Abstract
Bone morphogenetic protein-2 (BMP-2) is a potent osteoinductive factor; however, current clinical applications using
Introduction
The treatment of bone defects commonly involves the use of various bone grafts and carriers (or scaffolds), which provide structural support and facilitate bone regeneration over 3–6 months. Extensive research has explored the potential of several growth factors in enhancing this regenerative process, with bone morphogenetic protein (BMP)-2 emerging as the most extensively studied and widely recognized factor. 1 BMP-2 is one of over 20 BMPs within the transforming growth factor-beta superfamily, which also includes BMP-4, BMP-7, and BMP-9. BMP-2 is particularly notable for its capacity to induce bone formation.2 –4 It has also been demonstrated to markedly enhance the recruitment of osteogenic progenitor cells, thereby facilitating new bone tissue formation. 5 Notably, BMP-2 is the only BMP that has received U.S. Food and Drug Administration approval for clinical use as a recombinant human BMP-2 (rhBMP-2). 6
rhBMP-2 is primarily produced using either recombinant Chinese hamster ovary (CHO) cells or
Additionally, BMP-2 has the potential to disseminate into adjacent tissues, which can result in unanticipated outcomes, such as ectopic bone formation, exacerbated inflammatory responses, and edema.12,13 Therefore, the use of scaffolds is necessary to maintain the localized retention of BMP-2 at the target site. Although ongoing research continues to explore various carriers, including absorbable collagen sponge, as delivery systems,12,14 –19 most of these studies adhere to conventional approaches that involve applying purified rhBMP-2 to distinct carriers.
In this study, we aimed to develop an novel approach for bone regeneration by genetically engineering human retinal pigment epithelial cells to create human cell-derived BMP-2. These modified cells were cultured within a collagen-porcine bone graft material to allow the localized release of BMP-2 directly into the graft materials. Simultaneously, the efficacy of this approach was evaluated through in vivo tests to assess its effect on bone regeneration (Figure 1).

Schematic representation of the study.
Materials and methods
Plasmid constructs
The plasmids used for the recombinant constructs in this study are outlined in Supplemental Table 1.
Cell culture
ARPE-19 cells, a human retinal pigment epithelial cell line (ATCC: CRL-2302), were obtained from ATCC (Gaithersburg, Maryland, USA). The cells were cultured in Dulbecco’s modified Eagle’s medium/nutrient mixture F-12 (DMEM/F12; Gibco, cat# 11330-032, Seoul, South Korea) supplemented with 10% fetal bovine serum (FBS; Gibco, cat# 16140-089) at 37°C in a humidified atmosphere with 5% CO2. HEK293T cells, a human embryonic kidney cell line (ATCC: CRL-3216), were cultivated in Opti-MEM GlutaMax medium (Gibco, cat# 51985-091) containing 8% FBS (Gibco, cat# 16140-089) at 37°C in a humidified atmosphere with 5% CO2.
Lentivirus production and stable cell line
HEK293T cells (2 × 106cells) were seeded in a 100 mm dish (SPL, Pochon, Korea) and transfected with a plasmid mixture of pMD2.G, psPAX2, and pTK107 (1:1:2 ratio) after 24 h. To prepare the transfection mixture, polyethyleneimine (30 µg; 1 mg/ml in DW, Polysciences, cat# 23966, Pennsylvania, USA) was mixed with 10 µg of plasmid DNA, and the resulting mixture was added to the HEK293T cells. The supernatant containing pTK107-derived lentiviral particles was collected from the transfected cells after 48 h and filtered through a 0.45 µm CA syringe filter (Sartorius, Göttingen, Germany). ARPE-19 cells (5 × 105 cells) were seeded into a 6-well plate (SPL, Pochon, Korea) 1 day before transduction, and 2 ml of the filtered supernatant was added to the ARPE-19 cells. The transduced cells (ARPE19-TK107) were selected using DMEM/F12 supplemented with 10% FBS and 6 µg/ml puromycin for 2 weeks, with media changes every 3–4 days.
Bone graft preparation and BMP-2 ELISA
ARPE-19-TK107 cells (1 × 107 cells) were trypsinized, followed by centrifugation at 1200
Surgical procedure
In the in vivo experiments, 12 six-week-old Sprague-Dawley rats (male, 250–300 g; Orient Bio Inc., Gyeonggi-do, Korea) were used. Prior to the experiments, all rats were subjected to a 1-week acclimatization period. Protocols for animal experiments were approved by the Institutional Animal Care and Use Committee of the Seoul National University Bundang Hospital (approval number BA-2304-366-003-05). All animals were cared for in strict accordance with the Animal Research: Reporting of In Vivo Experiments guidelines, which outline the ethical treatment and care protocols for animal research subjects.
All surgical procedures were performed under general and local anesthesia. General anesthesia was induced through inhalation of 2%–3% isoflurane. Subsequently, tiletamin-zolazepam (Zoletil50, Virbac S.A., Carros, France) and xylazine (Rompun, Bayer Korea, Ansan, Korea) were administered intraperitoneally at doses of 20–30 mg/kg and 10 mg/kg, respectively, to ensure the required anesthetic time. Local anesthesia at the surgical site was achieved using 2% lidocaine and 1:100,000 epinephrine (Yuhan Co. Ltd., Seoul, Korea). For prophylaxis and analgesia, an intramuscular injection of 20 mg/kg gentamicin sulfate (Sp Gentamicin Injection, Shinpoong, Seoul, Korea) and ketoprofen (Ketoprofen injection, Bukwang, Seoul, Korea) were administered after surgery. Prior to the surgical procedure, the surgical site was disinfected with a chlorhexidine gluconate solution, and the scalp hair was removed. Using sterile instruments, following aseptic techniques, a midline cranial incision was made, extending from anterior to posterior. The subcutaneous tissue, musculature, and periosteum were dissected and reflected to expose the calvarium. Bilateral full-thickness defects, each 5 mm in diameter, were created symmetrically around the midline suture using a saline-cooled trephine drill. The defects were randomly assigned into one of three experimental groups: control, graft, or BMP-2. No material was applied in the control group after the defect formation, whereas porcine-collagen mixed graft material was applied to the defects in the graft group. In the BMP-2 group, the porcine-collagen mixed graft material was enriched with hBMP-2 (Figure 2). Subsequently, the periosteum and skin were closed using a 4–0 Vicryl suture (Ethicon, Menlo Park, CA, USA). After a 6-week recovery period, all rats were euthanized through CO2 asphyxiation, and the calvaria defects were harvested and fixed in formaldehyde for further analysis.

Insertion of hBMP-2 with collagen block in a rat calvaria defect model: (a) ARPE-19-TK107-injected bone graft in culture, (b) ARPE-19-TK107-injecteded bone graft was prepared prior to insertion into the calvaria defect model, and (c) application of the ARPE-19-TK107-injected bone graft to the calvaria defect.
Micro-computed tomography examination
Harvested calvaria bone was fixed onto a jig with parafilm for micro-computed tomography (CT) imaging. A total of 800 images were acquired using a tube voltage of 130 kV, a current of 60 µ, and a 1.0 aluminum filter. Cross-sections were reconstructed using NRecon software (Ver. 1.7.4.6, Kontich, Belgium). The acquired cross-sectional images were aligned for each section using Dataviewer software (ver. 1.5.6.2, Bruker-CT, Kontich, Belgium), and quantitative parameter values were calculated using CtAn software (version 1.19.4.0, Bruker-CT, Kontich, Belgium). The Micro-CT scans were conducted using the SkyScan1173 system (Bruker-CT, Kontich, Belgium).
Histology and histomorphometric analyses
The specimens were fixed in 10% neutral buffered formalin, dehydrated in 10% EDTA for 1 week, and subsequently cleared and embedded in paraffin. After deparaffinization, the specimens were stained with hematoxylin and eosin, followed by Masson’s trichrome staining. Images were captured using a microscope (Olympus BX51TF, Olympus Optical Co., Tokyo, Japan) and a digital slide scanner (PANNORAMIC 250 Flash III, 3DHISTECH Ltd., Budapest, Hungary). The resulting images were subjected to image metrology and analysis using a dedicated viewing program (Caseviewer, 3DHISTECH Ltd., Budapest, Hungary).
Statistical analysis
The data were analyzed using a
Result
Adverse events
No skin lesions, inflammatory complications (e.g. swelling), hematomas, wound dehiscence, infections, or osteolysis were observed in the rat calvaria treated with hBMP-2. Additionally, no fatalities occurred because of post-surgical complications.
Production of BMP-2
ARPE-19-TK107 cells represent multicistronic constructs that simultaneously expressed BMP-2, enhanced green fluorescent protein (EGFP), and a puromycin resistance gene. Therefore, the integrity of these stable cells can be verified through EGFP expression, which confirms the successful introduction of the gene delivered via the lentiviral vector. BMP-2 expression level in a 100 mm dish culture of confluent ARPE-19-TK107 was quantified to be approximately 4000 pg/10⁶ cells/day. Following the injection of 106 ARPE-19-TK107 cells into a 5 × 5 × 5 mm bone graft, BMP-2 expression was monitored for 1 month.
At 12 days post-injection, BMP-2 expression was approximately 9000 pg/day (4500 pg/ml × 2 ml), and this level gradually increased to 30,000 pg/day (15,000 pg/ml × 2 ml) at 30 days post-injection. Notably, ARPE-19-TK107 cells demonstrated effective proliferation within the bone graft (Figure 3).

BMP-2 production per day. BMP-2 secreting cells were applied to a collagen bone graft, and the accumulated BMP-2 was measured at 3-day intervals, from days 12 to 30. The total amount of BMP-2 produced during each interval was then divided by three to obtain the average amount released.
Micro-CT and Histomorphometric analysis
Micro-CT analysis revealed significant differences in several parameters between the BMP-2 and graft groups compared to the defect group without bone grafting, including bone volume per total volume (BV/TV), bone surface density (BS/TV), bone mineral density (BMD), and trabecular number (TbN). Although the BMP-2 group exhibited higher BV/TV, BS, and BMD values than the graft group, these differences were not statistically significant. However, the TbN exhibited borderline significance (

Results of micro-CT analysis after 6 weeks: (a) bone volume per tissue volume, (b) bone surface density, (c) bone mineral density, and (d) trabecular number.
Histological and histomorphometric analysis corroborated the micro-CT findings. High-resolution histological images further highlighted new bone formation surrounding the bone graft materials in the BMP-2 group compared to that in the bone graft group. (Figure 5(a)–(c)) The BMP-2 group exhibited a significantly greater area of new bone formation than the bone graft group (
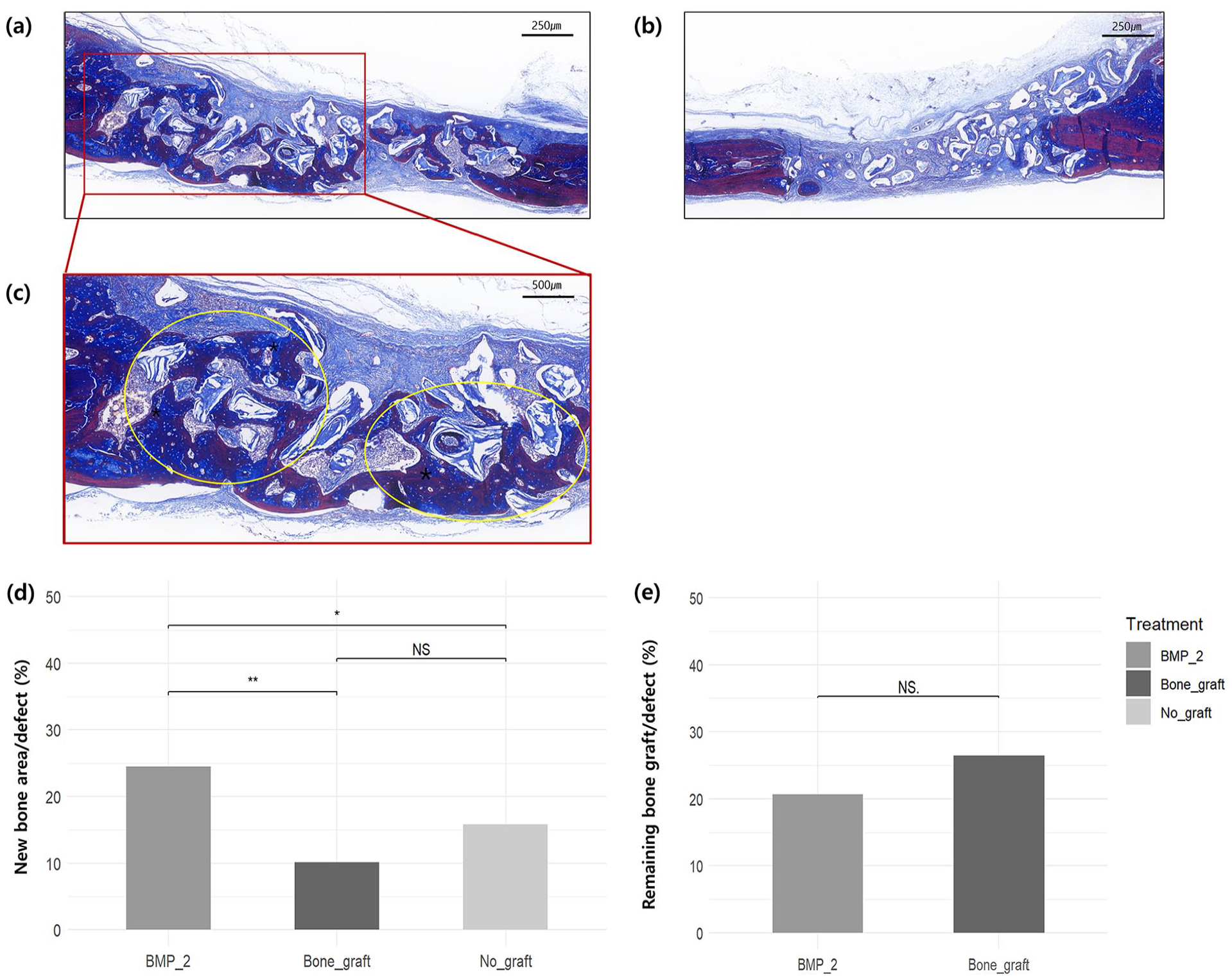
Representative histological images and histomorphometric analysis after 6 weeks: (a) image of the BMP-2 group, left side of the calvaria defect, 5× magnification, (b) image of the bone graft group, right side of the same calvaria defect, 5× magnification, (c) image of the BMP-2 group, left side of the calvaria defect, 10× magnification. The yellow circles indicate the staining of new bone formation around the bone graft, (d) percentage of new bone area per defect, and (e) percentage of remaining bone graft per defect.
Discussion
ErhBMP-2 is widely used in clinical settings because of its efficacy in stimulating de novo bone formation, promoting bone marrow growth, and enhancing vascularity at grafted areas. Bone grafting procedures that employ the concomitant use of rhBMPs have demonstrated efficacy in patients with compromised bone-healing capacity or limited donor sites, where accelerated healing periods are necessary, or in cases of large bone defects. However, this approach has inherent limitations, such as the lack of post-translational modification mechanisms
The poor stability and in vivo reactivity of ErhBMP-2 often necessitate high-dose applications, with several studies reporting an effective concentration of 1.5 mg/ml.14,25 However, a higher dosage has been demonstrated to result in increased leakage into surrounding tissues, which may lead to a number of clinical complications, including ectopic bone formation, bone cyst formation, inflammatory complications, and an increased risk of tumor formation.12,13
Ex vivo gene delivery is a cell-based gene delivery method that employs recombinant vectors, such as adenoviral, adeno-associated viral, or lentiviral vectors, to deliver a target gene into patient cells. The ex vivo gene delivery of BMP-2 has been predominantly achieved using mesenchymal stem cells (MSCs), such as bone marrow stromal cells, muscle-derived cells, adipose-derived stem cells, periodontal ligament stem cells, and fibroblasts, 26 which are grafted with bone materials at the site of the lesion. This approach offers a solution to the structural limitations of ErhBMP-2 27 and which has demonstrated potential clinical benefits in several studies.28 –30 However, challenges such as the high cost, invasiveness of the procedure, and difficulties in standardization owing to individual differences and variations in handling processes hinder their real-world application. 31
To address the limitations of current clinical methods using ErhBMP-2 and to explore the potential of precision medicinal concept through ex vivo gene delivery to patient cells, we genetically engineered a non-tumorigenic human retinal pigment epithelium cell line, ARPE-19, to express human BMP-2. The ARPE-19 cell line has been well-documented for such in vivo application; they are characterized by a long lifespan, good viability, and non-tumorigenic properties and are associated with minimal immune responses. 32 The extraction of MSCs from the patient requires a high volume of cells, which is invasive and painful, and the process of centrifugation is challenging to standardize. 33 In contrast, the genetically engineered ARPE-19 cells enabled a more predictable expression of the target gene, enhancing the feasibility of large-scale production and eliminating the need for direct cell extraction from the patient. Encapsulating engineered cells for ectopic protein expression in various platforms was well demonstrated by series of studies.34 –36 Live cell encapsulation provides a more stable and long-lasting effect, that the engineered cells can produce and secrete fresh proteins in continuous or controlled manner.
Furthermore, ARPE-19-TK107 cells markedly reduced the required effective dose of BMP-2 compared to commercially available rhBMP-2 (1.5 mg/ml vs 15 ng). These results align with those of previous ex vivo gene delivery studies that showed that native BMP-2 can enhance bone regeneration even at low concentrations.37 –39 Furthermore, the use of a pre-existing, clinically approved collagen bone graft as a carrier for BMP-2 may streamline the process of industrialization and standardization. The consistent and predictable expression levels of the target gene in standardized ARPE-19-TK107 cells may also facilitate combinatorial therapy with other effective growth factors, such as human platelet-derived growth factor-BB or BMP-7, which may lead to synergistic effects and improved therapeutic outcomes
Conclusion
This study demonstrated that BMP-2 delivered via genetically engineered ARPE-19 cells significantly enhanced bone regeneration when compared with bone grafts alone. Micro-CT analysis revealed improved bone volume and density parameters in the BMP-2 group. Histological analysis confirmed a significantly greater level of new bone formation in the hBMP-2 group, but showed only a reducing tendency of residual graft material. The use of a clinically approved collagen bone graft material as a carrier for BMP-2 offered a streamlined and scalable alternative to the traditional gene delivery approach, addressing the structural limitations and high dosing challenges associated with commercial rhBMP-2. These findings highlight this method’s potential to improve therapeutic outcomes in bone regeneration; however, further studies are necessary to optimize conditions for maximizing bone regeneration.
Supplemental Material
sj-docx-1-jbf-10.1177_22808000251326799 – Supplemental material for Regenerative effect of human cell-derived BMP-2 cultured within a collagen bone graft in a rat calvaria defect model
Supplemental material, sj-docx-1-jbf-10.1177_22808000251326799 for Regenerative effect of human cell-derived BMP-2 cultured within a collagen bone graft in a rat calvaria defect model by Keun-Suh Kim, Euna Pi, Sang-Jun Park, Seungyoon Lee, Inyoung Choi, Taeuk Kim, Hyo-Jung Lee and Yang-Jin Yi in Journal of Applied Biomaterials & Functional Materials
Footnotes
Authors’ contributions
KSK contributed to the conception, design, data acquisition, analysis, interpretation, drafting, and critical review of the manuscript. EP contributed to data acquisition, analysis, interpretation, drafting, and critical review of the manuscript. SJP contributed to data acquisition, analysis, and interpretation. SL and IC contributed to data acquisition and data interpretation. TK contributed to the conception, design, data acquisition, analysis, interpretation, drafting, and critical review of the manuscript. HJL and YJY contributed to the conception, analysis, interpretation, and critical review of the manuscript. All authors contributed to the manuscript and approved the final version of the manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government [grant number 2021R1F1A106250413] and the Seoul National University Bundang Hospital Research Fund [grant numbers 14-2021-0024 and 18-2019-0002]. This study was partially supported by a Research Grant funded by the School of Dentistry, Seoul National University.
Ethical consideration
Protocols for animal experiments were approved by the Institutional Animal Care and Use Committee of the Seoul National University Bundang Hospital (approval number BA-2304-366-003-05). All animals were cared for in strict accordance with the Animal Research: Reporting of In Vivo Experiments guidelines, which outline the ethical treatment and care protocols for animal research subjects.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
