Abstract
Aim:
This study aimed to assess the time-dependent water sorption, solubility and hygroscopic expansion of experimental resin composites modified with three different types of ion-leachable glasses (ILGs): 45S5 Bioglass (BG), Fluoride-containing glass (F9) and Experimental fluoride-phosphate glass (F9X), incorporated in varying weight percentages (5%, 10% and 15%).
Materials and Methods:
A 50:50 Bis-GMA/TEGDMA-based resin matrix was loaded with each ILG type in 5, 10 and 15 wt% and compared against a control without filler. Disc-shaped specimens (
Results:
All ILG-containing composites showed significantly increased water sorption compared to the control, with the BG-15 group demonstrating the highest sorption (3.37% ± 0.09) and expansion. Solubility increased with ILG concentration, especially in the BG and F9X groups. Hygroscopic expansion correlated positively with water uptake. No significant changes were observed in specimen mass after desorption in low filler groups.
Conclusion:
The incorporation of ILGs into resin composites significantly altered their water uptake and dimensional stability. While these effects could compromise long-term mechanical properties, the resulting hygroscopic expansion may aid in reducing microgaps and secondary caries at restoration margins. Veneering ILG-containing composites with conventional materials is recommended to limit degradation. These findings contribute novel insights into time-dependent dimensional behaviour of bioactive composites.
Keywords
Introduction
The continuous releases of bioactive ions from restorative dental materials have the potential to delay or inhibit secondary caries at the tooth/filler interface.1
–4 Fluoride has been integrated into different forms due to its anti-cariogenic properties. Fluoride in the form of
Resin composites are widely used restorative materials in modern dentistry due to their aesthetic appeal and favourable handling characteristics. However, one of the persistent challenges affecting their clinical performance is the degradation of marginal integrity over time, which may lead to secondary caries and premature restoration failure. Among various approaches to mitigate this issue, the incorporation of ion-leachable glasses (ILGs) has emerged as a promising strategy due to their ability to release therapeutic ions, including calcium, phosphate and fluoride, which may contribute to remineralization and antibacterial activity.
Water sorption and hygroscopic expansion are key properties that significantly affect the mechanical properties of resin composites, 10 and are critical for the material`s long-term success. Resin composites (RC) should ideally be impenetrable to fluids. In oral environments, however, RC are violated with continuous exposure to oral fluids and may release harmful, unreacted monomers, consequently forming gaps and voids that are favourable for bacterial growth. 11 The quantity of water sorption by RC depends on the aggressiveness of the oral environment, copolymer structure, filler composition and geometry, coupling agent and degree of conversion of the resin composites.12,13 Water uptake by RCs after a prolonged period can cause silane hydrolysis in the matrix-fillers interface, 14 along with masticatory forces causing internal micro-cracks and failure of the restoration.11,12
Ion-leachable glasses such as 45S5 Bioglass (BG), fluoride-containing glasses (F9) and phosphate-modified experimental glasses (F9X) have shown potential to interact favourably with the oral environment. These fillers not only aim to enhance biological interactions but also influence water-related behaviours such as water sorption, solubility and hygroscopic expansion. These properties, however, are double-edged: while the release of ions may provide bioactivity, excessive water uptake and dimensional changes may adversely affect mechanical integrity and longevity.
Within an oral environment, RC can experience a loss in mass due to the leaching of unreacted monomer15,16 and a gain in mass attributed to water uptake. The latter normally compensates for the loss in mass and leads to an overall increase in mass and volumetric expansion. Resin composites undergo progressive bulk expansion until they reach a plateau.11,17 Water sorption can change the modulus of elasticity of the polymer in resin composites. This alteration in elasticity, along with hygroscopic volumetric expansion, is considered partly compensate the undesirable polymerizing shrinkage 18 that developed during and briefly after the curing process of dental resin composites. Water sorption and volumetric expansion, on the contrary, requires weeks to partly compensate for the polymerization shrinkage. However, the continuity of water uptake may lead to alternative deleterious stresses.18,19
Previous studies, such as those by Oral et al. 29 and Par et al. 30 have investigated the physical properties of bioactive or ion-leachable glasses incorporated in dental composites. While these studies highlighted issues related to water uptake and mechanical deterioration, they either used silanised fillers or short-term immersion protocols. Moreover, time-dependent dimensional behaviour—particularly hygroscopic expansion—was not a primary focus. The current study aims to address these gaps by systematically evaluating the long-term water sorption, solubility and volumetric expansion of composites with varying ILG types and concentrations.
Volumetric hygroscopic expansion of resin composites has an important role in determining the durability of the material. Yet, this property has no ISO test for it, and has gained limited attention from researchers. In 2017, the Academy of Dental Materials (ADM) guidance held hygroscopic expansion as key property value of 1. The academy also valued the laser scanning technique as the most feasible way to measure the expansion of the material (rank 1). 20
This study addresses this gap by evaluating the time-dependent behaviour of resin composites loaded with varying concentrations of three ILG types. Specifically, the study aims to:
Quantify the water sorption and solubility of experimental ILG-based composites.
Assess hygroscopic expansion and its correlation with water uptake.
Explore the potential implications for marginal gap sealing and clinical relevance in preventing secondary caries.
By correlating these behaviours over an extended period using a modified ISO 4049 protocol, this work seeks to provide meaningful insights into the clinical translation of ILG-filled resin composites and guide their safe application.
Materials and methods
Formulation of ion-leachable resin composites
The compositions studied are presented in Table 1, the ion-leachable resin composites (IL-RCs) were formulated, and after fabrication, the IL-RCs were stored in lightproof containers at 5°C, and re-mixed using a speed mixer for 5 min prior to the experiments.
The formulated resin composite groups with different glass fillers (constant filler loading of 72 wt%): inert barium borosilicate glass (BBS), bioactive glass 45S5 bioactive glasses (BG), Fuji IX® active glass (F9) and Fuji IX Extra® active glass (F9X).
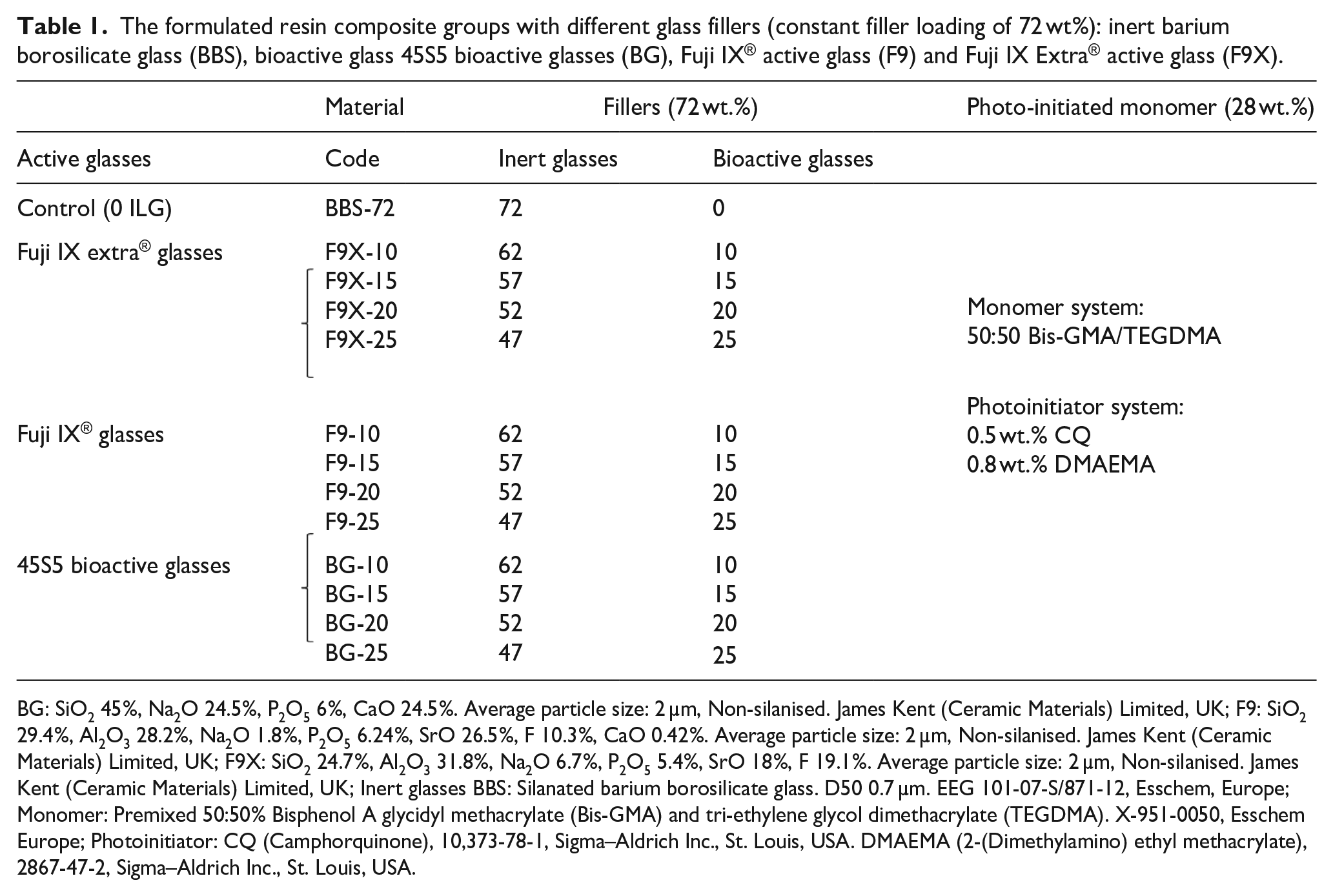
BG: SiO2 45%, Na2O 24.5%, P2O5 6%, CaO 24.5%. Average particle size: 2 µm, Non-silanised. James Kent (Ceramic Materials) Limited, UK; F9: SiO2 29.4%, Al2O3 28.2%, Na2O 1.8%, P2O5 6.24%, SrO 26.5%, F 10.3%, CaO 0.42%. Average particle size: 2 µm, Non-silanised. James Kent (Ceramic Materials) Limited, UK; F9X: SiO2 24.7%, Al2O3 31.8%, Na2O 6.7%, P2O5 5.4%, SrO 18%, F 19.1%. Average particle size: 2 µm, Non-silanised. James Kent (Ceramic Materials) Limited, UK; Inert glasses BBS: Silanated barium borosilicate glass. D50 0.7 µm. EEG 101-07-S/871-12, Esschem, Europe; Monomer: Premixed 50:50% Bisphenol A glycidyl methacrylate (Bis-GMA) and tri-ethylene glycol dimethacrylate (TEGDMA). X-951-0050, Esschem Europe; Photoinitiator: CQ (Camphorquinone), 10,373-78-1, Sigma–Aldrich Inc., St. Louis, USA. DMAEMA (2-(Dimethylamino) ethyl methacrylate), 2867-47-2, Sigma–Aldrich Inc., St. Louis, USA.
The 50:50 Bis-GMA/TEGDMA monomer system was selected for its well-documented balance between viscosity and cross-linking density. Bis-GMA provides structural rigidity, while TEGDMA enhances flow and polymerization efficiency. This specific ratio was chosen to simulate commercially relevant resin matrices and to allow for controlled water sorption behaviour. All ion-leachable glasses were used in their unsilanized form to permit optimal interaction with water and enable ion exchange. Silanization, while improving filler-matrix adhesion, would inhibit ion release which was central to the objective of this study.
Specimen preparation
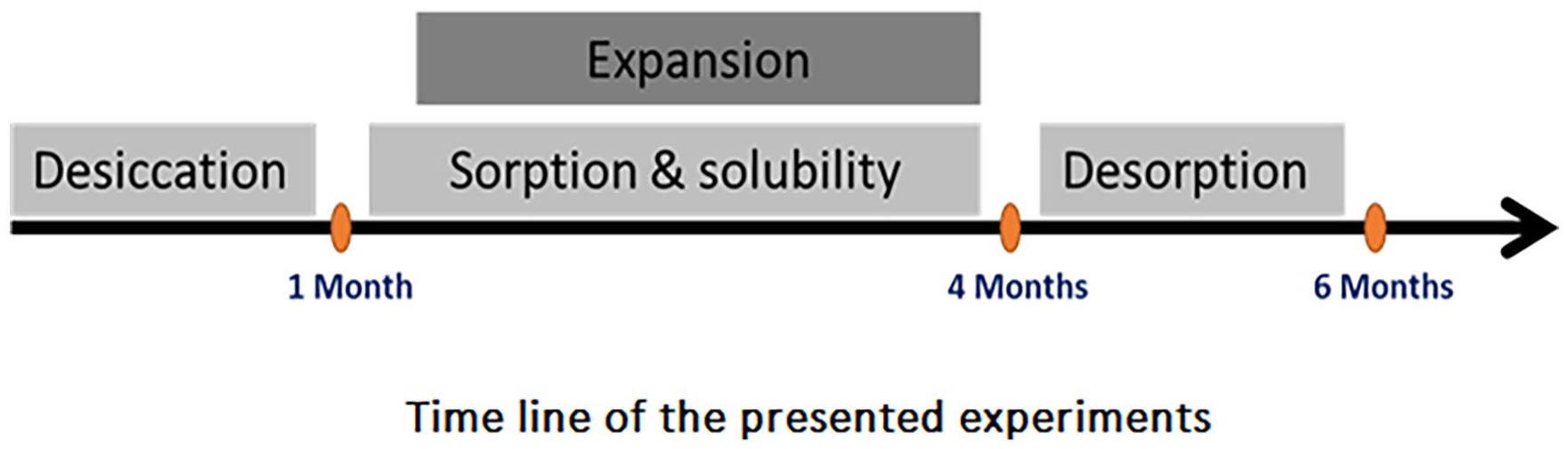
Water sorption, solubility and hygroscopic expansion were investigated using a modified version of ISO 4049. This study used a modified version of ISO 4049, adapted to allow long-term evaluation of dimensional changes over a 12-week immersion and 8-week desorption cycle. Additionally, specimen geometry and immersion intervals were adjusted to improve sensitivity in detecting gradual hygroscopic expansion, which is not covered in standard ISO protocols.
Teflon moulds with an internal diameter of 15 mm and a thickness of 2 mm were placed on top of a 2 mm thick glass slab and separated with a polyester strip (Mylar, Moyco Union Broach, USA). The IL-RC pastes were dispensed and overfilled in the moulds. The top surface of the mould was also covered with a polyester strip. Another glass slab (2 mm thick) was then placed on top of the mould and compressed to remove excess material and also minimize air-bubble entrapment.
Three specimens of each group (39 in total) were cured for 20 s using 5 overlapping sections from the top and bottom surfaces using a dental light cure unit (Elipar S10; 3M ESPE, USA). The specimens were then removed from the mould, finished (Dura white stone bur, Shofu, USA) and polished using polishing discs (Sof-Lex, 3M, UK).
Specimens were stored in separate glass vials filled with fresh silica gel. All vials were contained inside a desiccator at 37°C. The mass of the specimens was measured at 2-day intervals using an electronic microbalance (BM-252, AND company, Japan) with accuracy of 0.01 mg until the change in mass over a 24 h period was less than 0.1 mg.
After a constant mass was achieved (
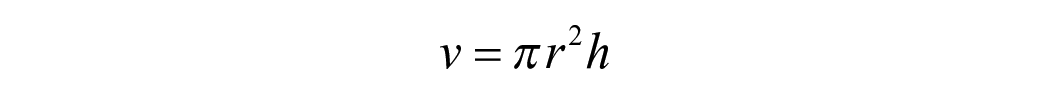
Sorption, solubility and hygroscopic expansion measurements
Sorption, solubility and hygroscopic expansion were measured at 1, 3, 7, 14, 21, 30, 40, 60 and 90 days after immersion in HPLC water. After the final measurement, specimens were subjected to a desorption cycle where specimens were placed in a 37°C desiccator with silica-gel and the mass of the specimens during the desorption cycle was measured at 3, 7, 14, 21, 30, 40 and 60 days. At 60 days, the final mass with change less than 0.1 mg over a 24 h period was recorded as the mass at the end of desorption cycle
For sorption and solubility measurements, specimens were removed from the vials, dried with filter paper until visible dry to the naked eye. The mass of the specimens was measured at each planned time points (outlined in the above paragraph,
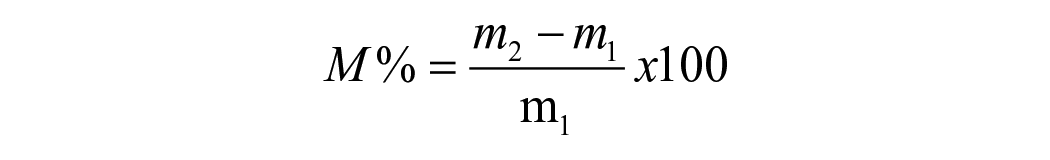
Sorption
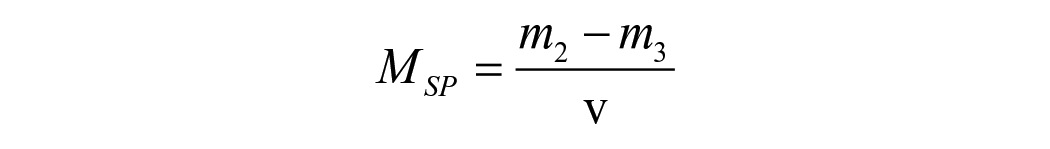
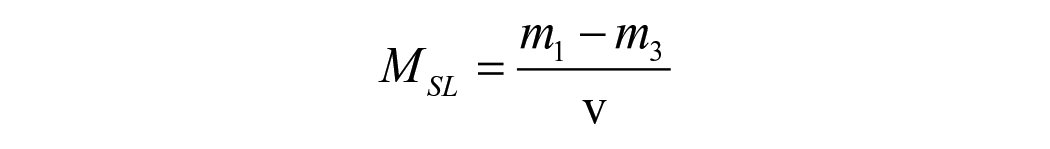
For hygroscopic expansion, the specimen’s dimensional change was taken immediately after the mass measurement at the same time intervals. The change in the horizontal diameter of each specimen was quantified using a laser scan micrometre system (LSM; measuring unit LSM-503a and Display unit LSM-6200, Mitutoyo Corporation, Japan). Each specimen was placed on a metal stub rotating 28 steps/s with the laser beam scanning the lateral edges of the specimens while rotating. The mean of 600 digital readings was used as the final value for each specimen. The percentage of radial expansion
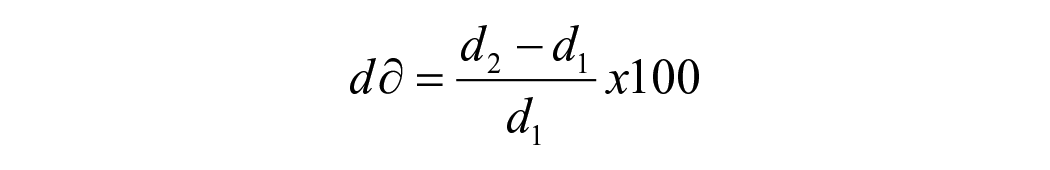
Where,
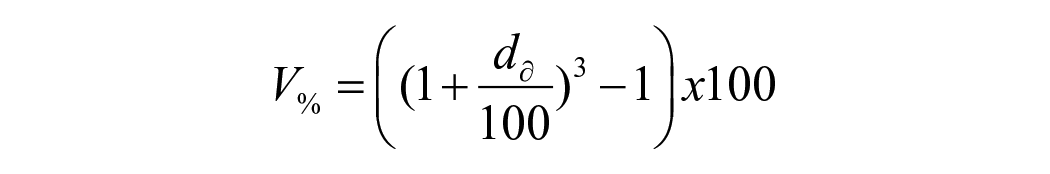
The data for all specimens was collected and analysed statistically using SPSS 22.0 (IBM SPSS Statistics, SPSS Inc., Chicago, IL, USA). Statistical analysis was conducted using one-way ANOVA to compare means among the 13 groups for each outcome variable (mass change, sorption, solubility and expansion). Degrees of freedom were calculated based on group number and sample size (df = 12, 26). Post-hoc comparisons were performed using Tukey’s test with α = 0.05. All
Results
Mass, sorption and solubility
Shapiro-Wilk’s test showed a normal distribution of variables (
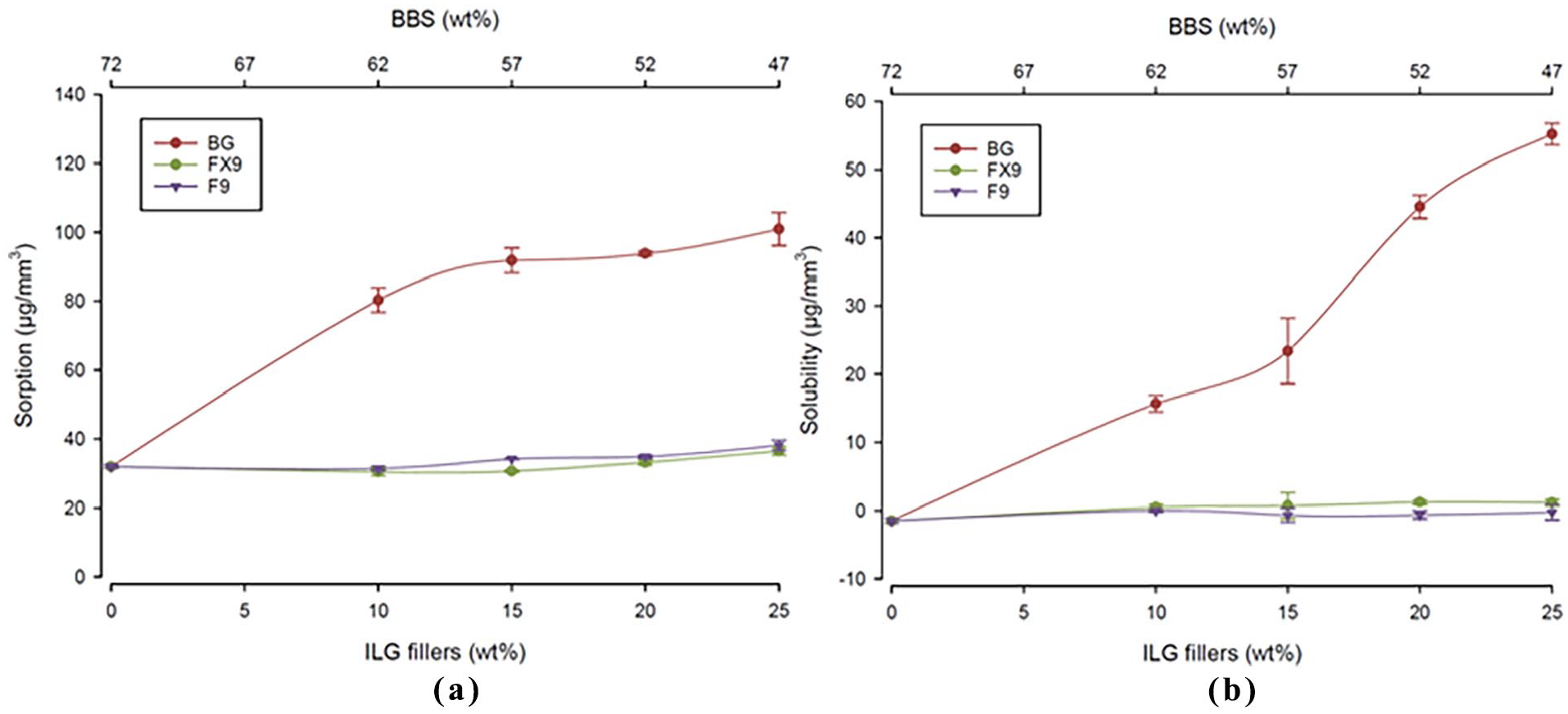
(a) Sorption and (b) solubility of the experimental resin composites after a 12-w immersion in water. The water sorption and solubility of F9X (green) and F9 (blue) resin composites was minimally affected by the increase in ILG content. While water sorption and solubility increased with the increase in BG concentration (red). The bottom and top
Mass change
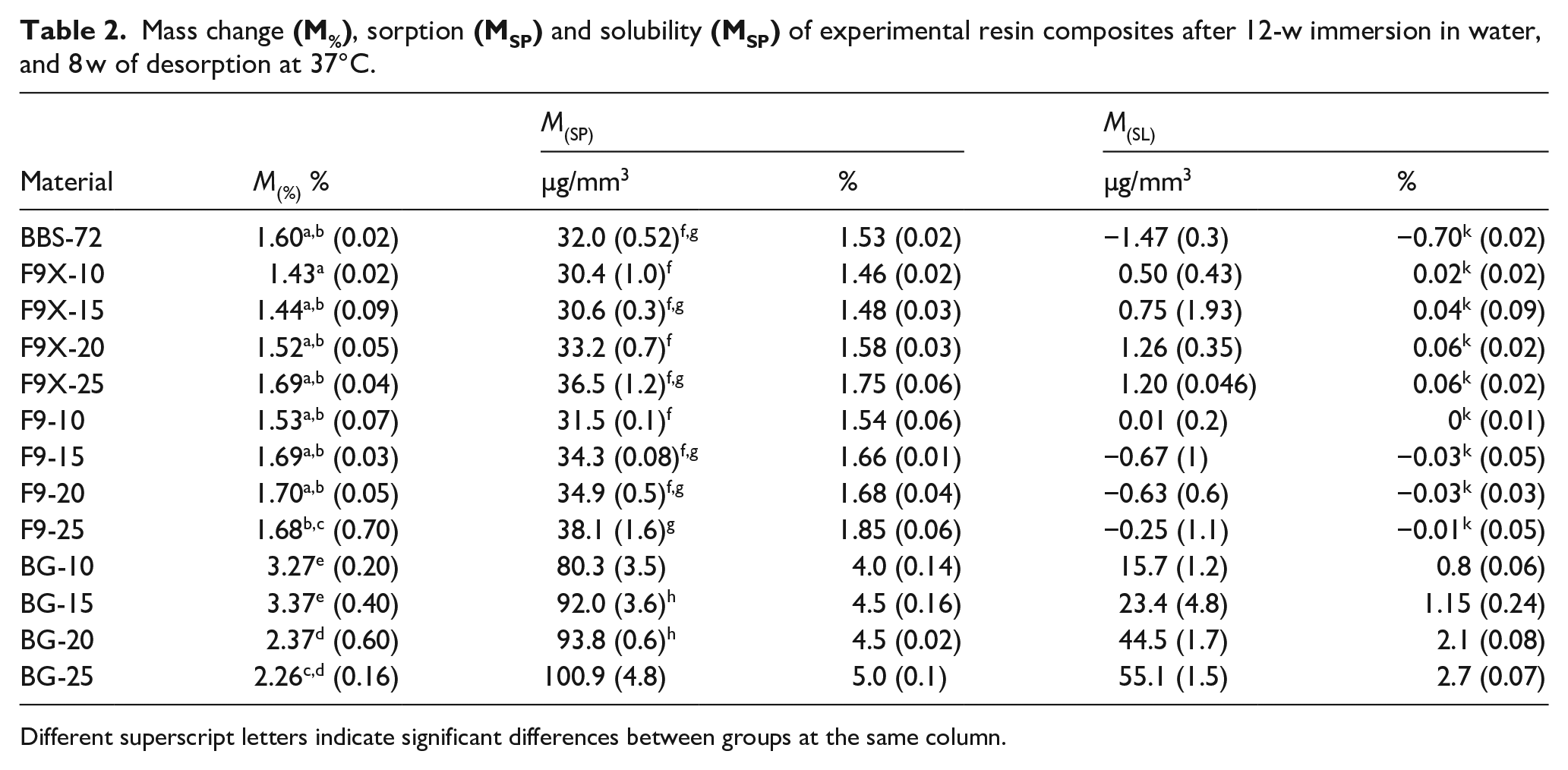
Different superscript letters indicate significant differences between groups at the same column.
Similar amount of water sorption and solubility was observed for F9X-RC and F9-RC (Table 2). Resin composite contains BG showed distinctly higher water sorption and solubility than other IL-RCs. F9X and F9-series resin composites showed similar behaviours to BBS-72 control group in sorption and desorption cycles. The materials showed a steady increase in mass for the first 3 w where they reached a plateau for the rest of the water sorption cycle (Figure 2).
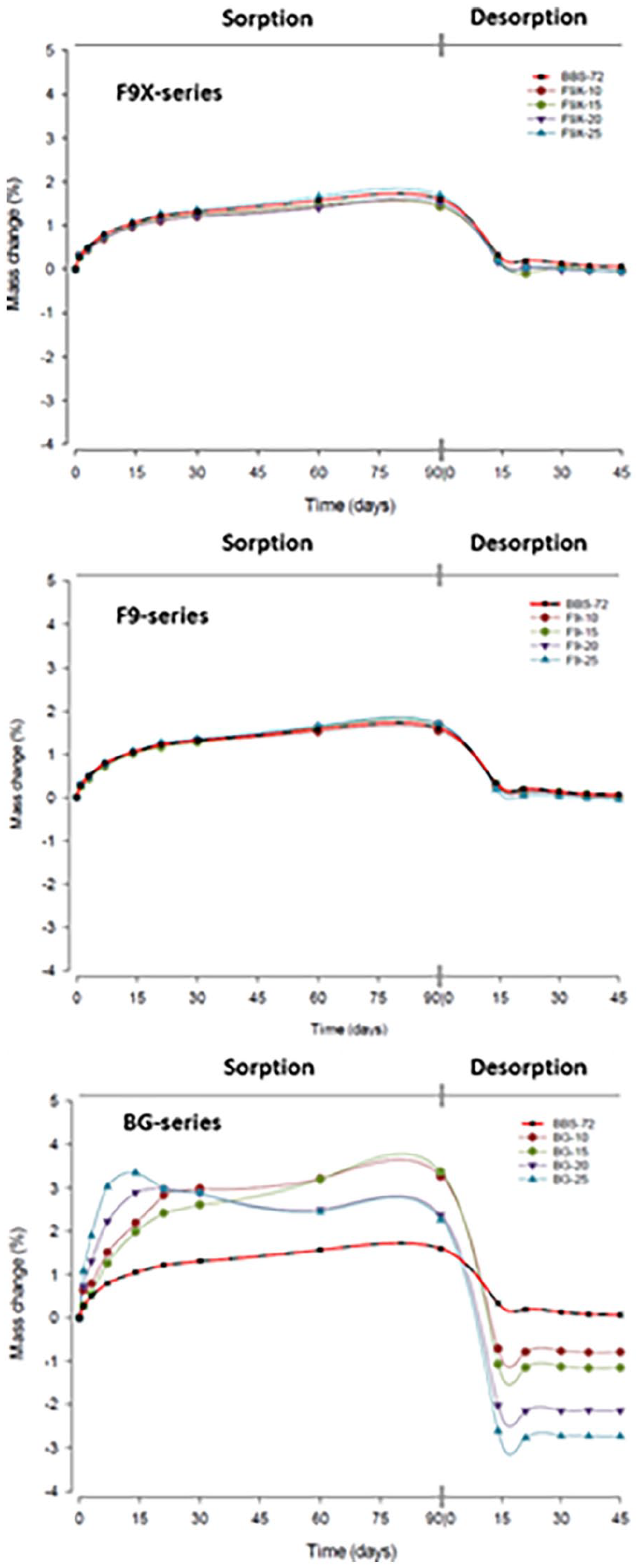
Mass change (%) of resin composite with BBS and different ILG fillers; F9X, F9 and BG after 12-w immersions in water and 8-w desorption. F9X and F9 resin composites expressed similar pattern in sorption and desorption cycles, where the mass changes was not affected by ILG type or content (wt%). BG-10 and BG-15 increased in mass for all sorption cycle, while BG-20 and BG-25 expressed mass increase in the first 2 w, followed with mass loss for the rest of sorption cycle.
The sorption for both fluoride release series (F9X- and F9-) ranged between 30 and 39 µg/mm3, with no statistically significance difference to the BBS-72 control group. The BG-series showed a higher sorption rate ranging from 4% to 5%. The highest sorption was 100.9 µg/mm3 found in BG-25, with F9X-10 resin composite has the lowest sorption of 30.4 µg/mm3.
BG-RCs demonstrated a distinctly different pattern with a more pronounced changes in mass than the counterpart BBS group, at all immersion time points (Figure 2). BG-10 and BG-15 showed continuous gradual increase in mass in the sorption cycle, while BG-20 and BG-25 expressed high mass increase for the first 2 weeks, an increase in mass followed with a reduction for the rest of the sorption cycle.
In desorption, F9X- and F9-RCs had a decrease in the mass over the first 2 w then mass remained constant over the rest of the period. Solubility of the experimental resin composites after 8 w ranged between −1.47 and 55.1 µg/mm3. F9X and F9-series had similar solubility with no significant difference to the BBS-72 control group (
The BG-series had a higher solubility positively correlated with BG filler proportions in the RC. The highest solubility was 55.1 µg/mm3 found with BG-25 and the lowest was −0.7 µg/mm3 for BBS-72 control group. The amount of ion leachable glasses had a quadratic relationship with the amount of water sorption; and linear relationship with the solubility of the materials as shown in (Figure 3). An optical image of the specimens after 90 days is shown in (Figure 4), colour changes was more clear with increasing ILG proportions, in which BG-series had higher changes in colour than the other groups.
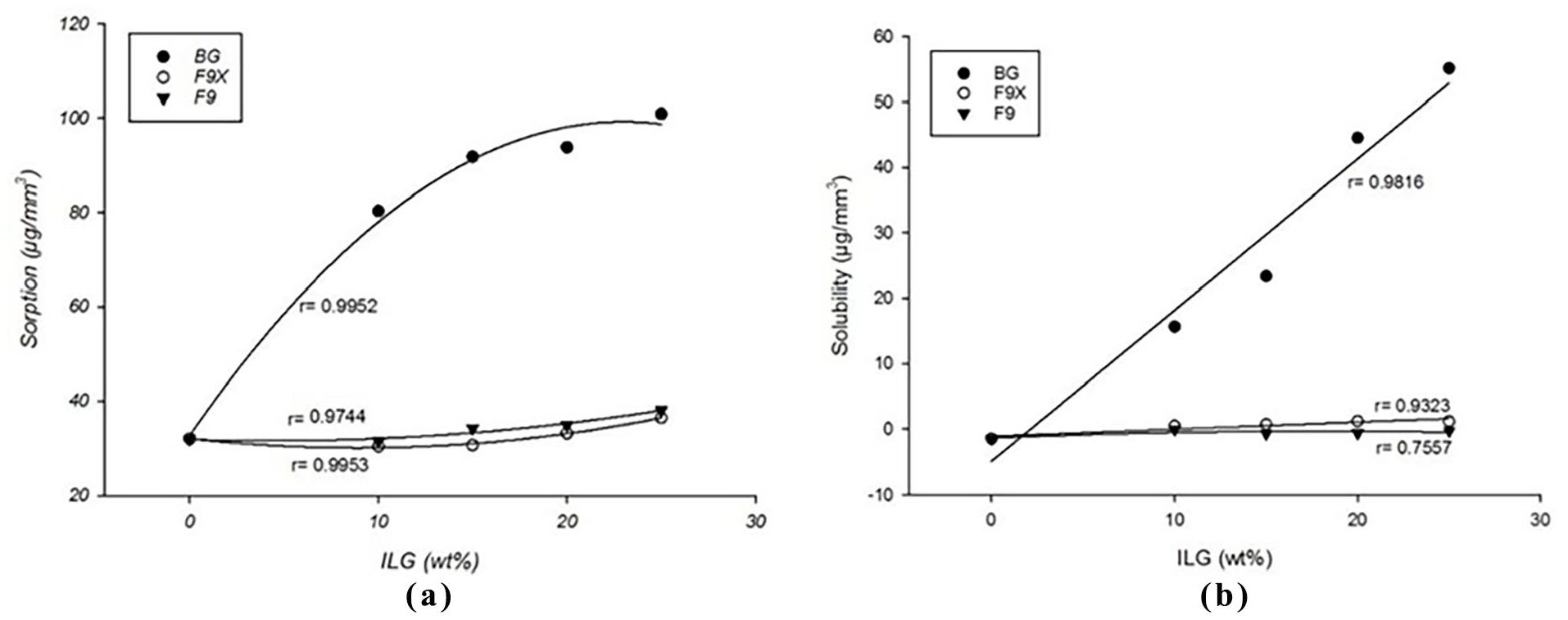
(a) Quadratic correlation between water sorption, (b) linear correlation between solubility and the amount of ion leachable glasses (wt.%).
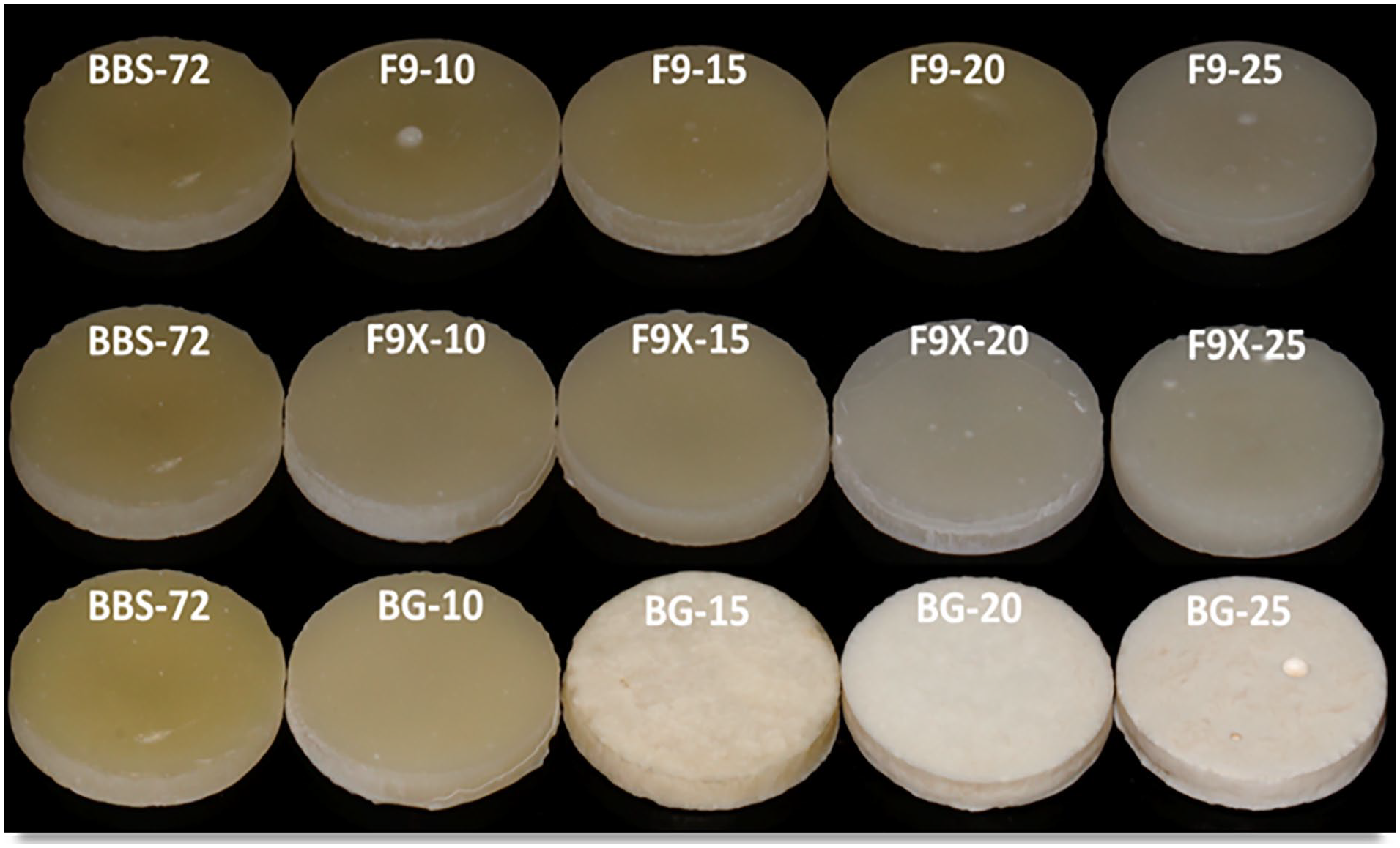
Visual inspection of colour changes in resin composites after 90 days of water immersion. F9-series displayed the lowest changes in colour, and BG-series had the highest change.
Different superscript letters within each column denote statistically significant differences between groups (one-way ANOVA,
A significant effect of filler type on water solubility was observed (
Hygroscopic expansion
Linear and volumetric expansion of the experimental ILG resin composites after 90 days of immersion in HPLC water are presented in (Table 3 and Figure 5). F9X- and F9-series resin composites showed significant expansion in the first 30 days, followed with minimal increase for the rest of the cycle. Interestingly, increasing in the proportion of F9X and F9 glasses (wt.%) did not influence the percentage of expansion (
Linear and volumetric expansion of IL-RCs after 90 days immersion in water.
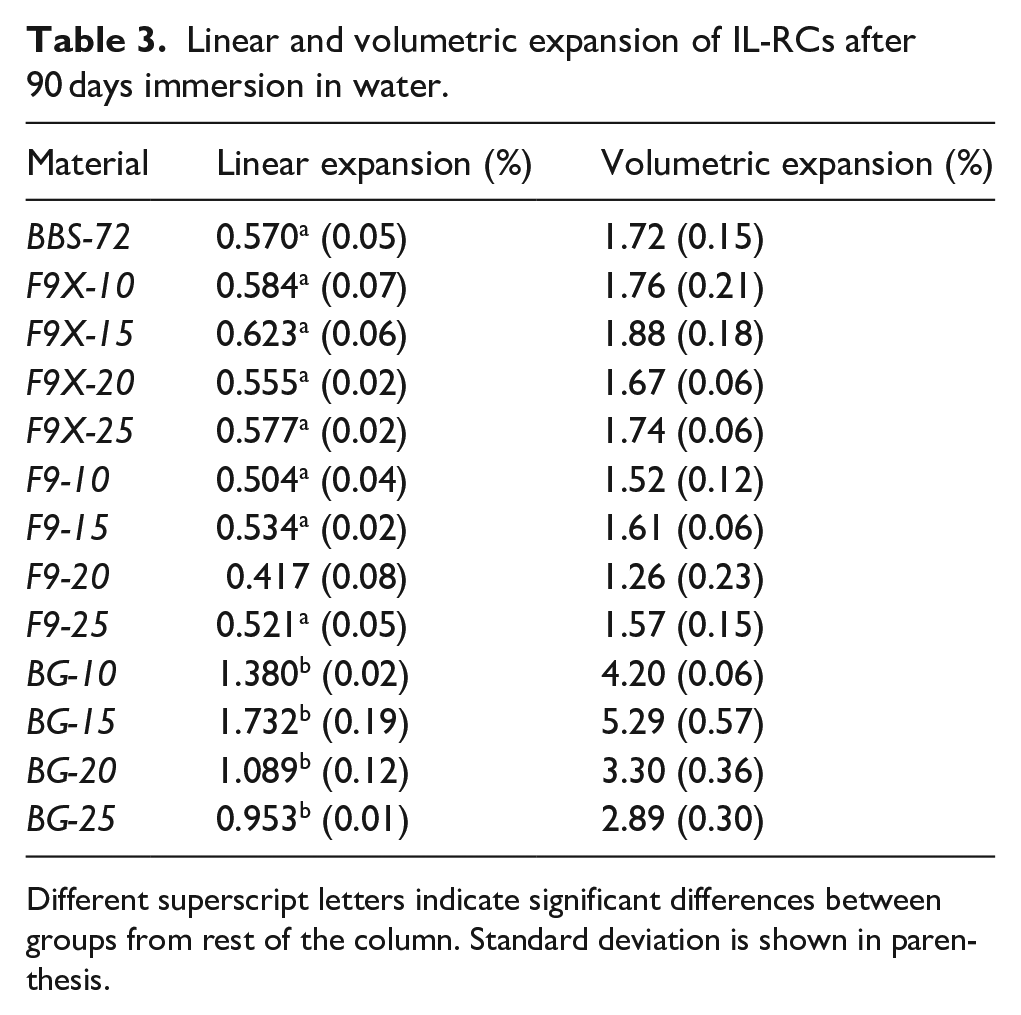
Different superscript letters indicate significant differences between groups from rest of the column. Standard deviation is shown in parenthesis.
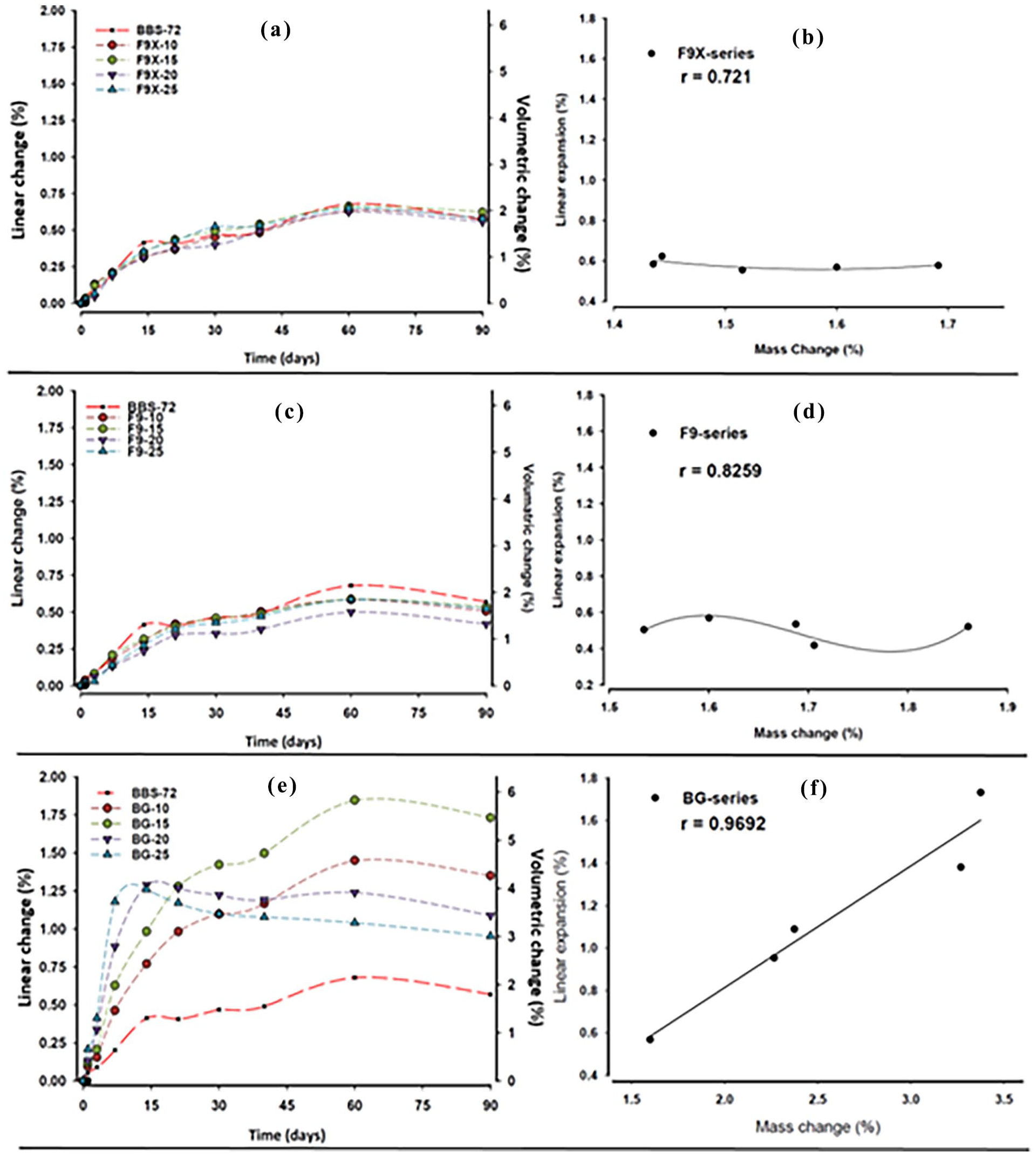
Linear and volumetric expansion of (a) F9X, (c) F9 and (e) BG resin composites over time. All resin composite specimens were immersed in HPLC water for 90 days. The relationship between the linear expansion and the changes in mass of the three resin composite groups are presented in (b), (d) and (f).
By contrasts, BG resin composites displayed different behaviours, with an expansion correlated to the BG content (wt.%). BG-10 and BG-15 displayed a sharp increase in the first 20-day, followed with a more steadily increase for the rest of sorption cycle. BG-20 and BG-25, on the other hand, had course of very rapid expansion in the first 15-day, followed with course of steadily shrinkage for the rest of the experiment. BG-series was statistically significant from BBS-72 (control, 0-ILG %) and the other IL-RCs (
Discussion
Study model
The aims of this study were to isolate and evaluate the solubility of ILG filler on RCs. The water up-take by resin composites is influenced by structural and environmental factors. The structural (intrinsic) factors such as matrix monomers, fillers solubility and matrix/filler interface have the largest influence on sorption. 21 There are also environmental (extrinsic) factors affecting water sorption, such as storage time and media,22–24 temperature 25 and applied stress. 26 Accordingly, all factors were kept standardize apart from the ILG filler contents.
Mass change
In an aqueous environment, conventional resin composites have the ability to absorb water and leach glasses and unreacted monomer, resulting in time-dependent mass and volumetric changes of the materials. BBS-72 (control) resin composite demonstrated similar behaviour to that of conventional resin composites, by means of gaining mass rather than losing.
27
This was also the case for F9X and F9 resin composites (
Mass changes in BG resin composites did not follow a constant pattern. Incorporating 45S5 bioactive glasses in low wt% (BG-10 and BG-15) showed an increase in the mass of resin composites (Figure 5). The mass changes over 60-day were 3.27% and 3.37% for BG-10 and BG-15, respectively. This increase in mass was the highest of all experimental groups. Yet the increase in mass dropped by adding more BG fillers in BG-20 and BG-25 (2.37% and 2.26%, respectively). This drop in mass was noticed after 30-day immersion in water, and may be linked to higher resin composites solubility caused by bioactive glasses. This behaviour of BG fillers in resin composites was also reported by Par et al., 30 where it was shown that 20 and 40 wt% of bioactive glasses reinforced with 50 and 30 wt% barium silica inert fillers (2:1), impeded in Bis-GMA:TEGDMA resin (60:40) have similar pattern in mass-drop up to 300-day of water immersion. 30 Similar behaviour was also found with S53P4 Bioactive glass contain composites. 29 One may conclude that resin composites with high soluble glasses have the tendency to reach a peak after mass increase, followed with drop rather than sustaining mass equilibrium. 29 Similar behaviour, though less pronounced, was found with self-adhering resin composites caused by monomer elution due to higher hydrophilicity of the monomer system. 31
Resin composites with F9X or F9 glasses reached equilibrium in mass after 14 days, regardless of ILG-fillers content. This may indicate that ion-leaching was limited to the first 2 w of immersion in water. The former suggests that any leaching continued after this time-point may have been very limited and masked by the amount of absorbed water. However, the fact that F9X- and F9-series resin composites have no statistically difference from the BBS-72 control group may indicate that the materials had no or uncountable ion-release. These results are in accordance with noticed behaviours in previous mechanical experimentation, in which F9X- and F9-series had no statistical reduction in flexural strength after 30 days of immersion in water. The presented results highlight the importance of analysing the ion-release of these glasses.
Sorption and solubility
Water sorption of resin composites depends on their composition such as the hydrophilicity of resin matrix, fillers and the presence of coupling agent. 32 The hydrophilicity of resin matrix is determined by the presence of polar functional group such as hydroxyl groups (−OH).29,33,34 The matrix system in the presented study consisted of 50:50 Bis-GMA/TEGDMA. Bis-GMA monomers have 2-Hydroxyl groups/molecule, which have the ability to attract and retain water by the formation of hydrogen bond. TEGDMA, in the other hand, is able to absorb more water owing to ether linkage. 33
The aforementioned ability of monomer systems to absorb water were majorly decreased in conventional resin composites by including less hydrophilic monomers such as Bis-EMA. 29 Another way to decrease water sorption is simply increase the amount of silanised fillers, thereby lowering the overall matrix-ratio in the resin composites. 32 However, reactive glasses require hydrophilic monomer to promote water permeability and initiate ion-leaching. For example, hydrophilic hydroxyethyl methacrylate (HEMA) was utilized in Xeno CF® Resin Composite (Sankin Dentsply, Japan) and in Reactmer® Giomer (Shofu Inc., Japan) to improve water permeability.
Coating fillers with coupling agents has the effect of decreasing the hydrophilicity of the material. Coating ILG fillers is difficult, since its ability to leach ions comes from water-contact. Oral et al. 29 attempted to study the effect of salinizing S53P4 bioactive glasses (315–1000 μm, SiO2 53 wt%, Na2O 23 wt%, CaO 20 wt% and P2O5 4 wt%) on water sorption and solubility, flexural strength and fracture toughness after 60 days of immersion in water. In their study model, 3, 6, 9 or 12 wt% of S53P4 bioactive glasses (particle size 315–1000 μm) were silanised with 2% MPS-silane and impeded in autopolymerising methyl methacrylate and ethylene glycol dimethacrylate (95:5, w/w) monomer system. The results showed increasing the quantity of un-silanised glasses reduced the weight after 60 days of water immersion, and material’s sorption increased proportionally with silanised glass filler-load. Salinization of S53P4 glasses resulted in significant reduction in solubility values, and had no significant different in flexural strength and fracture toughness values compared to un-silanised S53P4 resin groups. Unfortunately, the effects of salinizing BG glasses on the ion-release was not attempted. It is reasonable to say that increasing the content of unsilanised ion-leachable glasses would increase water sorption, shadowed with a higher solubility and mass loss. This was clearly affirmed from the sorption and solubility values of BG-RC.
Unlike Par et al., who observed sustained mass increase over 300 days, our BG-filled composites showed an initial peak followed by loss. This discrepancy may be due to differences in matrix composition and particle dispersion. 30
Resin composites containing two
Oral et al. found salinized bioactive fillers reduced solubility. Our use of unsilanized fillers allowed direct comparison of ion release–related solubility, explaining the higher values observed. 29
Sorption of the IL-RCs ranged between 29 and 106 µg/mm3, with none of the experimental resin composites exceeding the limits for sorption after 1 -w (40 µg/mm3) set by ISO standards for resin composites. It is worth mentioning that the thickness of specimens used in this study was twice what ISO 4049 suggests which may have affected water sorption.
The majority of elution is related to unreacted free monomers. TEGDMA is the monomer that is most susceptible to elution due to its higher hydrophilicity and mobility. 36 Van Noort and Barbour reported that high filler contents with weak matrix-filler interface have the maximum negative effect on the stability of the material. 32 Thus, unsilanised bioactive glasses are expected to be the predominant cause of solubility along with free monomers, and solubility would increase with the ILG proportions.
BBS-72 (0-ILG wt.%) showed negative solubility (−0.7 %), and was not statistically different to F9 nor F9X-series resin composites. The negative solubility values could be attributed to incomplete dehydration of the materials with time. These values cannot conclusively indicate that no solubility occurred, yet hint at low solubility. Negative and low solubility could be explained by the formation of metal hydroxide which results from the hydrolytic reaction of metal elements and water. 33 By contrast, BG-series demonstrated solubility at least twice the limit set by the ISO standard. The solubility was positively correlated with the increase in BG filler quantity (wt.%). A possible explanation is that the increase in BG content causes more glass to dissolve and ions to leach, forming an easier path for water to extend far down the deep layers of resin composites; thus, causing more components to leach-out.
It is important to note that this study did not include a quantitative assessment of ion release from the incorporated glasses. Since the primary mechanism of bioactivity in ion-leachable fillers is through sustained ion exchange with the surrounding environment, future studies should employ techniques such as inductively coupled plasma (ICP) spectroscopy or fluoride-specific ion electrodes to evaluate ion release profiles over time. This data would complement the water sorption and solubility results and offer deeper insight into the material’s therapeutic potential.
Hygroscopic expansion
Optical scanning, 37 electronic or laser micrometres,38,39 microscopy 40 and some other techniques have been applied to measuring dimensional expansion.41,42 Non-contact laser micrometres with high accuracy and reproducibility were used in this study. The method has proven to be suitable for measuring small dimensional changes such as those encountered in resin composites.24,38
Linear expansion of the experimental resin composites was positively correlated to mass change. In conventional resin composites, expansion is multi-factorial, where monomer type, hydrophilicity and degree of conversion are the major influences on sorption and consequent expansion. 43 However, these reasons cannot be fully applied to ion-release resin composites as dissolving glasses create micro-voids to accommodate water with or without changing the dimensions of the material. 24
The specimens size of the F9 and F9X-series increased significantly during the first 14-day, followed by a gradual increase until the end of the experiment. Mass and volumetric changes were more pronounced in BG-resin composites. The most important findings were with BG-20 and BG-25, where the materials demonstrated a large increase in mass and volume for the 14-day, followed by a reduction (0.25%–0.5%) in mass, and shrinkage in volume. This behaviour is due to the same reasons outlined in the discussion on sorption above.
Clinical application of the results
Marginal gaps and secondary caries at the tooth-filling interface are the main reasons for RC failure. This study presents a possible approach to minimize the onset of caries by continuous leaching of ions able to disrupt bacterial activity. It has been reported that the hygroscopic expansion of RC may not only compensate for polymerization shrinkage but also partially close the marginal gaps. In a previous study using confocal microscopy, the hygroscopic expansion of seven different types of RC (e.g. Spectrum TPH®, Esthet-X®, Dentsply, Germany) was able to reduce the marginal gaps produced by polymerization shrinkage and improve the cavity sealing after 8 w of water immersion. 44 Although IL-RCs have shown higher expansion than some commercial resin composites, there is no definitive evidence on the ability of hygroscopic expansion to prevent or reduce the onset of caries and further studies are needed to validate this relation.
The increase in the hydrophilicity of the materials suggests these materials should be veneered with conventional resin-composites. This technique was suggested with other resin composite materials such as bulk-fill resin composites. Veneering ILG-resin composites can retain ILG-resin composites inert, where they only activate and start leaching ions when marginal gaps form and saliva reaches the down-layers of filling restoration.
Conclusion
This study demonstrated that incorporating ion-leachable glasses (ILGs) into resin composites significantly influences their water sorption, solubility and dimensional stability. Among the tested fillers, 45S5 Bioglass (BG) led to the greatest increase in both water uptake (up to 3.37%) and volumetric expansion (up to 5.29%), particularly at higher concentrations (15–25 wt%). These findings suggest that while BG-containing composites may enhance ion release and marginal sealing via hygroscopic expansion, they also pose a risk for structural degradation due to increased solubility (up to 55.1 µg/mm³).
In contrast, composites containing F9 and F9X
The observed positive correlation between water uptake and expansion implies a compensatory effect for polymerization shrinkage, potentially contributing to the reduction of marginal gaps—a known precursor to secondary caries. However, the clinical benefit of this mechanism remains to be confirmed.
Based on these outcomes, veneering BG-filled composites with conventional resin composites is recommended to limit exposure and control degradation, activating ion release only in the event of marginal compromise. Future studies should focus on quantifying ion release kinetics and evaluating antibacterial and remineralizing effects in vivo to fully assess the clinical viability of ILG-filled materials.
Footnotes
Correction (December 2025)
In this article, special collection heading has been corrected to “Biomaterials for Caries Prevention” from “Advanced Biomaterials for Regenerative Dentistry in Oral Health Care”.
Author Contributions
All the authors equally contributed to this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported via funding from Prince Sattam Bin Abdulaziz University project number (PSAU/2024/R/1445).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data presented in this study will be available on request from the corresponding author upon reasonable request.
