Abstract
Objective:
To determine and characterize the antitumor activity of a hydrogel loaded with octenidine hydrochloride (OCTH) and its action mechanism on human tumor cells.
Material and methods:
The antibiofilm property of OCTH against a clinical methicillin-resistant Staphylococcus aureus isolate (MRSA-CI) was evaluated by disk diffusion assays and a human bone-biofilm model in combination with the BacLight™ staining. The antitumor activity of soluble OCTH and OCTH-loaded biodegradable hydrogels was assessed in human tumor cell lines (MCF-7, CAL-27, FaDu, and A549) under different experimental conditions. Cell viability was measured with the MTT assay, and membrane permeability after OCTH-loaded hydrogel exposure was evaluated with the Calcein AM assay. Genotoxicity was evaluated with comet assay and fluorescence microscopy.
Results:
A stable, transparent, and biodegradable OCTH hydrogel with a pH of 7.2 was successfully developed. Biofilms exposed to 50 μm OCTH for 24 h exhibited only dead bacteria (red fluorescence), comparable to the effect of 100 μm vancomycin. Soluble OCTH demonstrated dose-dependent inhibition of tumor cell growth in all tested cell lines, with significant effects observed from 1 μm. The LD50 values for soluble OCTH across the tumor cell lines ranged from 2.32 to 5.1 µm. Notably, hydrogel loaded with 50 μm OCTH significantly reduced cell growth by 44% after a 15-min exposure and by 97% after 120 min. The Calcein AM assay revealed that OCTH’s antitumor mechanism involves the disruption of tumor cell plasma membrane permeability.
Conclusion:
This study is the first that describes the antitumor activity of octenidine hydrochloride on different human tumor cell lines. A biodegradable hydrogel loaded with octenidine hydrochloride constitutes an innovative, low-cost alternative for the topical treatment of cancer.
Keywords
Introduction
Cancer continues to be one of the most important challenges in modern medicine worldwide. 1 Among the most prevalent types of cancer are breast, cervicouterine, prostate, and colon cancers. 1 Oral cancer incidence ranks 16th with 389,485 new cases per year, whereas its mortality ranks 15th with 188,230 annual deaths worldwide. 1 Among the oral cancers, oral squamous cell carcinoma (OSCC) is most commonly diagnosed, followed by nasopharyngeal cancer. 2 The International Agency for Research on Cancer (IARC) reports that Mexico ranks 21st with incidence and mortality rates of 1.2 and 0.43 per 100,000, respectively. The occurrence of OSCC shows a male predominance, and is typically diagnosed in patients older than 60 years. 3 Histologically, OSCC presents as an ulcer with abnormal edges; initially asymptomatic but developing into a painful inflammation. 4
The first treatment option is surgery, followed by radiotherapy and/or intravenous chemotherapy. 5 Interestingly, there is no oral antitumor agent against OSCC. In order to relieve pain caused by oral mucositis in cancer patients an indomethacin mouthwash has been suggested. 6 It has also been suggested that polyphenols and chitosan could serve as active ingredients in anticancer mouthwash formulations.7–9
Surgical site infections (SSI) are a common complication in cancer patients following tumor removal surgery to remove tumor, contributing to increased morbidity rates. 10 Methicillin-resistant Staphylococcus aureus (MRSA) and enterobacteria are among the main pathogens responsible for these infections. 11
Octenidine hydrochloride (OCTH) is an antimicrobial antiseptic belonging to the bipyridine family. It is commonly used as an active ingredient in mouthwashes, 12 dentifrices, 13 and surface antiseptics. 14 In comparative studies, mouthwashes containing 0.1% octenidine showed greater disinfectant efficacy than those with 0.2% chlorhexidine after 3 months of use. 15 OCTH has demonstrated effectiveness in nonsurgical endodontic retreatment, yielding comparable results to those achieved with chlorhexidine or sodium hypochlorite. 16 OCTH has also been proposed for use in body surface decolonization to help reduce the risk of SSI and minimize wound-related complications. 17 Additionally, octenidine gargles have shown promising results in the treatment of acute pharyngitis, providing faster relief compared to saline solution. 18 However, as far as we know, there are no reports on the antitumor potential of OCTH. Here, we describe for the first time the development an OCTH-loaded biodegradable hydrogel and its antitumor and antibiofilm activities as an innovative alternative of low cost for topical treatment of cancer. The strategy of use of hydrogels to locally delivery antitumor drugs against cancer is an actual them of study. It has the advantage of delivering the antitumor drug in the place where it is needed, increase its concentration and reduce exposition to non-tumor cells. Among previous reports we can found Shabana et al. 19 developed a thermosensitive and biodegradable hydrogel for co-delivery of gemcitabine and paclitaxel, which demonstrated antitumoral effectiveness against tumor cells.
Materials and methods
OCTH preparation
OCTH (octenidol®; Schulke y Mayr GmbH, Norderstedt, Germany) was freshly prepared by dilution with sterile distilled water. Across all concentrations used, the solution maintained a pH of 7.1.
Synthesis and characterization of the OCTH hydrogel
The OCTH hydrogel was prepared by dissolving 0.5 g Carbopol (Sigma Aldrich; St. Louis, MO, USA) in 50 mL of sterile distilled water using vortex mixing at 70°C. The OCTH solution was then added, and the pH was adjusted to 7.0 using triethanolamine (Sigma Aldrich; St. Louis, MO, USA). Sterile distilled water was added to bring the final volume to 100 mL, resulting in a hydrogel solution with a final OCTH concentration of 4 µm. The pH and stability of the OCTH hydrogel were evaluated by measuring potential weight loss over time, and the formulation was freshly prepared before their use.
Drug release profile
OCTH release was quantified after different times employing a Float-A-Lyzer G2 Dialysis Device (MWCO: 500–1000 D; Spectra/Por, USA) and measured by UV-Vis spectrophotometry (295 nm; Optizen Pop: APEX CIENTIFIC, China). 5 mL of OCTH hydrogel were put on a Float-A-Lyzer G2 Dialysis Device. Following, the Dialysis Device plus OCTH hydrogel were introduced in a bottle with 300 mL of deionized water and maintained at 37°C with constant agitation. Samples were taken at different times to measure the quantity of OCTH delivered.
MRSA clinical isolate
A clinical isolate of MRSA was obtained from the Infectious Diseases Laboratory of the “Dr. José Eleuterio González” University Hospital, a tertiary teaching hospital in Monterrey, Mexico. Species identification was performed by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS, Microflex LT system, Bruker Daltonics, Bremen, Germany) 20 according to the manufacturer’s instructions. First, the isolated bacteria were cultured on blood agar plates and incubated for 24 h at 37°C. Then, a single colony was plated onto the wells of a 96-spot stainless steel target plate (Bruker Daltonics), where 1 µL of 70% formic acid and 1 µL of alpha-cyano-4-hydroxycinnamic acid matrix solution (Sigma-Aldrich, Toluca, Mexico) were added. The plate was then analyzed using Biotyper 3.0 software (Bruker Daltonics) to detect possible matches with the spectral profile database. Identification was performed at the genus (score 2000–2299) and species (2300–3000) levels.
Susceptibility test of clinical isolated MRSA
Antimicrobial susceptibility was determined by broth microdilution using the VITEK 2.0 (bioMérieux, Marcy-l’Étoile, France) 21 according to the manufacturer’s instructions. Bacteria were first incubated on trypticase soy agar plates at 35°C overnight, and a bacterial suspension was prepared in 0.45% NaCl adjusted to a McFarland standard of 0.5. This suspension was then introduced into the equipment, and AST-GP75 VITEK cards for Gram-positive species were subsequently inoculated. After incubation at 37°C, the minimum inhibitory concentration (MIC) was obtained, and isolates were classified as susceptible, intermediate, or resistant according to the current Clinical and Laboratory Standards Institute (CLSI) breakpoint criteria (CLSI, 2023).
Bacterial culture
The MRSA clinical isolate (MRSA-CI) was grown on trypticase soy broth agar (BD DIFCO, Sparks, MD, USA) at 37°C for 24 h under aerobic conditions.
Bactericidal activity of OCTH against MRSA-CI
The bactericidal activity of OCTH was analyzed based on the methodology described by Tian et al. 22 A disk diffusion assay was conducted using MRSA-CI cultures. Briefly, 100 µL of a bacterial suspension (0.5 MacFarland) was grown onto Petri dishes containing trypticase soy broth agar (37°C; O2+; 18 h). OCTH was tested at a concentration of 4 μm. Vancomycin (100 μm vancomycin; Sigma Aldrich; St. Louis, MO, USA) served as the positive control of inhibition, while sterile double-distilled water served as the negative control. The experiment was performed in triplicate.
Antibiofilm test of OCTH
Based on the protocol described above, the antibiofilm activity was evaluated. A 12-h MRSA-CI biofilm, formed either in a 96-well plate or on a sterile human bone (BHT bone and tissue bank; Medicine faculty, Autonomous University of Nuevo Leon, UANL), was exposed to 4 μm OCTH for 24 h at 37°C. As positive control of the inhibition, 100 μm of vancomycin was used. After exposure, the remaining MRSA-CI biofilm was washed three times with 10 mm phosphate-buffered saline, pH 7.3 (PBS) and stained with the BacLight™ Live/Dead Viability Kit (Thermo Fisher SCIENTIFIC; Waltham, MA, USA) 23 : Each well received 100 μL of staining solution containing 6 μm SYTO 9 and 30 μm propidium iodide, followed by incubation at room temperature for 30 min in the dark. After a final wash with sterile distilled water, biofilms were observed using an EVOS Cell Imaging System (Thermo Fisher SCIENTIFIC; Waltham, MA, USA) equipped with FITC and rhodamine filters.
Cell culture and drug exposure
Human OSCC cell lines CAL-27 and FaDu, breast cancer cells (MCF-7), and a human cell line derived from a colon adenocarcinoma Caco-2 (HTB-37) were obtained from the American Type Culture Collection (ATCC; CRL-2095 and HTB-43; Rockville, MD, USA). Human fibroblasts (HFs; a kind donation of Dr. Higinio Arzate, Dentistry Faculty, UNAM) were employed as non-tumor control cells. Tumor and non-tumor cells were cultured in Dulbecco’s Modified Eagle’s Medium/Ham’s F12 (DMEM/F12) supplemented with 10% fetal bovine serum and antibiotics at 37ºC with 5% CO2 as previously reported. 24
CAL-27, FaDu, and HFs were seeded in 96-well plates (1 × 105 cells in 100 µL DMEM/F12 per well) and incubated at 37ºC with 5% CO2 for 24 h. After washing with PBS, OCTH (final concentration range, 0.1–12 µm; concentrations as indicated in results) was added employing FBS and antibiotics-free cell culture medium. Drug-free culture was used as a growth control, while 500 µm doxorubicin (DOX; Doxolem, Teva Lab, Madrid, Spain) served as a cytotoxicity control. Incubations at 37ºC, 5% CO2 lasted for 0.5, 1, 2, 4, 6, or 24 h and were stopped by washing with cold PBS.
MTT and median lethal dose (LD50) assays
To evaluate the antitumor effect of OCTH and its potential cytotoxicity toward HFs, the MTT cell viability assay25–27 (Biotium, Hayward, CA, USA) was employed. Following drug exposure, MTT was added to each well and incubated for 2 h at 37ºC and 5% CO2 according to the supplier’s instructions. Formazan crystals were then solubilized with dimethyl sulfoxide, and absorbance was measured at 570 nm using a 96-well microplate reader (BioTek, Winooski, VT, USA). Experiments were done in triplicate, and results were expressed as percentages of viable cells.
The selectivity index (SI) of OCTH was calculated using the formula: SI = IC50 (non-tumor cells)/IC50 (tumor cells), 28 where IC50 represents the drug concentration required to inhibit 50% of cell viability.
The lethal dose 50 (LD50) of OCTH was determined using the same assay on CAL-27 and FaDu cells, employing OCTH final concentrations ranging from 0.1 to 12 µm.29,30
Cell permeability (Calcein AM) assay
To verify whether altered cell membrane permeability could be a cytotoxic mechanism of OCTH, the Calcein AM assay was performed in conjunction with fluorescence microscopy. CAL-27 and FaDu cells were exposed to12 µm OCTH for 30 min, 2 h, and 24 h. After exposure, cells were incubated with 2 µm Calcein AM (Biotium, Hayward, CA, USA) for 30 min. at 37ºC, 5% CO2. After incubation, excess dye was removed, cells were washed with PBS and air-dried in the dark. Cells were analyzed with the EVOS Cell Imaging System (Thermo Fisher Scientific, CA, USA) using a FITC filter. Fluorescence intensity was quantified by using the software ImageJ.
Genotoxicity of OCTH by comet assay and fluorescent microscopy
The genotoxic potential of OCTH was evaluated in FaDu cells using the comet assay (Cell Biolabs, INC, San Diego, CA, USA), following the supplier’s instructions. Briefly, after drug exposure (6 μm OCTH), cells were detached, washed with sterile double-distilled water, and incubated with cold 70% acetone for 5 min. Genomic DNA was stained with 4′,6-diaminidino-2-phenylindole (DAPI; 1 µL/mL; 100 µL/well). DNA damage was assessed by fluorescence microscopy using the EVOS system (Thermo Fisher Scientific, CA, USA) and the DAPI filter.
Statistical analysis
Linear regression analysis was used to evaluate the effects of concentration gradients and exposure times. One-way ANOVA with Tukey’s correction was used to compare between groups. A significance level of α = 0.05 was considered for all statistical tests.
Results
Characterization of OCTH hydrogel
The carbopol-based hydrogel synthesized with the active ingredient presented a colorless, transparent, appearance with sufficient fluidity to be applied with a 3 mL syringe (Figure 1). Over time, the weight and pH of 7.2 of the OCTH hydrogel were stable. It was stored at room temperature until use.

Transparency and viscosity of biodegradable hydrogel loaded with 4 µm OCTH.
Drug release profile
When OCTH release was analyzed, the obtained results displayed that after 3 h of incubation of the hydrogel (37°C) 50% of OCTH was delivered (Figure 2). These results corroborate that the use of hydrogels as vehicle to specifically delivery antitumor drugs in the target site where is need it to obtain a therapeutic benefit.

Drug release profile of OCTH hydrogel was done employing a Float A Lyzer G2 dialysis device and UV-Vis spectrophotometry.
Characterization of MRSA-CI
The MRSA-CI strain was the S. aureus isolate 23-2672 obtained from a breast lesion in a patient in the Internal Medicine ward during 2023 (Figure 3). When analyzed for possible resistance, it only showed susceptibility to oxacillin, that is, methicillin-resistant and susceptibility to ciprofloxacin, levofloxacin, moxifloxacin, clindamycin, daptomycin, erythromycin, tetracycline, trimethoprim, and linezolid.

MALDI-TOF mass spectrum of MRSA-CI after flexAnalysis (Brucker Daltonics).
Antibiofilm activity of OCTH solution and hydrogel against MRSA-CI
Although the antibiofilm activity of OCTH is well known, it had not yet been evaluated in an MRSA-CI biofilm model on human bone. To this end, a 12-h MRSA-CI biofilm was established on a sterile human bone monolayer at 37°C under aerobic conditions. After removing the culture medium, the biofilms were exposed to 24 h at 37ºC to either the OCTH hydrogel or 100 µm vancomycin, used as a positive inhibition control. Drug-free culture medium served as a growth control. Figure 4 shows the antibiofilm activity of the OCTH hydrogel, with only a red fluorescence (dead bacteria) observed in biofilms exposed to OCTH and the inhibition control vancomycin, unlike the green fluorescence (live bacteria) in the growth control. These findings show the effectiveness of the OCTH hydrogel against MRSA biofilms on human bone, supporting its clinical potential.

MRSA biofilm on human bone and efficacy of antibiofilm compounds. MRSA biofilm after a 24-h exposure to drug-free medium (MRSA-CI), OCTH (4 μm OCHT-hydrogel) and 100 μm vancomycin (VAN). Bar, 50 μm.
Antitumor activity of soluble OCTH
The potential anticancer effect of soluble OCTH was studied on two human OSCC cells: CAL-27 and FaDu, and two other cancer cell lines: MCF-7 and Caco-2 using the OCTH concentration range 0.1–12 µm. A dose-dependent decrease in tumor growth was observed as of 0.1 µm OCTH with a maximum growth inhibition of 96% at 12 µm OCTH (p < 0.0001) (Figure 5). The effectiveness of OCTH was 41 times greater than the one obtained with the positive inhibition control, doxorubicin (Figure 5).

Antitumor activity of OCTH on different human cancer cell lines. The MTT cell viability assay was used to evaluate the anticancer properties of OCTH on oral cancer cells. 1 × 105 CAL-27, FaDu, MCF-7, and Caco-2 cells were exposed (37°C; 5% CO2; 24 h) to a range of OCTH concentrations (0.1–12 µm); 500 µm doxorubicin (DOX) was used as a positive inhibition control, while drug-free medium served as a growth control. The antitumor effect of OCTH is expressed as percentage of cell viability.
The LD50 of soluble OCTH on the different tumor cell lines ranged from 2.32 to 5.1 µm (Table 1). The SI of OCTH, determined in vitro with FaDu and HFs cells, was 2.84. This value SI indicates that OCTH is selective for tumor cells in comparison to healthy cells. Together, these data demonstrate the reproducibility of the antitumor activity of soluble OCTH on different types of human cancer cells with the same dose-dependent pattern.
LD50 of OCTH on different human cancer cell lines.

Antitumor effect of OCTH hydrogel at short exposure times
Once identified the antitumor property of soluble OCTH we develop a hydrogel loaded with 4 µm OCTH. Their efficacy was evaluated following short-term exposure, ranging from 5 min to 2 h, to FaDu tumor cells under standard conditions (37°C; 5% CO2). Cell viability was measured using the MTT assay and expressed as a percentage relative to untreated controls. A time-dependent reduction in cell viability was observed, beginning with a 31% reduction at 5 min post-exposure (Figure 6), reaching 50% reduction at 18 min, and culminating in a 97% reduction at 120 min (Figure 6). These findings confirm previous reports on the potent antitumor activity of octenidine as the active ingredient of a biodegradable hydrogel.

Antitumor effect of OCTH at short exposure times. FaDu tumor cells were exposed to the OCTH-loaded hydrogel (37°C; 5% CO2, O+) for 5, 15, 30, 60, and 120 min. Cell viability was assessed by the MTT assay, and the results were expressed as percentages based on the growth control in drug-free medium alone (0 min).
Effect of OCTH on cell permeability of FaDu cells
To study the potential mechanism of action of OCTH, plasma membrane permeability in FaDu cells was assessed using the Calcein AM assay combined with fluorescence microscopy. Following exposure to OCTH-hydrogel for 2 and 24 h, notable morphological changes were observed. The cells became rounded and released fluorescent Calcein AM, indicating loss of plasma membrane integrity (Figure 7(a)). This effect was more pronounced after 24 h exposure to OCTH-hydrogel, at which point only cellular debris remained. These findings are consistent with the results of the MTT cell viability assay, where 95% cell death was observed at 12 µm OCTH. Figure 7(b) shows the fluorescence intensity measured as of images displayed in Figure 7(a). In all, the cytotoxic effect may be due to membrane disruption.

Membrane permeability of FaDu cells after exposure to OCTH-hydrogel: (a) the membrane integrity of oral tumor FaDu cells was assessed using the Calcein AM assay combined with fluorescence microscopy. Calcein AM is a cell-permeant dye that only fluoresces when retained within intact cells. A time-dependent loss of fluorescence following OCTH-hydrogel exposure suggests decreasing plasma membrane integrity over time (bar, 75 µm) and (b) fluorescence intensity was quantified employing the software ImageJ.
Genotoxic assay of OCTH on FaDu cells
Potential genomic DNA damage in FaDu tumor cells was analyzed using the Comet assay and fluorescence microscopy. A 0.1% chlorhexidine solution served as a positive control for DNA break induction in tumor cells. In contrast to chlorhexidine-treated cells, which displayed characteristic “comet” tails indicative of genomic damage, cells exposed to OCTH-hydrogel showed no such DNA fragmentation (Figure 8). These data indicate that genomic damage is not involved in the mechanism of action of OCTH.

Genotoxicity of OCTH on FaDu cells. Calcein AM assay and fluorescence microscopy. The comet assay was used to determine the potential genotoxic effect of the OCTH on FaDu tumor cells; 0.1% chlorhexidine served as a positive genotoxicity control (CTRL); drug-free medium as a growth control (FaDu). Bar, 50 µm.
Cytotoxicity of OCTH on human fibroblast
Once the antitumor effectiveness of OCTH hydrogel had been demonstrated, its potential cytotoxicity toward normal human cells was assessed using a primary culture of human fibroblasts. After 24-h exposure to OCTH hydrogel, fibroblasts maintained 83% viability (Figure 9), indicating minimal toxic effects. In contrast, the positive control of tumor inhibition, doxorubicin (DOX), significantly reduced cell viability by 77%, consistent with its well-documented high cytotoxicity (Figure 9). These results are promising for the clinical use of the OCTH hydrogel as a selective and safe topical cancer treatment without undesirable side effects on healthy tissues.
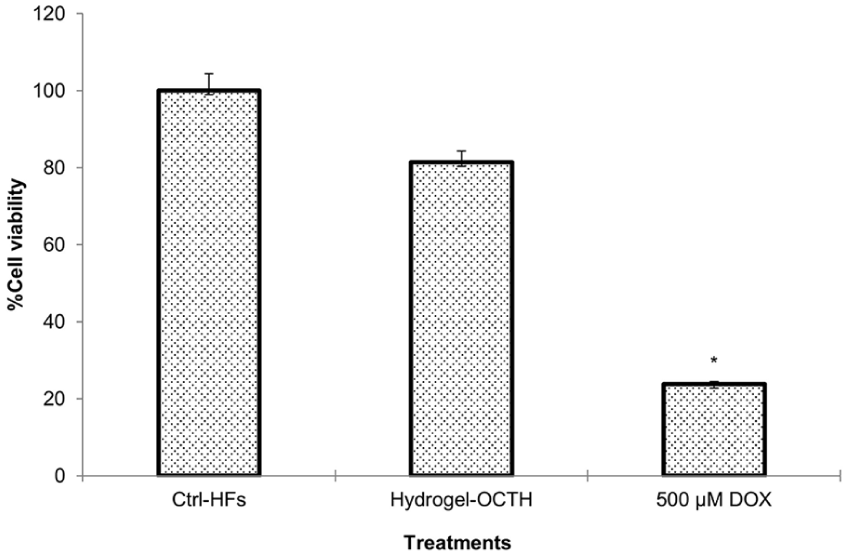
Cytotoxicity of OCTH on human fibroblasts (HFs). The MTT cell viability assay was used to evaluate the cytotoxic properties of OCTH on HFs. HF cells (1 × 105) were exposed (37°C; 5% CO2; 24 h) to the hydrogel containing 4 μm OCT. HF in drug-free medium were used as growth controls. The cytotoxic effect of OCTH is expressed as percentage of cell viability.
Discussion
OCTH has a broad range of applications in infection prevention, including use on hands, skin, wound, mucous membranes, and surfaces of equipment and instruments. It also serves as an active ingredient in mouthwash and as disinfectant in clean rooms, as well as industrial and medical settings. The bactericidal and antibiofilm activities of OCTH31–34 are well documented in the literature, but now we report a novel biological property that makes OCTH an even more versatile and promising molecule. Here, we describe for the first time the antitumor properties of OCTH against various human cancer cell lines. Furthermore, we extend the well-known antibiofilm activity of OCTH,35–37 to a bone model biofilm employing a clinical MRSA isolate (MRSA-CI) to assess OCTH efficacy in a clinically relevant context. The results from the BacLight™ Live/Dead assay corroborate the antibiofilm potential of OCTH against MRSA-CI, which only stained red after treatment with OCTH, very similar to vancomycin exposure. The same compound was also evaluated for its antitumor activity across several human tumor cell lines. Our findings show that OCTH inhibited tumor cell growth in a dose-dependent manner. Previously, our group reported the antitumor properties of chlorhexidine, another cationic disinfectant and widely applied ingredient of oral mouthwash. 38 Interestingly, chlorhexidine also exhibited a dose-dependent growth inhibition of tumor cell lines. OCTH and chlorhexidine share a cationic nature which likely contributes to their affinity for bacteria and cancer cells. Remarkably, a 2-h exposure to OCTH-hydrogel, led to a 97% reduction in FaDu, which suggests a rapid inhibitory action mechanism. Early have been described the efficacy of using hydrogels for topical delivery of 5-fluorouracil against human epidermoid carcinoma. 39 In this study, 50% of OCTH was released after 3 h of incubation at 37°C, suggesting the employment of hydrogels as a good vehicle to topical delivery. Also, Pazyar et al., 40 demonstrated the potential of topical 5-fluorouracil as an innovative strategy for vitiligo.
When the cell permeability of FaDu cells was assessed with the Calcein AM assay, damage of the FaDu cell membrane was evident after only 2 h of exposure to OCTH. This timeline is consistent with the inhibition of growth. This profile of OCTH is similar to the one of chlorhexidine as described previously by our team in 2019. 38 Recently, the antitumor activity of chlorhexidine against HCT-116 colorectal cancer cells in a xenograft model are consisted with our findings. 41
Comet studies on FaDu cells after OCTH exposure yielded no evidence of genotoxicity. We hypothesize that OCTH exerts its tumor toxicity so fast (less than 2 h) that there is not time for the compound to be internalized and interact with intranuclear molecules like DNA. The similar compound chlorhexidine has been shown to induce apoptosis though the caspase pathway. 41 To evaluate the cytotoxicity of OCTH was determined on non-cancerous cells, human fibroblasts were exposed to 4 μm OCTH for 24 h (37°C, 5% CO2, O + ). Under these conditions, cell viability remained high at 83%. This datum is particularly noteworthy, because the same concentration (4.8 µm or lower) represents the OCTH LD50 for various human tumor cell lines. In contrast, several reports have described the toxicity of chlorhexidine on human fibroblasts,42–44 including to tendon leading to decrease tenocyte viability. 45 Unfortunately, chlorhexidine’s cytotoxicity has limited its potential for broader clinical application. OCTH, on the other hand, shows promise as a topical treatment after tumor resection and surgery, potentially helping to prevent infection and cancer recurrence.46,47 Currently, no topical treatment exists to avoid cancer recurrency. OCTH represents an innovative, low-cost, and potentially effective option for applications into medical gels, solutions, and membranes.
In summary, these results support our hypothesis that OCTH has a strong antitumor activity against various human tumor cells with consistent effectiveness. This report constitutes the first evidence of the antitumor activity of OCTH, including its action mechanism which targets the tumor cell’s membrane selectively. Further studies should analyze its biocompatibility in animal models. Our results support the potential of OCTH for future clinical application in cancer treatment.
Conclusions
In this study, we describe for the first time the development of a biodegradable hydrogel loaded with octenidine with antitumor and antibiofilm properties, which constitutes an innovative, low-cost alternative for the topical treatment of cancer without undesirable side effects.
Footnotes
Acknowledgements
The authors thank the Sectorial Fund for Education Research grant of the CONACyT. Also, authors want to thank Nayely Pineda-Aguilar from Advanced Materials Research Center, CIMAV Unidad Monterrey, Nuevo Leon, Mexico for their support in drug release assays.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings in this study are available within the article.
