Abstract
Citrate, widely used as an anticoagulant in transfusion and extracorporeal therapies, disrupts calcium-dependent coagulation by chelating ionized calcium, resulting in hypocalcemia and severe coagulopathy. This interference elevates bleeding risks and may trigger hemorrhagic shock. Current treatments for local bleedings in the context of citrate-induced coagulopathy include topical hemostats that either provide a physical barrier to the bleeding or activate platelets and the coagulation cascade. This study investigates the potential of calcium carbonate (CaCO3) to counteract the citrate coagulopathic effect by promoting coagulation through calcium ion release and platelet aggregation. In vitro assays demonstrated that various CaCO3 polymorphs significantly enhance coagulation in citrated blood. Both coral-derived aragonite and synthetic calcite reduced coagulation time, with calcite particles achieving up to a 2-fold reduction. Prothrombin and Partial Thromboplastin Times decreased by 13% and 24%, respectively, with calcite treatment. Moreover, calcite reduced circulating platelet counts by 13% while directly binding platelets, indicating effective recruitment, and raised free calcium levels in plasma by 2.6-fold compared to controls. These dual effects—calcium elevation and platelet concentration—suggest that CaCO3 is a promising hemostatic agent for addressing bleeding in citrate-induced coagulopathy, offering innovative solutions for transfusion medicine and critical care.
Introduction
The challenge of bleeding management in transfusion and extracorporeal therapies using citrated blood
Citrate, widely used as an anticoagulant in transfusions and extracorporeal therapies, poses significant challenges when present at toxic levels in the bloodstream.1,2 By chelating ionized calcium, citrate disrupts calcium-dependent coagulation, leading to hypocalcemia and severe bleeding risks. 3 Calcium is a vital cofactor for multiple steps of the coagulation cascade, including the activation of clotting factors and platelet aggregation, and its depletion can severely impair hemostasis complicating surgical and clinical interventions.4,5 The clinical challenge in large transfusion and hemodialysis is further exacerbated as large volumes of citrated blood products are administered, potentially overwhelming the body’s capacity to metabolize citrate and maintain adequate ionized calcium levels.6,7 This not only predisposes patients to bleeding complications but may also contribute to cardiac dysfunction due to ischemia-derived impaired myocardial contractility5,8 and coagulopathy.1,9 Moreover, the liver plays a pivotal role in citrate metabolism. Patients with liver damage—whether preexisting or acute—have a diminished capacity to clear citrate from the bloodstream. 10 The resulting accumulation of citrate exacerbates hypocalcemia and coagulopathy, further increasing the likelihood of hemorrhagic events. 9 In these circumstances, the impaired clearance of citrate leads to compromised clot formation due to calcium depletion and an increased risk of uncontrollable bleeding. 11
Another blood coagulation mechanism that is disturbed by citrate is platelet-mediated clotting. Upon vascular injury, platelets adhere to the exposed subendothelial matrix, become activated, and aggregate to form a primary hemostatic plug. 12 This process is facilitated by various surface receptors and intracellular signaling pathways, many of which are calcium dependent.13,14 Therefore, in the context of citrate toxicity, platelet function can be impaired due to hypocalcemia, further exacerbating bleeding risks.6,15
The standard approach to counteracting citrate-induced hypocalcemia involves the administration of intravenous calcium, which serves to replenish ionized calcium levels in the bloodstream. 1 However, intravenous calcium supplementation does not directly address the persistent accumulation of citrate in the bloodstream, 16 which continues to sequester calcium ions and further disrupt hemostasis.17,18 Moreover, local bleeding in patients with citrate-induced coagulopathy—whether due to trauma or during surgery—is particularly dangerous and difficult to control. Because systemic measures alone are often insufficient in these scenarios, the use of adjunctive therapies becomes indispensable. Topical hemostatic agents provide a physical barrier and actively promote local clot formation, which is critical when the coagulation cascade is compromised. 19 In patients with citrate-induced coagulopathy, where there is no alternative to restore hemostasis effectively, these local interventions become essential for controlling hemorrhage and ensuring patient safety during critical procedures.20,21
Topical hemostatic agents for bleeding arrest in citrate-induced coagulopathy
Research has explored agents that promote coagulation through mechanisms independent of systemic calcium levels, examining synthetic polymers, biologically active compounds, and inorganic minerals. 17 These alternative agents offer the potential to initiate or support clot formation directly at the site of bleeding, circumventing the limitations imposed by systemic hypocalcemia.
Examples of topical hemostatic agents used in citrate coagulopathy include oxidized regenerated cellulose (ORC),22,23 gelatin sponges, 24 and fibrin sealants. 25 ORC acts as a physical matrix, facilitating platelet adhesion and activation, while gelatin sponges absorb blood and expand to exert localized pressure on the bleeding site. Fibrin sealants, which combine fibrinogen and thrombin, mimic the terminal steps of the coagulation cascade, thereby promoting rapid clot formation at the site of hemorrhage. In situations where systemic correction of coagulopathy is insufficient, these topical interventions have proven critical for achieving hemostasis. 26
Calcium carbonate polymorphs and blood coagulation
Calcium carbonate, a naturally occurring mineral, is a unique biomaterial with significant potential for effective hemorrhage control in coagulopathic conditions due to its hemostatic properties, acid sensitivity, and adhesiveness. Calcium carbonate exists in several polymorphic forms—most notably calcite and aragonite—with each form offering distinct crystalline structures.27,28 Owing to its excellent biocompatibility,29,30 inherent bioactivity, and wide availability, calcium carbonate has been successfully utilized in various medical applications, including drug delivery systems30,31 and bone regeneration.32,33 Furthermore, these properties have spurred research into its use as a hemostatic agent, with studies demonstrating its ability to accelerate blood coagulation.34–36
Calcium carbonate’s sensitivity to acids makes it a strong candidate as a topical hemostat for citrate coagulopathy. When exposed to strongly acidic conditions, calcium carbonate breaks down into CO2 and Ca2+. This property has been exploited to target specific hemorrhaging sites within acidic environments, such as tumors, where the local pH is often lower than in normal tissues.35,37 Studies have shown that citrate, a weak acid, can etch the surface of calcium carbonate crystals, releasing additional calcium ions into the surrounding environment.35,38 In such scenarios, the localized increase in calcium concentration at the bleeding site when using calcium carbonate would be sufficient to neutralize the citrate effect, offering a novel approach to controlling citrate coagulopathy.
Moreover, calcium carbonate is cell-adhesive and supports the attachment of various cell types.39,40 It also promotes cellular differentiation, proliferation, and growth.41–43 Additionally, there is evidence that calcium carbonate can bind and activate platelets. 44 Therefore, incorporating calcium carbonate crystals into citrated blood may serve as platelet-binding cores that trigger platelet-dependent clotting mechanisms, thereby bypassing the need for systemic calcium ion supplementation.
The primary objective of this study was to evaluate the efficacy of calcium carbonate as a hemostatic agent in mitigating the challenges of citrate coagulopathy. Our results demonstrate that calcium carbonate exhibits significant hemostatic capacity in both murine and human blood in vitro by binding platelets and releasing calcium ions, even in the presence of citrate. These findings underscore the potential of calcium carbonate for clinical applications in critical care and surgery of coagulopathic patients and transfusion medicine, offering a promising solution for managing coagulopathy and persistent local bleeding associated with citrate.
Materials and methods
Experimental design
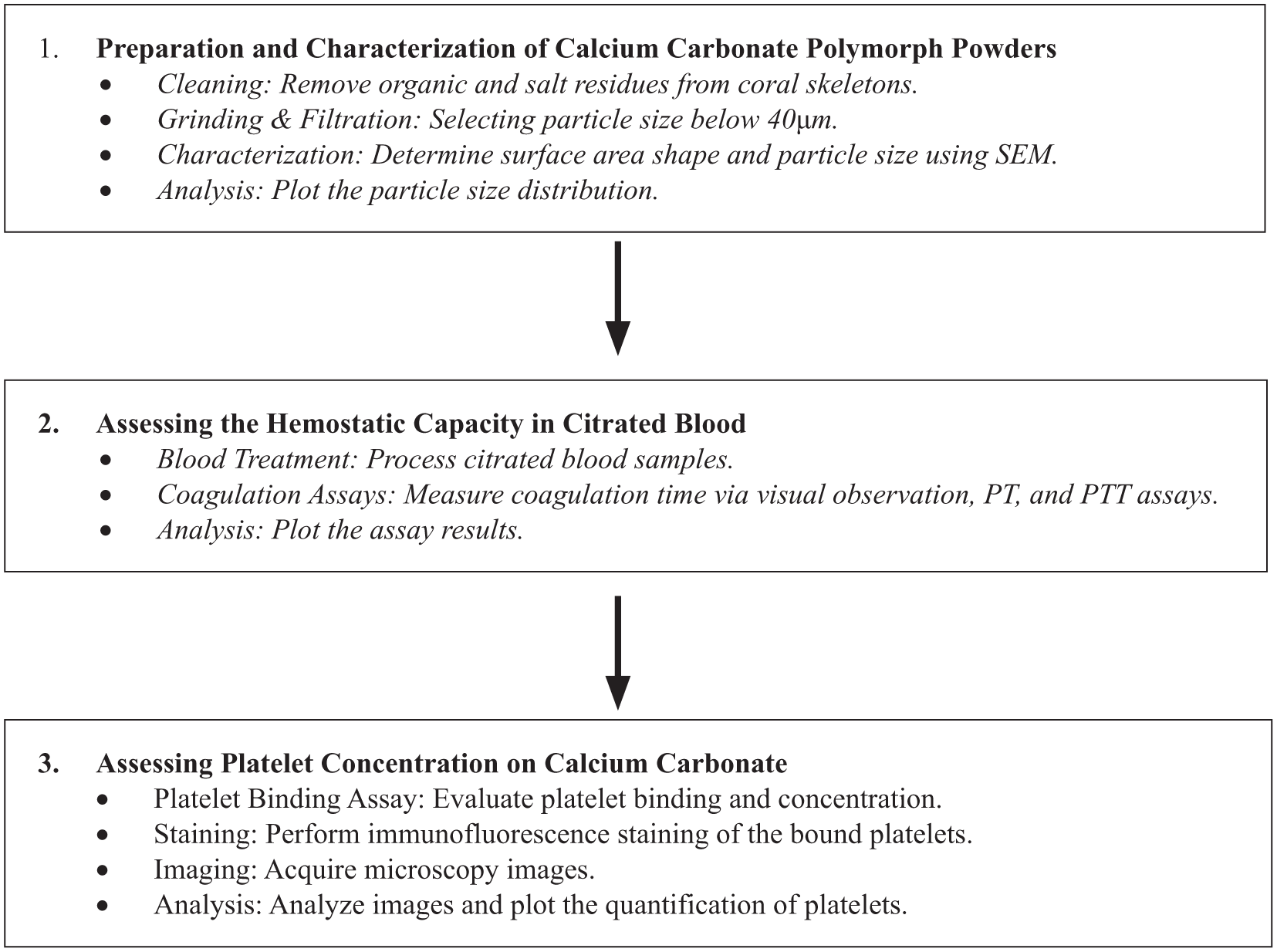
The objectives of the experimental setup were: (a) To evaluate whether calcium carbonate polymorphs can induce coagulation in vitro despite the presence of citrate. (b) To demonstrate that the hemostatic effect of calcium carbonate is attributable to its ability to bind and concentrate platelets, as well as to release calcium ions.
To achieve these objectives, calcium carbonate polymorphs were first ground, and the shape and size of the resulting grains were measured. Their effect on coagulation time was then compared to that of untreated citrated blood using visual observation, PT, and PTT assays. Platelet binding and concentration were determined and quantified through an in-vitro binding assay coupled with fluorescence microscopy. Finally, calcium ion release into the blood was measured using a specific assay kit.
Experimental design chart
Preparation of coral skeletons and calcite
The exoskeletons of the corals Fungia, Porites Lutea and Trachyphyllia geoffroyi, were sectioned into 1–2 cm sections and treated with 10% (v/v) sodium hypochlorite (Romical, Israel) at room temperature (RT) to remove organic residues and washed with double distilled water (DDW). Next, they were treated with 1 M NaOH (Sigma Aldrich) solution for 5 min at RT to digest any adhering organic matter. The fragments were then washed with DDW and transferred into a H2O2 (Merck, USA) solution (35% v/v aq, 10 min, RT,) and further rinsed three times with DDW, air-dried, and ground using a Smart Dentin Grinder (KometaBio, Cresskill, NJ, USA) to grains not larger than 200 µm in size, and autoclaved. The particles were sieved into different size fractions: smaller than 40 µm and smaller than 70 µm. For all experiments, particles smaller than 40 µm were used, except for analysis in Figure 2(d). For comparison, commercially available calcite (Glentham, Cat. #GK3384) was used, also containing particles smaller than 40 µm.
Scanning electron microscopy
For scanning electron microscopy (SEM) imaging, the particles were affixed to conductive carbon discs, coated with a thin layer of gold, and examined using a Quanta 200 ESEM/SEM (FEI).
In vitro clotting assay
Eight-week-old ICR mice were euthanized using CO2, and blood was collected via cardiac puncture with a 23G needle and placed in citrated tubes (~1 mL/mouse, 3.2% citrate). The designated treatment at a concentration of 50 mg/mL was placed at the center of a 60-mm Petri dish, 300 µL of blood were added and gently mixed with the particles. For each experimental series, an untreated control sample (300 µL of blood) was included to account for potential variability. Clot formation was assessed visually and by tilting the dish to evaluate related physical changes. Since blood without anticoagulant clots within several minutes when removed from the body and citrated blood doesn’t clot, we concluded the experiments after 15 min.
Coagulation panel
Coagulation assays, prothrombin time (PT), partial thromboplastin time (PTT), fibrinogen levels, and platelet counts were conducted at Hasharon and Hadassah Medical Centers, Israel. Human blood was collected into 3-mL citrate tubes, treated with Calcite (5 mg/mL), or CaCl2 (0.13 mg/mL), or a combination of both, and mixed gently.
For platelet counts, samples were centrifuged at 150 g for 3 min to obtain platelet-rich plasma (PRP), which was then analyzed using the ADVIA 2021i system (SIEMENS). For PT, PTT, and fibrinogen assays, samples were centrifuged at 2500 g for 5 min to obtain platelet-poor plasma (PPP) and then analyzed using the CS-5100 system (SIEMENS).
Platelet aggregation assay
Glass coverslips (12 mm-diameter) were coated with calcite by mixing DDW with Calcite particles at a concentration of 50 mg/mL, applying 40 µL of the mixture onto the coverslips, and heating them on a heating plate (40ºC) until water evaporation. 100 µL of mouse blood without anticoagulant were added to the coverslips and incubated for 3 min. The coverslips were then washed twice with Phosphate-Buffered Saline X1 (PBS X1) and fixed with 4% formaldehyde solution (RT, 10 min, Sigma-Aldrich).
Immunofluorescence
For platelet visualization after fixation with 4% formaldehyde, coverslips were immersed for 1 h in a blocking solution containing 3% Normal goat serum. The samples were then incubated overnight at 4ºC with an Anti-CD41 monoclonal antibody (rabbit, cat. ab134131, Abcam), washed, and further stained with a secondary antibody Alexa 555-conjugated Goat Anti-Rabbit (cat. ab150118, Abcam) for 1 h at RT. The coverslips were then mounted on slides using Fluoromount (Sigma-Aldrich) and analyzed using a fluorescence microscope.
Fluorescent microscopy
Fluorescent images were acquired using an inverted Zeiss Axio-observer Z1 microscope equipped with four objectives: X10/0.13, X20/0.45, X40/0.10, X60/0.85 and with the fluorescent FITC and Rhodamine filter cubes.
Calculation of PT/PTT/fibrinogen levels
A comparative analysis of PT/PTT/fibrinogen levels in calcite-treated versus untreated CB, as shown in Figure 3(c), was performed as follows (example for fibrinogen):
Calcium release assay
To determine the kinetics and pH-dependence of calcium release from Calcite, human blood was collected into 3 mL citrate tubes, treated with calcite (50 mg/mL) and incubated at RT for 5 min under gentle 360° tilting. Next, PPP was obtained (by centrifugation at 2500 g for 5 min) and analyzed for free calcium ions using a colorimetric assay kit (cat. MAK022, Sigma-Aldrich). Calcium concentration was determined via absorbance at 575 nm (based on the chromogenic complex between calcium ions and o-cresolphthalein), with blanks used to subtract systemic calcium levels and background interference.
Statistical analysis
Student’s t-test was used when comparing two groups if the variables did not significantly depart from a normal distribution according to a one-sample Kolmogorov–Smirnov test. Otherwise, a Mann–Whitney U-test was used with Pearson correlation. Multiple comparison analyses were performed either by one-way ANOVA followed by Fisher’s Least Significant Difference test (LSD) for parametric analyses with normal distributions, or by Kruskal–Wallis ANOVA followed by Dunn’s test for nonparametric analyses. Results presented with mean ±SD; *p < 0.05; **p < 0.01; ***p < 0.001. ns = non-significant.
Results
To examine the influence of calcium carbonate (CaCO3) from various sources on the coagulation of citrated blood, crystals were obtained from different coral skeletons and synthetic calcite. The upper row of Figure 1(a) displays the skeletons of three distinct types of corals, all of which are organized as aragonite. These coral skeletons serve as the source of the particles depicted in the second row. The particles were produced using a Smart Dentin Grinder. The fourth panel in the middle row represents synthetic calcium carbonate, which is organized in the calcite polymorph, providing a comparison to the naturally sourced aragonite.

Types of calcium carbonate particles used to test hemostasis: (a) (upper row) stereoscopic images of coral skeletons; (middle row) scanning electron micrographs of particles derived from the coral skeletons and synthetic calcite; (bottom row) higher magnification images of particles from the middle row. Some particle images were tilted clockwise or counterclockwise for visualization. (b) Particle size distribution of the various samples. Scale: (upper row) Fungia: 1.3 cm; P. Lutea: 1 cm; Trach: 1.8 cm; (middle row) 100 µm; (bottom row) 30 µm.
SEM images (Figure 1(a), middle and bottom rows) and a quantitative analysis of the distribution of particle length (Figure 1(b)) revealed that coralline crystals manifest a larger average length of 59.3 µm, compared to Calcite with an average of 11.09 µm. Among the coralline crystals, P. Lutea had the smallest average length of 36.2 µm, Fungia with 70.3 µm, and Trachyphyllia, having the largest particles with an average length of 104.5 µm. Length distribution analysis (Figure 1(b)) showed that calcite exhibits a pronounced peak at around 10 µm, indicating a high frequency of fine particles. P. Lutea has a broader distribution with most particles ranging from 20 µm to 100 µm, suggesting more variability. Trachyphyllia and Fungia display the widest length range among all CaCO3 sources tested, mostly below 100 µm, without pronounced peaks, showing lower frequency and particle consistency, compared to calcite. Further examination of the surface morphology of the particles under SEM showed distinct surface morphologies across the CaCO3 sources (Figure 1(a), middle and bottom rows). Fungia manifested a relatively smooth, planar surface with fine striations, suggesting minimal fragmentation. P. Lutea displayed a slightly rougher surface with visible microcracks. Trachyphyllia, though featuring sharper edges, exhibits a surface with less pronounced roughness, compared to P. Lutea and Fungia, suggesting some degree of smoothness but with defined structural elements. The calcite stands out with the roughest surface.
To evaluate citrated blood coagulation in the presence of CaCO3 grains, an in-vitro visual clotting test was developed. Mice blood was collected into a citrated tube and the anticoagulated blood was then transferred into a dish containing Calcite/Aragonite at a ratio of 50 mg/mL (CaCO3 to blood) and mixed (Figure 2(a)). Blood clotting was evaluated by tilting the dish and observing the physical changes indicative of clot formation (Figure 2(b) and (c)). Graph 2D illustrates the effect of different CaCO3 sources on the time required for blood coagulation. The control group (Citrated Blood; CB) exhibited the longest coagulation time, taking 15 min. In contrast, the addition of different coral skeletons significantly reduced the coagulation time. Specifically, the addition of Trachyphyllia reduced the coagulation time by 1.6-fold to 9.5 min, P. Lutea by 2-fold to 7.34 min, and Fungia, which had the strongest effect among the corals (aragonite), reducing the coagulation time by 3-fold to 5 min. Similarly to P. Lutea, Calcite had reduced the coagulation time by 2-fold to 7 min. Figure 2(e) illustrates the impact of different calcite grain sizes on the coagulation time of citrated blood. The control group exhibited a 15-minute coagulation. Calcite grains smaller than 70 µm and 40 µm significantly reduced the coagulation time to 7.6 min (2-fold reduction) and 6.4 min (2.3-fold reduction), respectively. These results suggest that the coagulation-enhancing effect of calcite particles increases with decreasing grain size.

Hemostasis by the calcium carbonate polymorphs in the presence of citrate: (a) schematic representation of the in vitro blood coagulation assay, (b) visual outcomes of the assay: non-coagulated blood, (c) coagulated blood, (d) coagulation time for blood exposed to particles derived from different coral skeletons and calcite, and (e) coagulation time for blood exposed to calcite particles of two different sizes.
Given its widespread abundance and stability, calcite represents the most accessible and practical form of calcium carbonate, justifying its selection for subsequent experiments. Following the visual assessments demonstrating the hemostatic effect of calcite in the presence of citrate, a coagulation test panel was conducted to evaluate its impact on the coagulation cascade, including measurements of Prothrombin Time (PT), Partial Thromboplastin Time (PTT), and Fibrinogen levels in citrated blood. Treatment of citrated blood with Calcite significantly reduced the PT and PTT by 13%, and 24%, respectively (Figure 3(a) and (b)). By contrast, fibrinogen levels significantly increased by 4% (Figure 3(c)), suggesting a possible augmentation of fibrinogen availability or stability in the presence of calcite.

Calcite activates the intrinsic and extrinsic pathways of coagulation in citrated blood: (a) partial thromboplastin time (PTT) assay to evaluate activation of the intrinsic pathway, (b) prothrombin time (PT) assay to assess activation of the extrinsic pathway, (c) fibrinogen levels in blood (see calculation formula in the methods section).
To further delineate the mechanism of calcite-driven hemostasis under citrate, we asked whether calcite (i) binds and activates platelets and (ii) alters the availability, kinetics, and pH-dependence of free Ca²⁺. Fresh citrated whole blood was exposed to calcite-coated coverslips (3 min), washed, and stained for CD41. Fluorescence imaging revealed abundant platelet binding on calcite surfaces, with numerous CD41⁺ platelets (red) interspersed among calcite particles (green) (Figure 4(a) and (b)).

Calcite promotes platelet adhesion/aggregation and elevates free Ca²⁺ in citrated blood. (a) Wide-field fluorescence image of citrated whole blood on calcite-coated coverslips: platelets (CD41, red) over calcite particles (green autofluorescence). Scale bar, 30 µm. (b) Higher-magnification view of (a). Scale bar, 5 µm. (c) Platelet counts in citrated blood (CB) with or without calcite. Counts are normalized to the CB baseline (mean ± SEM; n = 3). Statistics: One-way ANOVA. *p < 0.05. (d) Free Ca²⁺ level following exposure to calcite (colorimetric assay). Bars show mean ± SEM; n = 3. Statistics: Two-tailed t-test; ***p < 0.001. (e) Kinetics of free Ca²⁺ accumulation after adding calcite to CB; values are expressed relative to t = 0 (100%), n = 3. Statistics One-way ANOVA; ns > 0.1. (f) pH-dependence of free Ca²⁺ release; values are expressed relative to pH 7.4 (100%), n = 3. Statistics: One way ANOVA; **p < 0.002.
To determine whether calcite’s platelet-mediated pro-coagulant activity persists when citrate inhibition is clinically neutralized, we quantified platelet counts in citrated whole blood under four conditions: CB, CB + CaCl2 (recalcification), CB + CaCO3, and CB + CaCl2 + CaCO3. Recalcification with CaCl2 alone produced platelet levels comparable to the CB baseline (Figure 4(c), ns), indicating that restoring free Ca²⁺ did not by itself reduce circulating platelets. In contrast, addition of CaCO3 to recalcified blood induced a significant decrease in platelet counts relative to both CB and CB + CaCl2 both when CaCl2 was absent (CB + CaCO3) or present (CB + CaCl2 + CaCO3) (p < 0.01; Figure 4(c)), consistent with particle-mediated platelet activation/aggregation and consumption. These findings demonstrate that CaCO3 promotes coagulation even in the presence of excess Ca²⁺, thereby separating its effect from simple calcium replenishment.
In parallel, calcite increased free Ca²⁺ in blood by ~2.6-fold (n = 3, p < 0.001; Figure 4(d)). Kinetic profiling showed that free Ca²⁺ rose to a plateau within ~1 min after calcite addition (Figure 4(e)), and decreasing pH below 7.4 produced a modest additional increase in free Ca²⁺ (Figure 4(f)). Together, these data support a particle-dependent mechanism whereby calcite both recruits/aggregates platelets and promptly generates bioavailable Ca²⁺ under physiologically relevant conditions.
Discussion
This study demonstrates the hemostatic potential of calcium carbonate as a topical agent in citrate coagulopathy by inducing coagulation via calcium ion release and platelet aggregation. Both calcite and aragonite polymorphs successfully initiated coagulation even in the presence of citrate. The calcium carbonate surfaces promoted platelet adhesion and aggregation while releasing free calcium ions into citrated blood. These findings highlight calcium carbonate’s ability to overcome the inhibitory effects of citrate by restoring localized calcium levels and stimulating platelet aggregation. This dual mechanism positions calcium carbonate as a promising hemostatic agent for managing bleeding complications in citrate-induced coagulopathy. Importantly, this effect persisted in recalcified citrated blood, supporting a particle-mediated, platelet-dependent mechanism rather than simple Ca²⁺ replenishment.
The experiments support the potential use of CaCO3 particles in vivo, in settings of impaired hemostasis. However, systemic presence of CaCO3 particles within the circulation could increase embolic risk. Consequently, systematic in-vivo safety testing—including platelet counts, standard coagulation panels, and global viscoelastic assays—is required to ensure safety and preclude thrombosis or microvascular occlusion.
Polymorphism-independent hemostatic efficacy of calcium carbonate
Due to their unique structural and mechanical properties, as well as their excellent biocompatibility and biodegradability, coral skeletons have been extensively studied for applications as bone graft substitutes and in tissue regeneration.32,33 The findings of this study demonstrate that coral skeletons reduce coagulation time compared to the controls and suggest a novel application of coral skeletons in promoting hemostasis.
The findings show that all tested coral skeletons and calcite reduced coagulation time by approximately 50% on average, compared to controls (Figure 2(d)). This efficacy is attributed to their shared chemical composition, particle sizes, and rough surfaces, which promote platelet adhesion and aggregation. Variations in crystalline form (aragonite vs. calcite) and surface roughness do not significantly affect the hemostatic potential of calcium carbonate, highlighting its broad applicability in hemostatic applications.
Dosing considerations and translation
The concentrations used in our ex vivo whole-blood assays (50 mg/mL in mouse; 5 mg/mL in human) were selected to provide robust, reproducible readouts under citrate and are not intended as clinical doses. For topical use, dosing is better expressed as area-based loading (mg cm⁻²) with particle immobilization in a matrix or dressing and defined contact time/flow, which together can achieve effective local activity at substantially lower bulk particle densities than implied by mg/mL in blood. To enable translation, future studies will include concentration–response mapping under topical-like conditions (matrix-embedded CaCO3, controlled flow, and standardized application areas) to identify minimal effective ranges while maintaining safety.
The role of calcium carbonate’s particle size in hemostasis and potential mechanisms
The lack of significant hemostatic differences between 40 and 70 µm particles (Figure 2(e)) may be due to minimal variation in available surface area. If the size difference does not substantially increase surface area, the hemostatic response remains similar. In contrast, smaller particles (e.g., submicron or nanometer scales) enhance hemostasis because of a higher surface area-to-volume ratio, which improves interactions with platelets and coagulation factors.18,45 The similar performance of 40 and 70 µm particles may indicate an optimal range for particle-induced hemostasis, with both eliciting near-maximal responses. Future research on particles outside this size range could help clarify the thresholds for hemostatic efficacy.
The impact of calcium carbonate on coagulation machinery
The 13% reduction in PT (Figure 3(a)) indicates a significant acceleration in the extrinsic coagulation pathway, likely due to calcium ion release from calcium carbonate. This decrease indicates clotting factor activation and fibrin formation. Mechanistically, it suggests calcium ions overcome citrate’s chelating effect, restoring activity in calcium-dependent factors like factor VII and the tissue factor complex. 46
The 24% reduction in PTT (Figure 3(b)) reflects significant stimulation of the intrinsic and common coagulation pathways. This greater reduction compared to PT likely stems from calcium ions’ involvement in multiple factors, including VIII, IX, XI, and XII, and amplification loops in the intrinsic pathway. 47 The disparity between PT and PTT reductions highlights calcium carbonate’s stronger impact on the intrinsic pathway, driven by its reliance on sequential calcium-dependent activations.
The 4% increase in fibrinogen levels (Figure 3(c)) suggests stabilization or a slight rise in availability, likely due to interactions with calcium carbonate surfaces. Elevated fibrinogen indicates its release from activated platelets. 48 However, the modest increase implies that calcium carbonate mainly acts through platelet aggregation and activation and calcium ion release rather than affecting fibrinogen synthesis or turnover.
These findings underscore the dual action of calcium carbonate in restoring coagulation processes impaired by citrate coagulopathy. The differential impact on PT and PTT suggests a targeted enhancement of intrinsic pathway components, while the minimal increase in fibrinogen levels emphasizes the effects of calcium ion release and platelet aggregation. Future studies could further delineate these mechanisms by comparing the effects of calcium carbonate with other hemostatic agents in vitro and in vivo.
Calcium carbonate mediated platelet reduction and its implications for coagulation
Around 13% reduction in platelet number observed in Figure 4(c) signifies a substantial depletion of circulating platelets, reflecting their active aggregation at the site of calcite interaction. From a quantitative perspective, assuming an average platelet count of 300,000 per microliter in human blood, 49 a 20% reduction equates to approximately 75,000 platelets aggregating per microliter. This is a remarkably high number, suggesting robust platelet recruitment to calcite surfaces to initiate hemostasis.
Studies on platelet aggregation in response to various triggers, such as ADP, collagen, or thrombin, have demonstrated that the magnitude of aggregation depends on the stimulus and its concentration. For instance, strong agonists like thrombin can activate and aggregate a larger proportion of platelets compared to weaker agonists like ADP. 50 The high platelet aggregation observed underscores calcite’s potency as a hemostatic trigger, driven by calcium ion release and surface-mediated activation. Exploring the link between platelet number and clotting efficiency in calcite-induced aggregation could optimize its clinical hemostatic use.
Implications of enhanced calcium release from calcite in coagulation
The 2.6-fold increase in calcium ion release in Figure 4(d) highlights calcite’s significant contribution to local free calcium, a critical coagulation cofactor. This increase reflects calcite dissolution in response to acidity or citrate, with citrate partially chelating the released ions. Similar phenomena are reported in other systems. Calcium-based scaffolds like hydroxyapatite and calcium phosphate show size-dependent enhanced ion release, 51 which may be sufficient to bypass systemic chelation and promote coagulation. Controlled dissolution of calcium-rich substrates creates localized environments conducive to biological processes, even with compromised systemic calcium levels. Alternatively, the calcium release may result from citrate-induced surface disruption of calcite particles,50,52 accelerating ion release. Interaction with blood proteins or platelets could further amplify coagulation by concentrating calcium ions near activated platelets. The increase in free calcium highlights calcite’s potential as a hemostatic agent, overcoming citrate’s anticoagulative effects.
Summary and conclusions
Calcium carbonate exhibits a dual hemostatic mechanism, enhancing coagulation even in the presence of citrate. The study showed reduced PT and PTT, about 13% drop in circulating platelets, and a 2.6-fold increase in free calcium ion levels in the calcite-treated blood, demonstrating calcite’s ability to counter citrate-induced calcium chelation and support coagulation. These results showcase calcite’s unique ability to act as a topical hemostatic agent, bypassing systemic calcium deficiencies.
These findings have important implications for basic and clinical science. Mechanistically, they highlight calcite’s dual action: restoring local calcium levels through dissolution and promoting platelet aggregation via surface interactions. Therefore, they strongly contribute to understanding the benefits of calcium-based hemostats. Clinically, calcite’s ability to counter citrate-induced coagulopathy highlights its potential for managing bleeding during transfusion medicine and critical care, where citrate toxicity complicates coagulation. The observed calcium release and platelet aggregation provide a basis for developing innovative strategies for impaired coagulation, advancing treatments for bleeding disorders and surgical interventions.
Given that clinical hemostasis often benefits from combining modalities, CaCO3 particles could be formulated as an adjunct to established classes of topical hemostatic agents. In principle, co-application or incorporation of CaCO3 into porous matrices, adhesive/sealant systems, or pro-coagulant gels/foams may yield additive or synergistic effects—shortening time-to-hemostasis, increasing clot firmness, and improving adherence on oozing surfaces. The optimal pairing is likely context-dependent (e.g., capillary ooze vs. brisk bleeding; smooth vs. irregular tissue) and warrants standardized head-to-head testing to define performance and safety. Future work will systematically evaluate representative modalities by class and optimize formulation parameters (particle size control, loading, immobilization) to maximize benefit while maintaining safety. Delivery options include dry powder, matrix-embedded/coated dressings, and pastes/foams to enable area-based dosing, local immobilization, and compatibility with existing hemostats. Optimal format is context-dependent and should be defined via standardized comparative tests (time-to-hemostasis, clot firmness, adherence, safety).
Footnotes
Acknowledgements
The authors acknowledge Drs. Eyal Canji and Ayala Gancz for their assistance during the early stages of the work, and they thank Dr. Wayel Orfali for his help in drafting the initial manuscript and editing the final version.
Abbreviations
CB: citrated blood
PT: prothrombin time
PTT: partial thromboplastin time
SEM: scanning electron microscope
Trach: Trachyphyllia geoffroyi
P. Lutea: Porites lutea
Author contributions
Roni Hendler: Investigation, data curation, formal analysis, writing, visualization. Orly Weiss: Investigation, data curation, formal analysis, writing, visualization. Bhuwan Bhaskar: Formal analysis, writing. Akansha Kothidar: Formal analysis. Liat Hammer: Investigation, data curation. Romi Feigelman: Data curation, formal analysis. Danny Baranes: Conceptualization, resources, supervision, project administration, funding acquisition, writing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by the “Kamin” project and the Israeli Innovation Authority, and partially by Life-Force Labs (Ben Gurion 1, Ramat-Gan, Israel).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

