Abstract
Growing ecological and public health issues brought on by the increasing presence of novel organic contaminants in wastewater need the development of innovative remediation solutions. It’s usually challenging for conventional treatment methods to effectively collect these contaminants, which include pharmaceuticals, personal care products, and industrial chemicals. Scientists are, therefore, concentrating on innovative material to increase the efficiency of adsorption and removal. Because they facilitate interaction with a range of organic pollutants, 2D MXenes’ unique structural and chemical properties have drawn interest from these materials. MXenes are very excellent adsorbents for a variety of contaminants because of their large surface area, many terminal groups, and distinctive 2D layer architectures. Polyfluoroalkyl substances (PFAS), dyes, antibiotics (tetracycline, sulfonamides, and ciprofloxacin), amitriptyline, verapamil, carbamazepine, 17α-ethinyl estradiol, antibiotic resistance genes (ARGs), diclofenac, ibuprofen heavy metals, and other contaminants have all been claimed to be eliminated by MXenes. Recent studies propose the formulation of MXene-based biocomposites, which not only harness the high surface area and electrical conductivity of MXenes but also integrate biodegradable components to promote eco-friendliness. This work explores the potential of novel 2D MXenes biocomposites in addressing the critical challenge of wastewater treatment, focussing on their efficiency, and sustainability in removing emerging contaminants.
Introduction
Accelerated industrial progress, technological innovations, and economic expansion have led to worldwide environmental contamination, excessive depletion of raw material resources, incorrect release of dangerous pollutants into terrestrial, atmospheric, and aquatic systems, with discharge quantities continuing to rise. 1 Water degradation is among the most significant environmental issues of the 21st century, endangering ecosystems, human health, and sustainable development. The causes, effects, and possible remedies for the water contamination problem are examined in this paper. Water pollution encompasses common contaminants such as fluorides, nitrates, heavy metals, radioactive elements, insecticides, pesticides, and hazardous chemicals, along with the degradation of the quality of a stream, river, lake, ocean, aquifer, or other water bodies due to the presence of deleterious substances, typically chemicals or microorganisms. Steroids, hormones, medications, personal care items, endocrine disruptive substances, artificial sweeteners, surfactants, and more are examples of emerging water contaminants. In addition to potentially having unanticipated synergistic impacts on human and environmental health, this combination of water pollutants might cause serious health issues.
Water contamination is a widespread problem that jeopardises human health. Annually, contaminated water causes more fatalities than war and all other forms of violence combined. Our reserves of potable water are constrained; however, we can access fewer than 1% of Earth’s freshwater. If no action is taken, the issues will exacerbate by 2050, when global freshwater demand is anticipated to increase by one-third compared to current levels.2 –6
Furthermore, according to the research UN Water Summary Progress Update 2021: SDG 6, 7 more than two billion individuals reside in water-stressed nations and are compelled to consume contaminated water, including faecal matter, a predicament anticipated to worsen in certain regions due to climate change. Diarrhoea is estimated to cause one million deaths annually. More than 251.4 million cases of schistosomiasis, a serious and chronic illness brought on by parasites acquired through contact with rodent-contaminated water, were reported in 2021. Providing enough drinkable water has become a major problem for humanity, and the only way to meet the growing demand and depleting supply of clean water is to repurpose tainted water, which necessitates improvements in wastewater remediation methods. Conversely, it is anticipated that almost one third of the world’s nations may encounter significant water shortages by 2040 (Figure 1). 8

Forecasts of the water shortage rankings of nations for the year 2040, as per the Water Resource Institute. 9
According to the UN, enhancing the socioeconomic status, health, and general well-being of people in developing nations requires access to safe and clean water. Humans rely on surface and groundwater for their everyday requirements. To reach SDG 6, they go through a number of processes to ensure consumers receive clean water.
Solutions to the water pollution crisis:
i. Promoting the adoption of environmentally friendly agricultural methods and enforcing stringent rules on industrial discharge are essential. In the United States, laws like the Clean Water Act have been effective in lowering pollution levels. 10
ii. Wastewater treatment technologies: Reverse osmosis, bioremediation, and advanced oxidation techniques can all greatly enhance the quality of water.
iii. Emerging materials: According to Ihsanullah, new materials such as 2D MXenes are demonstrating potential in improving water purification systems and adsorbing pollutants. 11
iv. Behaviour changes can be sparked by increasing awareness of pollution prevention and water conservation. In nations like Kenya and India, community-led projects and grassroots movements have shown promise. 12
v. To tackle this global crisis, international collaborations such as the Sustainable Development Goals (SDGs) emphasise fresh water and hygiene (Goal 6), aim to consolidate resources and expertise. 13
The growing global population demands extensive freshwater usage across agricultural, industrial, and domestic sectors, leading to significant contamination of surface and groundwater due to human activities such as pesticide overuse, improper waste disposal, and industrial effluent discharge. These activities introduce emerging organic contaminants (EOCs), newly detected organic pollutants, and their derivatives into aquatic environments. Emerging organic contaminants bioaccumulate in aquatic environments, negatively impacting both aquatic creatures and humans via the food chain.
Groundwater, the primary freshwater source for billions, is vital for sustaining perennial rivers and ecosystems. However, it is susceptible to EOC pollution due to its interaction with surface water. Conventional wastewater treatment plants (WWTPs) are inadequate in removing EOCs like carbamazepine, resulting in downstream contamination. Advanced monitoring and management are necessary for groundwater, as its contamination can have long-term ecological and health impacts.
Pesticides, medications, surfactants, food, personal care items, and industrial additives are examples of organic compounds derived from humans that are used on a daily basis in a variety of industries around the world. These substances are categorised as emerging organic contaminants (EOCs; Figure 2), when they are present in the environment in undesired or harmful concentrations. 14 As research has advanced, several emerging organic contaminants (EOCs) have been identified in various sources of potable water in time. The situation is alarming because of the undetermined limits of their presence in water sources and the frequent difficulty in assessing their toxicological consequences.

The detrimental effects of EOCs on soil ecosystems, water systems, air quality, and human health are interrelated. (Reproduced with copyright permission.) 15
The advancements in nanotechnology over the past few decades have been beneficial to numerous sectors, including biological research, food science, agriculture, medical studies, cleaning up the environment, and countless more. Developments in nanotechnology have expedited use of nanomaterials to purify water due to their remarkable physicochemical features, which include elevated absorbances, porosity, and a large surface-to-volume ratio. Nanomaterials are used in water filter membranes to effectively and economically treat contaminated water. This raises the possibility of employing nanomaterials to remove pollutants and aid in wastewater reprocessing. 16 Nanomaterials are frequently employed as novel adsorbents. Examples include carbon nanotubes, magnetic nanoparticles, materials that are nanometre sized, and mesoporous materials. 17
The possible applications of MXenes in several spheres have been investigated exhaustively. MXenes’ remarkable adsorption capacity and ability to be functionalised for specific pollutants have made them very promising in the wastewater treatment sector in terms of the removal of EOCs. Their hydrophilic character helps them to strengthen their interaction with water molecules and organic pollutants, so they perform effectively as adsorbents in wastewater purification processes.
Even though MXenes alone have amazing qualities, their effectiveness in water remediation applications can be further improved by creating biocomposite materials that combine MXenes with biopolymers or other natural materials. Biocomposites combine the benefits of both sustainability and biocompatibility of biopolymers with the structural stability and adsorption power of MXenes.
When paired with MXenes, biopolymers including cellulose, alginate, and chitosan provide a number of advantages. While adding more functional groups that strengthen adsorption contacts with EOCs, they can also increase the composite material’s mechanical qualities. Additionally, because biocomposites are less hazardous and biodegradable than synthetic materials, they can help alleviate some environmental issues.
This review emphasises contemporary trends and future opportunities regarding the utilisation of MXenes and MXene-based materials for the elimination of hazardous pollutants. It addresses recent developments and impending challenges associated with these appealing two-dimensional materials and their potential applications in the removal of hazardous organic substances, such as pesticides, pharmaceutical compounds, etc., from wastewater.
MXenes
MXenes’ composition and structure
Numerous two-dimensional (2-D) materials have drawn a great deal of curiosity from the scholarly community since the discovery of 2-D graphene in 2004 because of its amazing features. Researchers at Drexel University identified a unique kind of 2D transition metal carbides, nitrides, and carbonitrides in 2011 that they called MXenes (pronounced “maxines”). From the moment they were found, MXenes have fascinated scientists and academics with their myriad fascinating mechanical, magnetic, electrical, and chemical properties. The general formula for MXene nanomaterials is Mn + 1 X n T x (n = 1–3), where M is an early transition metal (e.g. tungsten (W), molybdenum (Mo), chromium (Cr), tantalum (Ta), vanadium (V), niobium (Nb), hafnium (Hf), zirconium (Zr), titanium (Ti), yttrium (Y), or scandium (Sc)); X is carbon and/or nitrogen; T is the termination group on the surface (e.g. fluorine (–F), oxygen (=O), chlorine (–Cl), and hydroxyl (–OH)); and x is the number of surface functionalities (Figure 3). The value of n in MXenes (Mn + 1 X n T x ) determines the thickness of MXenes, which is typically in the range of 1 nm. In accordance with the literature,18 –22 MXenes are primarily produced by selectively etching the A layers from the precursor MAX phase (Mn + 1 A X n ), where n equals 1, 2, or 3, and A generally refers to any element from groups 12 to 16 (e.g. cadmium (Cd), aluminium (Al), silicon (Si), phosphorus (P), sulphur (S), gallium (G), germanium (Ge), arsenic (As), indium (In), tin (Sn), thallium (Tl), lead (Pb)), where X stands for C and/or N. A layer with a strong M single bond X bond and a somewhat weak M–A bond is typically encased in an octahedral Mn + 1 X n in the MAX phase. Hydrofluoric acid (HF) was used to specifically etch the Al atoms in layered hexagonal ternary carbide, Ti3AlC2, at room temperature to create the first known MXene, which was made of 2-D titanium carbide (Ti3C2).
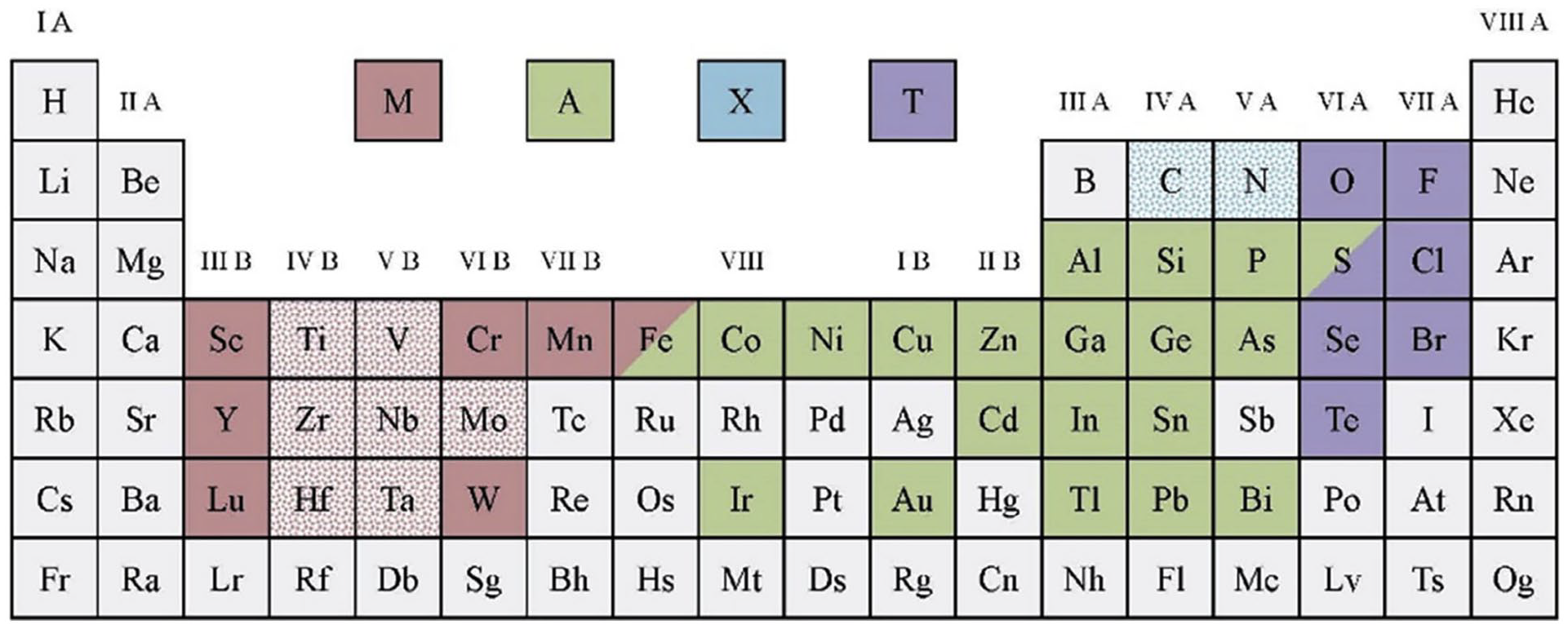
Systematic diagram represents the constituent elements of MAX phases and MXenes within the periodic table. The pink, green, and blue hues represent the M, A, and X elements in synthesised MAX phases, respectively. The pink and green hues with a shaded backdrop represent the M and X elements in synthesised MXenes, respectively. The purple hue signifies the typical functional end of synthesised MXenes. 23
Thanks to their two-dimensional shape and distinctive layered structure, MXenes are ideal for creating composites with other materials that improve their performance. Recycling, energy storage, electronics, sensors, water splitting, and catalysis are just a few of the many applications that benefit greatly from MXenes’ exceptional properties, which include a high surface area, activated metallic hydroxide sites, biocompatibility, ease of functionalisation, high metallic conductivity, and hydrophilicity. MXenes can be utilised in membranes, electrochemical separation, photocatalysis, capacitive deionisation and as adsorbents for water treatment and purification. 24
Synthesis methods: Selective etching, top-down and bottom-up approaches
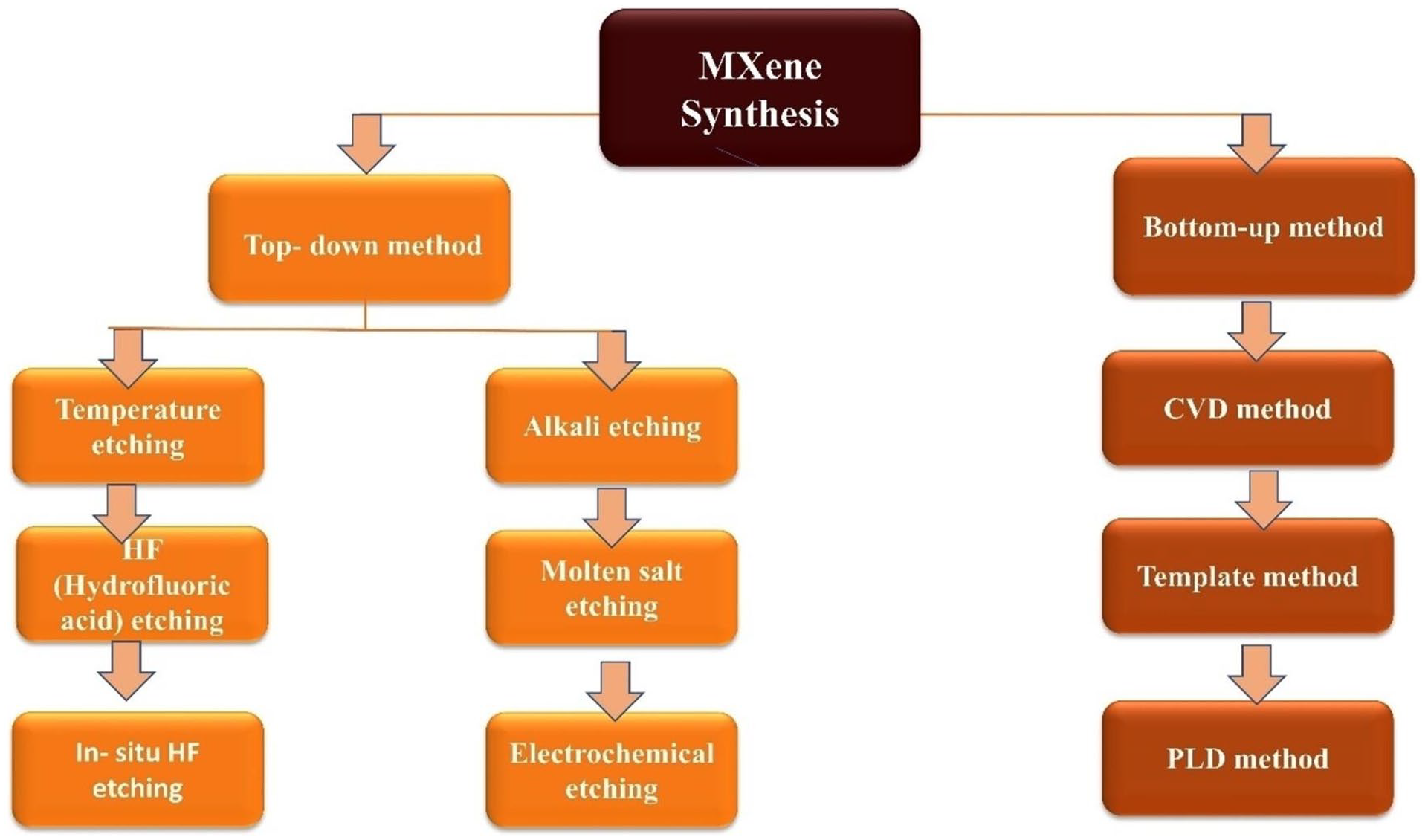
Since now, various methods have been adopted for the synthesis of MXenes done by delimination as shown in Figure 4. The current section describes various synthetic approaches, including their advantages and challenges.
Major synthesis process of MXene. 25
The processes for the production of MXenes significantly impacted their electrical properties, physicochemical characteristics, and application spectrum. Over 20 MXenes have been synthesised using the chemical selective etching of several atomic fragments obtained from pretreatment compounds, including carbide, nitride, and carbonitride (Figure 5).
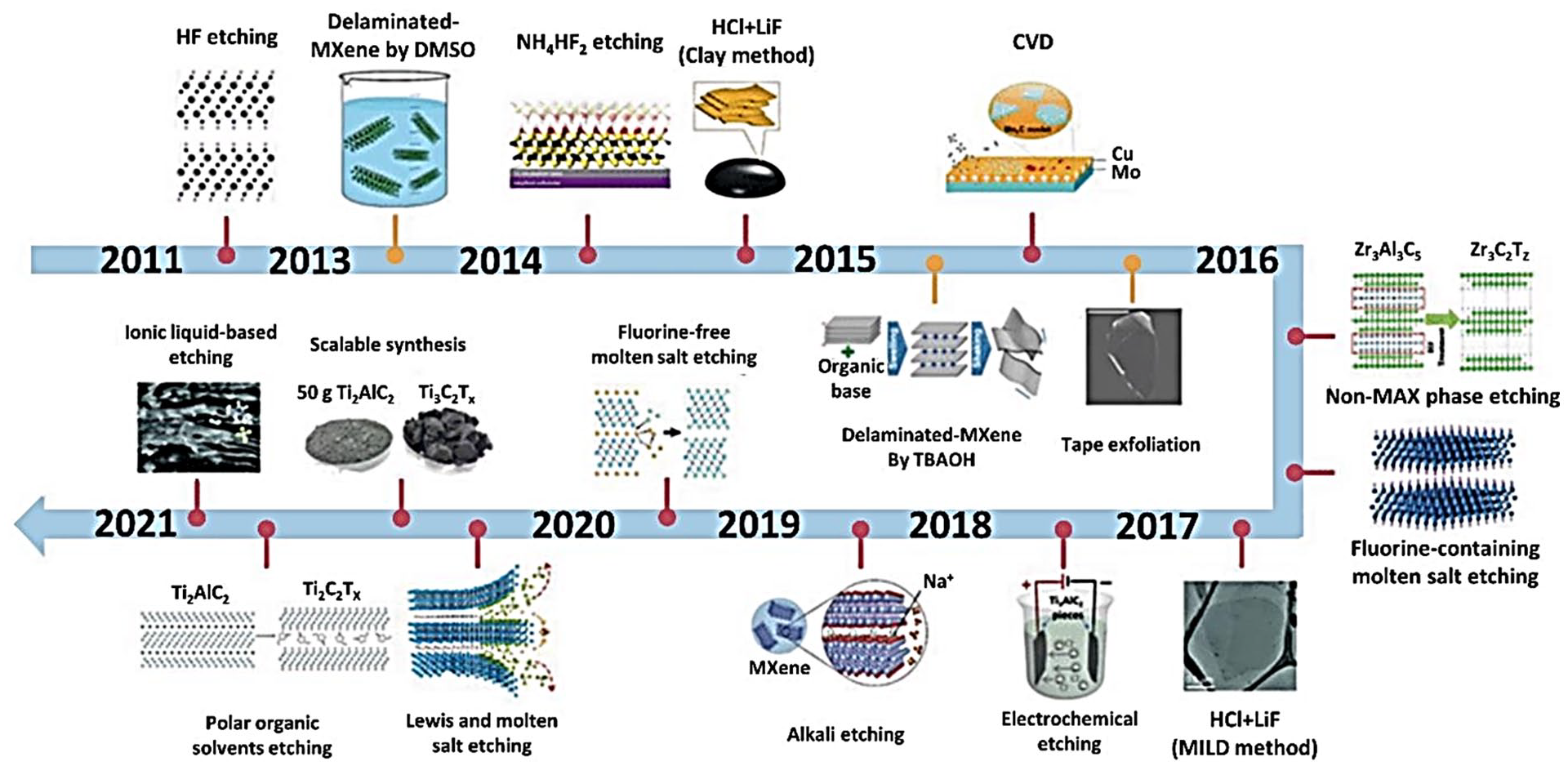
Advancements in MXenes synthesis utilising various methodologies. (Reproduced with copyright permission.) 25
The three main techniques for creating MXene are top-down, bottom-up, and etching. 26 Below, Figure 6 summarises each strategy.

Illustration of MXenes production via HF etching and its morphology. (a) The schematic representation of MXenes obtained through HF etching. (b) The synthesis and structural diagram of MXenes. (c) SEM picture of Ti3C2 27 ; following washing with (d) distilled water; (e) ethanol; (f) in situ synthesised MXenes. 28
Wet etching from the precursor MAX phase is usually the first step in synthesising MXenes. Bulk crystals are then layered to create single-layer or multilayer MXenes. For this, hydrofluoric acid (HF) is frequently employed as an etchant. The three steps of this MXenes synthesis process are delamination, intercalation, and etching (Figure 7). 29 First, MXenes layers are formed with the reaction of acid usually HF with the A layers of MAX phases. Polar functional groups –OH, –O, and/or –F takes the place of the A atoms in the MAX phases in the second step, which negatively charges the MXenes surface and promotes the creation of stable dispersions. However, in the final stage, ultrasonic treatment compromises the bond between the Mn + 1X n layers, leading to the development of MXenes with loosely arranged layered structures. 30

Schematic representation of (a) acid etching synthesis, involving various stages including etching and intercalation, to produce delaminated MXenes and (b) general mechanism of the molten salt etching technique using KCl and LiCl salts through electrolysis.25
The most widely used MAX precursor to produce the most researched Ti3C2T x MXene is Ti3AlC2. A multitude of new MXenes has been synthesised utilising this HF etching technique. However, there are risks associated with using HF etchant, and utilising it needs extra caution. Furthermore, stable nitride-based MXenes (Tin + 1 N n ) cannot be prepared using this approach. Consequently, it is essential to create more adaptable and ecologically friendly processes for the synthesis of MXenes.
In the top-down method, large or massive material is broken down into smaller sheets with this technique; these sheets may be single-layered or monolayered. Bulk precursors that will be transformed into sheets may have a crystalline structure. Top-down techniques include ball milling, 31 liquid exfoliation, heating with a hydrothermal or solvent aid, ultrasonication, and microwave-assisted exfoliation. Conditions like chemical etching or acid reflux are suitable for the top-down method. Furthermore, a lot of material is produced using this easy-to-implement strategy. There are challenges, though, like the restricted output and the necessity for particular interventions. In the HF etching process, because of the metallic nature of the M–A molecules, mechanical shearing is not feasible, which separates the A from the MAX by a chemical reaction. The chemically active character of M–A, as opposed to strong M–X, makes chemical etching (with HF, HCl, etc.) feasible. Figure 7(a) drowns out the HF’s process and Figure 7(b) displays the schematic representation of sonication method. The production of MXenes involves the chemical processes listed in Scheme 1. 16

Common HF etching process.
By reacting LiF with HCl, the Minimally Invasive Layer Delamination (MILD) approach produces HF in situ. This makes it possible to create regulated etching conditions that enhance the final MXenes’ quality and homogeneity, increasing their suitability for use in later processes. Ultrasonic treatment is frequently used to further exfoliate the MXenes after etching. This procedure makes it easier to incorporate them into biocomposites and improves their dispersion in solution. To guarantee that MXene nanosheets are evenly dispersed throughout a polymer matrix, ultrasonic vibrations aid in the dissolution of agglomerates.
In the bottom-up method, contrary to the top-down strategy, which uses bulk as its beginning material, this approach uses molecular material. In Chemical Vapor Deposition (CVD) deposits metal precursors onto a substrate and then allows a reaction to make MXenes; CVD techniques can be utilised to create MXenes. High-quality films with regulated thickness and composition are possible using this technique. CVD is useful for creating large-area MXene films that can be used in sensors and electrical devices, among other uses. While Pulsed laser deposited (PLD) is employed to build a Mo₂C layer in Figure 8, CVD creates MXene using high temperatures and chemicals (Cu, Mo, CH₄) in a regulated environment. PLD creates MXene crystals at low temperatures, although CVD has greater conductivity and crystallinity. The synthesis is progressing daily.

Common mechanism of (a) CVD methods, from methane to the production of Mo₂C layer and (b) template MXenes synthesis methods, using crystals of salts for the production of MoN sheets. 25
Atomic Layer Deposition (ALD) is another bottom-up technique that allows for the atomic-level deposition of tiny material layers on surfaces. MXene films or coatings with particular qualities suited for biocomposite applications can be produced using this method. ALD is appropriate for complex geometries because of its exceptional uniformity and conformity. A bottom-up technique has numerous pluses over a top-down strategy, like the use of molecules or small precursors, increased atomic utilisation, customisable structural and functional characteristics, and faster functionalisation. Because it only needs one pot reaction, bottom-up synthetic process is easier than top-down synthetic process nonetheless, further study on the bottom-up synthesis technique is needed. 32
Hydrofluoric acid is unnecessary for contemporary MXene production methods such as chemical vapour deposition (CVD), 33 acoustic synthesis, and molten salt. 34 These techniques embody the latest advancements in MXenes production. Cai et al. 35 showed that the MAX phase’s A layers may also be extracted after the hydrothermal treatment process in H2SO4 for the production of Ti3C2. This approach takes a lot of time, though in order to shorten the time required for synthesis and enable expansion, the hydrothermal technique must be optimised by raising the solution concentrations. Other techniques exist as well, such as the anodic corrosion of Ti3AlC2, which involves making a brine mixture with hydroxide and chloride ions. The MAX phase absorbs cations and the neutral compounds that result, lengthening the gap between layers and enabling chloride ions to pierce and etch the A layer. The usage of other anions is still up for debate because chloride ions have not been able to entirely etch the Al layer from TiAlC. 36 As a result, these technologies have not yet been successfully proven for widespread use, despite their promise and possibilities.
Properties of MXene
High capacitance, large surface area, remarkable chemical stability, hydrophilic character, exceptional thermal and electrical conductivity, exclusive sorption–reduction capacity, and mechanical adaptability are the properties that make this material distinguish and suitable for water treatment (Figure 9).
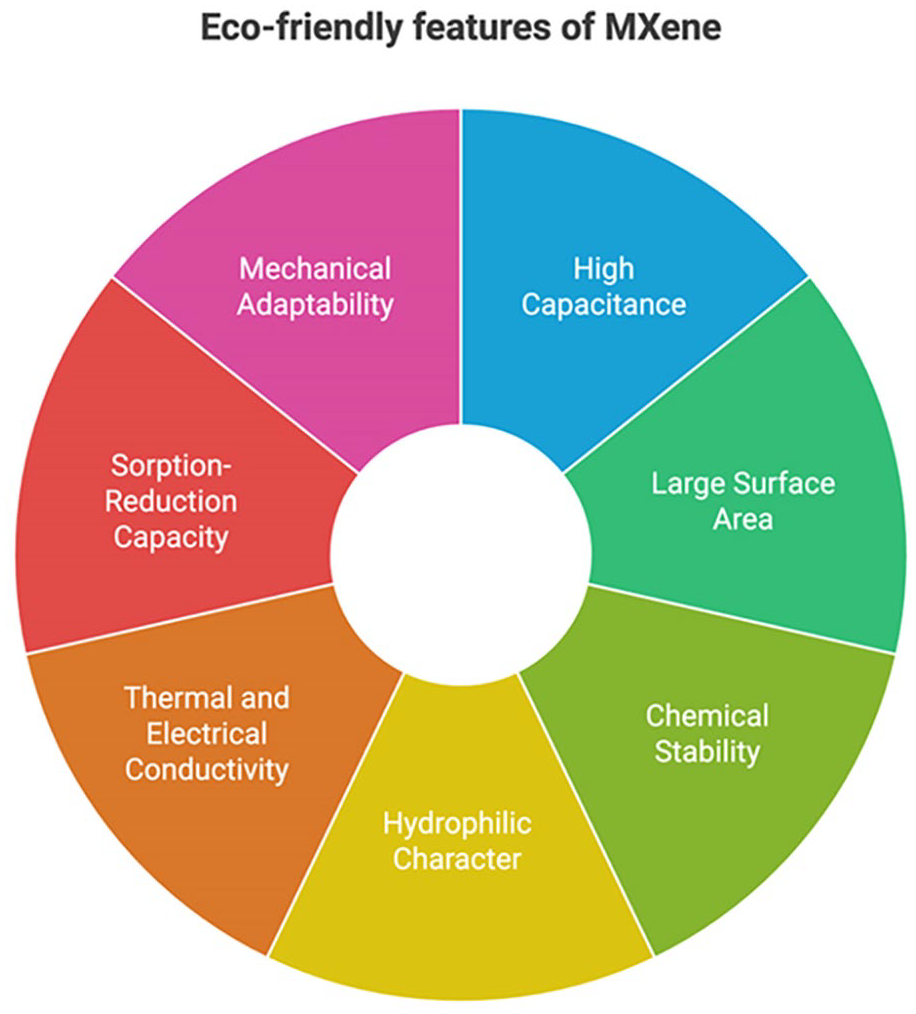
Diagram showing the properties of MXenes.
Capacitive characteristics
High capacitance is a result of MXenes abundance of oxidation-reduction sites. 37 Using Ti3C2T x MXenes as an example, the hydration of the terminal groups containing oxygen causes Ti’s oxidation state to fluctuate continually, giving the valence transition metal the ability to transfer charges. 38 Energy storage is another important function of MXene’s surface end groups. Due to their greater electron sharing with the element M, present in between the layers of MXene and the conversion between the =O and –OH terminal moieties while charging and discharging processes, which generates numerous active sites for redox reactions, =O active moieties are typically more steady than –OH and –F. 39 The multi-layered architecture of 2D MXene enhances ion intercalation and transmission, while its unique layered configuration yields an increased surface area. Furthermore, MXene’s layered structure enables it to adjust to different intercalating agents, increasing its electrochemical reaction activity relative to its surface area and improving cycling stability and pseudocapacitance. The high energy and power densities of the devices made with MXene demonstrate its good energy storage potential.
Hydrophilicity
Its layered architecture, including –OH and –F, gives MXene considerable hydrophilicity by including oxygen, hydrogen, and other functional groups, therefore rendering its surface highly polar. 40 By forming hydrogen bonds and other intermolecular interactions with water molecules, these polar functional groups show amazing hydrophilicity in water, 41 hence producing attractive contacts between MXene particles and water molecules. MXene’s multiple hydrogen bond acceptors in its interlayer structure help to increase the friendliness of MXene with water by enabling the hydrogen bond formation with water molecules. 42 Furthermore, MXene’s hydrophilicity can be enhanced by its properties, which include its higher specific surface area and porous design, which enable higher contact surface area between MXene and water, so improving their interaction. All of the several hydrophilic active moieties, hydrogen bonding sites, porous architecture, great specific surface area, and layered MXene arrangement help to explain its extraordinary hydrophilicity. This property gives MXene great dispersion, stability, and adaptability in aqueous systems, 43 hence preparing MXene for broad use in water-based supercapacitors and other domains.
Conductivity
MXenes have remarkable conductivity with a metallic value of up to ~24,000 S/cm. MXene is composed of many transition metal elements, including usually highly conductive titanium and molybdenum. These components generate conductive pathways inside the structure and enable electron transfer, 44 hence increasing the general conductivity of MXene. 45 MXene’s two-dimensional structure and great specific surface area help to improve electron accessibility. Freedoms of movement for electrons on the 2D plane reduces electron scattering losses and improves conductivity. MXene’s shape also significantly affects its conductivity; single-layer, large-sized flakes usually enhance conductivity more than multi-layered, small-sized flakes. 46 MXene has more chemical functional groups, including hydroxyl and fluorine atoms, than other two-dimensional materials including graphene or metal sulfides/hydroxides. MXene’s conductivity performance can be changed along with its electronic structure and charge transport properties by altering these functional groups. 47 All things considered, MXene’s unique structure—which comprises many chemical functional groups and transition metal elements—which makes it a highly conductive substance with a broad variety of conceivable uses—is the main factor influencing its increase in conductivity. High-power density supercapacitors can be developed thanks to MXene’s remarkable conductivity allowing fast electron transport. Not even necessary during the electrode manufacturing process are conductive agents and current collectors, therefore lowering the total energy density of the device.
Mechanical adaptability
MXene’s versatility in the field of mechanics is largely dependent on its layered structure. MXene is made up of many layers of two-dimensional nanosheets that are joined together via relaxed bonding. In addition to providing MXene with remarkable deformability and bending ability, the 2D nanosheets serve the purpose of enabling MXene to slide and shear under forces generated by external objects. 48 In the meantime, the layered structure of MXene can be significantly enhanced to create composite materials and increase its flexibility by intercalating other materials (such as liquids or polymers). 49 Chu et al. used a straightforward scratch approach and in situ polymerisation to create two-dimensional transition metal carbide/polyimide (Ti3C2T x MXene/PI) films, which demonstrated increased flexibility. The structural combination, size, flaws, density, imperfect nanosheet edges, surface end groups, and M–X elements determine the mechanical properties of MXenes.
MXene loaded biocomposites
MXene-loaded biocomposites combine the adaptability and eco-friendliness of biocomposites with the exceptional properties of MXenes. MXenes offer biocomposites—a kind of material made of a matrix enhanced with natural fibres or polymers—special qualities, and they may create a hybrid that is more efficient and biodegradable.
MXene-infused biocomposites exhibit significant potential as materials for water purification applications. The integration of polymer substituents produces a flexible MXene–polymer hybrid structure that can decrease interlayer spacing, minimise charge transport distance, and enhance electrical conductivity, biocompatibility, and flexibility. For example MXene-chitosan composites offer enhanced surface functionality, adsorption capacity, and biocompatibility for water pollution removal.50,51 Because of their unique characteristics, simplicity of manufacture, and cost, magnetic-MXene nanocomposites have great potential for reducing water pollution. 52 Developed for water purification and desalination, MXene-based composite membranes have high water flux and suitable rejection rates. 53 Among the toxins that biocomposites effectively remove from wastewater are organic dyes, drugs, and pesticides. 54
MXenes combined with other materials like magnetic nanoparticles and chitosan allows advanced water treatment technologies new possibilities. MXenes’ various surface-tailored functional groups, adsorption of electromagnetic interference radiation, light-to-heat conversion capabilities, enhanced metallic conductivity, and most importantly negative zeta potential are meant to improve their properties. It has proven possible to create MXene-biopolymer nanocomposites, including MXene/cellulose, MXene/chitosan, MXene/chitin, MXene/lignin, MXene/starch, and MXene/natural rubber. Due to inexpensive, renewable, readily available, hydrophilic, biocompatible, biodegradable, and contain adjustable functional groups, chitosan and chitin have been widely used. Chitin, which is composed of a chain of connected 2-acetoamido-2-deoxy-b-d-glucopyranose units, is the second most prevalent polysaccharide on Earth after cellulose. It is taken from the exoskeletons of some insects, molluscs, crustaceans, and fungus. It has a compact structure, is soluble in a limited number of solvents, and is hydrophobic by nature. Generally speaking, chitin is transformed into “chitosan,” a highly active derivative. Compared to chitin, chitosan is inherently more hydrophilic.
Polyacrylic acid (PAA) molecules are distinguished by the presence of carboxyl groups, which make it easier for them to form hydrogen bonds with other molecules. A particular study involved the combination of polyacrylic acid and MXene to produce a stable dispersion, which was then vacuum-filtered onto a polyacrylonitrile (PAN) nanofiltration film. Using the polyacrylic acid and MXene mixture to create a hydrogel that functions as a physical crosslinker to improve stability and avoid excessive bulging in water was the idea behind this. Rejection rates of 98.12% for AB90, 99.52% for AB8GX, 95% for CR, 90.63% for AM, and 98.92% for DR80 have been demonstrated for PAA-MXene/PAN film. Together with preparation, Table 1 provides a thorough analysis that aids in making well-informed decisions on other applications. 55
MXene polymer biocomposites’ preparation, characteristics, and possible uses.

CS: chitosan; CTAB: cetyltrimethylammonium bromide; DCM: dichloromethane; EMI: electromagnetic interference; MSCAs: micro-supercapacitor arrays; PANI: polyaniline; PDA: polydopamine; PLA: polylactic acid; PPy: polypyrrole; PVA: polyvinyl alcohol.
Polydopamine intimately interacted with a two-dimensional ultrathin nylon basement membrane to support an RGO/PDA/MXene composite material for oil-water separation and dye removal. The study’s results shown exceptional separation efficiency above 96% and elevated throughput (>200 L/m2 h) for various dyes, including Evans Blue, Congo Red, Methylene Blue, Methyl Red, and Methyl Orange.
The polypyrrole (PPy) substrate has positively charged nitrogen atoms and π-conjugated structures, and it has been shown that PPy serves as an effective adsorbent for the elimination of organic dyes. PPy possesses the capability to engage with pollutants in wastewater through many mechanisms, including π–π interactions, electrostatic interactions, and hydrogen bonding. Thus, it can be asserted that the adsorption capacity of PPy nanoparticles may partially counterbalance the potential reduction in adsorption capacity resulting from the diminished active sites on MXene nanosheets due to the presence of PPy nanoparticles. To exclude methylene Blue from the aqueous phase, Shi et al. 75 developed few-layered MXenes/PPy composite particles. The incorporation of PPy nanoparticles significantly enhances the exfoliation of MXene. As a result, the adsorption capacity of the MXene/PPy composites is significantly improved, reaching a maximum of 553.57 mg/g. Moreover, the MXene/PPy composites exhibit the ability to interact with both cationic and anionic dyes, while displaying remarkable selectivity for the adsorption and extraction of methylene blue from aqueous solutions. The MXene/PPy composite particles exhibit significantly enhanced MXene stability and little oxidation. The MXene/PPy nanocomposites exhibited remarkable selectivity by effectively eliminating only methylene blue from the aqueous phase, while accommodating both cationic and anionic dyes. This study presents a novel approach for fabricating MXene-based composite adsorbents that exhibit exceptional stability and can be utilised for wastewater treatment. 75
In 2022, Wu et al. developed Chitosan-functionalised porous carbon microspheres CPCM@MXenes, 76 a pod-inspired superabsorbent material. Chitosan-functionalised porous carbon microspheres (CPCM) and Ti3C2T x MXene formulae obtained from the Ti3C2T x MAX phase were used to create the composites in order to generate a huge surface area (>1800 m2/g) and to keep them from clumping together. CPCM@MXene, which has a very strong adsorbent ability, was studied to remove crystal violet from water 2750 mg/g was the greatest adsorption capacity. The comprehension of the adsorption mechanism is facilitated by the stacking effect, physical adsorption, hydrogen bonding, and electrostatic interactions. The remarkable adsorption efficacy of CPCM@MXenes for the removal of crystal violet in a model aquatic environment may be applicable to other molecular pollutants. The excellent recyclability of these super absorbents facilitates their reuse.
The Endocrine Society characterises endocrine disruptor chemicals (EDCs) as “an external substance, or a combination of chemicals, that disrupts any aspect of hormonal function.” In other words, compounds capable of negatively impacting the endocrine system are referred to as Endocrine Disruptor Chemicals (EDCs). The bulk of endocrine disruptors (EDs) and suspected endocrine-disrupting chemicals (EDCs) are anthropogenic and can be located in various materials. 77 These compounds frequently attach to endogenous receptors, such as oestrogen and steroid receptors, so affecting normal immunological, developmental, reproductive, and neurological activities.
Due to the ubiquitous presence of EDCs in various environments, including food, water, and air, individuals encounter them through multiple exposure pathways. Endocrine-disrupting chemicals can also penetrate the body via dermal touch. Natural water bodies are adversely affected by endocrine disruptor chemicals (EDCs), which are pervasive and hazardous pollutants typically present in discharged wastewater effluents. This is attributed to the inadequate analytical methods that fail to quantify EDC in treated wastewaters, hence hindering the advancement of wastewater treatment systems capable of fully mineralising the EDC. To adsorb triclosan, triclocarban, 2-phenylphenol, bisphenol A, and 4-tert-octylphenol from aqueous samples, Researchers have developed a novel micro-solid-phase extraction (μ-SPE) apparatus employing an MXene-based adsorbent encased in a polypropylene membrane. The optimal EDC extraction efficiency was attained with the addition of 2 mg of sorbent. The addition of oxygen and fluorine components imparted hydrophilic characteristics to MXene. Consequently, it was proposed that the O or F in MXene and the electropositive sites from the EDC must establish hydrogen bonds and participate in dipole-dipole interactions for the extraction and adsorption to occur.
Challenges of MXene-based biocomposites
Synthesis scalability
One of the primary hurdles is scaling up the synthesis of MXenes-polymer nanocomposites to meet industrial production requirements. Figure 10 shows some of the ways to scale up the MXene polymer nanocomposites. The scalability and economic feasibility of existing synthesis methods are constrained by the sometimes time-intensive and intricate processes they involve. 78
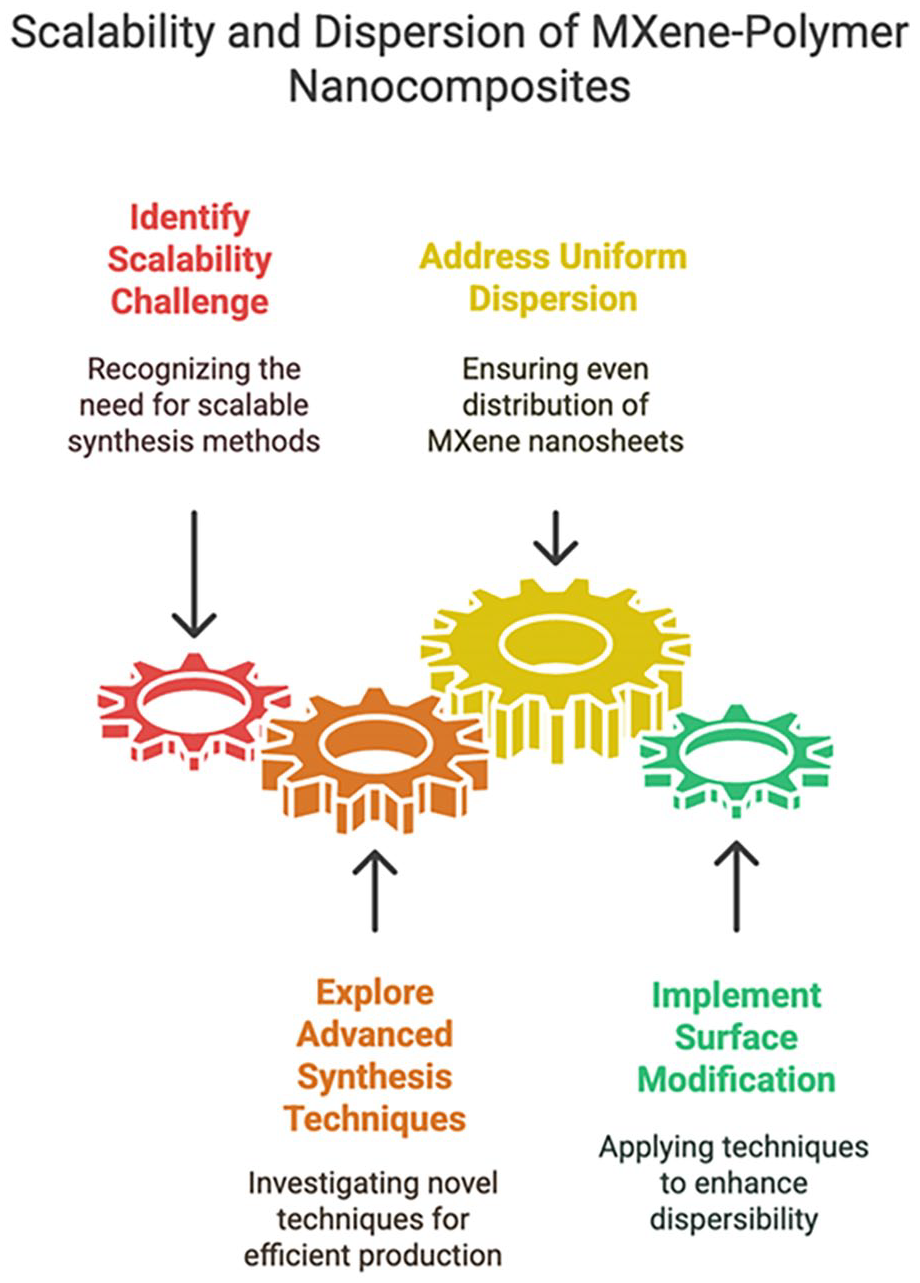
Ways to scale up the MXene polymer nanocomposites. 78
Even distribution
Maintaining the intended characteristics and functionality of nanocomposites requires that MXenes nanosheets be uniformly dispersed across polymer matrices. However, because MXenes nanosheets have a propensity to aggregate during processing, guaranteeing uniform dispersion continues to be difficult.
Adhesion between surfaces
To improve the mechanical characteristics and stability of nanocomposites, MXene nanosheets and polymer matrices must have strong interfacial adhesion. However, because MXenes and polymers have fundamentally different characteristics, it is still difficult to achieve good interfacial adhesion. Researchers are investigating methods to strengthen the bonding between MXene nanosheets and polymer matrices in order to boost the mechanical strength and stability of nanocomposites. 78 Techniques such as adding compatibilisers, conducting in-situ polymerisation, and using interfacial coupling agents are being applied to enhance interactions at the interface and improve overall nanocomposite performance.
Biocompatibility
Even though MXene-polymer nanocomposites have a lot of potential for use in biomedicine, it’s still difficult to make sure they’re biocompatible. To fully assess these nanocomposites’ biocompatibility and address any possible cytotoxicity or immunogenicity concerns, more investigation is required. 78 Current research is dedicated to thoroughly assessing the biocompatibility of MXene-polymer nanocomposites for use in biomedical settings. Investigations include testing for cytotoxicity, immunogenicity, and long-term biocompatibility using both laboratory and animal studies, with the goal of confirming the safety and effectiveness of these nanocomposites in biological systems.
Lifespan
Practical uses of MXene-polymer nanocomposites depend on their long-term durability in a range of environmental circumstances. But with time, problems including agglomeration, leaching, and degradation could compromise the stability and functionality of nanocomposites.
Cost-efficiency
MXenes-polymer nanocomposites’ affordability is still an issue, particularly for large-scale uses. The complicated synthesis procedures and expensive cost of MXene precursors may prevent their broad use in commercial applications. To lower the cost of producing MXenes-polymer nanocomposites, efforts are being made to create economical synthesis pathways and make use of less expensive precursor ingredients. Additionally, reducing total manufacturing costs and streamlining production processes are the goals of scale-up and process optimisation initiatives 79 (Figure 11).
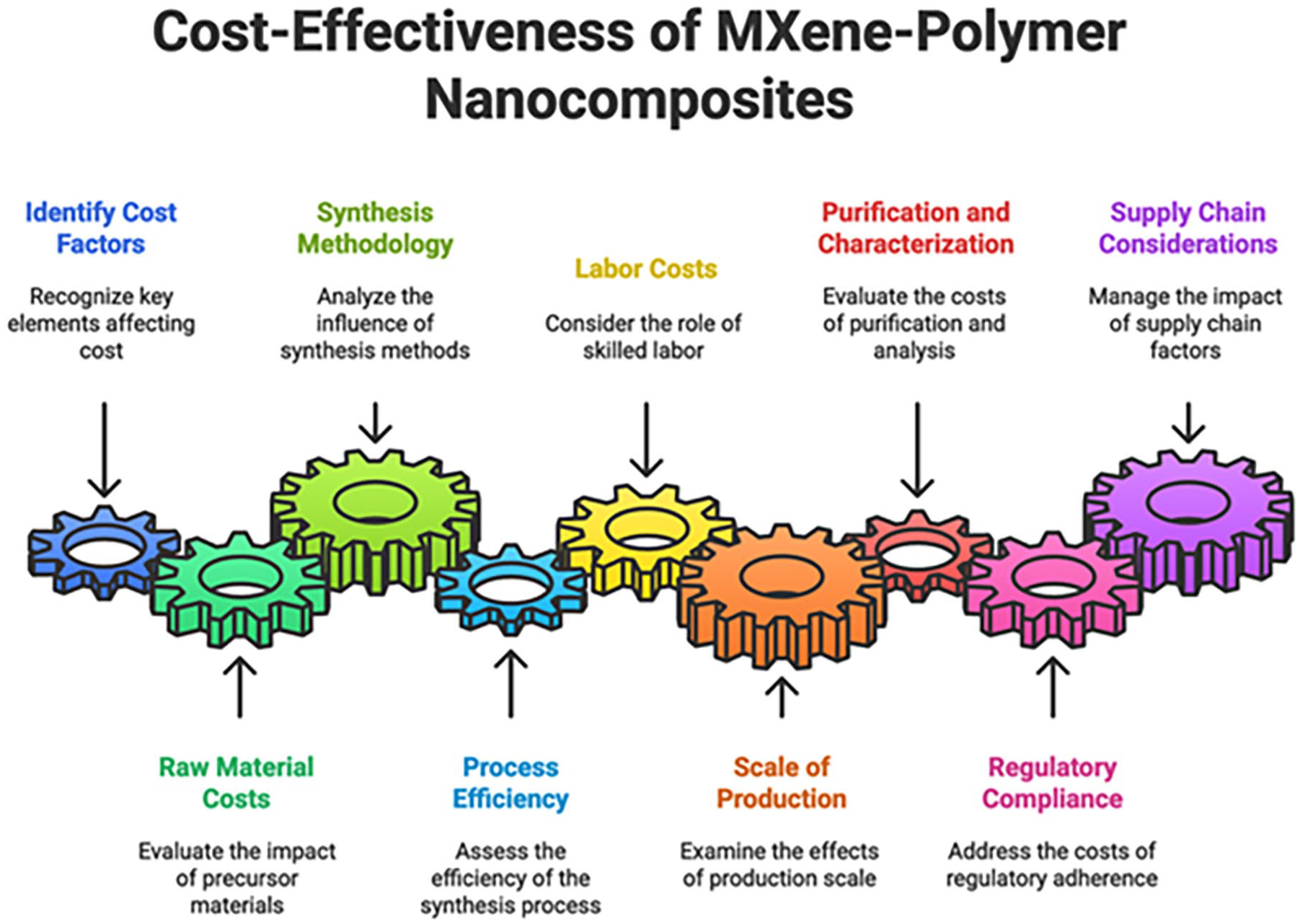
Cost-effecting factors of MXene polymer nanocomposites. 78
Regulatory clearance
Thorough safety evaluations and compliance with standardised testing protocols are being performed to secure regulatory approval for MXenes-polymer nanocomposites. The secure and ethical implementation of nanocomposites in commercial settings is guaranteed by collaboration with regulatory authorities and compliance with regulatory guidelines. Multidisciplinary research is essential to tackle these challenges, focussing on developing novel synthesis techniques, enhancing the properties of nanocomposite materials, augmenting scalability, improving biocompatibility, and conducting comprehensive performance evaluations. 78 To surmount these challenges and completely actualise the potential of MXenes-polymer nanocomposites across diverse applications, collaboration across academic institutions, industry, and regulatory entities is essential. Current research on MXene-polymer nanocomposites seeks to address prevailing challenges through innovative solutions, interdisciplinary collaborations, and improvements in synthesis, characterisation, and application methodologies. These projects facilitate the extensive application of MXenes-polymer nanocomposites across several sectors, including environmental remediation, electronics, energy, and healthcare.
Future perspectives
New trends and research topics that have the potential to further studies and applications are included in the field of MXenes-polymer nanocomposites’ future directions. Here are some important future directions.
Three-dimensional printing and additive manufacturing
This study will examine the incorporation of MXenes-polymer nanocomposites into additive manufacturing methods, such as 3D printing, to produce functional devices with specific properties and intricate structures. This method presents chances for on-demand manufacture of MXenes-based goods, fast prototyping, and customising. 80
Storage and energy conversion
MXenes-polymer nanocomposites have appeal for storage and energy conversion. 81 Because of their many benefits These cover mechanical flexibility, chemical stability, large surface area, and extraordinary electrical conductivity. Their characteristics make them effective electrode materials in many different energy conversion and storage systems. Furthermore, showing promise for improving fuel cell, lithium-ion battery, and other energy storage device efficiency are MXenes-polymer nanocomposites. By means of improved composition, structure, and processing methods, continuous research in this field aims to increase the energy storage and conversion efficiency of MXenes-based materials. 82
Medicines and biomedical equipment
MXene-polymer-based nanocomposites for medicines and biomedical tools will be developed under additional investigation. These nanocomposites might be used in drug delivery systems, tissue engineering scaffolds, biosensors, and medical implants depending on their biocompatibility, bioactivity, and controlled release characteristics. 78 The fast development of the biomedical devices and pharmaceuticals industry is driven by creative research and technological developments. Here are some future estimates.
Environmental clean-up
MXenes-polymer nanocomposites will be the main topic of future studies aiming at environmental cleanup and pollution control. These nanocomposites are fit for soil remediation, wastewater treatment, air filtration, and water purification since they have great adsorption capacity, catalytic activity, and selectivity for contaminants. One major future trend is the inclusion of cutting-edge technologies, including artificial intelligence, machine learning, and remote sensing, into environmental monitoring and cleanup plans. 83 By means of real-time data, predictive modelling, and automated decision-making capability, these technologies improve the efficacy and efficiency of pollution detection, site characterising, and cleaning activities. 84 Nature-based solutions for environmental cleanup and green infrastructure development are gathering steam. By leveraging ecosystems and natural processes to restore habitats, clean water, and store carbon, these technologies present affordable and sustainable substitutes for conventional remedial approaches.
Concerns regarding toxicity
When 2D MXene-based adsorbents and membranes are regarded as promising options for water and wastewater treatment, a systematic and analytical assessment of their environmental impact, particularly regarding potential risks to aquatic biota and ecosystems, is essential. The study investigated the toxicity of MXene nanosheets on developing avian embryos and their capacity for angiogenesis. 85 Exposure to MXenes resulted in a 46% mortality rate of embryos within 5 days and hindered angiogenesis. Analyses of gene activity in the brain, heart, and liver of treated embryos revealed alterations in seven genes critical for cell proliferation, survival, and angiogenesis. The results suggest possible developmental hazards associated with MXene exposure, underscoring the necessity for further research on long-term safety, degradation, and handling characteristics. 85
Current research on MXene toxicity remains limited, but one study investigated the biocompatibility of Ti3C2Tₓ using zebrafish embryos. Researchers examined how MXene aggregates form in seawater and their effects on embryonic development. 86 Findings revealed that zebrafish embryos absorbed MXenes in a dose-dependent manner, with concentrations tested at 25, 50, 100, and 200 μg/mL. The 96-h LC₅₀ (lethal concentration for 50% mortality) was calculated as 257.46 μg/mL, while the NOEC (no observed effect concentration) and LOEC (lowest observed effect concentration) were 50 and 100 μg/mL, respectively. At 100 μg/mL, mortality reached 21%, but no significant developmental abnormalities were observed. Neurotoxicity tests confirmed safety at 50 μg/mL, showing no adverse impacts on neuromuscular functions. Based on the FWS Acute Toxicity Rating Scale, Ti3C2Tₓ falls into the “practically nontoxic” category, suggesting that concentrations below 100 μg/mL may be safe for aquatic ecosystems. 86
Emerging organic contaminants (EOCs): Chemistry and challenges
Emerging organic contaminants (EOCs) are human-sourced organic chemicals, including pesticides, drugs, surfactants, personal care items, and industrial additives identified in the environment at hazardous amounts (Table 2). These chemicals can enter wastewater from a variety of sources, including stormwater, 87 industrial discharges, domestic sewage, and agricultural runoff. Common EOCs like carbamazepine, atrazine, caffeine, and metolachlor pose significant health risks to communities consuming contaminated water. The kinds and concentrations of contaminants in wastewater can be greatly influenced by its source. Many of these compounds are emerging pollutants of concern because they can have adverse impacts on human health and the environment, even at low concentrations. For example, Phenolic contaminants represent significant environmental pollutants that pose considerable risks to human health and the ecological environment. Efficient remediation of hazardous phenolic contaminants is essential to safeguard aquatic ecosystems and air quality. 88 Pesticide contamination has been identified as one of the most pressing global concerns in agriculture. Pesticides have mutagenic, teratogenic, carcinogenic, and cytogenic effects. Even trace quantities of pesticides can result in significant health issues, including epithelial toxicity, liver toxicity, immunotoxicity, neurotoxicity (total or partial disruption of the equilibrium between oxidants and antioxidants in neurones and nerves), and reproductive toxicity. It is estimated that one-third of the population in affluent nations is afflicted by foodborne infections, with the problem likely being more severe in developing countries. 89 For these different organic contaminants to be effectively removed, novel wastewater treatment technologies are often required in addition to conventional treatment methods. Monitoring and controlling these compounds in wastewater and the environment have become a top priority for environmental scientists and engineers. They are broadly classified based on their sources and chemical nature. 90
DEHP: di(2-ethylhexyl)phthalate; DES: diethylstilbestrol; EDCs: endocrine disrupting chemicals; HBCDs: hexabromocyclododecanes; MTBE: methyl-t-butyl ether; PBBs: polybromonated biphenyls; PBDDs: polybromonated dibenzo-p-dioxins; PBDEs: polybrominated diphenyl ethers; PBDFs: polybromonated dibenzofurans; PCPs: personal care products; PFOA: perfluoroctanoic acid; PFOS: perfluorotoctane sulfonates.
Conventional methods for EOC removal
Conventional wastewater treatment technologies have been employed to address the challenge of removing emerging organic contaminants (EOCs) from water sources. While these methods often face limitations, there have been notable successes and advancements in their application. Below (Figure 12) is an overview of these conventional methods, highlighting their effectiveness in EOC removal.

Diagram showing main treatment methods for EOC removal. (Reproduced with copyright permission.) 92
Adsorption techniques
Adsorption is a prevalent method for the elimination of EOCs, utilising materials such as activated carbon, zeolites, and metal-organic frameworks (MOFs; Table 3). Activated carbon is a conventional adsorbent recognised for its extensive surface area and porosity. It has effectively eliminated several emerging organic contaminants, including medications and personal care items. Nonetheless, its efficacy may be constrained by the presence of hydrophilic molecules, which exhibit a reduced propensity to adsorb onto the carbon surface. Recent studies have shown the efficacy of Metal-Organic Frameworks (MOFs) in eliminating EOCs, attributed to their adjustable pore dimensions and substantial surface areas. For instance, a zirconium–ellagate framework (SU-102) has been reported to remove cationic EOCs with efficiencies ranging from 79% to 99.6% from municipal wastewater treatment plant effluent. This framework works effectively due to its charge properties that enhance selectivity for certain contaminants while also serving as a photocatalyst for degradation under visible light (Figure 13).
Emerging contaminants along with their adsorbents.
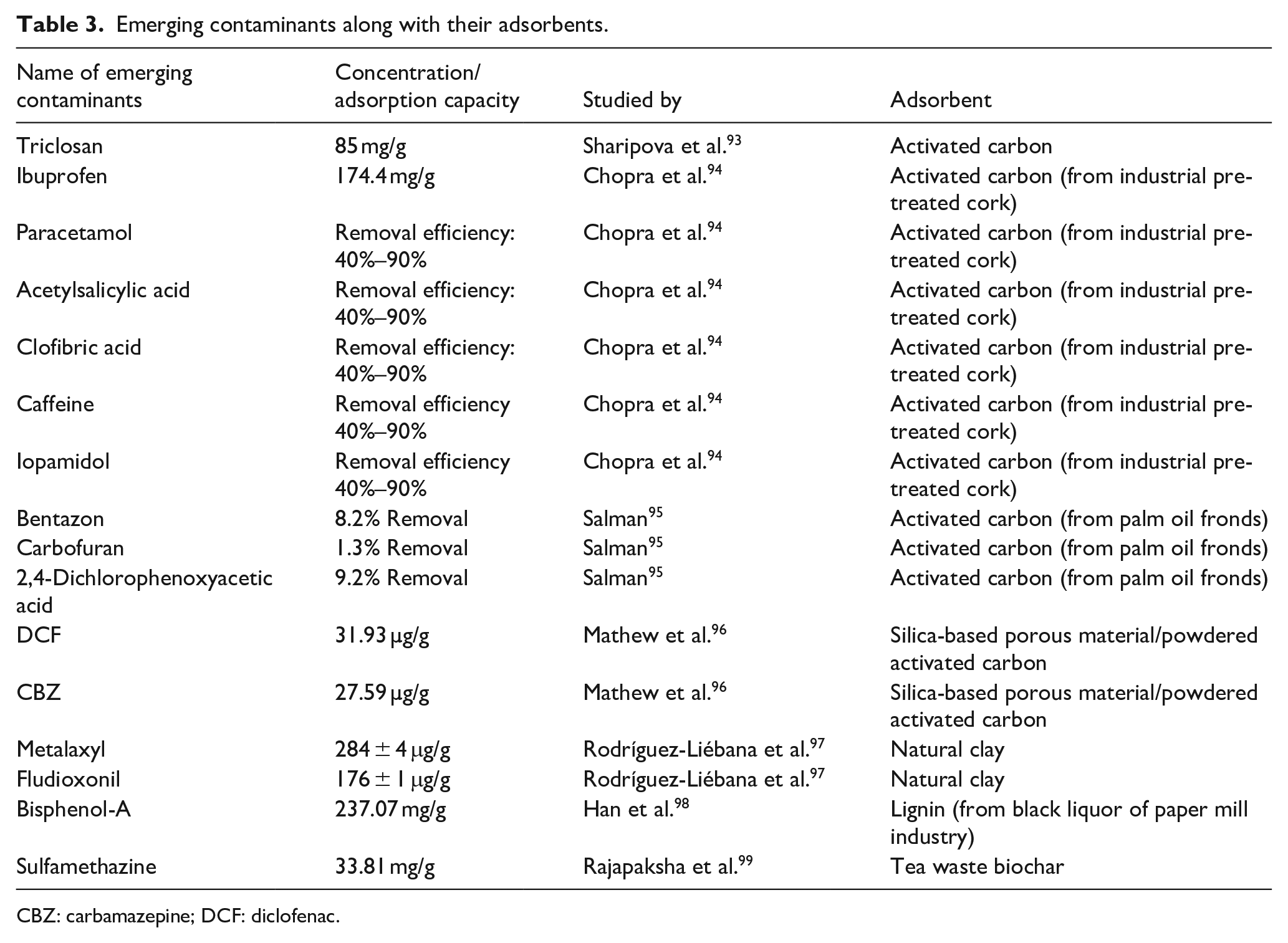
CBZ: carbamazepine; DCF: diclofenac.

Diagram showing different conventional methods for EOC removable.
Biological treatment
Biological treatment technologies are classified into two categories: aerobic processes and anaerobic processes. Active sludge, membrane bioreactors, and sequencing batch reactors are reliable aerobic treatment methods. Biological treatment technologies, especially those employing activated sludge processes, have been conventionally utilised to decompose organic contaminants (Table 4). The Activated Sludge Process utilises microorganisms to degrade organic debris, achieving an average removal effectiveness of over 85% for all 55 PPCPs examined. While effective for many biodegradable compounds, it often struggles with EOCs due to their low concentrations and recalcitrance. Research indicates that biological treatment can effectively remove certain pharmaceuticals when present at higher concentrations or when microbial communities are adapted to specific contaminants. Some EOCs can be degraded through co-metabolism, where microorganisms utilise other organic compounds as nutrients while simultaneously degrading EOCs. This method has shown promise but is limited by the requirement for suitable nutrient sources and microbial adaptability (Figure 13).
Various biological treatment of ECs and its efficiency of removal.
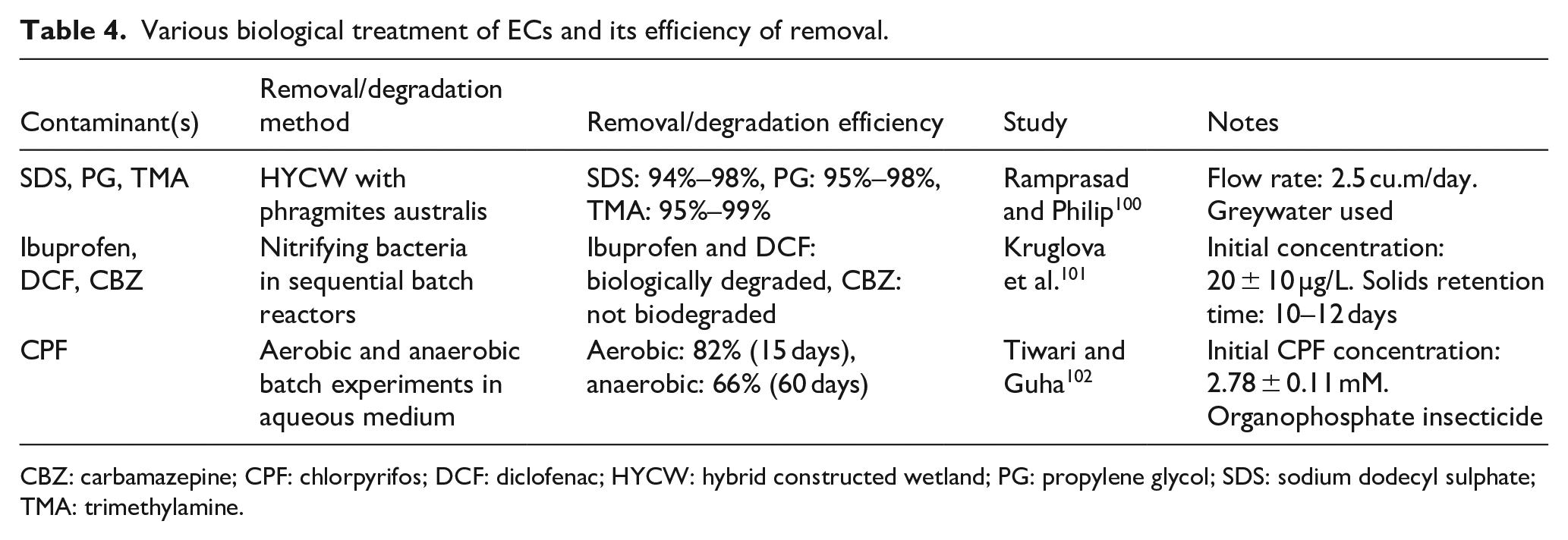
CBZ: carbamazepine; CPF: chlorpyrifos; DCF: diclofenac; HYCW: hybrid constructed wetland; PG: propylene glycol; SDS: sodium dodecyl sulphate; TMA: trimethylamine.
Advanced oxidation processes (AOPs)
AOPs are chemical treatment techniques that produce hydroxyl radicals (•OH) which can decompose various organic pollutants. Ozone is an effective oxidant that can degrade various EOCs, including pharmaceuticals (Table 5). Its application has been successful in achieving significant reductions in contaminant concentrations; however, it may produce harmful by-products. UV/H2O2 and Fenton’s Reagent—these AOPs utilise ultraviolet light or iron catalysts to generate hydroxyl radicals that oxidise organic pollutants effectively. Studies have indicated that these methods can mineralise EOCs efficiently but may require careful control of operational parameters to minimise by-product formation (Figure 13).
Emerging contaminants and its removable efficiency by AOP.

BOD5: biochemical oxygen demand, measured over 5 days; COD: chemical oxygen demand; DBCP: 1,2-dibromo-3-chloropropane; DCF: diclofenac; HRT: hydraulic retention time; NSAID: non-steroidal anti-inflammatory drug; O3: ozone; PCO: photocatalytic oxidation; SMX: sulfamethoxazole; WO3: tungsten trioxide; TOC: total organic carbon.
Membrane filtration
Membrane technologies have gained prominence for their capacity to segregate pollutants according to size and charge. Reverse osmosis (RO) membranes can efficiently eliminate many pollutants, including EOCs, via size exclusion and rejection processes. However, membrane fouling remains a significant challenge that can reduce operational efficiency and increase costs. Nanofiltration (NF) membranes have demonstrated effectiveness in removing smaller organic molecules and some ions from wastewater. Their performance can be improved through pre-treatment processes that reduce fouling potential (Figure 13).
Chemical precipitation
Chemical precipitation entails the addition of reagents to wastewater that interact with pollutants to generate insoluble precipitates. The heavy metal removal technique has shown effective for extracting heavy metals from wastewater, although it is less efficient for organic contaminants such as EOCs. The challenge lies in ensuring complete precipitation without generating toxic by-products or secondary waste (Figure 13).
Solutions for managing emerging organic contaminants
Table 6 examines a number of studies that discuss innovative methods for eliminating different kinds of pollutants. These studies are discussed according to the kinds of other than MXene-based biocomposites and methodologies used, such as biological and physicochemical methods.
List of materials used for the removal of emerging organic contaminants from wastewater, highlighting the contaminants targeted.

COD: chemical oxygen demand; EO-O3: electrochemical oxidation and ozonation; H2O2: hydrogen peroxide; MB: methylene blue; MBR: membrane bioreactor; N: nitrogen; NF: nanofiltration; O3: ozone; PBA: phenylboronic acid; PMS: peroxymonosulfate.
Limitations of conventional treatment technologies in EOC removal
Pharmaceuticals, personal care items, industrial chemicals, insecticides, and dyes are examples of emerging organic contaminants (EOCs) that are frequently difficult for conventional wastewater treatment systems to properly remove. The following are some of the main drawbacks of these conventional approaches (Figure 14).

Diagram showing the main drawbacks of these conventional approaches.
Inefficiency in removing EOCs
It has been demonstrated that, in comparison to other contaminants, many traditional techniques, including biological treatment systems and activated sludge processes, are less successful at eliminating EOCs. For example, biological processes can lower total nitrogen (TN) and total phosphorus (TP), but because of their intricate chemical structures and susceptibility to biodegradation, they frequently fall short in addressing EOCs. Depending on operational parameters like hydraulic retention time (HRT) and sludge retention time (SRT), the effectiveness of traditional systems can vary greatly. Variations in influent properties may result in uneven EOC elimination rates. 128
Formation of by-products
Hazardous Byproducts: Hazardous byproducts may be produced as a result of certain treatment procedures. Advanced oxidation processes (AOPs), for instance, have the potential to produce reactive intermediates that are much more hazardous than the initial pollutants. In a similar vein, chlorination may result in chlorinated byproducts that present further environmental hazards. 129
Operational challenges
Membrane fouling: Reverse osmosis and nanofiltration are two membrane-based technologies that are susceptible to fouling, which lowers their effectiveness and raises operating costs because they must be cleaned or replaced frequently. Their long-term suitability for treating wastewater with elevated EOC concentrations is limited by this problem.
High energy consumption: Due to their high energy requirements, processes such as electrodialysis and reverse osmosis are not as economically feasible for large-scale applications. Their extensive adoption may be discouraged by the high operating expenses linked to energy consumption. 128
Chemical dependency and waste generation
A lot of traditional techniques use chemical flocculants or coagulants, which can create secondary waste streams that need to be treated or disposed of further. This makes the entire therapy process more complicated and expensive. Production of Sludge: Biological treatments frequently produce an excess of sludge that needs to be handled and disposed of, adding another level of difficulty to wastewater management. Limited Applicability for Low Concentration Contaminants. Since many EOCs are found in wastewater at extremely low quantities, it is challenging to eliminate them using traditional techniques that are not intended for the removal of trace-level contaminants. Depending on the particular pollutants and their quantities, technologies such as biosorption can exhibit varying degrees of efficiency.
Regeneration issues
The regeneration capabilities of traditional adsorbents and resins used in ion exchange processes are frequently restricted. Instead of effectively regenerating after they are saturated with contaminants, they might need to be replaced at a high expense.
Regulatory issue
Regulations pertaining to different groups of emerging contaminants (ECs) are limited. Due to a lack of toxicological evidence, ECs are not properly regulated in many countries. Only a small number of pesticides are subject to regulatory limits for drinking water in India, according to Indian limits (IS 10500). The WWTPs and water treatment facilities are not checked for ECs. The World Health Organisation (WHO) has released comprehensive regulatory guidelines for a number of EC classes. Forty-five priority compounds with environmental quality standards that must be followed in aquatic environments have been designated by the European Union (EU) Water Framework Directive.
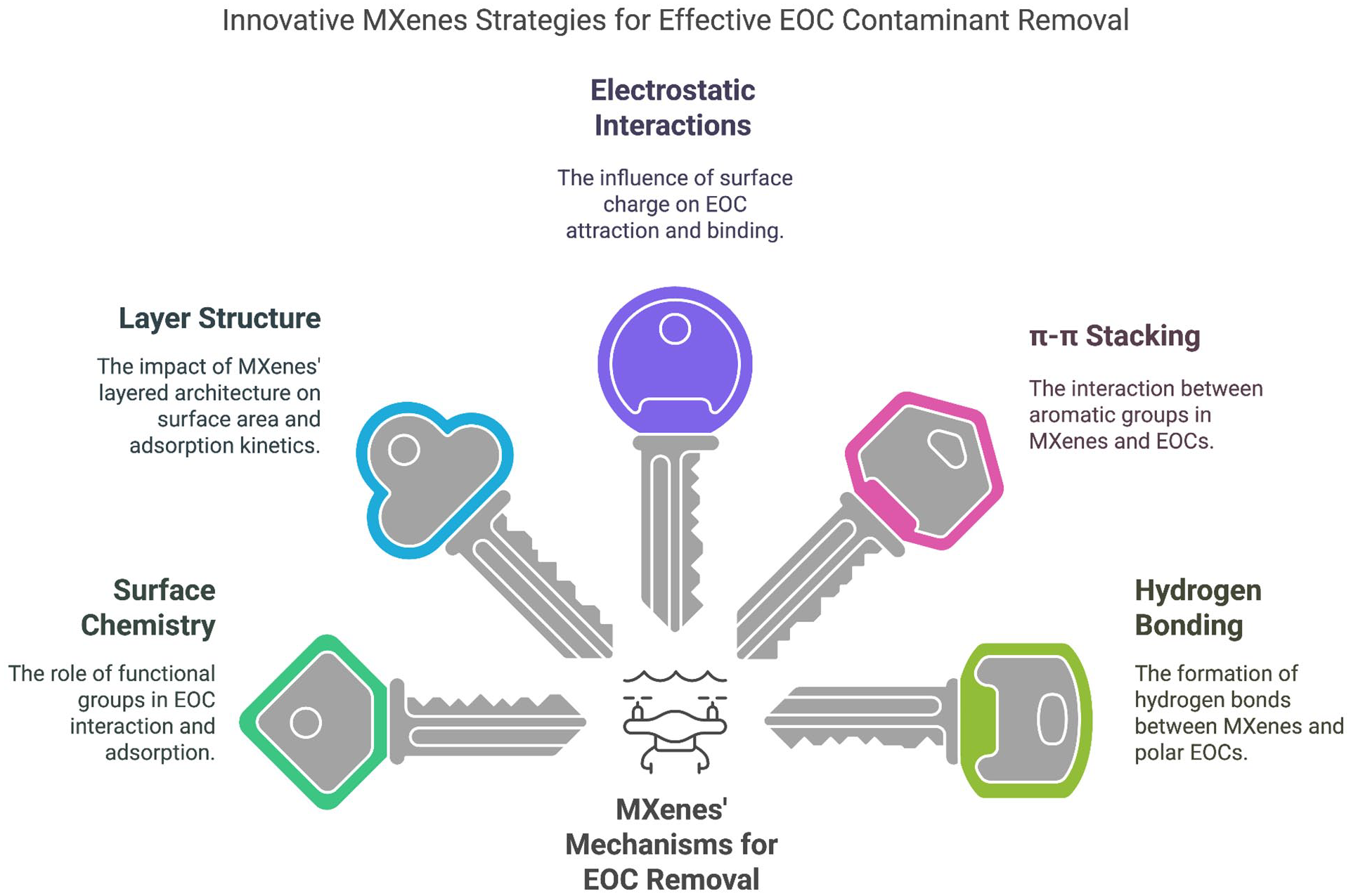
Mechanisms of EOC removal by MXenes
MXenes, a category of two-dimensional (2D) transition metal carbides and nitrides, have drawn considerable interest in recent years owing to their remarkable features, rendering them potential materials for diverse applications, including the elimination of emerging organic pollutants (EOCs). The distinctive characteristics of MXenes, especially their surface chemistry and layered architecture, are essential for their efficacy in eliminating EOCs. Due to the distinctive architectures of MXenes-based adsorbents, which enable the adsorption of various environmental contaminants, it is crucial to comprehend the mechanisms of adsorption and the interactions between adsorbents and pollutants. 130 To comprehend the precise probable uptake mechanism, it is necessary to conduct several spectroscopic approaches, adsorption isotherms and kinetics, as well as theoretical simulations employing density functional theory (Figure 15). The primary outcome of the interaction between organic pollutants and the functional groups of MXenes is the adsorption of contaminants onto the materials. Nonetheless, it was believed that the system’s adsorption efficiency was considerably influenced by physical characteristics, including hydrate diameter and agglomeration kinetics. The charges on both the adsorbent and the adsorbate are essential, regardless of whether the pollutant is organic or inorganic, as electrostatic interaction is the primary mechanism of adsorption. 131 An equilibrium of solute adsorption between the solution and the adsorbent is attained as the adsorption process advances. The subsequent formula was employed to determine the adsorption quantity of the molecules (qe, mmol/g at equilibrium):

Innovative MXene strategies for effective EOC contaminant removal.
In this context, V represents the solution volume (L); M denotes the mass of monolithic adsorbents (g); and Co and Ce indicate the initial and equilibrium adsorbate concentrations, respectively.
As a function of the equilibrium concentration in the bulk solution at a constant temperature, an adsorption isotherm shows how much solute is adsorbed per unit weight of adsorbent. Langmuir and Freundlich adsorption isotherms are commonly used to describe adsorption data.
The formula for the Langmuir equation is:

Ce denotes the equilibrium concentration of solute (mmol/L), qe signifies the amount of solute adsorbed per unit weight of adsorbent (mmol/g of clay), Xm represents the adsorption capacity (mmol/g), or monolayer capacity, and b is a constant (L/mmol).
The Freundlich isotherm characterises heterogeneous surface adsorption. The distribution of energy of adsorptive sites in the Freundlich isotherm approximates the real scenario using an exponential function. The intensity of energy at the adsorptive sites influences the rates of adsorption and desorption. The Freundlich equation is expressed as:

Here, k (mmol/g) and 1/n denote the constant parameters of the system.
Surface chemistry
Functional groups such as hydroxyl (–OH), carboxyl (–COOH), and amino (–NH2) on MXenes surface help to explain their increased surface reactivity. By means of multiple mechanisms—including electrostatic interactions, π–π stacking, and hydrogen bonding—these functional groups can interact with EOCs, therefore enabling the adherence of EOCs to the MXene surface. Changing the functional groups lets MXenes’ surface chemistry be tailored to improve their adsorption effectiveness for particular EOCs. 132
Layer structure
Weak van der Waals interactions split the transition metal carbide or nitride layers, forming MXenes’ layered structure. This multilayer construction allows high surface areas, which is beneficial for adsorption uses. Changing the interlayer gap between the MXenes layers permits EOCs with varying molecular sizes to be accommodated. Moreover, MXenes’ layered structure helps EOCs to diffuse into the material, hence improving the adsorption kinetics. 132
Interactions of electrostatic nature
MXenes’ surface charge can be changed by changing the functional groups or adding dopants, therefore influencing their electrostatically interactions with EOCs. MXenes with a positive charge, for instance, can attract negatively charged EOCs; those with a negative charge can attract positively charged EOCs. This electrostatic attraction could help EOCs to adhere better to MXenes surface. 132
π–π stacking
MXenes with aromatic functional groups—including pyridyl or phenyl groups—can interact with EOCs having aromatic rings by π–π stacking. This non-covalent interaction helps EOCs with planar molecular structures find it simpler to adsorb onto the MXenes surface.
Hydrogen bonding
EOCs with amino, carboxyl, or hydroxyl functional groups can create hydrogen bonds with MXenes having those functional groups. This hydrogen bonding might help EOCs—especially those with polar functional groups—to be more easily adsorbed onto the MXene surface.
Challenges and future perspectives
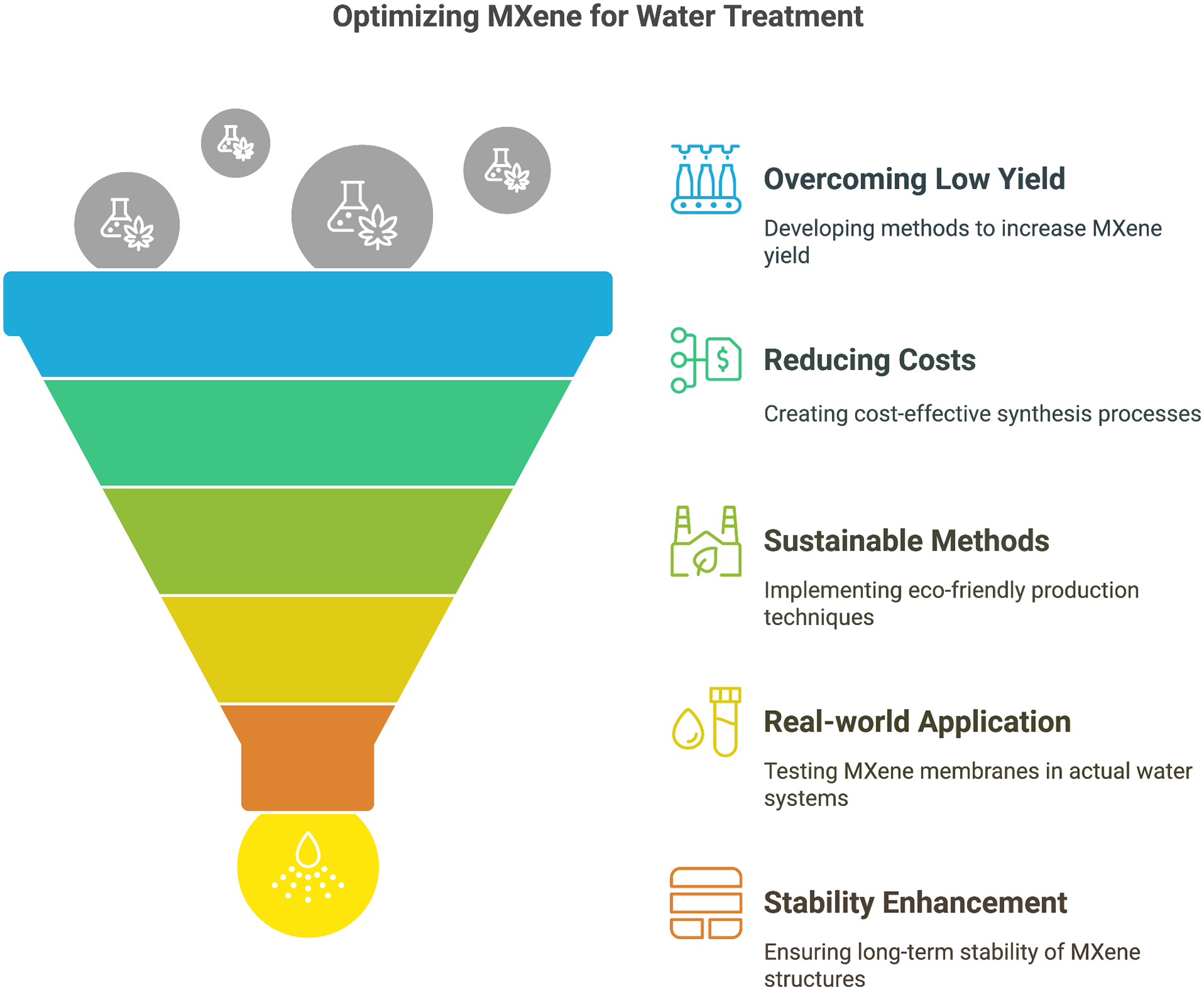
MXenes and MXene-derived materials, distinguished by their unique optical, magnetic, mechanical, and electrical properties, exhibit significant potential for many environmental applications. These encompass membrane-based ion separation, solar desalination, pervaporation desalination, and capacitive deionisation. 133 Their two-dimensional structure enables excellent application in filtration membranes, capacitive deionisation, photocatalysis, electrochemical separations, and as highly selective and reusable adsorbents for organic dyes and pharmaceuticals in water treatment. Nonetheless, numerous obstacles persist prior to the widespread adoption of these materials. Increasing production capacity and guaranteeing commercial feasibility for large-scale water treatment are significant challenges. Stacking MXene nanosheets into layered membranes can improve selectivity and permeability for desalination 130 ; nevertheless, challenges such as membrane swelling in water and restricted stability in aqueous settings need to be resolved. Moreover, existing MXene synthesis techniques frequently exhibit low yields, high costs, and the use of toxic chemicals, which raises environmental and health issues. Consequently, the advancement of more straightforward, economical, and environmentally sustainable synthesis methods is an essential research imperative.
Additional research is required to comprehensively evaluate the toxicity, biosafety, cytotoxicity, and biocompatibility of MXenes, along with their long-term environmental effects. The aggregation of MXene particles might diminish adsorption efficiency and surface area; therefore, comprehending and regulating their surface chemistry is crucial. While many MXenes have antimicrobial properties, rendering them appealing for water disinfection and anti-biofouling membranes, further investigation is necessary to elucidate their bactericidal mechanisms. 134 Theoretical investigations, including discrete Fourier transform simulations, can forecast MXene properties pertinent to desalination and environmental cleanup; nevertheless, these forecasts require experimental validation. Furthermore, it is essential to synthesise novel MAX phase precursors to broaden the spectrum of accessible MXene structures and to enhance the comprehension of the mechanisms influencing their etchability. Computational techniques, such as ab initio simulations, can elucidate exfoliation processes and inform the formulation of novel etching procedures for non-aluminium-based MAX phases. 135
MXenes are recognised for their hydrophilic surfaces, metallic conductivity, and adaptable surface chemistry; nonetheless, enhancing their stability is essential for practical applications. Functionalisation techniques, such as silylation or the incorporation of certain chemical groups, have demonstrated the capacity to improve the stability, dispersibility, and surface characteristics of MXenes. Functionalising Ti₃C₂ MXenes with phenylsulfonic groups enhances their dispersibility and augments their specific surface area. MXenes can be integrated with other materials, such as graphene oxide, to create hybrid structures that exhibit improved mechanical and electrical properties.
In conclusion, whereas MXenes present significant potential for improved water treatment and environmental remediation, further study is needed to overcome problems related to synthesis, stability, toxicity, and functionalisation to fully exploit their capabilities (Figure 16).
Important challenges in MXenes-based water treatment.
Conclusion and future outlooks
MXenes are highly versatile materials with notable uses in energy storage, electromagnetic shielding, gas and biosensing, photoelectrochemical catalysis, and water treatment, thanks to their large specific surface area, robust chemical stability, excellent thermal and electrical conductivity, antimicrobial effects, hydrophilicity, strong adsorption selectivity, and outstanding conductivity. Their easy surface functionalisation has sparked considerable research interest across fields such as environmental adsorption, biomedical applications, catalytic degradation, and energy storage. Moreover, MXenes offer flexibility and tunability in their composition and can be engineered with extremely thin layers for specific needs. While Ti3C2Tₓ is the most commonly studied MXene for water and wastewater treatment, there is a pressing need to create novel MXene structures and expand their environmental remediation roles—especially for MXene composites and functionalised MXenes. With surface modification, MXenes are emerging as next-generation materials for water purification, demonstrating high recyclability, enhanced stability, and superior adsorption capacity when conditions are optimised. Their unique physicochemical properties and ultrathin layered structure also provide powerful antimicrobial benefits, further highlighting their potential for advanced water and wastewater treatment applications.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

