived moieties, which were associated with enhanced colloidal stability through steric hindrance and hydrogen bonding interactions. 62 Retention of the carbonyl stretching vibration at 1694 cm−1 in the AgNPs-LA spectrum indicated that the carboxylic acid functionality of α-lipoic acid was preserved after surface attachment. This preserved feature likely contributed to the overall surface charge and facilitated biointeraction in physiological environments, as demonstrated in studies. 63
The attenuation of S–S stretching band at 732 cm−1 in the AgNPs-LA spectrum suggested reductive cleavage of the disulfide bond during synthesis. This transformation was interpreted as leading to the formation of thiolate species that subsequently formed strong Ag–S coordination bonds at the nanoparticle surface. Such chemisorption was consistent with high affinity of sulfur for silver and aligned with experimental observations reported by Benyettou et al. 64 As shown in Figure 2, the reduced intensity of the S–S band in AgNPs-LA relative to pristine LA supported this interpretation. Importantly, the persistence of C=O and O–H vibrational bands indicated that carboxyl and hydroxyl functional groups remained intact after binding. This spectral characteristic was consistent with an adsorption geometry in which sulfur-containing moieties interacted directly with the silver surface, while polar functional groups were oriented toward the surrounding medium, thereby contributing to colloidal stabilization and interfacial compatibility. 65
The complementary roles of α-lipoic acid and D-glucose in the green synthesis process yield a sophisticated surface architecture, in which α-lipoic acid provided strong covalent passivation via Ag–S bonding, while D-glucose contributed hydroxyl functionalities that enhanced hydrophilicity and reduced particle aggregation. This synergistic dual-ligand configuration was likely responsible for the enhanced stability and biological activity observed for the synthesized nanoparticles, consistent with reports by Singh et al. 66
Colloidal stability in this study was inferred indirectly from the dual-ligand surface chemistry and supported by molecular dynamics simulations, which indicated stable ligand binding and reduced solvent-induced destabilization at the nanoparticle interface. Nevertheless, comprehensive evaluation of colloidal stability under physiologically relevant conditions, particularly in high ionic strength and protein-rich media, was not performed and remained an important limitation. Parameters such as zeta potential, protein corona formation, and long-term dispersion stability were not assessed. Accordingly, future work will focus on systematic stability characterization under simulated physiological conditions. Experimentally, the AgNPs-LA suspensions remained visually stable, and no precipitation was observed during antibacterial testing.
Nanoparticle growth and size variation
The size variation observed across different synthesis durations provided insight into reaction kinetics. The increase in particle size between the 1 and 2 h syntheses suggested continued nanoparticle growth over time, consistent with trends reported for plant-mediated silver nanoparticle synthesis.67,68 However, the slight decrease in maximum particle size from 2 to 3 h (236 to 220 nm) warranted further investigation and may have indicated the onset of a stabilization regime. 69 The potential contribution of drying-induced agglomeration represented an important technical consideration, as sample preparation effects have been shown to influence nanoparticle size measurements. 70
UV-Vis spectroscopy provided additional insight into the nucleation and growth processes of AgNPs-LA. The observed SPR features and time-dependent peak shifts were consistent with a two-step nanoparticle formation mechanism involving initial nucleation followed by growth. 71 The SPR peak at 296 nm for the 1 h synthesis indicated the formation of smaller nanoparticles, while the red-shift to 458 nm at 2 h reflected continued growth and increased average particle size. The subsequent shift to 416 nm at 3 h suggested a stabilization phase, potentially accompanied by changes in size distribution or partial aggregation.51,53
In general, red-shifts in SPR peak position were associated with increasing particle size and plasmon coupling effects arising from closely spaced or partially aggregated nanoparticles. Conversely, blue-shifts accompanied by peak narrowing were associated with improved stabilization and reduced interparticle interactions. These optical trends were corroborated by surface morphology observations, and the combined UV-Vis and SEM analyses provided a coherent interpretation linking SPR evolution to nanoparticle growth kinetics and stabilization behavior.
Bacterial inhibition mechanism
To further contextualize the antibacterial evaluation, it is important to note that disk diffusion assays remain widely employed as an initial screening approach for nanoparticle-based antimicrobial systems due to their reproducibility, simplicity, and ability to provide rapid comparative insights across multiple bacterial strains. Unlike soluble antibiotics, nanoparticle diffusion in agar matrices is influenced by particle size, surface functionalization, and aggregation state, which may affect the apparent inhibition zone diameter. Consequently, inhibition zones should be interpreted primarily as indicators of relative antimicrobial response under standardized conditions rather than direct measures of bactericidal potency. In this study, the disk diffusion method was therefore employed as a qualitative comparative tool to establish concentration-dependent antibacterial trends and to benchmark the AgNPs-LA system against a clinically relevant antibiotic reference. While quantitative assays such as minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) measurements would provide higher-resolution antimicrobial characterization, the present screening approach remains appropriate for establishing the preliminary antibacterial potential of newly synthesized nanoparticle systems.
The findings demonstrated that antibacterial activity of AgNPs-LA increased with concentration, consistent with previous reports.55,57 A two-fold increase in concentration significantly improved the zone of inhibition and antibacterial efficacy against S. aureus and P. aeruginosa. At 40 µg/mL, AgNPs-LA exhibited antibacterial activity comparable to gentamicin, suggesting potential applications as an alternative antimicrobial agent. In contrast, at 20 µg/mL, the antibacterial effect of AgNPs-LA was not significantly different from that of absolute ethanol, indicating limited efficacy at lower concentrations. The reduced inhibition observed against E. coli was attributed to electrostatic repulsion, between negatively charged nanoparticles and the negatively charged bacterial outer membrane, which reduced interaction efficiency. 60 Nonetheless, hydrophobic domains on the AgNPs-LA surface may have contributed to bacterial adhesion through hydrophobic interactions. 72
Overall, the antibacterial response was found to be both concentration-dependent and microorganism-specific, reflecting differences in cell envelope structure, membrane permeability, and susceptibility to nanoparticle-mediated mechanisms. These findings highlighted the potential utility of AgNPs-LA as a topical antimicrobial alternative or adjunct, particularly for wound management applications where localized, high-concentration delivery was feasible.
Structural differences between Gram-negative and Gram-positive bacteria influenced the antibacterial response observed in this study. In Gram-negative bacteria, the highly negative surface charge associated with lipopolysaccharides (LPS) was expected to reduce nanoparticle adhesion through electrostatic repulsion and steric shielding, while the outer membrane barrier limited nanoparticle penetration and access to intracellular targets. 73 In contrast, Gram-positive bacteria lacked an LPS-rich outer membrane and possessed a thicker but more accessible peptidoglycan layer, facilitating closer nanoparticle-cell contact. 74 This structural distinction provided a mechanistic basis for the comparatively higher susceptibility of Gram-positive strains to AgNPs-LA under the tested conditions.
Based on inhibition trends and nanoparticle characteristics, membrane interaction and permeability enhancement were identified as the dominant antibacterial mechanisms of AgNPs-LA under the present assay conditions. The small particle size and surface functionalization facilitated close contact with bacterial cell envelopes, leading to membrane destabilization and increased permeability, consistent with the rapid inhibitory response observed in disk diffusion assays. Lipoic acid played a critical role by enhancing membrane permeability, disrupting cellular homeostasis, reducing intracellular adenosine triphosphate (ATP) levels, lowering internal pH, and inducing membrane hyperpolarization. 75 Molecular dynamics simulations supported effective LA coating of the AgNPs, which contributed to the overall antibacterial efficacy. This antibacterial action likely involved initial adsorption to the bacterial cell wall, accumulation at membrane sites, and subsequent disruption of the lipid bilayer, resulting in leakage of cellular contents and inhibition of cell multiplication, as illustrated in Figure 10. 60 The proposed antibacterial pathways are derived from well-established mechanisms reported for silver nanoparticles in the literature and should therefore be interpreted as mechanistically supported hypotheses rather than direct experimental observations within the present study.

Proposed interaction mechanism wherein AgNPs-LA silver ions and ROS react with bacterial cell wall.
Silver ion (Ag⁺) release and subsequent intracellular disruption were considered secondary mechanisms contributing to metabolic interference and enzymatic inhibition following initial nanoparticle–cell contact. Reactive oxygen species (ROS) generation and interactions with intracellular components such as DNA and proteins were proposed as supportive mechanisms that amplified antibacterial efficacy. AgNPs-LA may have disrupted bacterial signal transduction through protein dephosphorylation, ultimately leading to impaired cellular signaling. Released Ag⁺ ions increased membrane permeability and inhibited ATP production through ROS-associated pathways. 76 These ions exhibited strong affinity for phosphorus- and sulfur-containing biomolecules, enabling interactions with DNA that inhibited replication and cell division. 77 ROS-mediated oxidative stress further damaged genomic integrity. Within the cytoplasm, Ag⁺ ions interacted with ribosomes, proteins, and enzymes, where ribosomal destabilization inhibited translation and protein synthesis.60,76 Suppression of antioxidant defenses elevated ROS levels, ultimately contributing to bacterial death. 77
Molecular adsorption and interaction effect
The adsorption of α-lipoic acid (LA) onto the silver surface was characterized by an increasingly negative interaction energy over time, indicating progressive interfacial stabilization of the AgNPs-LA system. This stabilization was primarily attributed to the anchoring role of the disulfide (S–S) moiety, which oriented toward the Ag surface and facilitated strong interfacial attachment. The terminal carboxyl group remained exposed, enabling further interactions with the surrounding medium. Such structural arrangement contributes to enhanced surface passivation, reduced aggregation, and improved dispersion stability in aqueous environments. These features are consistent with the observed antibacterial activity of AgNPs-LA against Staphylococcus aureus, Pseudomonas aeruginosa, and Escherichia coli, where stable nanoparticle dispersion likely supports effective interaction with bacterial cells.
Molecular dynamics simulations further revealed that LA introduces hydrophobic domains near the metallic core while maintaining hydrophilic functional groups at the outer interface. This amphiphilic configuration provides a mechanistic basis for the biological behavior of AgNPs-LA. Hydrophobic regions may enhance interactions with bacterial membrane components, while exposed carboxyl and hydroxyl groups promote hydration and colloidal stability. The resulting balance between polar and nonpolar interactions supports stable dispersion without compromising membrane association, thereby strengthening the structure–function relationship between ligand chemistry and antibacterial performance.
Comparative adsorption analyses involving stearic acid, palmitic acid, and glucose highlighted ligand-specific interaction patterns on both bare AgNPs and LA-functionalized AgNPs. On bare AgNPs, long-chain fatty acids exhibited strong attraction and close packing due to van der Waals interactions along their hydrocarbon chains, promoting surface coverage and stabilization. 78 In contrast, glucose formed a more uniform polar layer that modulated surface activity and potentially acted as a diffusion barrier. 79 Upon LA functionalization, the adsorption behavior of these biomolecules changed markedly. Increased intermolecular distances observed for AgNPs-LA indicate that the LA coating modifies surface accessibility and interaction strength. The hydrophobic backbone of LA partially limits close approach of fatty acids, while the exposed hydrophilic groups mitigate excessive repulsion for polar molecules such as glucose. This ligand-dependent modulation demonstrates how surface functionalization governs subsequent interfacial interactions.80,81
The AgNPs-LA system therefore exhibits a heterogeneous amphiphilic surface architecture that adapts to environmental conditions. In aqueous or glucose-rich environments, hydrophilic groups dominate and promote colloidal stabilization. In lipid-rich environments, hydrophobic domains contribute more significantly to membrane association. This adaptive surface behavior provides a mechanistic explanation for the observed antibacterial efficacy while maintaining dispersion stability, supporting the proposed wound-healing application.
In general, the observed attraction of stearic acid, palmitic acid, and glucose, representative biomolecules of the skin lipid layer, to the AgNP surface supported their role as capping agents that stabilize nanoparticles via adsorption.82–84 Differences from the findings of Fabara et al. 85 may be attributed to the increased number of silver atoms in this simulation, which provided more realistic depiction of molecular interactions. For AgNPs-LA, the initial decrease in interaction distance followed by subsequent stabilization suggested dual behavior, an initial attraction due to orthogonal anchoring of the carboxylic acid group in LA, then followed by clearance stability that may have reduced nonspecific interactions and toxicity. 85 This dual behavior supported improved biocompatibility while preserving antimicrobial activity, enabling AgNPs-LA to interact with the skin lipid layer while maintaining dispersion stability and minimizing potential toxicity risks.
It is important to note that colloidal stability and cytocompatibility are critical for clinical translation. In the present study, stability was inferred from dual-ligand functionalization and MD-supported adsorption behavior. Biocompatibility considerations were discussed based on prior literature reporting reduced cytotoxicity of lipoic acid-coated AgNPs. However, zeta potential measurements and direct cytotoxicity evaluations (e.g., fibroblast viability or hemolysis assays) were not performed and represent limitations of this work. Future studies will incorporate electrokinetic stability measurements and in vitro cytocompatibility testing to validate safety under physiologically relevant conditions. Accordingly, the wound-dressing application proposed herein should be regarded as a potential application pending comprehensive biological validation.
Conclusion
This study demonstrated the successful green synthesis of α-lipoic acid-functionalized silver nanoparticles (AgNPs-LA) using D-glucose as a biogenic reducing agent. The infrared spectroscopy and molecular dynamics simulations confirmed a dual-ligand surface architecture in which α-lipoic acid anchors strongly to the silver surface through sulfur-silver interactions (Ag-S bonds), while D-glucose-derived hydroxyl groups enhance hydrophilicity and colloidal stabilization. This synergistic functionalization strategy resulted in predominantly spherical nanoparticles with tunable size distributions influenced by synthesis duration.
The variation in nanoparticle size observed at different synthesis durations provided insights into the kinetics of nucleation and growth. UV-Vis spectroscopy results were consistent with a two-step formation mechanism involving an initial nucleation stage followed by particle growth and stabilization. The time-dependent shift in surface plasmon resonance (SPR) peaks suggested continuous nanoparticle formation with a tendency toward stabilization at longer synthesis durations, although minor agglomeration during sample drying could not be excluded.
Antibacterial screening using agar disk diffusion assay revealed concentration-dependent inhibition of Staphylococcus aureus, Pseudomonas aeruginosa and Escherichia coli. At 40 µg/mL, AgNPs-LA exhibited antibacterial efficacy comparable to conventional antibiotics such as gentamicin against S. aureus and P. aeruginosa, highlighting their potential a as an antimicrobial platform. Reduced efficacy against Escherichia coli was attributed to electrostatic repulsion associated with the Gram-negative outer membrane, emphasizing the microorganism-specific nature of nanoparticle-cell interactions. Molecular dynamics simulations further elucidated the mechanistic basis of nanoparticle stability and biointeraction, revealing an amphiphilic surface structure that balances aqueous dispersion with membrane-active behavior.
Despite these promising findings, several aspects of the present study require further investigation to strengthen the translational relevance of AgNPs-LA. While current assessments were limited to qualitative agar disk diffusion, future studies will incorporate quantitative minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) determinations via broth-based assays to achieve higher-resolution antibacterial characterization.
Although this study focused on the integrated AgNP-lipoic acid system to reflect its intended use as a unified wound-dressing platform, it did not isolate the independent contributions of each component. Therefore, subsequent evaluations will include both non-functionalized silver nanoparticles and free α-lipoic acid. This comparative approach will allow for a clearer delineation of the individual and synergistic effects driving the system’s overall antimicrobial activity.
Comprehensive evaluation of colloidal stability under physiologically relevant conditions is an important consideration for the translational development of nanoparticle-based antimicrobial systems. In the present study, preliminary evidence of interfacial stabilization was inferred from the combined experimental and computational observations, including the dual-ligand surface chemistry and molecular dynamics simulations that indicated stable ligand adsorption and reduced solvent-induced destabilization at the nanoparticle interface. Nevertheless, quantitative electrokinetic characterization would provide stronger validation of nanoparticle dispersion behavior and stability.
Accordingly, the absence of electrokinetic measurements such as zeta potential and dynamic light scattering (DLS) analysis is acknowledged as a limitation of the present work and is identified as a priority for future investigations. Subsequent studies will incorporate systematic stability characterization, including zeta potential measurements to evaluate surface charge, DLS-based size distribution analysis to assess aggregation behavior, and stability testing under physiologically relevant ionic strengths and protein-rich environments. In addition, the formation of a protein corona and long-term dispersion stability will be examined to better understand nanoparticle behavior in biological systems. In parallel, cytotoxicity and hemocompatibility assessments will be conducted to further validate the biological safety of the AgNPs-LA system and support its potential application in wound-care technologies.
From a mechanistic standpoint, additional computational studies will be pursued to deepen understanding of nanoparticle-ligand and nanoparticle-biomolecule interactions. These will include extended simulation timescales, detailed analysis of adsorption layer structure and ligand orientation dynamics, and the application of more advanced computational approaches such as reactive force fields or electronic-structure-based methods. Furthermore, direct experimental validation of the proposed antibacterial pathways remains an important objective for future investigations. In particular, experimental confirmation of silver ion (Ag⁺) release and reactive oxygen species (ROS) involvement will be conducted to further substantiate the mechanistic framework proposed in this study. Quantification of Ag⁺ release kinetics will be performed using inductively coupled plasma mass spectrometry (ICP-MS), while ROS generation will be evaluated through scavenger-based assays to assess oxidative stress contributions to antibacterial activity.
These complementary analyses will provide deeper mechanistic insight into the relative roles of ion release and oxidative stress in the antimicrobial behavior of AgNPs-LA.
Footnotes
Acknowledgements
The authors would like to express their sincere appreciation to the Materials Research and Consulting Facilities of the University of the Philippines Department of Mining, Metallurgical and Materials Engineering.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Engineering Research Foundation and Development, Inc. (ERDFI) of the University of the Philippines College of Engineering and the Department of Science and Technology-Science and Education Institute (DOST-SEI). Additional support through a publication grant was provided by the Office of Vice Chancellor for Research and Development of the University of the Philippines Diliman (OVCRD-UPD).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available upon reasonable request from the corresponding author.