Abstract
The use of plant extracts for the synthesis of metal nanoparticles represents a promising direction in nanotechnology due to the high environmental friendliness and cost-effectiveness of the method. In this study, green synthesis of silver nanoparticles was carried out using Hibiscus sabdariffa and Aloe vera extracts. The synthesis was conducted by reaction between the extracts with a silver nitrate solution, leading to the formation of silver nanoparticles. The synthesized nanoparticles were identified using UV-visible spectroscopy, scanning electron microscopy, particle size determination, and X-ray diffraction (XRD) analysis. The UV spectra exhibited characteristic absorption in the range of 350–450 nm, confirming the presence of silver nanoparticles, while XRD analysis determined an average particle size of 6.11 nm. The size analysis demonstrated the stability of the nanoparticles over time and their retention within the nanometer range. Comparative antibacterial tests demonstrated that AgNPs synthesized using H. sabdariffa extract showed higher inhibitory activity against E. coli and S. saprophyticus compared to those produced with A. vera extract. The obtained results confirm that biogenic synthesis using plant extracts is an environmentally safe and highly efficient alternative to conventional chemical methods, offering promising applications in biomedicine, catalysis, and other high-tech fields.
Keywords
Introduction
Metal nanoparticles represent a significant advancement in nanotechnology due to their remarkable stability and exceptional physicochemical properties, including superior electrical conductivity, enhanced thermal resistance, and outstanding optical characteristics. 1 Their versatile characteristics make them suitable for a wide range of applications, including textiles, electronics, medical products, food packaging, and bio-labeling. They are particularly valuable in wound care, antimicrobial dressings, and as potential anti-tumor agents.2,3 Silver nanoparticles, which have garnered significant attention, are widely used in consumer products, electronics, and biomedical applications, including as anti-inflammatory, antiviral, and antidiabetic agents.4,5
Nanoparticles of metals and their oxides can be synthesized using physical, chemical, and biological methods. Physical synthesis methods are costly due to the need for high pressure and temperature, as well as continuous energy consumption. Additionally, these methods require high-throughput equipment and are often limited by time constraints.6–8 The chemical synthesis of nanoparticles often results in the formation of by-products that may be toxic, contribute to environmental pollution, and pose potential health risks, especially when used in food or medical applications.9–11 Hence, to address these challenges, biological synthesis of nanoparticles is preferred, as it utilizes plant materials or microorganisms that are cost-effective, eco-friendly, non-toxic, and readily available, while also eliminating harmful by-products.12–14 By avoiding costly chemicals and energy-intensive procedures, the use of biological reducing and stabilizing agents in green synthesis significantly lowers production costs. 15 Furthermore, biomolecules derived from plant extracts wrap nanoparticles with protective organic layers that improve their colloidal stability and lessen their propensity to aggregate and oxidize while being stored. 16 In particular, low-temperature synthesis is a promising approach, ensuring process stability, reducing energy consumption, and minimizing by-product formation. Green synthesis in nanoparticle production refers to the use of natural biological agents such as plant extracts, 17 microorganisms, 18 or enzymes 19 to reduce metal ions to their nanoparticle form. Unlike physical methods, which require high energy input, and chemical methods, which often involve hazardous reagents and generate toxic by-products, green synthesis is environmentally friendly, cost-effective, and typically operates under mild reaction conditions. 20
Silver nanoparticles synthesized through green methods have a broad range of applications across various fields due to their unique properties. In medicine, they are utilized for antimicrobial coatings on medical devices and implants, effectively reducing the risk of infections. 21 In the food industry, these nanoparticles serve as antibacterial agents, helping to extend the shelf life of food products. 22 In the field of water purification, green-synthesized silver nanoparticles effectively eliminate pathogens and remove toxic compounds, ensuring safer and cleaner water. 23 The environmental friendliness and biocompatibility of green synthesis make these nanoparticles highly promising for further research and practical applications. Using plant extracts for synthesis offers significant advantages, including energy efficiency, the absence of toxic by-products, and simplified process conditions compared to microbial-based methods.
The use of Hibiscus sabdariffa extract for the synthesis of silver nanoparticles has demonstrated promising antibacterial, antioxidant, and biochemical properties.24–26 Phytochemicals of the H. sabdariffa extract acts as both a reducing and capping agent during the green synthesis of silver nanoparticles, with its phenolic acids, flavonoids, and anthocyanins facilitating the rapid reduction of silver ions and ensuring nanoparticle stability. 27 The formation and structural characteristics of AgNPs were confirmed through analytical and spectroscopic techniques, revealing a spherical shape with an average size of 21.22 nm25 and 65.9 nm. 26 Antimicrobial studies showed that AgNPs were more effective against Salmonella typhi, Shigella dysenteriae, Staphylococcus aureus, Shigella boydii и Escherichia coli. The significant biochemical potential of H. sabdariffa extract was further demonstrated by its total phenolic and flavonoid content, measured at 84.9 mg/g and 41.5 mg/g, respectively. These findings suggest that silver nanoparticles synthesized from H. sabdariffa extract hold great promise for medicinal and biological applications.
Aloe vera leaf extract, contains phytochemicals, that are promising as stabilizing and reducing agent, is used for the eco-friendly synthesis of silver nanoparticles (AgNPs).28–30 The extract functions simultaneously as a reducing and stabilizing agent in silver nanoparticle synthesis, with phytocomponents such as acemannan and polysaccharides enabling silver ion reduction and forming a protective coating that enhances nanoparticle stability and bioactivity. 31 The nanoparticles range in diameter from 70.70 ± 22 nm to 192.02 ± 53 nm and exhibit a crystalline structure. Studies have shown that the temperature and duration of synthesis directly influence the size of AgNPs. When tested for antibacterial activity against Pseudomonas aeruginosa and Staphylococcus epidermidis, these nanoparticles demonstrated a significant inhibitory effect. 28 No significant cytotoxic effects were observed when human peripheral blood mononuclear cells were used to assess biocompatibility. 29 However, it should be noted that the cytotoxic behavior of silver nanoparticles strongly depends on their size and concentration. Noticeable toxicity is observed only at doses above 25 µg mL⁻¹, whereas at lower concentrations the particles do not significantly affect cell viability. 32 Furthermore, due to the higher release of silver ions from their surface, smaller nanoparticles (around 10 nm) exhibit stronger cytotoxic effects than larger ones (100 nm). In general, the biological safety of silver nanoparticles is size- and dose-dependent, and their non-toxic performance is limited to specific conditions.32,33 The research highlights that A. vera phytocomponents, such as acemannan, play a crucial role in stabilizing and coating AgNPs, thereby enhancing their antibacterial properties.30,34 These findings suggest that AgNPs synthesized from A. vera could have valuable applications in biotechnology, medicine, and pharmaceuticals.
Aloe vera and H. sabdariffa are well-known for their broad spectrum of beneficial properties, which have been well-researched in the literature.35–38 Both plants can function as reducing and stabilizing agents in the sustainable production of silver nanoparticles because they contain a wide variety of bioactive compounds, such as phenolic acids, flavonoids, and anthocyanins in H. sabdariffa24–26 and acemannan, polysaccharides, and antioxidants in A. vera.28–30,39 Comparative studies assessing the physicochemical and biological properties of silver nanoparticles derived from these two plant sources are scarce, despite the widespread acknowledgment of their medicinal potential and individual applications in nanoparticle synthesis. Closing this gap may provide important information about how to optimally utilize green synthesis methods for industrial and biomedical uses.
Since green-synthesized silver nanoparticles possess antibacterial, antioxidant, and biocompatible properties, they are particularly attractive for biomedical, environmental applications40–43 and sensor application. Plant-based synthesis approaches represent a promising alternative to conventional chemical methods, with potential uses in wound healing, 40 antimicrobial coatings, 41 drug delivery systems, 42 food packaging, 41 and water purification. 43
This study aims to comparatively analyze the physicochemical characteristics and biological activity of silver nanoparticles synthesized using H. sabdariffa and A. vera extracts while developing a low-temperature method for their production. This approach contributes to the creation of an environmentally safe, energy-efficient, and sustainable synthesis process that minimizes by-product formation and broadens the potential applications of nanoparticles in medicine, biotechnology, and other fields.
Materials and methods
Apparatus and reagents
Magnetic stirrer with heating function, water bath and filter paper were used for the experiments. Reactants included silver nitrate (AgNO₃, ⩾99.0%, Sigma-Aldrich, USA), deionized water, dried H. sabdariffa petals and fresh A. vera leaves. The dried form of H. sabdariffa was selected because controlled drying preserves anthocyanins and phenolic compounds, ensuring higher stability and reproducibility of the extract composition, 44 while A. vera gel was used in its fresh form to avoid degradation of thermolabile components during dehydration. 45
Preparation of plant extracts
The extraction of plant components followed these steps: raw material preparation, solvent selection, solution preparation, cooling, and filtration. For dried components (H. Sabdariffa) 1 g of plant material was used, while for fresh components (A. vera) 10 g was taken. The material was ground to a uniform consistency and mixed with distilled water. The mixture was heated in a water bath at 80°C for 4 h with continuous stirring. After cooling to room temperature, it was filtered through filter paper to remove solid particles. The extracts were stored in sterile, airtight containers in a dark and cool place.
Green synthesis of silver nanoparticles
Silver nanoparticles (AgNPs) were synthesized by mixing an initial silver nitrate (AgNO₃) solution with plant extracts obtained through aqueous extraction from H. sabdariffa and A. vera. The component ratio (plant extract to AgNO₃ solution) was 1:49, with the process carried out under constant stirring at a temperature of 30 ± 5°C. A schematic representation of the extraction steps and AgNPs formation is shown in Figure 1.
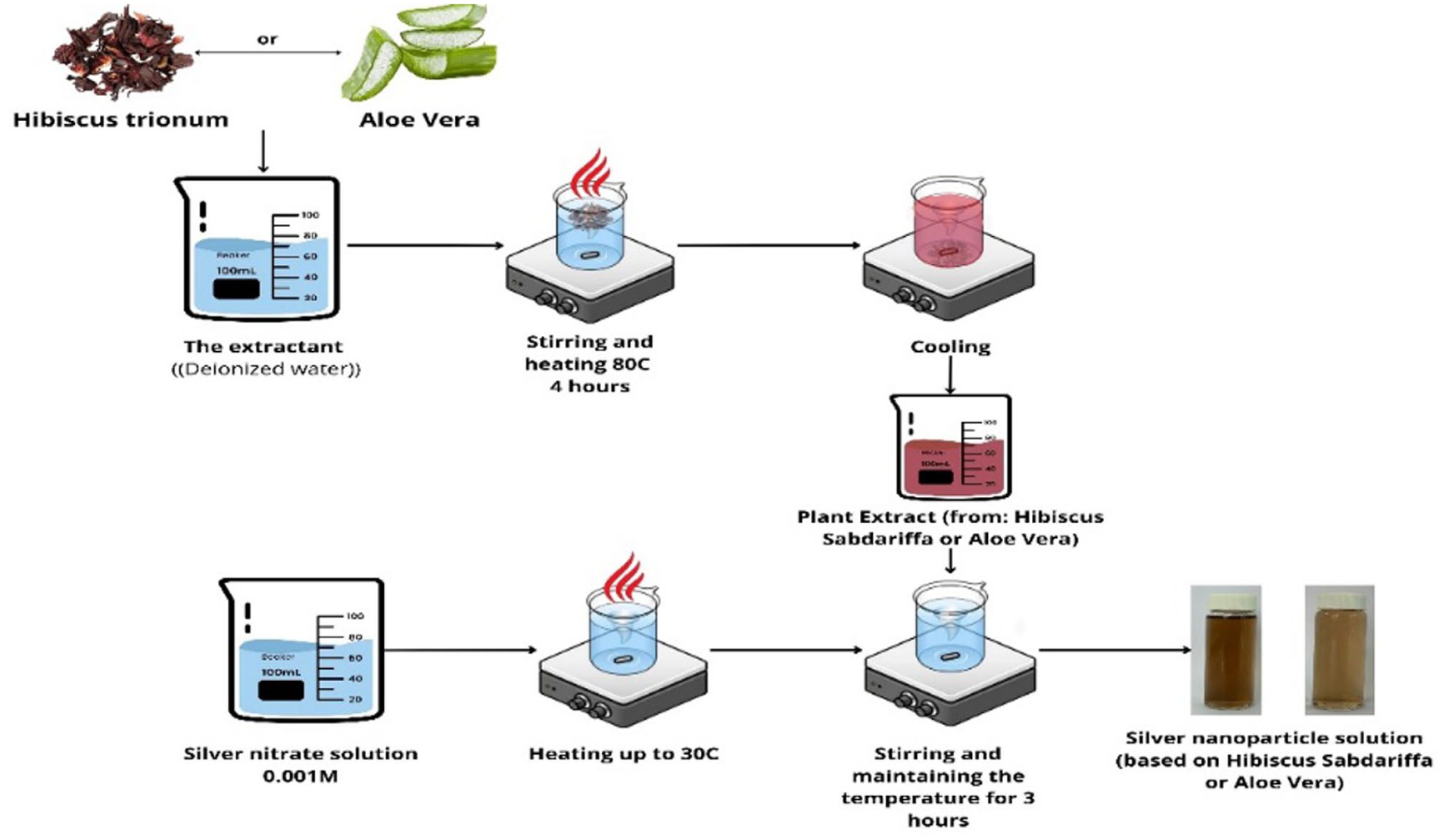
Step-by-step scheme of plant extract preparation and subsequent synthesis of AgNPs.
For the synthesis of AgNPs, an aqueous silver nitrate solution with a concentration of 0.001 M was prepared. The solution was transferred to a reaction vessel and heated to 30°C to enhance its reactivity. The plant extract (H. sabdariffa or A. vera) was then added, initiating nanoparticle synthesis. The process was monitored using UV-Vis spectroscopy by tracking color changes in the solution and the appearance of characteristic absorption peaks. After 3 h of reaction, nanoparticle size was measured using dynamic light scattering (DLS).
The synthesized AgNPs changed color within the first 60 min; however, the completion of the reduction reaction was observed after 3–24 h of incubation.
Characteristics of nanoparticles
UV-Vis spectroscopy
The synthesis of silver nanoparticles was confirmed using a UV-visible spectrophotometer (C-7000, Peak Instruments, USA) in the wavelength range of 300–800 nm, recording characteristic absorption peaks. The colloidal solution of silver nanoparticles was utilized for the analysis without any further preparation.
Scanning electron microscopy (SEM)
The structure of AgNPs were investigated by scanning electron microscopy. Imaging mode – 5 kW, scanning speed – 2, resolution – 1 to 100 μm with an Auriga Crossbeam 540 (Carl Zeiss, Oberkochen, Germany). The sample was prepared for analysis by centrifuging the colloidal solution containing silver nanoparticles for 20 min at 8000 rpm in order to separate the solid fraction. The precipitate that resulted was gathered, allowed to dry at room temperature, and then carefully crushed into a fine powder.
Particle size
The average particle size and zeta potential of AgNPs were determined using dynamic light scattering (DLS) with a Zetasizer NanoZS 90 (Malvern, UK) at 25 ± 2°C. The samples were analyzed immediately after synthesis and after 1, 7, 14, and 21 days of storage at room temperature to evaluate the effect of time on particle stability and size distribution. The silver nanoparticle colloidal solution was utilized directly for the analysis.
XRD analysis
The crystallinity of AgNP samples was studied using X-ray diffraction (XRD) on an X’PertPRO diffractometer (Malvern Panalytical Empyrean, Netherlands) with monochromatized copper (CuKα) radiation, a scanning step of 0.02°, and K-Alpha1 wavelength of 0.1542 Å. The measurement angle ranged from 10° to 45°, with a voltage of 40 kV, a current of 30 mA, and an X-ray tube voltage of 45 kV. The measurement time at each step was 0.5 s. A rectangular aluminum universal sample holder (PW1172/01) was used for reflection mode measurements. The sample was prepared for analysis by centrifuging the silver nanoparticles from the colloidal solution for 20 min at 8000 rpm, drying them, and then finely grinding them into a uniform powder.
Antibacterial analysis
The antibacterial activity was assessed using the agar well diffusion method. Gram-positive (Staphylococcus saprophyticus) and gram-negative (Escherichia coli) bacteria were used for the analysis. Wells with a diameter of 8 mm were punched (negative control – water). The wells were then filled with 30, 40, and 50 μL of the tested AgNPs and extracts, with a concentration of 5 mg/mL, respectively. All solutions were sterilized in ultraviolet light for 10 min before being placed in the wells. After incubation at 35°C for 24 h, the diameter of the inhibition zones was measured. For inoculation, 1 mL of a standardized microbial suspension was used, adjusted to 0.5 on the McFarland scale (≈1.5 × 108 CFU/mL; at 600 nm) of each microorganism under study, which was evenly distributed over the surface of the agarised nutrient medium. 46
Results and discussion
Absorption spectra of AgNPs by UV-vis spectroscopy
Key parameters, including reaction time, AgNO₃ concentration, and the silver nitrate to extract concentration ratio, were optimized for the synthesis of AgNPs. The formation of AgNPs was visually confirmed by a color change in the solution from pink to reddish-brown (Figure 1), indicating the reduction of silver ions (Ag⁺) to their metallic state (Ag⁰) and the subsequent formation of nanoparticles. Since the existence of a distinctive absorption peak in the 400–500 nm region suggests the presence of localized surface plasmon resonance typical for AgNPs, UV-visible spectroscopy was used to validate the production of silver nanoparticles. 46
The UV–V is absorption spectra of the synthesized AgNPs using the green method are presented in Figure 2. The appearance of a distinct surface plasmon resonance (SPR) band in the visible region confirmed the successful formation of metallic nanoparticles. 47 For H. sabdariffa AgNPs (Figure 2(a)), a dominant absorption peak was observed at ~455–460 nm, 48 which is characteristic of spherical AgNPs and indicates the reduction of Ag⁺ to Ag⁰. The gradual increase in peak intensity over time reflects a rising nanoparticle concentration, while the stability of the peak position suggests limited aggregation during the observation period. A secondary band around 340 nm is attributed to the presence of phytochemicals from the extract.49,50 In principle, two well-pronounced bands could be interpreted as evidence of transverse and longitudinal plasmon modes typical for rod-shaped or anisotropic nanoparticles, however, SEM analysis (Figure 4) revealed predominantly spherical particles, supporting the assignment of the main band to a single localized SPR. Similar conclusions regarding geometry – dependent plasmonic behavior of AgNPs have been reported in recent optical studies.51–54

UV spectra of AgNPs synthesized by the green method using plant extracts: (a) H. sabdariffa and (b) A. vera over time.
In contrast, the UV-Vis spectra of the reaction mixture containing A. vera extract (Figure 2(b)) showed a peak at 341 nm. This wavelength is well below the typical range for AgNPs, making it challenging to conclusively identify the nanoparticles. The peak at 341 nm could be attributed to the presence of smaller nanoparticles, organic compounds from the extract, or other optical phenomena. An increase in peak intensity over time suggests nanoparticle formation or aggregation; however, additional characterization is required to confirm the presence of AgNPs. Stability interpretation for A. vera is more complex due to the atypical absorption band at 341 nm.
Thus, a comparative analysis of the spectra demonstrates that the type of plant extract significantly influences the kinetics of silver nanoparticle formation and their stability.
Scanning electron microscopy (SEM)
Figure 3 shows SEM images of silver nanoparticles synthesized using H. sabdariffa (a) and A. vera (b) extracts. However, clear variations in particle aggregation and packing are visible. In the H. sabdariffa sample, the nanoparticles form a closely packed and relatively homogeneous layer, with smooth contours and a narrow size range of 12.21–19.67 nm. Aggregation is minimal, and individual particles are clearly discernible. In contrast, the A. vera sample, although displaying a comparable primary particle size range (13.07–19.25 nm), contains more irregular clusters with looser packing. Many particles appear to be partially fused or bridged, producing larger aggregate domains and less uniform interparticle spacing. These morphological differences suggest that the surface capping by H. sabdariffa phytochemicals maintains better particle separation, whereas A. vera components allow greater interparticle contact, facilitating aggregate growth.

SEM images of AgNPs obtained by reduction with plant extracts: (a) H. Sabdariffa and (b) A. vera.
Average hydrodynamic size of AgNPs
Dynamic light scattering (DLS) is a widely applied technique for nanoparticle characterization, enabling the determination of hydrodynamic diameter, size distribution, and colloidal stability by analyzing fluctuations in scattered light intensity caused by Brownian motion.55,56 In this study, DLS measurements confirmed the successful formation of AgNPs and revealed distinct size characteristics depending on the plant extract used. AgNPs synthesized with H. sabdariffa extract exhibited a narrow, monodisperse size distribution with an average hydrodynamic diameter of 52.5 nm, accounting for 99.5% of the particle population, indicating high stability immediately after synthesis. In contrast, AgNPs synthesized using A. vera extract showed a broader distribution from 58 to 164 nm, with the predominant peak at 91 nm (Figure 4), suggesting a higher degree of polydispersity. These initial size characteristics provided a baseline for assessing the stability and aggregation behavior of the nanoparticles during storage.

Particle size of AgNPs obtained by reduction with plant extracts: (a) H. Sabdariffa and (b) A. vera.
Analysis of the size stability dynamics of silver nanoparticles synthesized using H. sabdariffa extract demonstrates a high degree of stability and gradual reduction in particle size over time (Figure 5(a)). Initially, the nanoparticles range in size from 37 to 78 nm. Within 24 h after synthesis, the particle size decreases to 4.1–8.7 nm, with a distribution peak at 5.6 nm. This reduction continues until the 14th day, when particle sizes range from 0.7 to 4.1 nm, with a peak distribution at 0.7 nm. By the 21st day, an initiation of particle aggregation is observed, leading to an increase in size to a range of 2.6–68.7 nm, with a distribution peak at 2.6 nm (Figure 4(a), record 63). This indicates intense disaggregation and stabilization processes, making H. sabdariffa extract an effective stabilizer for silver nanoparticles. 57

Time-dependent size distribution of AgNPs synthesized using: (a) H. sabdariffa and (b) A. vera extracts at room temperature (25 ± 2°C) over 21 days, determined by DLS.
In contrast, nanoparticles synthesized using A. vera extract exhibit a tendency for gradual growth and aggregation (Figure 5(b)). Initially, their sizes range from 58 to 140 nm. Over time, the average size increases, and the distribution range expands. On the 21st day after synthesis, their sizes range from 105 to 615 nm, with a peak distribution at 190 nm, indicating significant particle enlargement during storage (Figure 5(b)). This process is attributed to the presence of polysaccharides, proteins, and phenols in A. vera extract, which play an antiseptic role and contribute to aggregation. 58
Different stability tendencies are shown by a direct comparison throughout the duration of the 21-day storage period. In addition to maintaining reduced initial dimensions (37–78 nm), nanoparticles made with H. sabdariffa extract gradually reduce in size to 0.7–4.1 nm by day 14, with only a slight aggregation by day 21 (2.6–68.7 nm, peak at 2.6 nm). The nanoparticles made from A. vera, on the other hand, begin larger (58–140 nm) and increase steadily, reaching 105–615 nm by day 21 with a wider spread and peak at 190 nm. These findings suggest that while A. vera encourages progressive particle growth and aggregation, H. sabdariffa guarantees greater long-term stability and a narrower size dispersion.
The results of silver nanoparticle synthesis using H. sabdariffa and A. vera extracts show significant differences in size, stability, and formation kinetics. This is consistent with studies indicating that the chemical composition of plant extracts plays a crucial role in the morphology and stabilization of silver nanoparticles. 9 The H. sabdariffa extract’s phytochemicals proved to be more effective reducing agents, ensuring the synthesis of smaller particles, which is consistent with studies highlighting the high activity of anthocyanins in reduction processes. 59 Its rich content of anthocyanins (e.g. cyanidin-3-sambubioside, delphinidin-3-sambubioside), 60 flavonoids (quercetin, luteolin) 61 and phenolic acids (gallic, chlorogenic acids) 61 enables the formation of a dense organic layer on the nanoparticle surface, providing both steric hindrance and electrostatic repulsion, thereby preventing aggregation and maintaining long-term colloidal stability. 62
In the case of A. vera extract, particle growth was observed during storage, which may be associated with the predominance of high-molecular-weight polysaccharides, primarily acemannan – a linked polymannose – along with glucose- and galactose-containing heteropolysaccharides, glycoproteins, and phenolic compounds.63,64 While these components are capable of reducing silver ions and forming a stabilizing coating, their extended polymer chains can adsorb simultaneously onto multiple nanoparticles, promoting interparticle bridging and resulting in aggregate formation during storage. 65 Similar results have been reported in studies highlighting the significant influence of biopolymers on the aggregation and long-term stability of silver nanoparticles. 66
Thus, nanoparticles synthesized using H. sabdariffa and A. vera extracts exhibit fundamentally different size evolution trends. H. sabdariffa extract promotes stabilization and disaggregation of nanoparticles, whereas A. vera extract leads to particle growth. This effect can be attributed to differences in the composition of bioactive compounds: flavonoids and phenolic compounds in H. sabdariffa act as antioxidants and stabilizers, preventing aggregation, while polysaccharides, proteins, and phenols in A. vera facilitate bridging between particles and accelerate aggregation.
XRD analysis
The X-ray diffraction (XRD) pattern of silver nanoparticles (AgNPs) synthesized using green methods with H. sabdariffa and A. vera extracts is shown in Figure 6. The crystalline structure of the synthesized AgNPs was relatively determined based on the conducted studies.67–69 By comparing the measured diffraction peaks with standard reference data from the Joint Committee on Powder Diffraction Standards (JCPDS), the crystalline structure of the produced AgNPs was determined. The (111), (200), (220), (311), and (222) planes of face-centered cubic metallic silver are represented by the typical reflections at 2θ = 38.12°, 44.30°, 64.46°, 77.41°, and 81.56° (JCPDS No. 01-071-3762). The presence of metallic and oxidized phases in this sample was confirmed by additional weak peaks matched with silver oxide card (AgO) (JCPDS No. 01-076-1489).
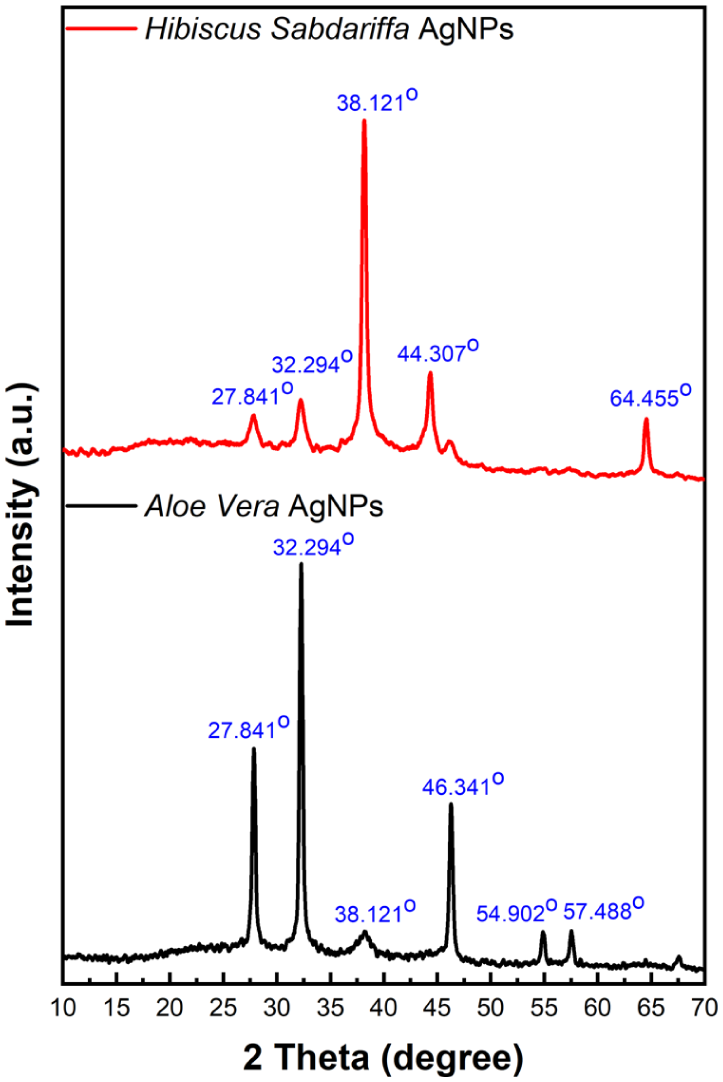
X-ray diffraction pattern of silver nanoparticles synthesized using H. sabdariffa and A. vera extracts.
The crystalline phase formation and structural characteristics of silver nanoparticles made with extracts from H. sabdariffa and A. vera differ significantly, according to a comparative XRD study. 70 With distinctive peaks at 38.121°, 44.307°, and 64.455° that correspond to the (111), (200), and (220) planes of face-centered cubic silver, H. Sabdariffa-mediated AgNPs demonstrated primarily pure metallic silver phases, demonstrating exceptional crystallinity and high phase purity.71–73 With additional peaks at 27.841°, 32.294°, 46.341°, 54.902°, and 57.488°, A. vera-synthesized AgNPs, on the other hand, showed a complex multi-phase structure that was attributed to the synthesis of silver chloride (AgCl) and silver oxide (AgO) phases in addition to metallic silver.74–76
This discrepancy implies that H. sabdariffa extract offers more selective reduction conditions that favor the crystallization of pure metallic silver, whereas A. vera extract contains chloride-containing compounds and a variety of phytochemicals that encourage mixed-phase development during synthesis. While the high phase purity of H. sabdariffa AgNPs suggests superior electrical and optical characteristics appropriate for particular technological applications, the presence of secondary phases in A. vera AgNPs, such as bio-organic complexes and inorganic silver compounds, may contribute to enhanced stability and multifunctional properties.
These results show that the choice of plant extracts essentially dictates the phase composition and functional characteristics of nanoparticles, offering essential information for the focused synthesis of silver nanoparticles with specific characteristics for various industrial and biological applications. In the context of silver nanoparticles, a crystalline structure is generally more desirable, as it ensures phase purity, higher structural stability, and consistent physicochemical properties.71,77
Antibacterial activity analysis
Antibacterial properties of A. vera and H. sabdariffa extracts, as well as their silver nanoparticle formulations, were determined against E. coli and S. saprophyticus using the agar well diffusion method. The mean inhibition zone diameters ± standard error of the mean (SEM; n = 3) are presented in Table 1.
Results of statistical processing of the determination of the zone of inhibition of antibacterial activity: mean values ± SEM* (n = 3), including a well diameter of 8 mm.

Inhibition zone ± Standard error of the mean, mm (amount of substance per well, μL).
A. vera extract produced inhibition zones of 8.0 ± 0.06 mm against both E. coli and S. saprophyticus, with no significant variation across tested volumes. H. sabdariffa extract showed a significant dose-dependent increase in activity: inhibition zones against E. coli increased from 10.0 ± 0.03 mm at 30 μL to 12.0 ± 0.09 mm at 50 μL. A similar trend was observed for S. saprophyticus, with inhibition zones increasing from 10.5 ± 0.17 mm to 12.5 ± 0.09 mm.
A. vera AgNPs exhibited slightly higher activity than the corresponding extract, with inhibition zones ranging from 8.5 ± 0.06 mm to 9.5 ± 0.25 mm. Hibiscus-derived AgNPs demonstrated a statistically significant enhancement compared to the crude extract, with inhibition zones of 14.0 ± 0.02 mm at 30 μL, increasing to 16.5 ± 0.07 mm at 50 μL.
Thus, H. sabdariffa formulations both extracts and AgNPs exhibited significantly greater antibacterial effects than A. vera formulations against both bacterial strains (p < 0.05). The results are consistent with the visual evaluation of inhibition zones shown in Figure 7.

Antibacterial activity evaluated by the agar well diffusion method against E. coli and Staphylococcus saprophyticus: (a) extracts of A. vera (Al-ex) and H. sabdariffa (Hb-ex) and (b) AgNPs synthesized from A. vera (Al-NP) and H. sabdariffa (Hb-NP).
Overall, the comparison indicates that H. sabdariffa provided more favorable synthesis conditions than A. vera. The higher content of anthocyanins, flavonoids, and phenolic acids in Hibiscus enabled rapid reduction of Ag⁺ ions, controlled nucleation, and effective capping, which resulted in smaller particle sizes, higher crystallinity, and long-term colloidal stability. In contrast, the predominance of polysaccharides and glycoproteins in A. vera extract promoted interparticle bridging and aggregation.
The water content in fresh plant materials amounts to 70% to 90% of their total weight depending on plant sections and environmental factors during growth. Scientists convert plant extracts into nanoparticles through a process which changes their chemical structure because of their high water content. When a fresh H. sabdariffa sample is used, much of the extractable volume consists of water along with dissolved minerals, sugars, and phytochemicals. The dried powder contains bioactive compounds in a concentrated state because it maintains essential phytochemicals which reduce metal ions after removing most free water.78–80
Conclusions
According to the comparative research, H. sabdariffa performs better than A. vera as a carrier of stabilizing and reducing agents for the environmentally friendly synthesis of silver nanoparticles. AgNPs made using H. sabdariffa extract showed a face-centered cubic crystalline structure verified by XRD, reduced particle sizes (12.21–19.67 nm by SEM), and long-term colloidal stability (4.1–8.7 nm after 24 h by DLS). These characteristics were attributed to the high concentration of phenolic acids, flavonoids, and anthocyanins, which provides strong capping effects, regulated nucleation, and rapid reduction. On the other hand, because polysaccharides and glycoproteins predominate, AgNPs produced from A. vera showed increased aggregation during storage, leading to larger particle sizes (up to 531.2 nm after 21 days) and a mixed-phase composition. Additionally, Hibiscus-derived nanoparticles’ improved physicochemical properties resulted in higher antibacterial efficacy, exhibiting noticeably bigger inhibition zones against S. saprophyticus and E. coli. Hibiscus-derived AgNPs’ outstanding crystallinity, stability, and bioactivity underscore their potential uses in food packaging, water purification, antimicrobial coatings, and wound healing. This emphasizes the significance of extract selection in tailoring nanoparticle properties for both industrial and biomedical applications. The comparative design of this study provides valuable insights into the influence of different plant matrices on nanoparticle formation and stabilization mechanisms, offering a scientific basis for selecting optimal biogenic sources in future research.
Footnotes
ORCID iDs
Author contributions
Conceptualization, Sana K. Kabdrakhmanova, Aigul Zh. Kerimkulova, and Asylkan Z. Dzhumanazarova; methodology, Dana Bolatkhan, and Esbol Shaimardan; software, Anastassiya D. Kukhareva, Kantay Nurgamit, and Madiar M. Beisebekov; validation, Sana K. Kabdrakhmanova, Dana Bolatkhan, and Asylkan Z. Dzhumanazarova; formal analysis, Esbol Shaimardan, Anastassiya D. Kukhareva, and Kantay Nurgamit; investigation, Aigul Zh. Kerimkulova, Daria Toktorbekkyzy, and Madiar M. Beisebekov; resources, Sana K. Kabdrakhmanova; data curation, Daria Toktorbekkyzy, Anastassiya D. Kukhareva, and Asylkan Z. Dzhumanazarova; writing—original draft preparation, Aigul Zh. Kerimkulova, Esbol Shaimardan, and Madiar M. Beisebekov; writing—review and editing, Sana K. Kabdrakhmanova, Kantay Nurgamit, and Asylkan Z. Dzhumanazarova; visualization, Daria Toktorbekkyzy, Dana Bolatkhan, and Anastassiya D. Kukhareva; supervision, Sana K. Kabdrakhmanova and Aigul Zh. Kerimkulova; project administration, Aigul Zh. Kerimkulova; funding acquisition, Aigul Zh. Kerimkulova.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been funded by the Science Committee of the Ministry of Science and Higher Education of the Republic of Kazakhstan (Grant No. AP19680576).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data presented in this study will be available on request from the corresponding author upon reasonable request.
