Abstract
Peri-implant diseases, such as peri-implantitis, affect up to 47% of dental implant recipients, primarily due to biofilm formation. Current decontamination methods vary in efficacy, prompting interest in polymeric nanoparticles (NPs) for their antimicrobial and protein-specific cleaning properties. This study evaluated the efficacy of polymeric nanoparticles (NPs) in decontaminating titanium dental implants by removing proteinaceous pellicle layers and resisting recontamination. Titanium discs were treated with saline water, PrefGel®, hydrogen peroxide (H2O2), GUM® Paroex®, or polymeric NPs, and analysed using SEM, EDX, XPS, and contact angle measurements to assess changes in surface composition, morphology, and hydrophilicity. Polymeric NPs significantly reduced nitrogen levels compared to PrefGel® (mean reduction: 2.6%, p < 0.05), indicating effective protein removal. However, their carbon reduction efficacy was similar to that of other agents. SEM images revealed that polymeric NPs disaggregated larger protein aggregates but did not fully decontaminate the surface. Contact angle analysis showed changes in hydrophilicity consistent with other treatments. Hydrogen peroxide performed best overall, achieving the lowest carbon levels post-recontamination (mean reduction: 13%, p < 0.01). While polymeric NPs exhibited unique protein-specific cleaning potential, their overall performance was comparable to traditional agents. Residual contaminants, including carbon and oxygen, persisted on all treated surfaces, indicating enhanced cleaning strategies were needed. These findings highlight the potential of polymeric NPs as an innovative approach to implant decontamination, particularly for protein-specific biofilm control. However, their efficacy in broader applications remains like that of conventional methods. This research contributes to developing targeted decontamination protocols to manage peri-implant diseases and improve long-term implant outcomes.
Introduction
Implant-related infections, especially those tied to dental implants, are a growing global health concern. 1 As the world’s population ages, 2 there’s a burgeoning demand for various prosthetics, from dental to hip and knee replacements. Unfortunately, in specific situations, such as during implantation surgery or in the postoperative period, these implants are exposed to potential bacterial contamination from diverse sources, such as the patient’s skin, medical tools, or healthcare providers. 3 In the oral cavity, dental implants are perpetually in contact with the bacteria of the oral environment.
Once attached to an implant, bacteria quickly develop into a structure known as biofilm. 4 This protective layer shields the bacteria from the body’s immune defences and antibiotics, exacerbating the problem of antibiotic resistance. 5 If left abandoned, these infections can lead to additional surgical procedures, ranging from simply cleaning the implant to its outright removal. 6
Dental implants have long been a trusted solution for replacing lost teeth, offering durability and success. However, the alarming rates of peri-implant diseases, with peri-implant mucositis affecting 19%–65% and peri-implantitis affecting 1%–47% of recipients, underline the urgency for effective interventions. The accumulation of bacterial biofilms on the implant components remains the principal causative factor. Our research aims to address this pressing issue and contribute to developing more effective treatments.
Oral peri-implant tissues present a complex challenge for the immune system to modulate inflammation and maintain a delicate balance between the body’s immune response and bacterial invaders7 –9 essential for optimal peri-implant health. Peri-implant diseases may be initiated due to the combined effects of various factors.10 –13 These include risk factors such as smoking, predisposing factors like poor oral hygiene, and precipitating factors such as a sudden increase in bacterial load.14 –18 Understanding and managing these factors is crucial for effectively preventing and treating peri-implant diseases. 19
While current treatment modalities revolve around surface decontamination through mechanical or chemical means or antimicrobial agents,20,21 their efficacy varies. Clinicians have little agreement about the most effective treatment methods.22,23 Hence, researchers have turned to pioneering solutions. One promising field is nanotechnology,24 –30 which is gaining traction in medical 31 and dental arenas, yet surface modification has not seemed to lower the progression of peri-implant disease. 32 Specifically, polymeric nanoparticles exhibit impressive antimicrobial qualities. Some are tailored to dispense antibacterial elements like silver, zinc, or doxycycline.33 –40 Present polymeric nanoparticles (NPs) are non-resorbable and exhibit carboxyl groups on their external surface, which may be functionalised with different molecules, 41 thereby enhancing the nanoparticles’ antibacterial properties and potential for promoting healing, effectively shaping the environment to elicit the desired response.42 –45
Our research delves deep into the efficacy of diverse chemical solutions in cleaning titanium dental implants. The initiation of biofilm formation involves establishing a pellicle layer on the surface, acting as a base for bacterial adherence and the ensuing growth of the biofilm. This process is further aided by the adherence of proteins to the surface, which promotes bacterial attachment and biofilm progression. 46 We were interested in understanding the residual effects of these cleaning and disinfection methods on titanium-acquired pellicles and their impact on pellicle reformation. The goal was to enhance our understanding and gain insights into future treatments for peri-implantitis.
The objective was to evaluate nanoparticles’ potential in managing peri-implant diseases. Our research primarily addressed their interaction with the pellicle on the titanium implant surface and compared it with conventional dental implant surface cleaning methods. The findings underscore the promising role of polymeric nanoparticles in the future of dental implantology, offering hope for more effective treatments for peri-implantitis.
Materials and methods
Preparation of titanium discs and decontamination groups
The setup is similar to earlier published studies. In the experimentation phase, commercially pure titanium discs with dimensions of 6.2 mm in diameter and 2 mm in height underwent a specific surface modification process to mimic the rough surface characteristics typically found on dental implants, closely resembling the commercially available OsseoSpeed® surface by Dentsply Sirona (Charlotte, USA). This surface treatment procedure adhered to established protocols as outlined by Lamolle et al. 47 To ensure uniformity and accuracy, the surface characteristics of all treated discs were meticulously analysed using a PLμ NEOX instrument (Sensofar-Tech S.L., Terrassa, Spain). A blue light laser profilometer and interferometer were employed, utilising a 50 × EPI confocal objective from Nikon (Tokyo, Japan) to capture an extended topographical view through 2 × 2 images. Each image covered an area of 253 μm × 190 μm, with a 20% overlap. Eight images were collected for each material. Subsequently, the gathered data underwent processing using the Sensomap 5.1 Plus software (Sensofar-Tech S.L., Terrassa, Spain) designed for dimensional and surface state metrology. Various surface amplitude parameters, including average roughness (Sa), total surface height (St), skewness of height distribution (Ssk), and kurtosis of height distribution (Sku), were meticulously analysed to ensure consistency across all surfaces. Five decontamination groups were evaluated (Table 1): PrefGel® (Institut Straumann AG, Basel, Switzerland), 3% vol H2O2 (Sigma-Aldrich, Norway), GUM® Paroex® (0.12% Chlorhexidine digluconate and 0.05% Cetylpyridinium chloride, Sunstar Suisse, Etoy, Switzerland) and polymeric nanoparticles (University of Granada, Granada, Spain).
Decontamination products used in this study.

Dental pellicle model—pellicle formation and decontamination
Saliva specimens were acquired from three individuals with good health, combined, and subsequently subjected to centrifugation. The titanium discs that had been prepared were positioned within 24-well plates (ThermoFisher, Waltham, USA), and the combined saliva was administered onto the surfaces of these discs. After this application, the discs underwent an incubation period of 30 min at 37°C, aimed at facilitating the formation of a pellicle layer on the surface. It is essential to highlight that the pellicle was not subjected to sterilisation procedures, although this study did not assess the presence of bacteria within the pellicle. Three discs were deliberately contaminated at three distinct time intervals for each decontamination group (n = 9) to warrant the reproducibility of the experimental design. Two sets of discs were employed for each sample in this study phase. One set underwent decontamination followed by analysis, while the other set was initially decontaminated, then subsequently re-contaminated with the pooled saliva, and finally analysed after re-contamination (see Figure 1). The methods used for analysis remained consistent for both groups of discs, intending to evaluate the propensity for pellicle formation on the surface following decontamination. Discs without any treatment, resulting in pellicle formation, served as negative controls, while discs lacking pellicles were used as positive controls.

Graphical illustration of how the pellicle study was performed.
Contact angle
This study assessed surface hydrophobicity/hydrophilicity using the static contact angle measurement method. The sessile drop technique was employed with the OCA 20 instrument from DataPhysics Instruments GmbH, Filderstadt, Germany. The analysis was conducted at room temperature, and ultrapure water (obtained from VWR, Oslo, Norway) was used as the wetting agent following the Young–Laplace fitting principle. Triplicate measurements were conducted for each sample (n = 3) to ensure accuracy and reliability.
SEM/EDX
The analysis of surface morphology was conducted using Scanning Electron Microscopy (SEM) equipment (model TM3030, manufactured by Hitachi, Germany), which was complemented by electron diffraction analysis (EDX).
XPS
The X-ray photoelectron spectroscopy (XPS) analysis was carried out using an Axis UltraDLD XP spectrometer (Kratos Analytical Limited, Manchester, UK). During the analysis, the photoelectrons were emitted from the sample at a 90° angle relative to the sample surface, and the X-rays were incident at an angle of 33.3° (or 56.7° between the direction of X-ray incidence and the direction of captured photoelectron emission). A hybrid lens mode with a slot aperture was employed, providing an analysis area of 700 × 300 µm². An 80 eV pass energy was used to acquire survey spectra, covering the binding energy (BE) range from 0 to 1100 eV. Detail spectra were explicitly recorded for O 1s, C 1s, Ti 2p, and N 1s, employing a 40 eV pass energy. The instrument’s resolution for the survey scans was 1.1 eV, while for the detail scans under these settings, it was 0.71 eV. This resolution was determined by measuring the full width at half maximum (FWHM) of the Ag 3d5/2 peak observed on sputter-cleaned silver foil. The samples were mounted on an insulating support to mitigate potential differential charging effects due to insulating surface layers, and low-energy electrons were applied for charge compensation. The spectra were reference-adjusted based on the position of the C 1s peak, attributed to aliphatic C-C/C-H bonds and set at 284.8 eV BE. In the context of our study, the analysis of X-ray photoelectron spectroscopy (XPS) data was conducted utilizing the CasaXPS software package developed by Casa Software Ltd, Teignmouth, UK.
Statistics
The dataset was checked for normality, and all variables were non-parametrically distributed; therefore, all data were presented as boxplots. The groups were statistically compared using multiple comparisons of the Kruskal–Wallis’s test. A probability of less than or equal to 0.05 was considered significant. Due to the high significance of some measurements, only selected significant groups were presented for easier visualization.
All data obtained were analysed using GraphPad Prism version 10.1 (GRAPH PAD Software Inc, CA, USA). All graphical representations were performed on GraphPad Prism and Biorender.
Results
The Scanning Electron Microscopy (SEM) images show a textured and rough topography characteristic of titanium dental implant surfaces. Differences in brightness and contrast across the images suggest variations in the surface composition and roughness after applying different debridement agents (Figure 1). The surface cleaned with polymeric NPs exhibits a unique texture compared to other cleaning agents. This could indicate the mechanical or chemical interactions between the polymeric NPs and the protein layer. There appear to be fewer large debris or protein remnants, which might suggest that polymeric NPs effectively disaggregated or removed larger protein agglomerates. Some areas still have darker spots or patches, indicating incomplete cleaning or regions where the polymeric NPs did not entirely remove the protein layer (Figure 2(e) and (k)). The overall uniformity of the surface is disrupted by several irregularities, possibly caused by the binding of polymeric NPs to the titanium surface or the remnants of the pellicle. When compared to other cleaning agents like “Saline Water” (Figure 2(a) and (g)), “PrefGel” (Figure 2(B) and (h)), “H2O2” (Figure 2(c) and (i)) and “Gum Paroex” (Figure 2(d) and (j)), the polymeric NPs cleaned surface exhibits a distinct pattern, which could be due to the inherent properties of the polymeric NPs or their specific interaction with the protein layer. The “Without Pellicle” control shows a clean and homogeneous surface, indicating the original state of the titanium before contamination (Figure 2(f)). The “With Pellicle” control exhibits a more cluttered and irregular surface, displaying the presence of the salivary pellicle as expected (Figure 2(l)).

SEM panel before and after recontamination. All are taken at the same scale bar.
Energy-dispersive X-ray spectroscopy (EDX) micrographs show the surfaces of titanium implants exposed to various cleaning agents to remove the acquired pellicle layer formed from human saliva. Four elements were selected to visualise the distribution of these elements on the titanium implant surface. These micrographs are colour-coded to indicate the presence and distribution of different elements: nitrogen (red), titanium (purple), oxygen (blue), and carbon (green) (Figure 3). In the “Without Pellicle” (Figure 3(f)) and “With Pellicle” rows Figure 3(l)), red indicates the presence of pellicle proteins, as nitrogen is the only element present in the pellicle, and not any of the decontamination is used. The green colour indicates carbon and, thus, the presence of polymeric or other organic contaminations. The green colour is notably less intense for the NP groups (Figure 3(e) and (k)) when compared to other cleaning agents like “Saline Water” (Figure 3(a)), “PrefGel” (Figure 3(b)), and “Gum Paroex” (Figure 3(d)). This suggests that polymeric NPs are relatively effective in expelling the protein layer from the titanium surface. When observing the titanium element, purple is most vital in the “Without Pellicle” control (Figure 3(f)), which is expected as there is no contamination. PrefGel and Saline Water have the least purple colour. The “polymeric NPs,” similar to H2O2 and Gum Pareox treated surfaces, imply exposure to the titanium surface. The carbon signal (green) is strong in some areas of the “Polymeric NPs” group, possibly indicating the presence of polymeric material from the cleaning nanoparticles. The lowest carbon seems to be for the H2O2 group (Figure 3(c) and (i)) and the highest for PrefGel (Figure 3(b) and (h)). The blue signal (oxygen) in the “Polymeric NPs” column appears to be comparable to the other groups, suggesting a similar degree of oxygen-containing compounds or residues on the titanium surface. Overall Efficacy: While the polymeric NPs seem to have an appreciable cleaning effect, especially regarding protein interception, they do not completely clear the surface, as indicated by the remaining carbon and oxygen signals. Compared to other cleaning agents, the Polymeric NPs present a unique profile with a distinct pattern of element distribution. These observations suggest that while polymeric NPs effectively reduce protein contamination, they do not fully restore the titanium surface to an uncontaminated state, as evidenced by the residual presence of carbon and oxygen signals. It is also noticeable that the cleaning efficacy of polymeric NPs is comparable to that of some other agents, particularly in nitrogen removal.
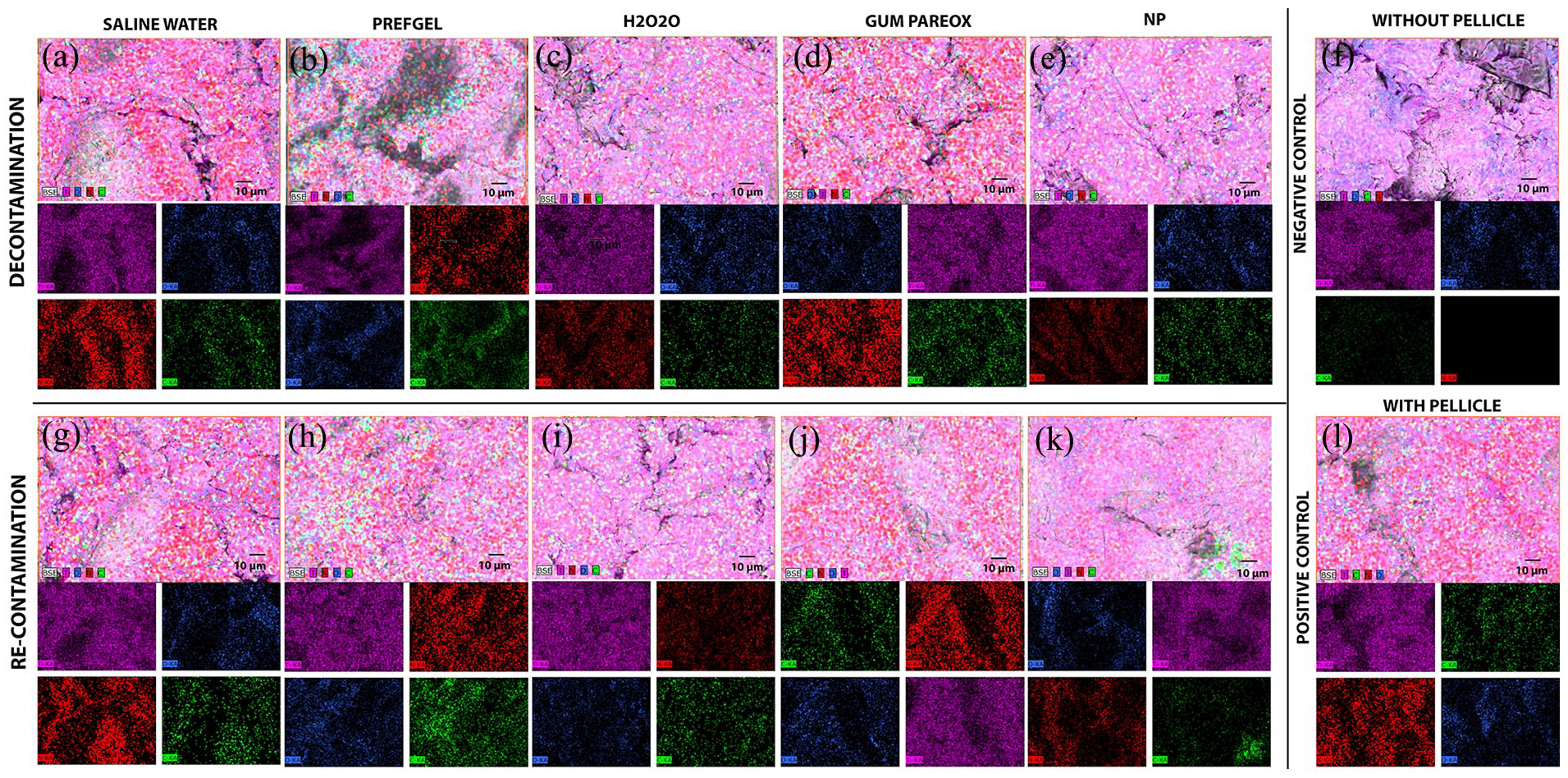
EDX panel before (top rows, a–e) and after recontamination (bottom rows, g–k), control: without pellucle (f) and with pellice (l). The upper panels are combined EDX images of four elements (Ti, C, O, and N). In contrast, the lower panel shows the elements of Titanium: Purple, Oxygen: Blue, Carbon: Green and Nitrogen: Red.
The impact of various cleaning methods was evaluated on the contact angle of titanium implants (Figure 4). After re-contamination, PrefGel showed a significantly higher contact angle compared to Gum Paroex (Mean Diff: 26.51, p = 0.0009) and “Without pellicle” (Mean Diff: 58.02, p < 0.0001). There was a significant at a lower contact angle when comparing PrefGel with the group “With pellicle” (Mean Diff: −28.70, p = 0.0028). Comparisons with H2O2, Saline water, and Polymeric NPs showed no significant difference. H2O2-Recontaminated (Mean Diff: −23.46, p = 0.0045), Saline water-Recontaminated (Mean Diff: −25.46, p = 0.0012), “With pellicle” (Mean Diff: −47.30, p < 0.0001), and a significantly higher contact angle when compared to “Without pellicle” (Mean Diff: 39.41, p < 0.0001). Saline water showed a significantly higher contact angle compared to Gum Paroex (Mean Diff: 26.51, p = 0.0009) and Without pellicle (Mean Diff: 58.02, p < 0.0001) and a lower contact angle compared to “With pellicle” (Mean Diff: −28.70, p = 0.0028). Polymeric NPs showed a significantly higher contact angle compared to “Without pellicle” (Mean Diff: 66.15, p < 0.0001) and Gum Paroex -Recontaminated (Mean Diff: +34.64, p < 0.0001). No significant differences were observed when compared with PrefGel, H2O2, Saline water, Gum Paroex, and other cleaning methods (Figure 4).

Contact angle for the titanium surfaces before and after recontamination (*p < 0.05, ***p < 0.001, ****p < 0.0001, n = 3).
An XPS analysis was performed on all titanium surfaces after decontamination (Figure 5(a)) and after re-contamination (Figure 5(b)), showing full survey scans in Figure 5. As several elements were detected, a detailed scan was performed for the Ti 2p (Figure 6(a) and (b)), C 1s (Figure 6(c) and (d)), O 1s (Figure 6(e) and (f)) and N 1s (Figure 6(g) and (h)). Additional Ca 2p (Figure 7(a) and (b)), Cl 2p (Figure 7(c) and (d)), and Na 1s (Figure 7(e) and (f)).

Selected XPS survey scans of the surface after surface contamination (a) and re-contamination (b) (n = 1).

Selected XPS high-resolution spectra for titanium (a,b), carbon (c,d), oxygen (e,f) and nitrogen (g,h). The left panel has a titanium surface after pellicle decontamination, and the right panel has a titanium surface after re-contamination with a pellicle (n = 1).

Selected XPS high-resolution spectra for calcium (a,b), chlorine (c,d) and sodium (e,f). The left panel titanium surface after pellicle decontamination, and the right panel titanium surface after re-contamination with pellicle (n = 1).
No significant difference in titanium level was seen between Polymeric NPs and Saline water (Mean Diff: −1.100, 95% CI: −5.345 to 3.145, p = 0.9979). Compared to PrefGel, polymeric NPs showed significantly higher effectiveness in cleaning the implant surface as they showed higher Ti levels (Mean Diff: −7.433, 95% CI: −11.68 to −3.188, p < 0.0001). There were no significant differences in the efficacy of polymeric NPs compared to H2O2 (Mean Diff: −2.933, 95% CI: −7.179 to 1.312, p = 0.3897) and Gum Paroex (Mean Diff: −2.133, 95% CI: −6.379 to 2.112, p = 0.7968) after surface decontamination (Figure 8(a)).

Selected elements, titanium (a), carbone (b), nitronge (c), oxygen (d), calcium (e) and chloride (f) with atomic percentage (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001, n = 3).
The mean carbon level reduction on titanium implants when cleaned with polymeric NPs compared to Saline water was not statistically significant (Mean Diff: −4.667, 95% CI: −14.18 to 4.848, p = 0.8190) after surface decontamination. This suggests comparable efficacy between these two methods. Compared to PrefGel, polymeric NPs showed a significantly lower carbon level after surface decontamination (Mean Diff: 11.33, 95% CI: 1.819–20.85, p = 0.0105). There are no significant differences in carbon level reduction between polymeric NPs and H2O2 after surface decontamination (Mean Diff: −1.000, 95% CI: −10.51 to 8.514, p > 0.9999). The comparison between polymeric NPs and Gum Paroex also showed no significant difference (Mean Diff: 5.667, 95% CI: −3.848 to 15.18, p = 0.5975). Notably, Polymeric NPs, when re-contaminated, showed a significant carbon reduction compared to PrefGel -Recont (Mean Diff: −13.00, 95% CI: −22.51 to −3.486, p = 0.0023). None of the cleaning methods produced a surface with carbon levels as low as the clean control (Figure 8(b)). After re-contamination, H2O2 gave surfaces the lowest carbon levels, significantly lower than PrefGel, polymeric NPs, and gum parox.
A significant reduction in nitrogen levels was observed when Polymeric NPs were compared to PrefGel (Mean Diff: 2.600, p = 0.0307) after decontamination: No significant differences were found between Polymeric NPs and various cleaning agents (Figure 8(c)) after re-contamination, however, all groups had higher nitrogen against the control samples “With Pellicle” (Figure 8(c)).
The difference in oxygen levels between polymeric NPs and PrefGel was not statistically significant (Mean Diff: −6.333, 95% CI: −13.08 to 0.4122, p = 0.0796), suggesting similar effectiveness after decontamination. Similarly, when compared to H2O2, the effect of Polymeric NPs on oxygen levels showed no significant difference (Mean Diff: −0.3333, 95% CI: −7.079 to 6.412, p > 0.9999) after decontamination. The influence of re-contamination on the efficacy of polymeric NPs was examined. The comparison between polymeric NPs and polymeric NPs after recontamination showed no significant difference (Mean Diff: 5.667, 95% CI: −1.079 to 12.41, p = 0.1602), indicating consistent performance even after recontamination. The highest level of oxygen after re-contamination was for H2O2; however, it was not significantly different from NP (Figure 8(d)). PrefGel was the most effective calcium remover and was significant against most groups, while saline water was the poorest (Figure 8(e)). Compared with PrefGel, polymeric NPs did not show a significant difference in calcium removal (Mean Diff: −0.8667, 95% CI: −3.103 to 1.369, p = 0.9530). Similarly, no significant differences were observed between polymeric NPs and other cleaning methods like H2O2 (Mean Diff: 1.700, 95% CI: −0.5362 to 3.936, p = 0.2655), Gum Paroex (Mean Diff: 0.1667, 95% CI: −2.069 to 2.403, p > 0.9999), and Saline water (Mean Diff: 2.367, 95% CI: 0.1305–4.603, p = 0.0315) (Figure 8(e)). The chlorine levels of the surface were low and almost negligible as none had higher median levels than 0.6 atomic percentage (Figure 8(f)).
There was no difference between different decontamination procedures for C=O/O-C-O%; all values were higher than the positive control (with pellicle) (Figure 9(a)). Polymeric NPs significantly increased O=C-OH% levels compared to PrefGel against most groups after decontamination (Figure 9(b)). A notable effect was observed when comparing H2O2 to Polymeric NPs, with a mean difference of −3.001 (95% CI: −5.008 to −0.9946, p = 0.0006), suggesting a more pronounced effect by H2O2. Polymeric NPs demonstrated a consistent impact on recontamination scenarios with the other groups and even lower value than after decontamination (Figure 9(b)). Regarding C-O-C*=O, the lowest value was seen for Gum Paroex; however, it was only significantly lower toward Saline water and comparable to the positive controls (Figure 9(c)). No significant changes were observed after recontamination.

Quantification of selected carbon chemical bonds (C=O/O-C-O and O=C-OH) from the XPS high-resolution spectra (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001, n = 3).
Polymeric NPs, when compared with PrefGel, did not show a significant difference in the reduction of C-C/C-H levels (Mean Diff: −0.8667, 95% CI: −3.103 to 1.369, p = 0.9530). The comparison with other cleaning methods such as H2O2 (Mean Diff: 1.700, 95% CI: −0.5362 to 3.936, p = 0.2655) and Gum Paroex (Mean Diff: 0.1667, 95% CI: −2.069 to 2.403, p > 0.9999) also showed no significant differences in the context of de or recontamination. All groups had lower values than both controls (Figure 10(a)). Polymeric NPs, when compared to PrefGel, showed no significant difference in the reduction of O-C=O levels (Mean Diff: 1.230, 95% CI: −3.368 to 5.828, p = 0.9972). Similarly, there were no significant differences in the effectiveness of Polymeric NPs compared to other cleaning methods such as H2O2 (Mean Diff: 1.522, 95% CI: −3.076 to 6.120, p = 0.9845) and Gum Paroex (Mean Diff: −2.150, 95% CI: −6.747 to 2.448, p = 0.8579). In recontamination contexts, the efficacy of Polymeric NPs did not show significant differences compared to the other groups. The effectiveness of polymeric NPs in reducing C-O-C/C-OH% levels was not significantly influenced by decontamination or re-contamination, although all groups had higher levels in both controls. Hydrogen peroxide was most effective in the decontamination phase (Figure 10(c)).
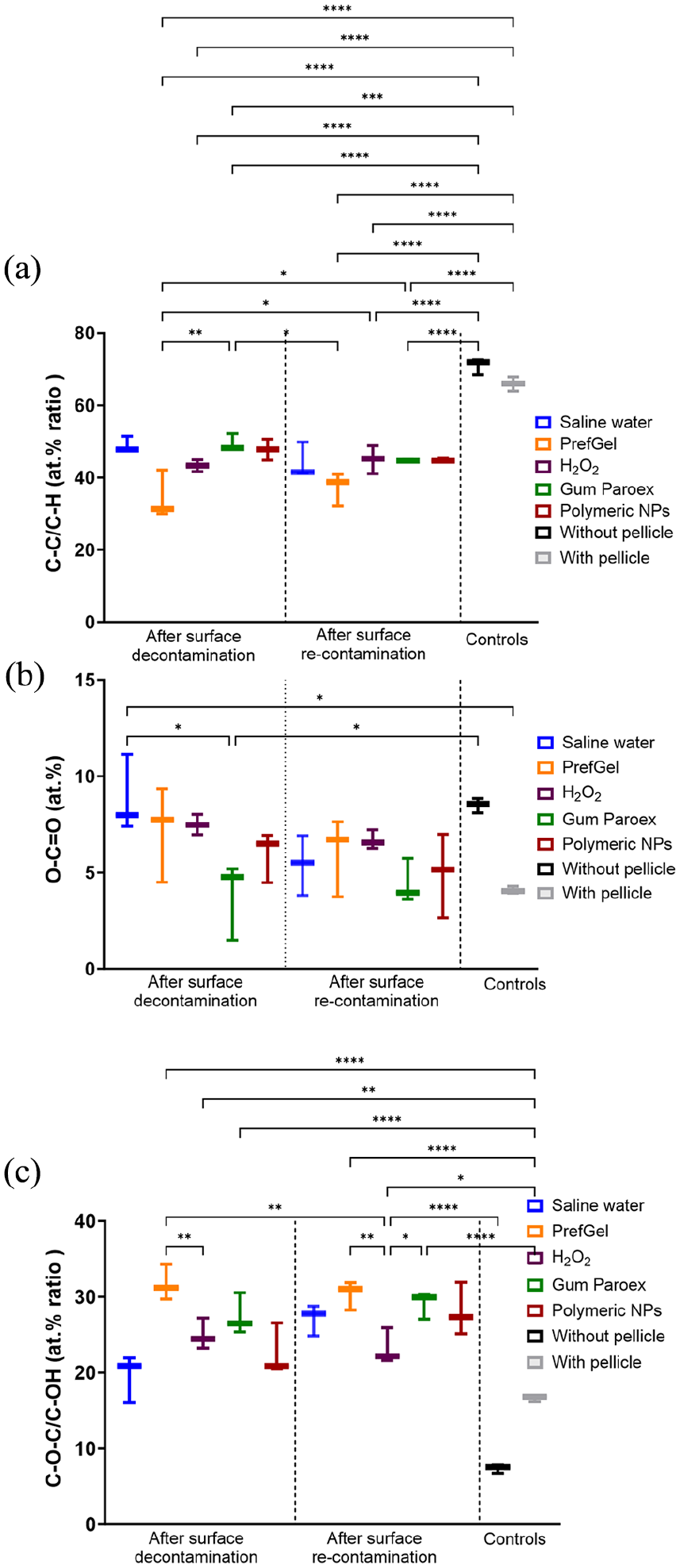
Quantification of selected carbon chemical bonds (C-C/C-H (a), O-C=O (b) and C-O-C/C-OH (c)) from the XPS high-resolution spectra (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001, n = 3).
Discussion
Various chemical agents have long been utilised in dental practice, with many now being repurposed for the decontamination of implant surfaces.21,48 The present research compares these agents’ impact on pellicle elimination and development on implant surfaces. The effectiveness of these existing agents on implant surfaces appears to be limited, as highlighted by the outcomes of this current laboratory investigation. Therefore, the creation of innovative decontamination methods holds significant importance and is anticipated by specialists in the field.22,49 The primary objective of this study was to evaluate the effectiveness of polymeric nanoparticles (NPs) in decontaminating titanium dental implant surfaces. This evaluation focused on assessing the ability of these nanoparticles to remove pellicle layers—a crucial factor in preventing peri-implant diseases. Previous studies have demonstrated that these nanoparticles (NPs) exhibit non-toxic and non-apoptotic properties when tested against a human fibroblast cell line 41 and do not interfere with osteoblasts proliferation/differentiation when used on titanium surfaces. 44 The composition of these NPs includes 2-hydroxyethyl methacrylate, ethylene glycol dimethacrylate, and methacrylic acid. 41 Notably, their synthesis is characterized by an efficient and environmentally friendly process devoid of hazardous solvents or residual non-polymerized compounds that could potentially disrupt the cellular biological activities of the NPs. These nanoparticles exhibited a mean particle size of 150 ± 10 nm, with a polydispersity index (PDI) of 0.2, indicating a uniform size distribution. Surface characterisation revealed the presence of carboxyl functional groups, which play a pivotal role in binding proteinaceous substances, such as the pellicle layer, via electrostatic and chemical interactions. Additionally, these nanoparticles’ high surface area-to-volume ratio enhances their adsorption capabilities, while a zeta potential of −35 mV ensures stable dispersion in aqueous environments and facilitates interactions with positively charged proteins. 41 Bueno et al. 42 have shown that these NPs could decrease the bacterial load in dental biofilms and alter their formation dynamics.
The study rigorously compared the performance of polymeric NPs with traditional decontamination methods, including PrefGel®, hydrogen peroxide, GUM® Paroex®, and saline water. The traditional agents utilised in this study displayed varied chemical properties and mechanisms of action. PrefGel®, a 24% EDTA-based hydrogel, is a chelating agent that disrupts protein-mineral bonds by removing calcium ions. 50 Hydrogen peroxide (H2O2) at a 3% concentration is a robust oxidising agent with proteolytic properties, effectively breaking down organic contaminants.51,52 GUM® Paroex® combines 0.12% chlorhexidine digluconate and 0.05% cetylpyridinium chloride, providing antimicrobial action particularly effective against biofilm-associated bacteria. 53 This comprehensive comparison aimed to provide a thorough understanding of polymeric NPs’ efficacy in dental implant hygiene and maintenance. By evaluating their performance against established cleaning protocols, the study offers valuable insights into the potential advantages and limitations of using polymeric NPs in dental implant care.
Efficacy of polymeric nanoparticles in pellicle removal
A key focus of polymeric nanoparticles (NPs) effectiveness in dental implant decontamination is their ability to reduce nitrogen levels on the titanium surface. 54 This reduction is particularly significant as it indicates the removal of protein components, which are integral to forming the pellicle layer. 55 The pellicle layer, in turn, is critical in the initial stages of biofilm formation, which can lead to peri-implant diseases.55,56
The study demonstrated that polymeric NPs were notably effective in decreasing nitrogen levels compared to traditional decontamination methods, such as PrefGel®. This finding suggests that polymeric NPs possess a substantial capacity to disrupt or remove the proteinaceous components of the pellicle. The potential role of polymeric NPs in inhibiting the initial steps of biofilm formation on dental implants is pivotal in preventing implant-related infections. The detailed characterisation of polymeric nanoparticles highlights their unique advantages, particularly their functional carboxyl groups and nanoscale interaction capabilities, which differentiate them from conventional agents. PrefGel® relies on chelation, while hydrogen peroxide exhibits broad-spectrum proteolytic activity. GUM® Paroex®, while effective against microbial biofilms, has limited efficacy in pellicle removal. This diversity in mechanisms underscores the complexity of dental implant decontamination, where a tailored approach, potentially combining nanoparticles and traditional agents, may provide superior outcomes. This observation underscores the promising potential of polymeric NPs in implant surface decontamination.
However, while polymeric NPs demonstrated this specific efficacy in nitrogen reduction, it is essential to consider this finding in the broader context of overall implant surface cleanliness and maintenance. The ability of polymeric NPs to reduce protein layers must be weighed against other critical factors, such as their impact on the implant’s surface morphology, overall cleanliness, and the potential for recontamination. Moreover, the comparison with traditional cleaning methods, which might be more effective in other aspects of decontamination, provides a comprehensive understanding of the potential advantages and limitations of using polymeric NPs in dental implant care.
The ability of polymeric nanoparticles to reduce nitrogen levels and thereby potentially diminish protein layers on dental implants represents a promising avenue in implant surface decontamination, as also seen by Al-Qarni et al., 57 where a resin with calcium phosphate nanoparticles showed protein-repelling behavior. However, further research and a balanced assessment of all relevant factors are essential to ascertain their role and efficacy in clinical applications fully. While polymeric NPs show promise, particularly in reducing nitrogen levels and thus potentially preventing initial biofilm formation, their overall efficacy compared to established agents like hydrogen peroxide may vary depending on the decontamination requirements. Hydrogen peroxide offers broad-spectrum cleaning, a recognised proteolytic activity 58 and a proven track record but might lack the targeted, nano-level action that polymeric NPs can provide. A similar finding has been reported by Jervoe-Storm et al., 59 where hydrogen peroxide was superior to all other irrigation solutions (saline water and GUM® Paroex®) overall. Therefore, the choice between these agents should be guided by the specific clinical scenario, desired outcomes, and understanding of their strengths and weaknesses. 54
Surface morphology and chemical composition changes
The SEM images suggest that while Polymeric NPs have an observable impact on protein removal, their cleaning efficacy varies and may not achieve complete removal across the entire surface. The SEM images also suggested that polymeric NPs effectively disaggregated or removed larger protein agglomerates. The carboxyl superficial functional groups in NPs may play a role in the produced protein disruption, as they may interact with amino radicals, forming peptide bonds [3R]. This points to their potential role in minimising the initial steps of biofilm formation. Despite their effectiveness, some areas showed incomplete cleaning, with darker spots or patches indicating residual contaminants. One must also consider that this is an in vitro pellicle model, and the dental implant biofilm from a clinical setting may be different and, as El Chaar et al. 60 and Qian et al. 61 reported, much more challenging to remove by chemical procedures and a combination of mechanical debridement is needed. The images also indicate that the interaction between polymeric NPs and the titanium surface could be complex, potentially affecting the surface topography. Further analysis, including quantitative surface roughness measurements and higher magnification imaging, would be beneficial to elucidate the detailed mechanisms of cleaning and the potential for surface modification by polymeric NPs.
The cleaning method significantly affects the contact angle of titanium implants, indicating that different methods alter the surface characteristics to varying degrees. Polymeric NPs show a pronounced increase in contact angle compared with the “Without pellicle” condition, suggesting a substantial impact on surface wettability. The variability in contact angle changes across different cleaning methods highlights the importance of method selection based on the desired surface properties of titanium implants post-cleaning. 62 Surface hydrophilicity is quantified by measuring the contact angle, with a pristine titanium surface being exceptionally hydrophilic, typically exhibiting a contact angle nearing zero. 63
Elemental reduction: Energy-dispersive X-ray spectroscopy (EDX) and X-ray Photoelectron Spectroscopy (XPS) analyses showed that polymeric NPs influenced the elemental composition of titanium surfaces. The reduction in elements like nitrogen and carbon was notable. XPS analysis revealed that while polymeric NPs effectively reduced protein contamination, they did not completely restore the titanium surface to an uncontaminated state, evidenced by residual carbon and oxygen signals. 64 In re-contaminated conditions, polymeric NPs maintained a relatively consistent performance, as indicated by the elemental composition of the surfaces.
Efficacy in cleaning: Traditional cleaning methods, such as PrefGel®, GUM® Paroex® and saline water, were less effective in altering the chemical composition. Chlorhexidine-containing products have also been shown to compromise the biocompatibility of titanium surfaces, 41 and GUM® Paroex® should, therefore, be used with care when detoxifying implants. While some were more effective in certain aspects (e.g., overall surface cleanliness), they might not provide the targeted action at the nano-level like polymeric NPs. Conventional methods might alter the surface characteristics more significantly than NPs. Both polymeric NPs and traditional methods left certain residual elements on the titanium surfaces, though the nature and extent of these residuals varied. Residual would hinder the osseointegration of the implant.65 –67 PrefGel®, a hydrogel-based solution, showed the highest residual left and the worst cleaning effect. The analysis indicates that polymeric NPs effectively reduce carbon levels on protein-contaminated titanium implants, albeit with varying efficacy compared to other treatments like PrefGel®, H2O2, and GUM® Paroex®. The presence of a pellicle significantly influences the cleaning efficacy of polymeric NPs, and their performance remains consistent post-recontamination. As reported by others, these findings underscore the importance of considering the specific cleaning method and surface conditions when managing protein contamination on titanium implants.68 –70
Future research on polymeric nanoparticles (NPs) for dental implant decontamination should take a comprehensive approach, focusing on immediate cleaning efficacy and long-term impacts on implant integrity, patient health, and the environment. Optimising NP formulations, such as adjusting size, shape, and surface properties, is critical to enhancing biofilm removal. Developing NPs for targeted antimicrobial delivery and controlled release could improve biofilm prevention and reduce peri-implant diseases. Studies on NP biocompatibility and toxicity are essential to ensure the safety of surrounding tissues. Long-term research should examine the durability of NPs in maintaining implant cleanliness and the effects of repeated use on titanium surfaces. Clinical trials are needed to assess patient outcomes, including peri-implant disease recurrence and implant longevity. Additionally, evaluating the cost-effectiveness and practicality of NP-based cleaning methods is essential for broader adoption.
Conclusion
The present study comprehensively evaluated the efficacy of polymeric nanoparticles (NPs) in decontaminating titanium dental implant surfaces, focusing on their ability to remove pellicle layers and prevent recontamination. Comparative analyses were performed against conventional decontamination methods, including PrefGel®, hydrogen peroxide, GUM® Paroex®, and saline water, utilising a range of analytical techniques such as Scanning Electron Microscopy (SEM), Energy-dispersive X-ray spectroscopy (EDX), and X-ray Photoelectron Spectroscopy (XPS).
Our findings indicate that while polymeric NPs demonstrate a certain level of effectiveness in cleaning titanium surfaces, their performance could be more consistently superior to traditional cleaning methods. Specifically, the study revealed that polymeric NPs showed no significant advantage in reducing titanium levels or carbon content compared to saline water and other standard cleaning agents. Furthermore, polymeric NPs’ efficacy in elemental removal (titanium, carbon, nitrogen, oxygen, calcium, and chlorine) was like conventional cleaning agents in initial decontamination and after-recontamination scenarios.
A critical observation was the significant reduction in nitrogen levels achieved by polymeric NPs compared to PrefGel®, highlighting their potential in protein removal. However, this advantage was not evident in the context of re-contaminated surfaces. Additionally, the impact of polymeric NPs on the contact angle of titanium surfaces suggested variations in surface hydrophilicity/hydrophobicity post-treatment. Still, these changes were not significantly distinct from those induced by other decontaminants.
In terms of reducing specific carbon chemical bonds, polymeric NPs performed comparably to other cleaning agents without significant differentiation in the effectiveness post-recontamination. However, caution should be taken regarding some of the examined chemical bonds, as both positive and negative controls were outside the range of the tested cleaning agents. The study concludes that polymeric NPs are comparable to other cleaning methods in terms of their effectiveness in removing from protein-contaminated titanium implants; however, overall, H2O2 performed better.
This research underscores the significant potential of polymeric NPs, which, in addition to performing comparably to other decontamination materials, demonstrate antibacterial properties and the capacity to release therapeutic substances, as shown in other studies 42 . These qualities collectively position them as a valuable tool for enhancing the management of peri-implant diseases. The study contributes valuable insights into the nuanced performance of polymeric NPs, enriching the current understanding of implant dentistry and materials science.
Footnotes
Acknowledgements
Martin F Sunding, Sustainable Energy Technology, SINTEF Industry, is acknowledged for aid and a great help in conducting XPS analysis and, subsequently, data processing and interpretation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics statement
All participants gave written informed consent to participate in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Research Council of Norway, “MISFAITH” Grant No. 331752, and “DEBRIGEL” Grant no. 332148.
Data access statement
Data is available upon request to the corresponding authors.
