Abstract
Background:
The adhesion of root canal sealers to dentin plays a decisive role in the long-term success of endodontic treatment. Activation of irrigants before obturation has been suggested to enhance sealer penetration and bonding effectiveness. This study aimed to evaluate the effect of different irrigation activation techniques on the push-out bond strength of two bioceramic-based sealers to root dentin.
Materials and methods:
Eighty extracted human single-rooted teeth were decoronated to a standardized length and instrumented using rotary files with sodium hypochlorite irrigation. Specimens were randomly allocated into four groups according to the irrigation activation technique: conventional needle irrigation (control; lateral side-vented needle), manual dynamic agitation (MDA), EndoActivator, and EDDY (n = 20 each). Each group was further subdivided based on the sealer used—BioRoot RCS or GuttaFlow Bioseal (n = 10 each)—and obturated using the single-cone technique. The roots were sectioned into coronal, middle, and apical thirds, and push-out bond strength was measured using a universal testing machine. Failure modes were analyzed under a stereomicroscope.
Results:
A statistically significant regional variation was observed only in the EDDY–BioRoot RCS group, with the highest bond strength recorded in the coronal third (p < 0.05). In intergroup comparisons, significant differences were detected among experimental and control groups in the coronal region (p < 0.001), with the EDDY–BioRoot RCS group demonstrating superior bond strength compared to both activated and non-activated GuttaFlow groups. No statistically significant differences were observed among groups in the middle and apical thirds (p > 0.05). When data were pooled across activation protocols, BioRoot RCS exhibited significantly higher bond strength than GuttaFlow Bioseal in the coronal and middle regions (p < 0.05).
Conclusions:
Both the irrigation activation technique and the choice of calcium silicate-based sealer significantly influence push-out bond strength, predominantly in the coronal region. Sonic activation with EDDY in combination with BioRoot RCS yields the most favorable bonding performance. The diminishing effect toward the apical region underscores the need for further optimization of activation strategies to enhance sealer–dentin adhesion throughout the entire root canal system.
Keywords
Introduction
The successful outcome of root canal therapy is determined by an array of interrelated factors that extend beyond thorough chemomechanical debridement. Paramount contributors include the integrity and quality of the coronal seal, the patient’s host inflammatory response, and the sustained stability of both the obturation and subsequent restorative procedures. Prolonged prognosis hinges on effectively eliminating viable microorganisms, preventing reinfection via adequate coronal restoration, and minimizing adverse host tissue reactions, thereby securing functional and biologic stability for the endodontically treated tooth over time. 1
However, the inherent anatomical complexity of the root canal system—including lateral canals, isthmuses, accessory canals, apical deltas, and dentinal tubules—poses substantial challenges to achieving complete debridement with mechanical instrumentation alone.2,3 Numerous studies have demonstrated that conventional canal shaping frequently leaves residual smear layer, necrotic pulp debris, and bacterial biofilms adherent to canal walls, particularly in areas inaccessible to endodontic instruments.4,5
In overcoming the anatomical complexities inherent to root canal systems, advanced irrigation protocols assume a pivotal role in endodontic disinfection by effectively diminishing microbial load and facilitating access to regions inaccessible to mechanical instrumentation. Passive irrigation is limited in its capability to penetrate the apical third and intricate lateral canal anatomies, thereby restricting its overall efficacy. To surmount these challenges, irrigation activation techniques have been engineered to augment the mechanical effects of irrigants. These modalities enhance the hydrodynamic properties of irrigation solutions, resulting in amplified fluid movement, improved interaction with dentin surfaces, and greater penetration within the root canal network, ultimately promoting the disruption and eradication of resilient microbial biofilms.6,7
Manual dynamic agitation (MDA) is recognized as a straightforward and economically efficient technique for enhancing irrigant exchange and debridement of root canal walls. The method involves introducing a precisely fitted gutta-percha cone to the established working length, followed by controlled vertical push–pull movements. This action facilitates hydrodynamic activation of the irrigant, thereby improving its distribution and interaction within the canal system. 8
To enhance the effectiveness of irrigation during endodontic procedures, considerable research has focused on the development of activation systems based on sonic and ultrasonic energy. 7 Among these, the EndoActivator system (Dentsply Tulsa Dental Specialties, Tulsa, OK, USA) is one of the most extensively investigated sonic irrigation devices and was specifically designed to promote hydrodynamic irrigant activation within the root canal system. Its flexible, non-cutting polymer tips allow controlled and safe irrigant agitation, thereby significantly reducing the risk of procedural errors such as canal transportation, ledge formation, or unintended dentin removal during irrigation.9,10
Another advanced sonic activation system, EDDY (VDW, Munich, Germany), enhances irrigant activity through the generation of cavitation and acoustic streaming within a frequency range of 5,000–6,000 Hz.7,11,12 Similar to the EndoActivator, EDDY utilizes non-cutting polyamide tips, which enable efficient irrigant agitation while minimizing the risk of iatrogenic damage to the dentinal walls.12,13 In contrast, passive ultrasonic irrigation (PUI), although effective in improving fluid dynamics and debris displacement, employs oscillating metal tips that may unintentionally contact canal walls, potentially leading to uncontrolled dentin removal and unwanted alterations in the original canal morphology.14,15
Due to these limitations, sonic activation systems with non-cutting polymer tips have attracted considerable attention in the endodontic literature. Their ability to safely and efficiently deliver irrigants into the intricacies of the root canal system without compromising dentin structure has positioned them as promising alternatives to traditional ultrasonic methods.
Sonic (e.g., EndoActivator), ultrasonic (e.g., PUI), positive pressure delivery systems, and MDA are among the most commonly investigated techniques aimed at improving irrigation efficacy. These methods not only enhance smear layer removal but also significantly improve sealer infiltration into dentinal tubules, contributing to a more hermetic and durable root canal obturation.4,16 Moreover, several studies have reported that such activation techniques improve the adaptation of the sealer to the canal walls and reduce the potential for microleakage—an essential factor for the long-term success of endodontic therapy.17,18
Emerging evidence substantiates that the advantages of irrigation activation in endodontic procedures extend beyond mere disinfection. Contemporary systematic reviews and randomized trials demonstrate that active irrigation methods, such as sonic or ultrasonic activation, enhance canal and isthmus cleanliness, improve the sealing ability of filling materials by promoting irrigant movement and penetration, and can result in reduced postoperative pain. Furthermore, activation techniques have been shown to minimize adverse effects on dentin properties and debris removal from anatomical complexities inaccessible to standard needle irrigation.19,20 These techniques have been shown to positively influence the bond strength of root canal sealers, promote deeper penetration of bioceramic-based materials, and improve the overall quality of obturation. 18 Accordingly, irrigation activation is increasingly recognized as a critical procedural step with substantial impact on both the biological and mechanical outcomes of root canal treatment.
Bioceramic-based sealers have recently gained prominence in endodontics due to their inherent bioactivity, biocompatibility, and potential to chemically interact with dentin. These materials typically release calcium hydroxide during the setting process, resulting in an alkaline environment that promotes hydroxyapatite precipitation at the sealer–dentin interface, enhancing physical and chemical bonding.21,22 Among these, BioRoot RCS—a hydraulic calcium silicate–based sealer—exhibits sustained calcium ion release, high initial pH, and immunomodulatory bioactivity, making it a robust choice for durable adhesion. 21 In contrast, GuttaFlow Bioseal features a silicone-based matrix enriched with dispersed bioceramic particles. While it offers favorable handling properties and adequate flow characteristics, its alkalinizing activity is moderate, and its bioactive potential is comparatively limited.22,23 These physicochemical differences underscore the rationale for comparative evaluation of their adhesive performance in endodontic contexts.
In light of these considerations, the present study aims to comparatively assess the push-out bond strength of two bioceramic-based root canal sealers—BioRoot RCS and GuttaFlow Bioseal—following the application of different irrigation activation techniques, including MDA, EndoActivator, and EDDY systems. The null hypothesis of the study is that the type of irrigation activation method applied prior to obturation does not result in a statistically significant difference in the bond strength of the tested sealers. This hypothesis was tested using push-out bond strength, a validated method for assessing sealer–dentin adhesion. To our knowledge, no previous study has systematically compared the effects of MDA, EndoActivator and EDDY on the bond strength of BioRoot RCS and GuttaFlow Bioseal. This study therefore provides novel insights into the interaction between irrigation activation protocols and the adhesive performance of contemporary bioceramic-based sealers.
Materials and methods
In this study, 80 extracted human single-rooted, single-canal mandibular premolars (n = 80) with straight morphology, free of caries, fractures, cracks, and restorations, and extracted for orthodontic or periodontal reasons were utilized. After extraction, the teeth were immersed in 0.1% thymol solution and stored at room temperature until use to prevent microbial growth and dehydration. The root morphology and canal configuration were verified radiographically in both buccolingual and mesiodistal directions to confirm the presence of a single root and single canal and to ensure canal curvature less than 10° according to Schneider’s method. All teeth were obtained following informed patient consent. This study received ethical approval from the Bezmialem Vakıf University Ethics Committee (Approval Number: 183111). The study protocol was reviewed and deemed ethically appropriate.
Sample size calculation
An a priori power analysis was performed (GPower v3.1.9.7, Heinrich-Heine-Universität Düsseldorf) to determine the minimum total sample size for the primary comparison of coronal push-out bond strength across eight subgroups (four irrigation protocols × two sealers: control, MDA, EndoActivator, and EDDY × BioRoot RCS or GuttaFlow Bioseal). Because GPower does not directly model the Kruskal–Wallis test, a one-way fixed-effects ANOVA (omnibus) was used as a conservative parametric proxy. Assuming a medium-to-large effect size of f = 0.40 (based on pilot observations and previous reports comparing calcium silicate- and silicone-based sealers), with α = 0.05 and power (1−β) = 0.80, the required total sample size was N = 80 (i.e., n = 10 per subgroup). To compensate for possible specimen loss during preparation or sectioning, this total was planned a priori and equal allocation across groups was maintained. Although the final hypothesis testing was carried out using non-parametric procedures because of deviations from normality, the ANOVA-based power model is widely accepted as an approximation for Kruskal–Wallis designs with comparable group sizes and provides an adequate justification of study power for the primary endpoint.
Sample selection
The study included a total of eighty extracted human teeth, selected based on specific inclusion and exclusion criteria to ensure standardization and clinical relevance. Teeth were obtained from patients aged between 18 and 65 years, following informed consent. Only single-rooted mandibular premolar teeth extracted for orthodontic or periodontal purposes were included. Eligible teeth exhibited an intact coronal structure, minimal caries, and no existing restorations.
Teeth were excluded if they were obtained from patients outside the specified age range or if they exhibited multi-rooted anatomy, additional canals, open apices, calcified canals, or curved roots. Additionally, teeth with extensive caries, restorations, structural fractures, or visible cracks were not considered for inclusion in the study.
Root canal preparation, irrigation, and obturation
To standardize root length to 18 mm, the crowns of all teeth were sectioned using a diamond disc under water cooling. All specimens were embedded in self-curing acrylic resin blocks to ensure standardized stabilization during canal preparation and sectioning procedures. Root canal instrumentation was performed using an X-Smart Plus endodontic motor (Dentsply Sirona, Ballaigues, Switzerland) in combination with VDW ROTATE rotary files (VDW GmbH, Munich, Germany). All instruments were operated strictly according to the manufacturer’s recommended speed and torque settings to ensure controlled and standardized canal shaping.
During instrumentation, each canal was irrigated with 2 mL of 5.25% sodium hypochlorite (NaOCl; Merck KGaA, Darmstadt, Germany) between each file, resulting in a total irrigant volume of approximately 15 mL per canal. After completion of instrumentation, a final irrigation protocol was applied using 5 mL of 17% EDTA for 60 s to remove the smear layer, followed by 5 mL of distilled water to eliminate any chemical residues.
Specimens were randomly allocated into four primary groups (n = 20), based on the irrigation activation technique employed:
Group 1—Conventional needle irrigation (control)
Irrigation was performed using a 30-gauge side-vented needle (NaviTip; Ultradent Products Inc., South Jordan, UT, USA), positioned 2 mm short of the working length (WL). A total of 5 mL NaOCl was delivered through gentle in-and-out movements over 60 s, followed by a final rinse with 5 mL distilled water. No activation was applied.
Group 2—MDA
For MDA, a size 30/0.04 gutta-percha cone was used to generate a controlled hydrodynamic piston effect within the canal, as recommended in previous studies employing small-diameter cones for effective manual irrigant agitation.7,8,24,25 After delivery of 5 mL NaOCl, activation was performed for 60 s using approximately 100 gentle push–pull strokes, with the cone positioned 2 mm short of WL.
Group 3—Sonic irrigation with EndoActivator (Dentsply Sirona, Ballaigues, Switzerland)
Operated at 167 Hz with a size 25.04 red polymer tip, irrigant activation was performed in two rounds of 60 s each, with the tip positioned 2 mm short of the WL. A final rinse with 5 mL distilled water was performed.
Group 4—Sonic irrigation with EDDY (VDW GmbH, Munich, Germany)
Performed with a 25.04 polymer tip attached to an air scaler (TA-200; Micron, Tokyo, Japan) operating at 6000 Hz. Irrigation involved 5 mL NaOCl activated in three cycles of 20 s each, with the tip positioned 2 mm short of WL.
Following final irrigation, all canals were dried with #40 paper points but were intentionally left slightly moist to promote optimal adhesion of calcium silicate-based sealers. 26
Each primary group was subdivided into two subgroups (n = 10), according to the sealer used:
BioRoot RCS (Septodont, Saint-Maur-des-Fossés, France): A calcium silicate-based hydraulic cement characterized by calcium hydroxide release, bioactivity, and hydrophilicity. Obturation was performed using the single-cone technique with matching gutta-percha cones to ensure consistent adaptation along the canal walls.
GuttaFlow Bioseal (Coltène/Whaledent AG, Altstätten, Switzerland): A silicone-based sealer containing dispersed bioceramic particles, selected for its superior flow properties and ease of clinical handling. Obturation was also performed using the single-cone technique, following the manufacturer’s instructions.
All specimens were stored at 37°C with 100% humidity for 2 weeks to allow complete setting of the sealers before conducting push-out bond strength testing.
Section preparation
All obturated teeth were embedded in acrylic resin and allowed to set completely. Each specimen was then transversely sectioned using a low-speed diamond wafering disc (Isomet 1000 Precision Saw; Buehler Ltd., Lake Bluff, IL, USA) under continuous water cooling. Sectioning was performed at a controlled speed and load to minimize thermal and mechanical stress on the specimens. From each root, six transverse slices, approximately 1 mm in thickness, were obtained—two slices from each root third (coronal, middle, and apical)—to ensure standardized representation across all regions. 27
Push-out test
Slices obtained from the eighty prepared specimens were subjected to push-out bond strength testing using a universal testing machine (Shimadzu AG-X Plus; Shimadzu Corp., Kyoto, Japan). Each slice was positioned centrally on the testing jig, and a compressive load was applied in an apical-to-coronal direction using custom-made cylindrical plungers. The custom-made plunger diameters (0.5, 0.8, and 1.2 mm) were individually selected according to the canal lumen diameter at each root level to ensure that the applied load was directed exclusively to the filling material without any contact with the surrounding dentin walls.28,29 The load was applied at a constant crosshead speed of 1 mm/min until bond failure occurred.
The maximum load at failure, recorded in Newtons (N), was used to calculate the push-out bond strength in megapascals (MPa) by dividing the load by the bonded area of the root canal surface (mm2), following the standard formula The lateral area of the root canal space was derived using the formula for a conical frustum, which is as follows:
A compressive force was then applied to each section using a cylindrical plunger mounted on a universal testing machine until bond failure occurred at the sealer-dentin interface. The maximum force required to dislodge the filling material was recorded in Newtons (N) and subsequently converted to Megapascals (MPa) to determine the push-out bond strength.
Failure mode analysis
Three specimens from each experimental group were randomly selected for failure mode analysis. Following the push-out bond strength test, the root slices were examined to determine the type of bond failure, using criteria consistent with methodologies described in previous studies.30,31 Failure mode classification was performed under a stereomicroscope (Leica Microsystems, Heerbrugg, Switzerland) at 16× magnification. Representative micrographs were captured at 2.5× magnification to display the entire canal circumference. Scale bars were added post-acquisition based on calibration with a stage micrometer imaged using the same optical configuration.
Adhesive failure: debonding at the sealer-dentine interface, where the canal walls appeared free of sealer.
Cohesive failure: fracture within the sealer material, with canal walls remaining completely coated with sealer.
Mixed failure: a combination of adhesive and cohesive failure patterns, where both sealer-coated and uncoated areas were observed on the canal walls.
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics version 22 (IBM Corp., Armonk, NY, USA). The distribution of the data was assessed using the Shapiro–Wilk test. Variables that did not conform to a normal distribution were expressed as median, minimum, and maximum values. For comparisons among six independent groups, the Kruskal–Wallis H test was used. Where significant results were observed, post hoc pairwise analyses were conducted with the Mann–Whitney U test with Bonferroni correction. A p-value of <0.05 was considered to indicate statistical significance These non-parametric tests were chosen because the data did not follow a normal distribution and all statistical analyses were performed based on independent observations at the slice level. The use of Bonferroni correction ensured control of type I error in multiple comparisons.
Results
The descriptive statistics of push-out bond strength values for all experimental and control groups are presented in Table 1. The EDDY–BioRoot group exhibited the highest median bond strength in the coronal region (11.48 MPa), whereas the lowest median values were recorded in the EDDY–GuttaFlow and EndoActivator–GuttaFlow groups. The MDA groups demonstrated intermediate bond strength values between the EDDY and EndoActivator protocols in all root regions, without showing a consistent superiority over the other activation techniques. The control groups showed moderate bond strength values, with the BioRoot control group (CB) consistently exhibiting higher medians than the GuttaFlow control group (CG) across all root regions.
Descriptive statistics of push-out bond strength values (MPa) for all experimental and control groups at the coronal, middle, and apical thirds of the root canal.
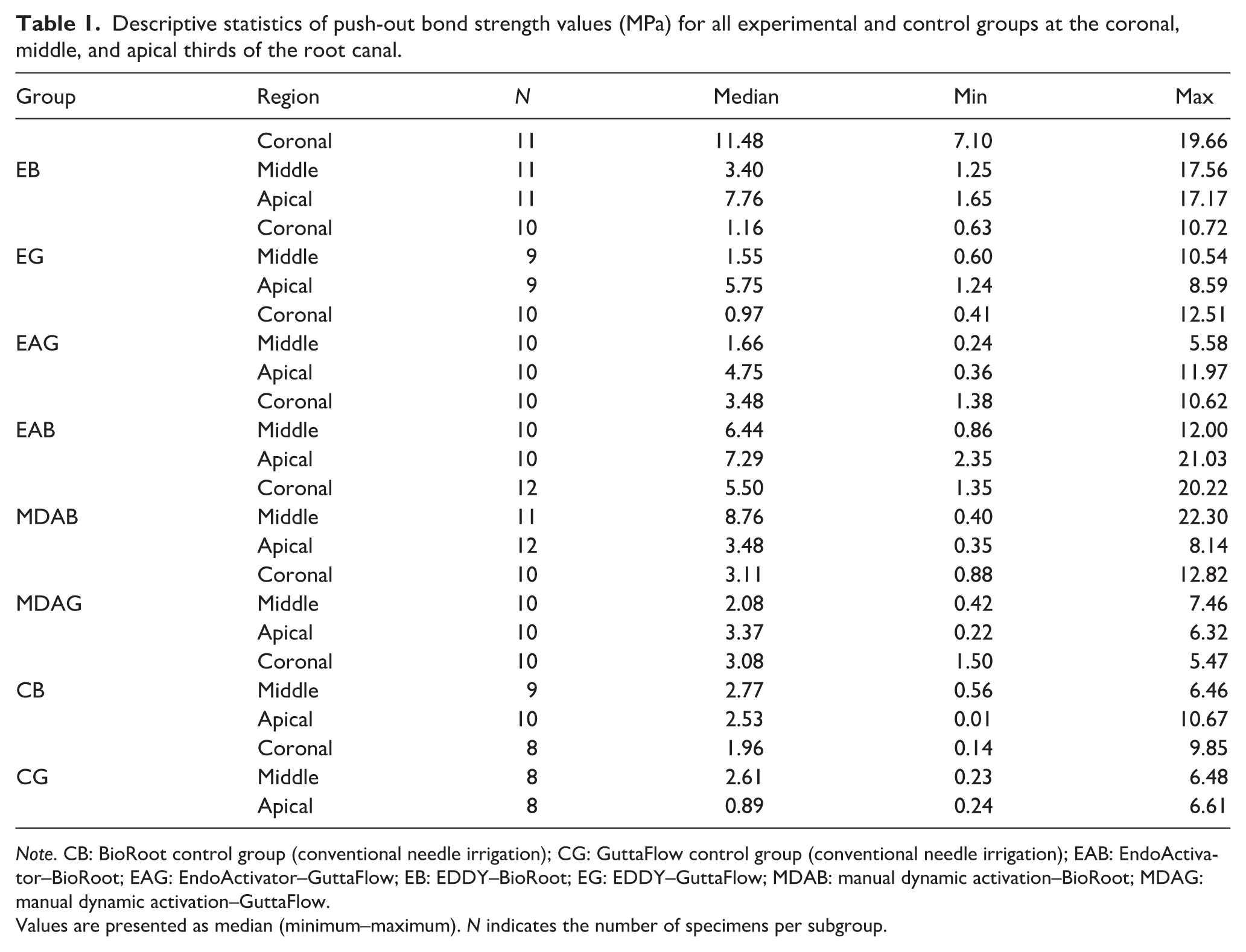
Note. CB: BioRoot control group (conventional needle irrigation); CG: GuttaFlow control group (conventional needle irrigation); EAB: EndoActivator–BioRoot; EAG: EndoActivator–GuttaFlow; EB: EDDY–BioRoot; EG: EDDY–GuttaFlow; MDAB: manual dynamic activation–BioRoot; MDAG: manual dynamic activation–GuttaFlow.
Values are presented as median (minimum–maximum). N indicates the number of specimens per subgroup.
Intergroup comparisons including both experimental and control groups were performed using the Kruskal–Wallis H test (Table 2). A statistically significant difference was observed among the groups in the coronal region (χ2 = 24.31, p < 0.001). Post hoc comparisons indicated that the EDDY–BioRoot group demonstrated significantly higher coronal bond strength values compared with GuttaFlow-based experimental and control groups.
Comparison of coronal, middle, and apical bond strength values among experimental groups.

Note. Values are presented as median (minimum–maximum). Statistical analysis was performed using the Kruskal–Wallis H test (p < 0.05). Post hoc comparisons with Bonferroni correction are indicated in the “d” column. In the coronal region, the EDDY–BioRoot group exhibited significantly higher bond strength values compared to GuttaFlow-based experimental and control groups (1 > 2, 3, 7).
In the middle third, the Kruskal–Wallis test also revealed statistically significant intergroup differences (χ2 = 17.82, p = 0.022). BioRoot-based groups, particularly MDA–BioRoot and EDDY–BioRoot, exhibited higher median bond strength values compared with both GuttaFlow experimental groups and the CG control group.
In the apical third, a statistically significant difference among the groups was likewise detected (χ2 = 16.94, p = 0.030). Although overall bond strength values were lower in this region, BioRoot-based experimental and control groups tended to outperform GuttaFlow groups.
Overall, these findings demonstrate that both the irrigation activation technique and the type of bioceramic sealer significantly influence push-out bond strength across all root canal regions, with the most pronounced differences observed in the coronal and middle thirds.
Taken together, these findings indicate that both the irrigation activation technique and the choice of calcium silicate-based sealer exert their most pronounced influence on bond strength in the coronal third, whereas their effect becomes less evident toward the apical region. This corono-apical gradient highlights the need for further optimization of activation protocols and material formulations to improve adhesion throughout the entire root canal system.
Failure mode distribution
In the control groups, the GuttaFlow control group showed a predominance of mixed failures (45.8%), followed by cohesive (29.2%) and adhesive (25.0%) failures (n = 24). In contrast, the BioRoot control group was characterized by a higher proportion of mixed failures (51.8%), closely followed by adhesive failures (44.8%), whereas cohesive failures were rare (3.4%; n = 29).
Within the MDA groups, the MDA–BioRoot subgroup exhibited predominantly adhesive failures (55%), followed by mixed (27%) and cohesive (18%) failures (n = 11). A similar trend toward adhesive dominance was observed in the MDA–GuttaFlow subgroup, where adhesive failures accounted for 50% of cases, followed by cohesive (33%) and mixed (17%) failures (n = 12).
In the EndoActivator groups, the EndoActivator–BioRoot subgroup demonstrated an even distribution between adhesive and mixed failures (both 43%), with cohesive failures being least frequent (14%; n = 14). Conversely, the EndoActivator–GuttaFlow subgroup displayed a predominantly adhesive failure pattern (67%), while cohesive and mixed failures each constituted 17% (n = 12).
In the EDDY–activated groups, a clear shift toward cohesive failure was observed. The EDDY–BioRoot group exhibited a marked predominance of cohesive failures (73%), with mixed (18%) and adhesive (9%) failures occurring less frequently (n = 11). Similarly, the EDDY–GuttaFlow group showed a dominance of cohesive failures (58%), followed by adhesive and mixed failures at equal frequencies (21% each; n = 14).
Overall, these findings indicate that irrigation activation—particularly sonic activation with EDDY—shifts the failure pattern toward a more cohesive mode, especially in calcium silicate-based sealers, reflecting enhanced sealer–dentin interfacial integrity compared with both control and manual activation protocols.
Representative stereomicroscopy micrographs of the three failure modes are shown in Figure 1(a): mixed; Figure 1(b) and Figure 1(c): cohesive; Figure 1 (d): adhesive, while the group-wise distributions are summarized quantitatively in the 100% stacked bar chart (Figure 2). All images were obtained under the same optical setup (acquisition at 2.5×; failure-mode classification at 16×).

Representative stereomicroscopic images of failure modes after push-out bond strength testing: (a) mixed failure; (b) cohesive failure; (c) cohesive failure; (d) adhesive failure.

Distribution of failure modes by group (100% stacked bars).
Discussion
This in vitro study evaluated the influence of different irrigation activation protocols—conventional needle irrigation (control), MDA, EndoActivator, and EDDY
Recent evidence highlights that the effectiveness of bioceramic based sealers is closely linked to the quality of smear layer removal and the efficiency of the irrigation protocol used prior to obturation. Smear layer remnants and adhered debris can hinder the penetration of calcium silicate-based sealers into dentinal tubules, reducing micromechanical retention and compromising the formation of a stable interfacial bond. Enhanced irrigant activation improves the chemical and physical cleansing ability of the irrigating solutions, increasing dentin surface wettability and facilitating deeper sealer infiltration. Studies comparing various irrigation protocols have shown that activation-assisted smear layer removal significantly increases the push-out bond strength of calcium silicate sealers by promoting superior intratubular penetration and a more homogeneous sealer–dentin interface. This has been confirmed by Al Omari et al., 33 who demonstrated that activation-enhanced irrigation protocols substantially improved the bonding performance of both TotalFill and AH Plus sealers. These findings collectively indicate that the interaction between irrigant activation and bioceramic sealer adhesion is not incidental but mechanistically interconnected, thereby reinforcing the clinical relevance of selecting an optimized irrigation strategy prior to placement of calcium silicate-based sealers.
The predominance of cohesive failures in BioRoot groups—especially with EDDY activation—indicates that the adhesion to dentin was sufficiently strong that debonding occurred within the material itself rather than at the interface. Clinically, this suggests a more reliable long-term sealing ability, as cohesive failures are less likely to compromise the sealer–dentin interface. On the other hand, the higher incidence of adhesive and mixed failures in GuttaFlow groups suggests a weaker interfacial bond, which may predispose to microleakage and reduced long-term sealing efficiency. 34 This finding aligns with the material’s silicone-based composition, which prioritizes handling properties over chemical bonding capacity.
The role of irrigant activation—particularly using the EDDY sonic system—has been shown in new 2025 in vitro and clinical studies to significantly improve irrigant penetration, sealer infiltration, and smear layer removal, especially in the coronal and middle thirds of the canal. These advantages are attributed to enhanced acoustic streaming and cavitation inherent to EDDY’s design. 35
When comparing the two sealers, BioRoot RCS consistently exhibited higher bond strength values than GuttaFlow Bioseal in the coronal and middle regions, while no significant difference was observed in the apical third. These findings are in agreement with previous reports demonstrating the superior adhesion of BioRoot RCS compared to silicone-based sealers in push-out testing.36–38 This enhanced performance is primarily attributed to its bioactive characteristics: during setting, BioRoot RCS releases calcium hydroxide, which promotes the nucleation of hydroxyapatite crystals and establishes a chemical interaction with dentin, forming a mineralized interfacial layer that reinforces long-term adhesion.39,40 Furthermore, its hydrophilic nature allows reliable adaptation in moist environments, a clinically relevant feature in cases where achieving complete canal dryness is challenging. These material-specific properties collectively explain the consistently higher bond strength values of BioRoot RCS observed in the present study. Recent findings confirm that calcium silicate-based sealers, such as BioRoot RCS, are associated with superior interfacial adaptation, bond strength, and long-term sealing ability compared to resin- and silicone-based alternatives, with randomized clinical trials showing improved outcomes and healing rates over a 4-year follow-up period. 41
By contrast, GuttaFlow Bioseal—although marketed as a bioceramic-containing sealer—relies primarily on a silicone matrix within which calcium silicate particles are dispersed. This distinctive formulation undoubtedly provides certain practical advantages, including excellent flowability and reliable compatibility with the single-cone obturation technique, thereby facilitating efficient and convenient clinical handling. Nevertheless, it must be emphasized that these benefits come at the expense of adhesive capacity. Its comparatively lower ion release and restricted potential for establishing true chemical bonding to dentin significantly limit its ability to form a stable and durable sealer–dentin interface when compared with calcium silicate-based sealers. The weaker bond strength observed for GuttaFlow Bioseal in the present study is, therefore, not incidental but rather an inherent reflection of its material composition—one that prioritizes handling convenience over sustained biological and chemical integration with dentin. 42
These material-dependent differences clearly emphasize the pivotal role of sealer selection in achieving predictable long-term root canal obturation quality. Recent systematic reviews and well-controlled experimental studies consistently demonstrate that calcium silicate-based sealers exhibit superior sealing ability, enhanced dentin adaptation, and greater bioactivity compared with conventional alternatives. Their intrinsic capacity to induce hydroxyapatite nucleation and to chemically interact with dentinal substrates facilitates the formation of a stronger, more stable, and biologically active interfacial layer.43,44 Although both BioRoot RCS and GuttaFlow Bioseal may be considered clinically acceptable materials, the superior adhesion, improved interfacial stability, and sustained bioactivity associated with BioRoot RCS confer distinct and clinically meaningful advantages in maintaining long-term sealing integrity. This perspective is further supported by emerging clinical evidence reporting improved obturation outcomes and enhanced radiographic healing when calcium silicate-based sealers are used in comparison with resin-based sealers, 45 as well as by comprehensive reviews underscoring the decisive influence of material properties on overall endodontic treatment prognosis. 46
Systematic reviews published in 2025 further endorse the use of calcium silicate-based sealers for their bioactive properties, including the formation of a mineralized interfacial layer and hydroxyapatite nucleation, promoting durable adhesion even under challenging clinical moisture conditions.47,48
The superior outcomes obtained with the EDDY–BioRoot RCS protocol further suggest a synergistic effect between a highly effective sonic activation system and a calcium silicate-based sealer. Sonic activation methods, such as EDDY, are known to improve irrigant penetration and enhance sealer infiltration into dentinal tubules through acoustic streaming and cavitation, ultimately reinforcing the sealer–dentin interface.49,50 Other in vitro investigations, such as the study by Tomar et al., 51 corroborate these findings by reporting that sonic and ultrasonic activation significantly increase sealer penetration compared with conventional irrigation methods.
BioRoot RCS consistently outperformed GuttaFlow Bioseal in the coronal and middle thirds, consistent with studies showing the superior bioactivity and bonding of calcium silicate sealers. 52 Despite these favorable outcomes, the apical third remained a persistent limitation, with no significant differences detected among groups. This finding is consistent with previous studies, which reported that the apical region is less amenable to irrigant penetration and sealer adaptation due to reduced tubule density, smaller canal diameters, and limited irrigant dynamics. 17 The inability to achieve predictable adaptation in this region highlights the need for continued development of irrigant delivery systems and modified sealer formulations tailored specifically to overcome apical anatomical challenges.
From a clinical perspective, these findings suggest that combining EDDY with BioRoot RCS may enhance the adhesive performance of root canal sealers, particularly in the coronal and middle thirds, thereby improving long-term sealing and treatment outcomes. Nevertheless, the results should be interpreted cautiously. The in vitro design of this study does not replicate the complex oral environment, and variables such as occlusal forces, saliva contamination, or thermal fluctuations were not simulated. Moreover, the push-out test, although widely used, provides only a simplified assessment of adhesion and does not fully represent the functional stresses encountered in vivo. 53 Chen et al. 54 previously highlighted that push-out testing, while valuable, may overestimate adhesive strength due to its simplified mechanics.
This study has certain limitations that should be acknowledged. First, its in vitro design cannot fully replicate the complex biological, thermal, and mechanical conditions of the oral environment; therefore, factors such as occlusal loading, thermal cycling, saliva contamination, and long-term aging were not simulated. Second, although the push-out test is a widely accepted method for evaluating sealer–dentin adhesion, it represents only one aspect of sealer performance and does not fully reflect the complex stress distribution encountered under clinical conditions. Additionally, anatomical variability of dentinal tubules and canal morphology, particularly in the apical third, may have influenced the regional bonding outcomes. Passive ultrasonic and laser-activated irrigation techniques were not included in the present study, which may limit direct comparison with studies employing these activation methods. Minor inequalities in subgroup sample sizes were unavoidable due to specimen loss during preparation; however, non-parametric statistical methods were applied to reduce potential bias.
Future investigations should incorporate long-term aging protocols, such as thermocycling and cyclic mechanical loading, and advanced imaging techniques, including micro-computed tomography and confocal laser scanning microscopy, to provide a more comprehensive evaluation of sealer penetration, interface stability, and failure modes. Well-designed clinical trials are also required to determine whether the observed in vitro advantages can be reliably translated into long-term clinical success.
Conclusion
Within the limitations of this in vitro study, both the irrigation activation technique and the choice of bioceramic sealer significantly influenced push-out bond strength predominantly in the coronal region. The EDDY–BioRoot RCS combination yielded the highest bond strength values. BioRoot RCS performed better than GuttaFlow Bioseal in the coronal third, while no significant differences were observed in the middle and apical regions. These findings suggest that sonic activation may enhance coronal sealer–dentin adhesion under controlled laboratory conditions; however, bond strength represents only one parameter of sealer performance and should not be interpreted as a direct predictor of clinical success.
Footnotes
Ethical considerations
The research was carried out in accordance with the Declaration of Helsinki and received approval from the Non-Interventional Clinical Research Ethics Committee of Bezmialem Vakif University (Protocol Number: 2025/22, Approval Number: 183111, dated February 5, 2025) for studies involving human participants.
Author contributions
Conceptualization—F.B.P., H.Ç.; Investigation—F.B.P., A.N.; Writing—original draft—F.B.P.; Writing—review and editing—F.B.P., H.Ç., A.N.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated and analyzed during the current study are included in this published article. Further inquiries can be directed to the corresponding author.*

