Abstract
Background:
As the major inorganic component of natural bone, nano-hydroxyapatite (n-HA) on its own is limited in its use in bone repair, due to its brittleness. Chitosan (CS) and sodium alginate (SAL) are used to reduce its brittleness and tendency to degradation. However, the compressive strength of the composite is still low, and its biological performance needs further study.
Methods:
Nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) composite was prepared via an in situ synthesis method. Further, we prepared the n-HA/SAL/CS self-setting bone repair material by mixing n-HA/SAL/CS powder with a curing liquid (20 wt.% citric acid). In addition, the in vitro bioactivity and cell cytotoxicity were also explored.
Results:
Transmission electron microscopy photos revealed that the n-HA crystals were uniformly distributed throughout the polymer matrix. Infrared IR spectroscopy indicated that the HA interacted with the COO− of SAL and NH2− of CS. The compressive strength of the n-HA/SAL/CS bone cement was 34.3 MPa and matched the demands of weight-bearing bones. Soaking in vitro in simulated body fluid demonstrated that the composite material had reasonably good bioactivity, while cytotoxicity tests indicated that the n-HA/SAL/CS cement could promote cell proliferation and was biocompatible.
Conclusions:
Compressive strength of n-HA/SAL/CS can satisfy the needs of cancellous bone, and in vitro bioactivity and cytotoxicity tests results indicated that the n-HA/SAL/CS composite could act as an optimal bone repair material.
Introduction
The major components of natural bone are hydroxyapatite and collagen (1), and nano-hydroxyapatite (n-HA) is its major inorganic component, thus n-HA has good biocompatibility and bioactivity in the body. However, n-HA is relatively brittle, thereby limiting its therapeutic applications. To imitate bone structure and function, n-HA can be combined with other materials (2), which is reflected in a variety of studies published by international biomaterial research groups.
Chitosan (CS) is a natural biodegradable polycation polysaccharide, and its degradation product is nontoxic and noncarcinogenic. CS degradation provides a foundation for cell and tissue growth, while also promoting new bone formation. Unfortunately, HA/CS composites are inefficient in their bonding at interfaces, and their dispersion is not uniform (3).
Sodium alginate (SAL) is a polyelectrolyte polysaccharide that possesses high negative charge density, which is water-soluble, nontoxic, biodegradable and biocompatible. In addition, SAL can promote interactions between HA and CS to form a stronger composite (4, 5).
Nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) ternary composites have been prepared using several methods, and these composites were homogeneous, with optimal chemical binding between the inorganic n-HA and organic components (5-10). However, the compressive strength of the composites was still low, and they require further development (5, 9).
In the present study, the synthesis of n-HA/SAL/CS composite was prepared via in situ synthesis. Furthermore, a self-setting bone cement was prepared by mixing n-HA/SAL/CS powder with a curing liquid (citric acid). In previous research, the biological performance of such a composite has not been studied. In this work, the biological performance of bone cement was evaluated. Bioactivity of bone cement composite was tested by in vitro soaking in simulated body fluid (SBF). Compressive strength was tested by a universal testing machine. It was shown that the n-HA/SAL/CS bone cement had good biological activity and mechanical properties. In addition, the preparation of bone cement was simple and safe, suggesting that it has wonderful potential in bone repair.
Materials and methods
Materials
The chemicals used were calcium nitrate (Ca(NO3)2·4H2O; Tianjin Hedong Hongyan Chemical Reagent Co. Ltd., China), citric acid monohydrate (Tianjin Dengke Chemical Reagent Co. Ltd., China), diammonium hydrogen phosphate ((NH4)2HPO4) and sodium alginate (Tianjin Kemiou Chemical Reagent Co. Ltd., China), and chitosan (Jinan Haidebei Marine Bioengineering Co. Ltd, China). All reagents were of analytical quality.
n-HA/SAL/CS composite preparation
At a 1.67 molar ratio of Ca to P, 5 M Ca(NO3)2 solution and 3 M (NH4)2HPO4 solution were prepared with deionized water. Based on a 7/3 mass ratio of HA to SAL/CS and a 5/1 mass ratio of CS to SAL, the mass of each material was assessed. CS powder was dissolved in acetic acid solution, while SAL powder was dissolved in deionized water. After CS was dissolved completely, the sodium alginate solution was added to the CS solution. After stirring for 5 hours, Ca(NO3)2 solution was poured into the CS/SAL solution, followed by dropwise addition of (NH4)2HPO4 solution at 40°C. After addition, the pH was >10. After stirring for 15 hours, the obtained slurry was aged for 24 hours, and supernatant was removed by filtration. The precipitate was dispersed in ethanol using an ultrasonic dispersion method. After an additional 10 hours of stirring, the solution was filtered again and dried at 80°C for 24 hours, ultimately resulting in n-HA/SAL/CS composite.
n-HA/SAL/CS bone cement preparation
The curing liquid was different concentrations of citric acid (17-21 wt.%). The cement paste was made by mixing the n-HA/SAL/CS composite powder with different concentrations of curing liquid at the powder to liquid ratio of 1:1.4 (wt./vol.). Finally, the cement paste was poured into a homemade cylindrical mold to take shape; the size of the mold was Φ10 × 10 mm.
SBF solution preparation
Conventional SBF was prepared in accordance with Kokubo and Takadama’s specifications and instructions (11). The SBF solution was prepared by dissolving reagent-grade NaCl, KCl, NaHCO3, MgCl2·6H2O, CaCl2 and KH2PO4 in distilled water and buffering it to pH = 7.25 with Tris and 1 M HCl at 36.5°C. The SBF was refreshed every 24 hours.
In vitro cell cytocompatibility assay
Based on the GB/T16886.5-2003 standards, CCK-8 cells were utilized for the extraction assays. Specimens were prepared for extraction and bone cements were washed 3 times with ultrasonic cleaning in a 75% ethanol solution for 20 minutes each time. The specimens were then irradiated with UV for 4 hours. The bone cement specimens were incubated with minimum essential medium (MEM, 10% embryo bovine serum and 1% penicillin/streptomycin) at 37°C in a 5% CO2 incubator for 24 hours. The leaching ratio was 6 cm2/mL.
Cryopreserved L929 fiber cells were put through a cycle of recovery, then cultivation, then subculture and finally cultivation. MEM was used with 10% embryo bovine serum and 1% double medium, then the cells were pancreatic enzyme digested and counted. The cells were cultured at 37°C in a 5% CO2 incubator with 5,000 cells/well in 96-well plates. The MEM was removed after 24 hours, and 100 µL/well of MEM was added as the control, while 100 µL/well of bone cement extract was added to experimental samples. After the cells were incubated for 24, 48 and 72 hours, the medium was removed and replaced with 100 µL of serum-free MEM supplemented with 10% CCK-8. The 96-well plates were then incubated for 1 hour, the specimens were harvested, and the optical density (OD) was measured at 450 nm with a microplate reader.
Sample characterization
The morphology and microstructure of the precipitated n-HA/SAL/CS composite was examined by transmission electron microscopy (TEM; JEM-2100 (URP); Jeol, Japan).
The samples phase composition and crystal structure of n-HA/SAL/CS composite were analyzed using an X-ray diffractometer (XRD; D8ADVANCE; Bruker, Germany) equipped with a monochromator for beam diffraction. The diffraction spectra were recorded from 5° to 80° using CuKα (wavelength = 0.154056 nm, 40 mA, 40 kV) radiation with a 0.05° step size, and a 1-second step duration.
The bonding between inorganic phase and polymer phase was determined by Fourier transform infrared spectroscopy (FT-IR; Shimadzu 2000; Shimadzu, Japan) and Raman spectroscopy (RS; inVia; Renishaw, UK). FT-IR spectra were recorded using KBr pellets with a Shimadzu-2000 spectrometer that had a 2 cm-1 spectral resolution. For excitation and analysis of Raman spectra, the 514.5 nm emission (20 mW) of an Ar+-ion laser was used with 1 cm-1 resolution.
Mechanical properties were evaluated using a WDW-2C electronic universal testing machine (Jinan Kehui Test Equipment Co. Ltd., China). The average values of 3 or more tests were reported.
In vitro studies were performed by soaking the samples in SBF solution at 37°C for 4 weeks. At regular intervals (1, 2, 3 and 4 weeks), samples were removed, carefully washed with deionized water, and dried at room temperature. The samples were then tested by XRD and field emission gun scanning electron microscopy (FEG-SEM; FEI Quanta 250) coupled to an X-Max 30-mm2 detector energy dispersive X-ray spectrometer (Bruker Quantax 200 X flash 6 | 30 EDS).
Results
TEM analysis
The n-HA/SAL/CS composite powder was imaged by TEM (Fig. 1A), while the n-HA/SAL/CS diffraction pattern was analyzed by selected area electron diffraction (SAED) (Fig. 1B), which indicated that the HA was rod-like at the nanoscale, and the grain size was about 5-20 × 30-50 nm. SAED patterns were observed to be of different radii in concentric circles, which indicated that HA was polycrystalline. The electron diffraction pattern composition was further determined by calculating the interplanar spacing using Equation [1]:

Where d is the crystal interplanar distance, K is the camera constant and R is the radius of the diffraction rings. Comparison of the calculated interplanar distance with a powder diffraction file (PDF) standard card, confirmed the HA composition of the synthetic crystal. The crystal planes, proceeding from inside the crystal to the surface, were [002], [211] and [213], respectively. These crystal planes were also presented in the XRD pattern.

Transmission electron microscopy (TEM) image of nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) composite (
XRD analysis
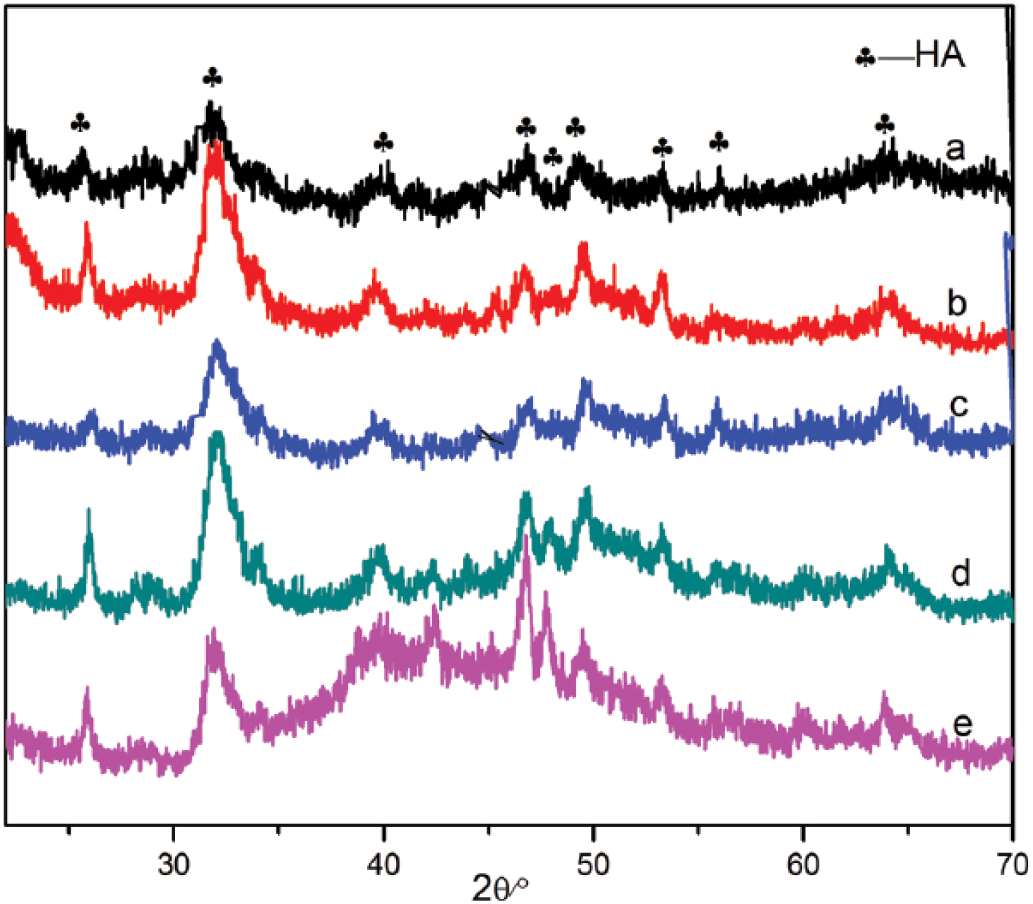
The XRD patterns of SAL, CS and the n-HA/SAL/CS composite were recorded (Fig. 2). SAL had a strong characteristic peak at 31.7° (12), while CS had a broad characteristic peak at 20.2° (13). The major diffraction peaks identified for the n-HA/SAL/CS composite were in accordance with the standard data for HA (PDF# 09-0432; Tab. I). The composite was weakly crystallized, but the characteristic peaks of n-HA were all present in n-HA/SAL/CS composite. The characteristic peaks of CS and SAL were not represented in the XRD patterns, which could be explained by the low crystallinity that is derived by interactions between HA, SAL and CS, a state that is similar to natural bone. Using Scherer’s equation (Lhkl = Kλ/βcosθ) (14) and the crystal plane [002] at 25.835° to calculate the average crystallite size of the C axis, the size was found to be ~20.01 nm. Using the same method, the crystallite size of the crystal plane [211] at 31.92° was 9.83 nm. These results indicated that the n-HA composite crystal existed at the nanometer scale.
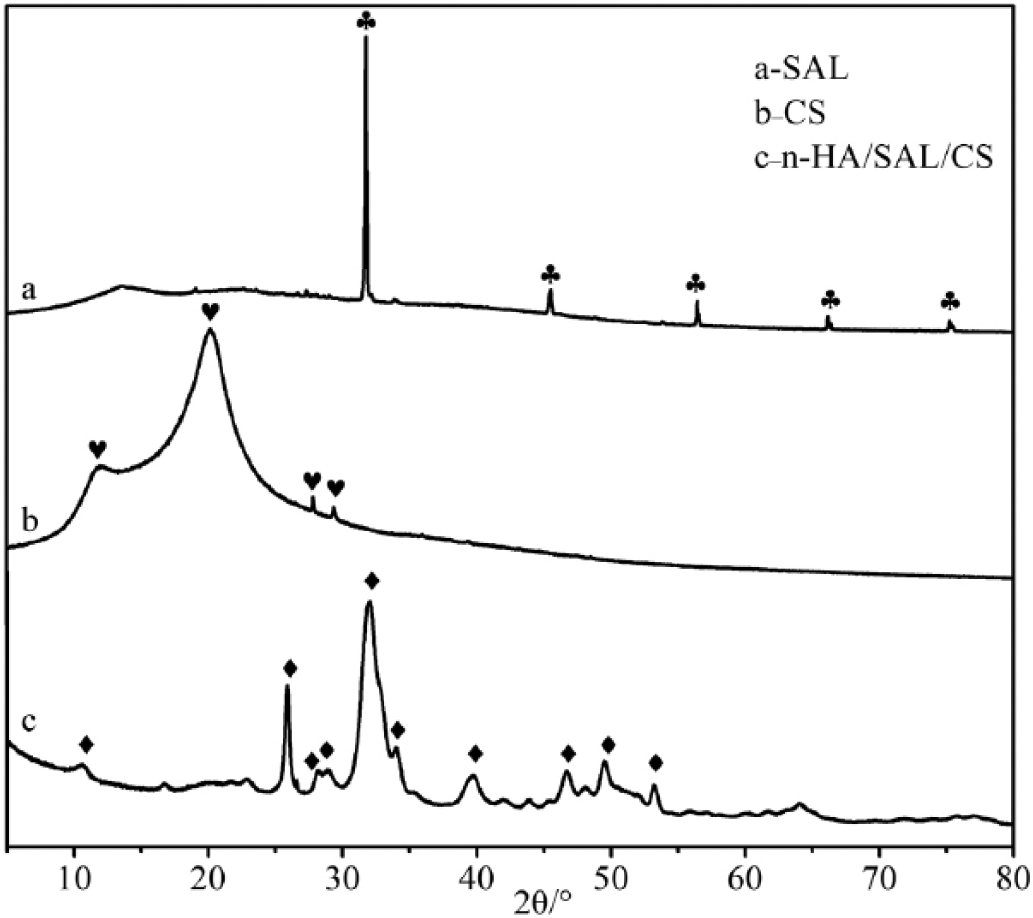
X-ray diffractometer (XRD) patterns of sodium alginate (SAL), chitosan (CS) and the nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) composite.
Diffraction angle (2θ) and corresponding crystal indices of standard HA and n-HA/SAL/CS
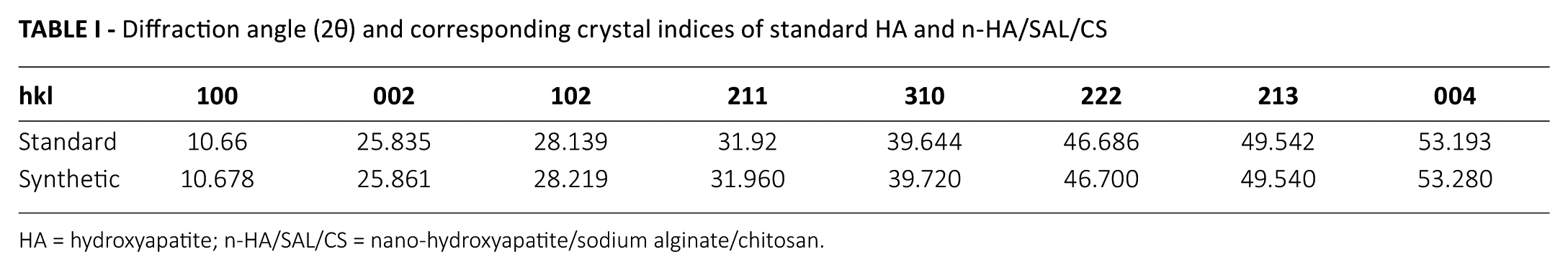
HA = hydroxyapatite; n-HA/SAL/CS = nano-hydroxyapatite/sodium alginate/chitosan.
FT-IR analysis
The FT-IR spectra of SAL, CS, n-HA, the n-HA/SAL/CS composite, citric acid and the n-HA/SAL/CS cement were measured (Fig. 3). In the FT-IR spectra of SAL, 1,416 cm-1 and 1,637 cm-1 corresponded to the COO− symmetric and asymmetric stretching peaks, respectively (15). The 2,924 cm-1 peak represented CH2−, while 3,440 cm-1 was the H2O absorption peak. In the FT-IR spectra of CS, 1,608 cm-1 was the NH2− bending vibration absorption peak, and 1,650 cm-1 was the C=O stretching vibration peak of amide Ι. In the FT-IR spectra of the n-HA/SAL/CS composite, the 568 cm-1 and 607 cm-1 peaks were the bending vibrations of PO43-, 1,040 cm-1 was the asymmetric stretching vibration absorption peak of P-O (16), 1,383 cm-1 was probably the NO3− absorption peak that remained in the system, 1,640 cm-1 was the O-H stretching vibration of liquid water, 1,552 cm-1 represented the correlation absorption peak of secondary amide in the composite. Interestingly, the COO− peak of SAL and the NH2− vibration peak of CS disappeared, indicating that the SAL COO− crosslinked with the CS NH2− (17). Meanwhile, the OH− vibration peak of SAL and CS disappeared, as a hydrogen bond may be formed between the OH− and PO43-.
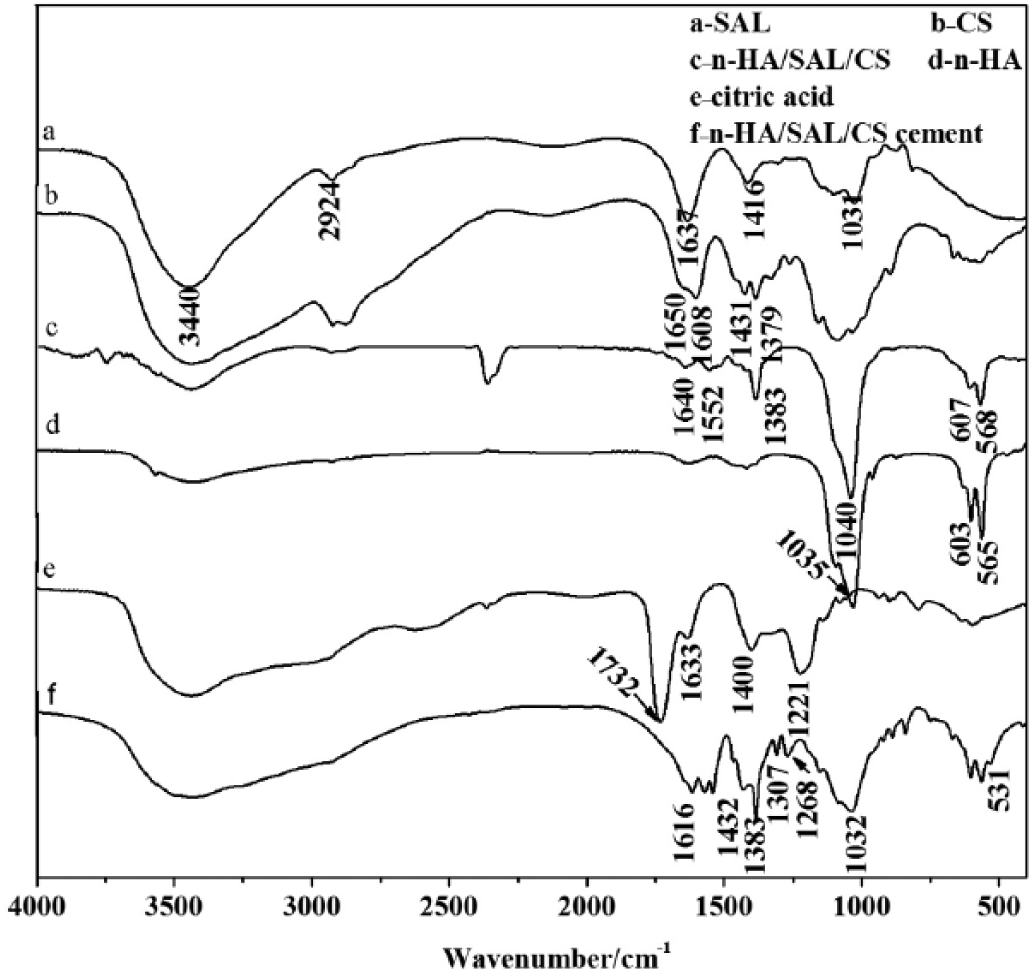
Fourier transform infrared spectroscopy (FT-IR) spectra of sodium alginate (SAL), chitosan (CS), the nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) composite, citric acid and the n-HA/SAL/CS cement.
In FT-IR spectra of citric acid, 1,732 cm-1 belongs to the C=O stretching vibration peak of RCOOH, 1,633 cm-1 was the characteristic peak of COOH, 1,400 cm-1 was the O-H in-plane bending vibration peak of COOH and 1,221 cm-1 was the C-O stretching vibration peak of COOH. In the FT-IR spectra of the n-HA/SAL/CS cement, the 1,732 cm-1 peak disappeared, and 2 new peaks appeared at 1,616 cm-1 and 1,432 cm-1, which likely belonged to the asymmetric and symmetric peaks of COO−, respectively. The peak at 1,307 cm-1 was the C-O vibration peak of C-OH, 1,268 cm-1 was the C-O stretching vibration peak of COO−. The 2 peaks shifted in comparison with the citric acid peaks, demonstrating that the n-HA/SAL/CS composite reacted with citric acid. The peak at 531 cm-1 was the Ca-O vibration peak, indicating that the COO− of citric acid coupled with the Ca2+ of HA (18).
Raman analysis
The Raman spectra of CS, n-HA/SAL/CS composite, n-HA/SAL/CS cement and citric acid were recorded (Fig. 4).In CS, 351 cm-1 represented the C-N in-plane bending vibration, and 893 cm-1 was the characteristic peak of a β-glycosidic bond, 1,083 cm-1 was the C-N stretching vibration peak and 1,377 cm-1 was the C-H bending vibration. The peak at 1,592 cm-1 was the N-H bending vibration peak of NH2−, while 2,883 cm-1 belonged to C-H vibration peak of CH2OH. In the n-HA/SAL/CS composite, the 427 cm-1 and 584 cm-1 peaks corresponded to the PO43- symmetric and asymmetric bending, respectively. The peaks at 961 cm-1 and 1,045 cm-1 were the respective P-O-P symmetric and asymmetric stretching vibration peaks of PO43. The CS at 1,592 cm-1 (N-H) peaks disappeared, while a weak C=O vibration at the 1,635 cm-1 peak appeared for the amide bond in n-HA/SAL/CS composite. This indicated that the CS NH2− reacted with the SAL COO−, consistent with the FT-IR results.
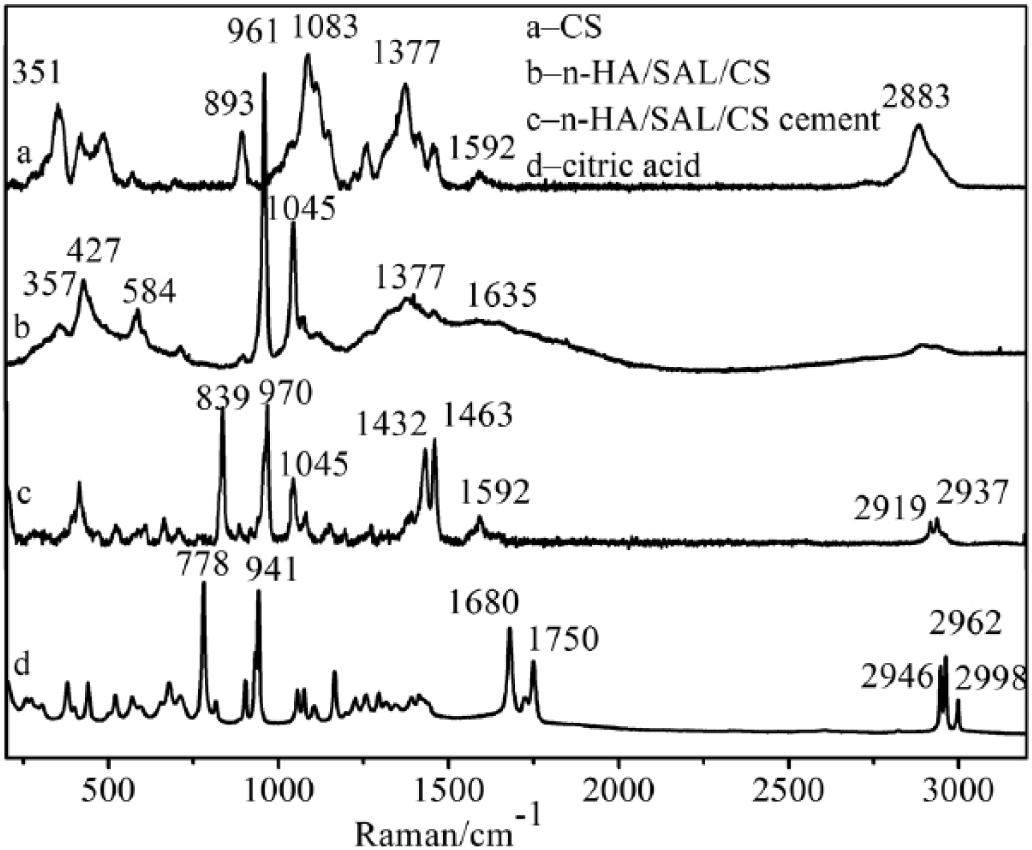
Raman spectra of chitosan (CS) (a), the nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) composite (b), the n-HA/SAL/CS cement (c) and citric acid (d).
In the Raman spectra of citric acid, 778 cm-1 belonged to the O-H out-of-plane bending peak and 941 cm-1 belonged to the C-COOH vibration peak. The peak at 1,680 cm-1 was the asymmetrical stretching vibration of COOH, while 1,750 cm-1 was the C=O vibration of COOH. In the n-HA/SAL/CS cement, the 778 cm-1 and 941 cm-1 peaks shifted to 839 cm-1 and 970 cm-1 and 1,432 cm-1 belonged to the C-O of COO− and 1,592 cm-1 to the C=O of COO−. The characteristic peaks of 1,680 cm-1 and 1,750 cm-1 in citric acid disappeared, indicating that the COOH of citric acid reacted with the n-HA/SAL/CS composite.
Mechanical properties
The compressive strength of the n-HA/SAL/CS composite bone cement was investigated as a function of citric acid concentration (17-21 wt.%; Fig. 5). As the concentration of citric acid increased, the strength of the cement also increased, although this effect was reversed at >20% citric acid. The greatest compressive strength (34.3 MPa) was observed at 20% citric acid, which is in the range of cancellous bones’ strength (19).
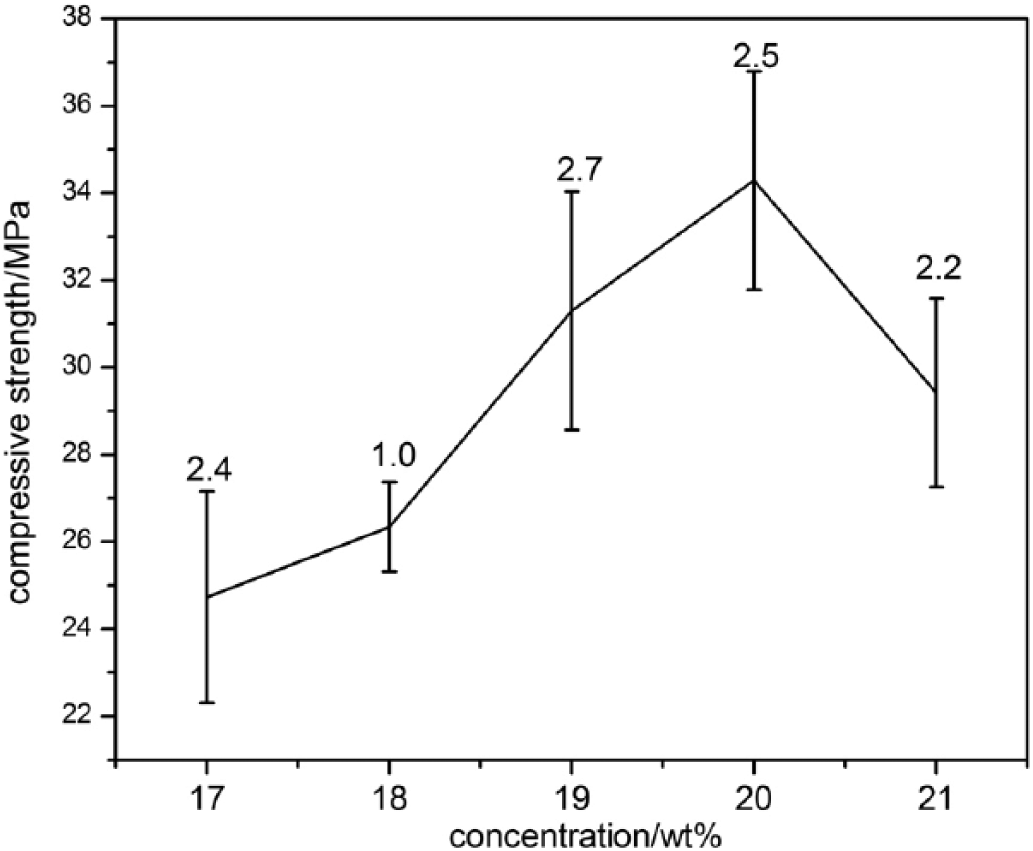
Compressive strength of the nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) cement with different concentrations of citric acid.
In vitro bioactivity
The apatite-forming ability of the n-HA/SAL/CS bone cement was assessed at the optimum citric acid concentration (20 wt.%) for various cycles at 37 ± 0.5°C.
The surface XRD patterns of the n-HA/SAL/CS bone cement in 20 wt.% citric acid solution were measured after soaking in SBF for different lengths of time: 0, 1, 2, 3 and 4 weeks (Fig. 6A-E, respectively). Based on the pattern from nonimmersed samples, the characteristic peak of SAL (2θ = 31.7°) and HA (2θ = 32.0) appeared on the surface of the n-HA/SAL/CS bone cement in 20 wt.% citric acid solution, based on the weakly crystallized apatite structure. The XRD pattern differed in correlation to the time it was soaked in SBF (1, 2, 3 and 4 wk). At 1 week immersion, the HA diffraction peak at the bone cement surface was weakened. As the immersion time lengthened, the degree of weakening was reduced, and the HA crystal structure was gradually normalized. By the fourth week, the HA structure was similar to that of the nonimmersed HA, and it was also characterized by weak crystallization. These results indicate that, at the beginning of immersion, n-HA on the composite surface may have been initially dissolved. Meanwhile, Ca2+ and PO43- ions in the SBF were deposited on the composite surface, initially forming amorphous CaP salt and later gradually transitioning back to HA. In addition, the HA of natural bone is also weakly crystalline (20). This indicates that, during immersion, the formative HA on the composite surface is similar to the HA of natural bone, which suggests that the composite may have positive qualities as a repair and replacement material for bone.
Surface X-ray diffractometer (XRD) patterns of the nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) bone cement immersed in simulated body fluid for different times: (a) 0 week, (b) 1 week, (c) 2 weeks, (d) 3 weeks and (e) 4 weeks. The curing liquid was 20 wt.% citric acid solution.
The surface morphology of the n-HA/SAL/CS bone cement (20 wt.% citric acid) was assessed after immersion in SBF (0, 1, 2, 3 and 4 weeks; Fig. 7). The n-HA/SAL/CS bone cement surface experienced the apatite formation at various SBF immersion times, but the amount of apatite formed and the surface coverage were variable. At 0 week, there was no apatite on the surface of the n-HA/SAL/CS bone cement, but the n-HA of the composite was dispersed uniformly (Fig. 7A). At 1 week, the surface was not completely covered with apatite growth (Fig. 7B). There was irregular apatite growth of n-HA/SAL/CS after 2 weeks of SBF immersion (Fig. 7C). After the sample had been immersed in SBF for 3 weeks, the n-HA/SAL/CS surface was completely covered by apatite (Fig. 7D), and the apatite showed a short rod-like structure. After soaking the sample in SBF for 4 weeks, a dense apatite layer was formed on n-HA/SAL/CS (Fig. 7E). The rod-like apatite gradually became indistinct, and tended to aggregate. This apatite layer on the n-HA/SAL/CS sample can improve the osteoconduction and osteointegration properties (21). Hence, the apatite-forming ability of the n-HA/SAL/CS was optimal at 3 and 4 weeks of immersion, whereas 1 week of immersion was not sufficient for complete surface coverage. SEM images indicated that as immersion time was increased, the number of crystals on the surface also increased. EDS analysis demonstrated that the crystals were composed of Ca, P and O, and providing evidence that the synthesized bone cement composite possessed bioactivity in vitro.

Surface scanning electron microscopy (SEM) and energy dispersive spectrometer (EDS) images of nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) (20 wt.% of citric acid solution) bone cement immersed in simulated body fluid for different times: (
Cytotoxicity test
The cytotoxicity of n-HA/SAL/CS cement and control was tested (Fig. 8). In comparison with the control, cell viability with the n-HA/SAL/CS cement was enhanced. However, the viability of cells treated with the n-HA/SAL/CS cement decreased as culture time increased. After 24 hours of incubation, Levene’s test was used to assess variance homogeneity (F = 18.628, p = 0.003), and independent samples t-test (t = 5.831, p = 0.004) results indicated that there were significant differences between n-HA/SAL/CS cement and control. However, for the 48- and 72-hour cultures, the p was >0.05, indicating that there was no significant difference between samples. These results indicated the n-HA/SAL/CS cement could promote cell proliferation and had good biocompatibility.
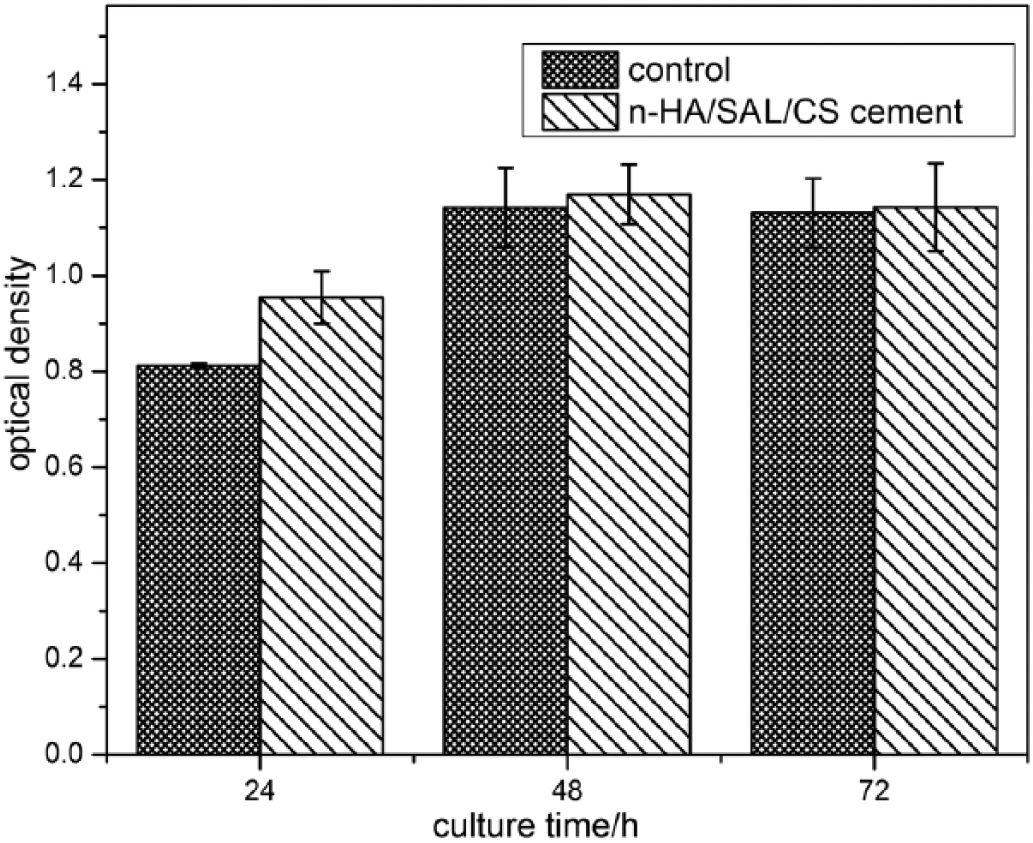
Cytotoxicity of the nano-hydroxyapatite/sodium alginate/chitosan (n-HA/SAL/CS) cement and control at various time points. Optical density was measured at 450 nm.
Discussion
N-HA/CS composites have good bioactivity, biocompatibility and inductivity and an appropriate degradation rate. However, neither dispersion nor the interface bonding between n-HA and CS are ideal (6, 22-24). In this study, an n-HA/SAL/CS composite with 70 wt.% of n-HA was synthesized using an in situ synthesis method. In this study, we selected citric acid as the curing liquid, and the compressive strength of n-HA/SAL/CS cement was up to 34.3 MPa, which can satisfy the requirements for partially weight-bearing bones. Operations to insert the cement into bone are more convenient. Moreover, the bioactivity of the cement was good. We also tested the cytotoxicity test again, and it was found that n-HA/SAL/CS cement could promote cell proliferation and was biocompatible.
SAL contains carboxyls, while CS contains primarily aminos, indicating that SAL and CS could form an ion exchange network structure via attraction between positive and negative charges. Meanwhile, hydroxyl residues in the organic phase can interact with the PO43- of n-HA via hydrogen bonds. Therefore, as the bonding strength between organic and inorganic phase increases, nanoparticle deposition increases.
In the bone cement, the compression strength increased in correlation with the citric acid concentration, suggesting that the strength of the curing product was, in part, determined by the product’s microstructure. As citric acid concentration increased, the solution pH decreased, and the phosphate solubility improved. In addition, the carboxylate group of citric acid can react with the Ca2+ of HA, thereby enhancing the strength of the n-HA/CS/SAL bone cement. Nakamoto (18) hypothesized that COO− could coordinate with metal ions as either a unidentate ligand, bidentate ligand or bridging ligand. Nakamoto suggested that the coordination mode of COO− could be judged by the value of Δν(νas- νs): if the value of Δν(νas- νs) was greater than the Δν of free carboxylic acid ions, it was an unidentate ligand coordination; the Δν(νas- νs) of a bidentate ligand is less than the Δν of free carboxylic acid ions; the Δν(νas- νs) of a bridging ligand is closer to the Δν of free carboxylic acid ions. The Δ of sodium citrate is 196 cm-1, while the Δ of n-HA/SAL/CS cement was νas(C=O) = 1,616 cm-1, νs(C=O) = 1,432 cm-1, Δ = 184 cm-1. The Δ of n-HA/SAL/CS cement was less than the Δ of free citrate, suggesting a bidentate ligand coordination mode.
Figure 9 is a representation of the reaction process. First, the SAL carboxyl and the CS aminos form an amide bond. Next, the hydroxyl group of the organic phase and the HA phosphate form a hydrogen bond. Finally, the citric acid carboxyl combines with the HA Ca2+ to form a complex.
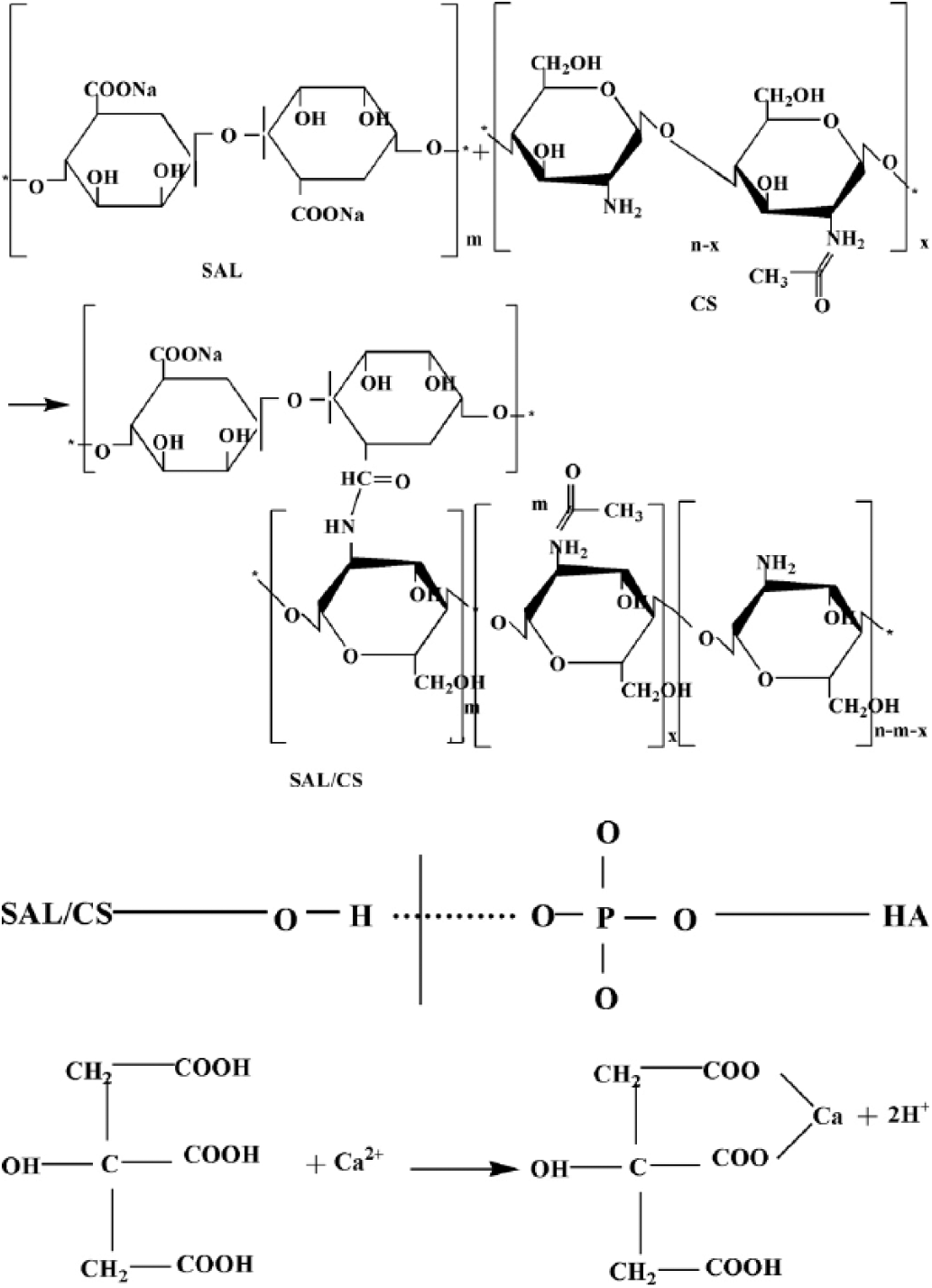
Reaction mechanism of the nano-hydroxyapatite/sodium alginate/chitosan (n-HA/CS/SAL) bone cement.
In addition, the decreased crystallinity of n-HA is similar to that of human bone and has good bioactivity. SAL and CS are easy to degrade, providing pores in the material after degradation, which is beneficial for nutrient transfer and elimination of metabolites.
Conclusion
This study involved in situ synthesis of an n-HA/SAL/CS composite and the preparation of n-HA/SAL/CS cement for which citric acid was used as a curing liquid . From the FT-IR and XRD results, the COO− of SAL crosslinked with the NH2− of CS. Hydrogen bond was formed between the OH− of SAL, CS and PO43-. The compressive strength of the n-HA/SAL/CS bone cement was up to 34.3 MPa, which matches the strength observed in cancellous bone tissue. Citric acid as the curing liquid possesses many carboxyl groups, which react with Ca2+ of HA. In vitro bioactivity showed that the n-HA/SAL/CS composite material has good bioactivity, as HA formation on the surface of n-HA/SAL/CS bone cement is a process of dissolution and redeposition. The n-HA/SAL/CS bone cement surface releases Ca2+, PO43- and other ions to enter the SBF on reaching a specific concentration, which are then absorbed and accumulated on the solid surface, this ultimately results in the formation of an apatite layer similar to bone apatite. Cytotoxicity tests indicated that n-HA/SAL/CS cement can promote cell proliferation and has good biocompatibility. Which suggests that n-HA/SAL/CS bone cement may serve as an optimal bone repair material.
Footnotes
Disclosures
Financial support: Funding was provided by the National Natural Science Foundation of China (grant no. U1304820, China); Key Technology Research Project of Henan Province (grant no. 152102210103, China); and National Natural Science Foundation of China (grant no. 31370999, China).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
