Abstract
Improving bone-graft substitutes and expanding their use in orthopedic and spinal surgery leads to shorter surgical times, fewer complications, and less pain among patients both in human and veterinary medicine. This study compared an elastic porous β-tricalcium phosphate/poly(L-lactide-co-ε-caprolactone) (β-TCP/PLCL) copolymer scaffold (composite scaffold) and a commercially available β-TCP/PLCL bone-graft substitute (chronOS Strip) in a rabbit calvarial defect. A bilateral, 12-mm circular defect was created in the parietal bones of 12 rabbits. Both graft materials were soaked in bone marrow aspirate before implantation, and the usability of the material was recorded during surgery. After a follow-up time of 24 (n = 5) and 48 (n = 7) weeks, artificial intelligence- (AI-) assisted micro-CT imaging was used to evaluate the bone formation and β-TCP distribution. Bone formation, implant material decomposition, and tissue reactions were further investigated through histopathology and AI-assisted histomorphometric analyses. Both materials supported tissue ingrowth and vascularization and modest 10%–16% new bone formation through the implant. In both materials the degradation advanced during the follow-up time, but there was implant material visible 48 weeks after implantation. Typical long term foreign body reaction with histiocytes, giant cells and lymphocytes, was seen in both materials being more pronounced in composite scaffold. The benefit of the new composite scaffold was its superior usability during surgery.
Introduction
Bone-graft harvesting is a common procedure in orthopedic and spinal surgery, performed when there is a need to support new bone formation in a defect or to repair large bone defects.1 –3 Autologous bone grafting has been the gold standard for years, although fairly high complication rates of between 7% and 39%.4,5 Synthetic bone-graft substitutes, propagated by advancements and innovations in material technology and tissue engineering, show great promise in reducing possible complications and decreasing pain for the patient, with no need for another invasive procedure.
β-tricalciumphosphate (β-TCP), which is used in commercially available bone-graft substitutes, is a ceramic that has the same calcium phosphorus ratio as native bone and has well-documented biocompatibility and osteoconductive properties.6 –9 The degradation of pure β-TCP in human body is slow and takes over a year. It occurs primarily through dissolution, while there is also some cell-mediated degradation.10 –12 The disadvantage of pure ceramics lies in their brittleness and often-reported difficulty in handling and shaping them during surgery.5,9,13 By contrast, poly(L-lactide-co-ε-caprolactone) (PLCL) is a biocompatible elastic copolymer composed of lactide and ε-caprolactone, which degrades in tissues through nonenzymatic hydrolysis.14,15 This co-polymer has been used in multiple fields in tissue engineering, such as urology and vascular procedures, as a scaffold for stem cell applications, suture materials, and bone-graft substitutes.16 –20 By mixing β-TCP and PLCL, degradation time and features of the bone-graft substitute can be altered, and create elastic and moldable composite scaffold. 21 Foaming the material with supercritical CO2 is cost effective, non-toxic and simple method for creating porous homogenous composite scaffold material with interconnected pores and high ceramic concentration.15,22 We have tested hydrolytic degradation, cytocompatibility and osteogenic capacity as well as mechanical properties of β-tricalciumphosphate and poly(L-lactide-co-ε-caprolactone) (β-TCP/PLCL) in vitro with promising results. 15 The modulus of this composite decreased significantly while compressing the material and also when it was heated up to 37°. 15 The composite proved to be elastic and easy to handle during surgery in our in vivo study in critical-sized rabbit calvarial model. 23 We also showed that this new composite was biodegradable, biocompatible, and promoted new bone formation through the defect. 23
The primary aim of the present study was to compare this elastic β-TCP/PLCL composite scaffold with a commercially available composed β-TCP/PLCL bone-graft substitute in a 12-mm calvarial defect in rabbits with 24- and 48-week follow-up periods. In addition, we sought to objectively and quantitatively assess micro-CT and histological data employing deep learning artificial intelligence (AI) for image analysis. We hypothesized that both materials promote bone and tissue ingrowth, support long-term new bone formation, and exhibit a good biocompatibility, with the new material proving more beneficial given its superior usability during surgery.
Materials and methods
β-tricalcium phosphate/poly(L-lactide-co-ε-caprolactone) composite scaffold
The composite scaffold was manufactured by melt-mixing the β-TCP (TCP, Plasma Biotal Ltd., Buxton, United Kingdom) and polylactide-co-ε-caprolactone 70L/30CL (Purasorb PCL 7015, Corbion Purac Biomaterials, Gorinchem, The Netherlands), which consisted of 50 wt-% β-TCP. The composite was foamed with supercritical CO2. The porosity was 65% and the average pore size was 380 µm ± 150 µm measured by micro-CT (MicroXCT-400, Zeiss). The foamed preforms were cut into 2.4-mm-thick (±0.5 mm) plates and sterilized using γ-radiation at a minimum dose of 25 kGy.
Commercially available bone graft substitute
As a control we used a commercially available bone-graft substitute (ChronOS Strip, DePuy Synthes, West Chester, PA, USA) composed of β-TCP and the copolymer poly(lactide)-co-ε-caprolactone, where the β-TCP granules are imbedded to the copolymer matrix. The information about the porosity or β-TCP content of this product, is not publicly available. This material is designed for use as a bone-graft substitute in spinal surgery. The manufacturer recommends moistening the implant with bone marrow or blood before implantation. In our study, we used a 3-mm-thick plate.
Surgical procedures
This study was approved by the Animal Experiment Board in Finland (ESAVI/5398/04.10.07/2014) and complied with Finnish legislation on animal experimentation and European Union Directive 2010/63/EU.
In total, 14 adult female New Zealand White rabbits (weight 3–4.4 kg, mean weight 3.8 kg) underwent surgery. Procedures were performed under general anesthesia achieved through the administration of 0.3 mg/kg medetomidine and 35 mg/kg ketamine. A total of 5–10 mg/rabbit intravenous propofol and/or isoflurane was used if necessary, during the operation. An intravenous dopamine infusion of 3–10 µg/kg/min was used to raise the blood pressure during surgery if needed. In addition, 15 mg/kg trimethoprime-sulfa was administered preoperatively. The rabbits received intravenous fluids (0.9% NaCl cum 5% glucose) during surgery.
Routine surgical-field preparation was performed, and strict aseptic protocols were followed during the operation. A midline skin incision was made to the skull and 12 mm, circular, full-thickness craniotomy defects were drilled on both parietal bones with a burr under continuous saline irrigation. Both materials were press-cut into a 12-mm-diameter disc during the surgery, and before implantation they were moistened with bone marrow aspirate collected from the medial femoral condyle. The moistening of the implants was completed in a squeezing pack. The right defect was filled with a composite scaffold and the left one with a chronOS Strip. After implantation the periosteum, subcutis, and skin were closed with 4–0 poliglecaprone 25 (Monocryl, Ethicon) using a continuous suture pattern.
Postoperatively, atipamezole was administered to reverse the effect of the medetomidine. For postoperative pain, the rabbits were treated with 0.03 mg/kg buprenorphine and 4 mg/kg carprofen preoperatively, with treatment continuing for 2–3 days following surgery. In addition, 0.2 mg/kg metoclopramide was administered twice daily until their intestinal function returned to normal. Rabbits were followed against infection or inflammation/tissue reactions in surgical field during the whole follow-up time. Animals were single cages during the recovery phase and after that they were housed at large group cages with enrichments. Hay and water were ad libitum.
Usability during the procedure
The ability to shape the materials intraoperatively, crumbling of the material during handling, the ability to fill the defect, and the visual evaluation of the bone–scaffold interface was all evaluated subjectively by the surgeons during the procedure.
Specimen collection
None of the rabbits were lost due to surgery, but two rabbits were excluded from the study because of ambiguous tattoo markings. Five rabbits were euthanized 24 weeks and 7 rabbits 48 weeks after the surgery. Euthanasia was performed with a lethal combination of ketamine, medetomidine, and pentobarbital. Parietal bone blocks were harvested with a diamond-coated saw.
Micro-CT imaging
A micro-CT study (MicroXCT-400, Zeiss) was performed for all of the harvested bone blocks (voltage 110 kV, current 91 µA, exposure time 4 s). From each bone block, 1600 projections were taken with a 19.97 µm voxel size. Projections were reconstructed using the manufacturer’s software (XM Reconstructor, Zeiss, Oberkochen, Germany).
AI-assisted image analysis of the micro-CT images
A deep, convolutional neural network-based artificial intelligent platform (Aiforia Create, Aiforia Technologies, Helsinki, Finland) was used to generate an AI model to recognize bone and β-TCP particles inside the calvarial defect.
For the AI model, 3D-images of the defects were transformed into multiple horizontal two-dimensional (2D) images. A series of 30 images per defect were uploaded to the software. The AI model was trained by using 37 different images including both time points and materials (Figure 1). The model was trained to recognize the defect, bone and β-TCP (Figure 1). Very radiodense (white) material were considered as a β-TCP and light grey area were considered as bone. Black radiolucent areas were considered as soft tissue or polymer and were trained to be a background. The AI model was considered ready when apparent visual improvement was not seen anymore. The total amount of 720 images were analyzed.
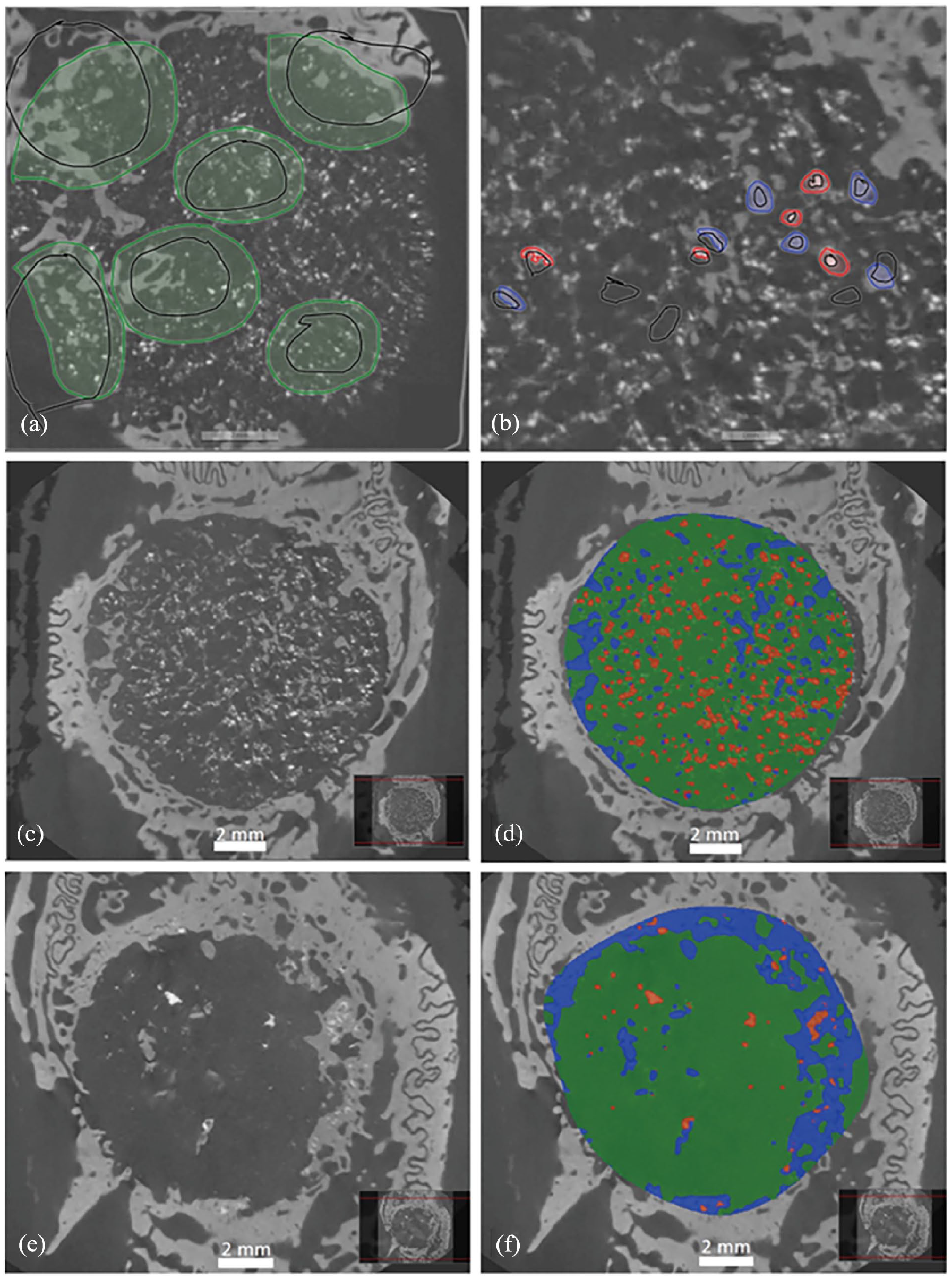
Examples from the micro-CT image analysis using Aiforia Create. Researchers defined the training regions (marked with black lines) which the AI software used for learning (a and b). Training regions marked with green (a) represent the defect, blue ones bone (b) and red ones β-TCP (b). No color represents background (a and b). 2D micro-CT images from composite scaffold and chronosStrip at 24 weeks are presented in (c and e), respectively. The same micro-CT images are shown after AI-analysis in (d and f). The colored areas show the analyzed defect, where blue color represents bone, red β-TCP and green radiolucent soft tissue or polymer.
After analyzing the 2D images with the AI model, 3D reconstruction from these analyzed images (Figure 2) were created by using multiple softwares. From the 3D reconstructed images, the distribution of β-TCP particles and the pattern of new bone formation were visually evaluated.
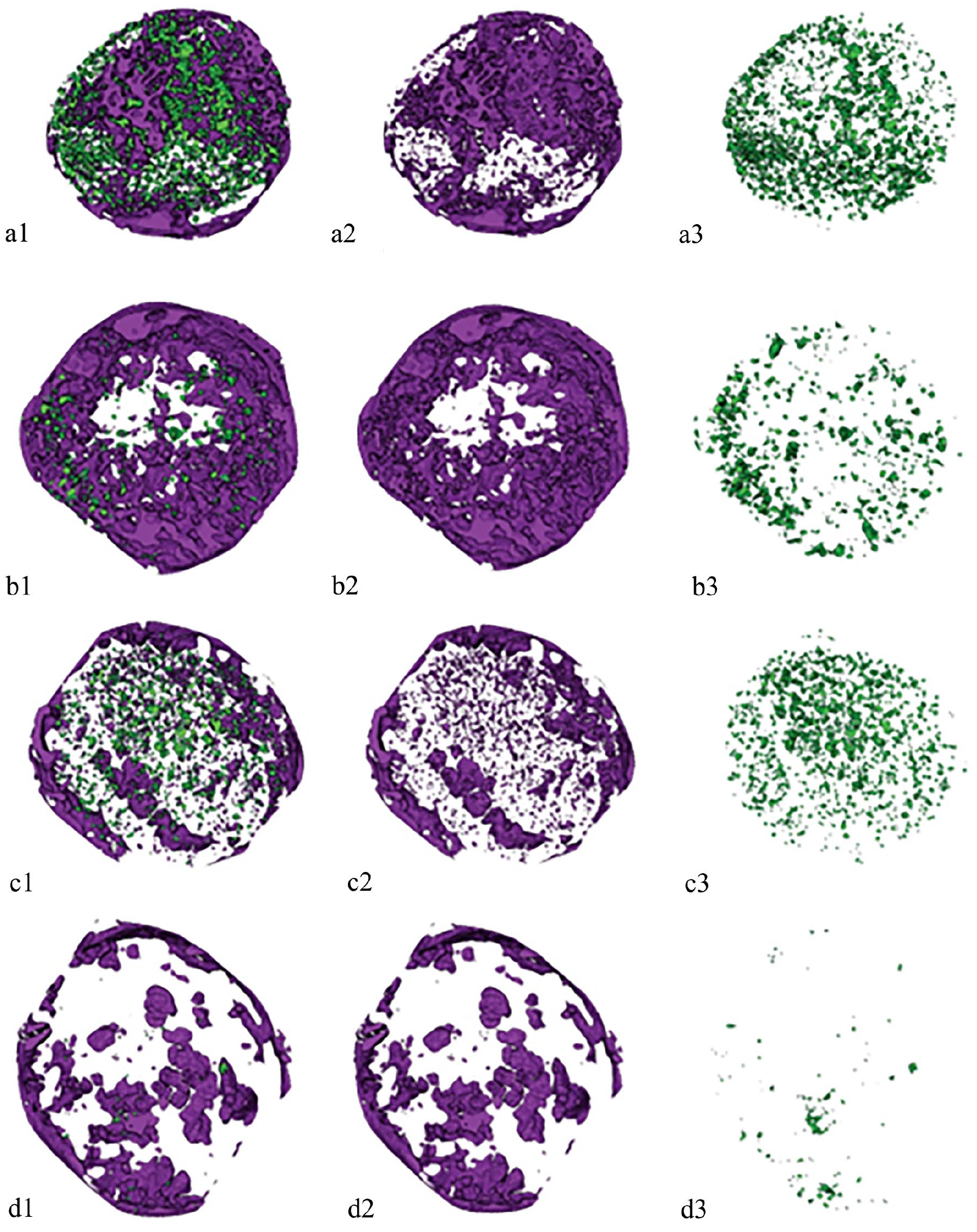
Representative 3D images of the defects reconstructed from AI-analyzed micro-CT images of the composite scaffold at 24 weeks (a1) and 48 weeks (c1) and corresponding images of the chronOS Strip (b1 and d1). In the reconstruction bone was chosen as purple and β-TCP green in order to enhance discernibility. Bone ingrowth was more prominent from the defect walls in the chronosStrip (b2 and d2). In the composite scaffold–filled defects (a2 and c2), smaller separate islands of bone are distributed throughout the defect, but in the chronOS Strip (b2 and d2) bone appears in fewer, but larger clumps. Degradation of the β-TCP advanced in both materials between 24 (a3 and b3) and 48 weeks (c3 and d3) being more prominent in the chronOS Strip. β-TCP particles are more evenly distributed in the composite scaffold (a3 and c3) than in the chronOS Strip (b3 and d3).
A more detailed description of the method is provided in the Supplemental materials.
Sample preparation for histopathological analysis
The parietal bone blocks were fixed in 10% formalin and decalcified in 20% EDTA, pH 7.2 for 9 weeks before paraffin embedding. Eight 5 µm sections from the midline of the defect were sectioned for each animal. The slides (containing two sections) were stained employing Weigert Van Gieson (WVG) and Masson–Goldner’s trichrome (MGT) methods and digitized to whole-slide images (WSIs) using a scanner (Pannoramic 250 FLASH, 3DHISTEC, Budapest, Hungary). The slide with best technical quality from each staining was selected for the descriptive analysis and the WVG-stained WSI was employed for AI-assisted image analysis and histomorphometry.
Histopathology and bone bridging
A veterinary pathologist blinded to the material identities subjectively evaluated the histopathological findings. The focus of the evaluation was on areas occupied by implant material, the bone, osteogenesis areas, and macrophages, as well as multinucleated foreign-body giant cells constituting the histiocytic reaction decomposing the implant. Specifically, the osteogenesis areas were defined as the greenish extracellular–treated material and blast-like cells in the MGT staining, and crisscross and/or speckled-collagen staining with plump blastoid cells in the WVG staining; microscopic islands of woven bone, capillaries, and osteoids were sometimes also present in the osteogenesis areas.
In order to determine which percentage of the defect was bridged by bone, the areas with bone were projected on the x-axis, as described in the literature. 24 Bone bridging percent was calculated from the histological slides using measuring tools in Aiforia Create software, no AI-technology was used.
AI-assisted histological image analysis
Aiforia Create was used in the histological image analysis and histomorphometry of the WVG-stained WSIs. A total of 33 WSIs representing both time points and materials, and different staining qualities were selected for AI training (Figure 3). An AI model was generated to recognize the defect areas and inside them the remaining implant material and seven tissue classes: (1) bone, (2) osteogenesis areas, (3) fibrosis, (4) histiocytic (macrophages and multinucleated giant cells) reaction, (5) adipose tissue, (6) lymphocyte infiltrates, and (7) blood vessels. Validation of the AI model was performed with a validation tool in the software and cross-check with descriptive histopathology was also performed.
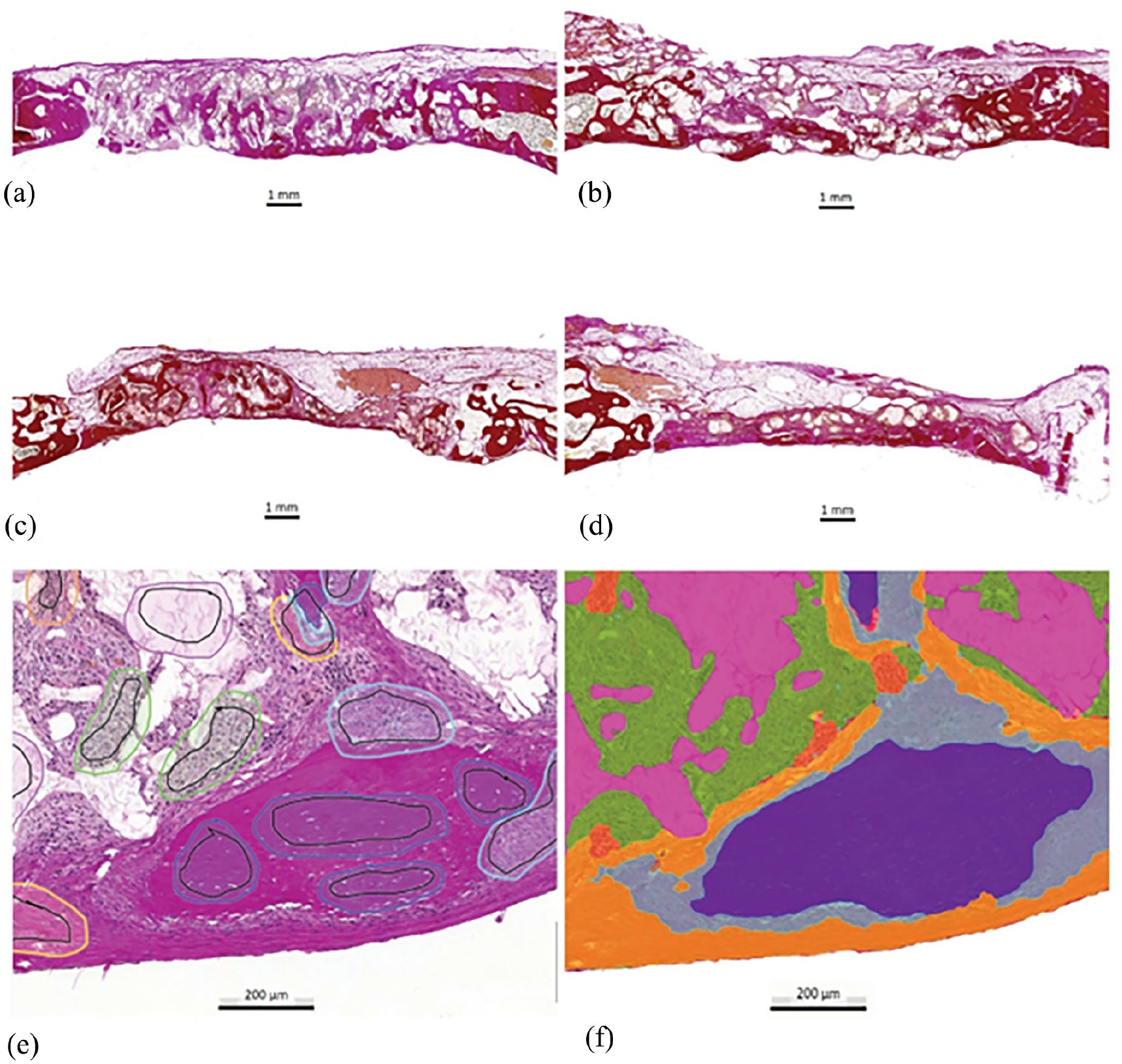
A typical histopathological appearance in the composite scaffold (a) and chronOs Strip (b) filled defects at 24 weeks. The new bone formation primarily advances along the dura, but also deposits along the defect walls and inside the implant material. A typical histopathological appearance in the composite scaffold (c) and chronOs Strip (d) filled defects at 48 weeks. The degradation of the implant material has proceeded. New bone formation is seen along the dural side and inside the implant. Training (e) of different tissue types and implant materials in the Aiforia Cloud. Black circles represent the training region and different colors represent different tissue types or implant materials. AI analysis (f) from the image (e) shows the results of different tissue types and implant materials. Blue/purple = bone, light blue = osteogenesis areas, green = histiocytic reaction, orange = lymphocytic reactions, yellow = fibrosis, and pink = implant material.
For the final analysis, one representative histological section for each defect per animal with a good technical quality was selected, totaling 24 analyzed images. The AI model marked recognized tissue types with different colors into the images and calculated defect area, the exact surface areas of the remaining implant material and the tissue classes in each section producing typical histomorphometric data. The results were used to calculate implant material/different tissue types as a percentage of the total defect. The percentage was used for statistical analysis.
A more detailed description of the method is provided in the Supplemental materials.
Statistical analyses
We performed statistical analyses (SPSS Statistics 26.0, IBM, Armonk, NY, USA). A Kolmogorov–Smirnov test was used to test the normal distribution of the data. The nonparametric Wilcoxon signed-rank test was applied for the comparison of the defect size, different tissues, and implant materials in the composite scaffold and the chronOS Strip at 24 and 48 weeks, respectively, and the Mann–Whitney test was used in the comparisons between 24 and 48 weeks. We considered a p-value under 0.05 (p < 0.05) as statistically significant.
Results
Usability of the material during the surgery
No visible particle loosening was observed from the composite scaffold during the procedure. During the cutting and moistening of the chronOS Strip material, β-TCP-particles were detached from the surface of the implant in most cases.
Because of the resilient structure of the composite scaffold, it was easier to implant to convex and slightly irregular defects than the more rigid chronOS Strip. The composite scaffold achieved a more uniform contact with the bone, whereas gaps were detected between the chronOS Strip and the bone rim along the defect walls.
AI-assisted image analysis of the micro-CT images
At 24 weeks in the composite scaffold, numerous small β-TCP pieces were scattered throughout the defect, whereas fewer and larger clusters of β-TCP were present in the chronOS Strip (Figure 2). New bone advanced from the defect walls towards the center from both materials, but the advancing bone appeared to be more copious in the chronOS Strip. The composite scaffold showed numerous small bony islands throughout the defect, while fewer and larger islands were present generally in the middle of the defect in the chronOS Strip.
At 48 weeks, the distribution of the β-TCP particles remained visually similar to those at 24 weeks (Figure 2), although the amount of β-TCP diminished. New bone emerged from the defect walls as at 24 weeks, although with less intensity. The composite scaffold generally formed small bony islands throughout the defect, whereas fewer but larger islands were present in the chronOS Strip.
Histopathological evaluation and AI-assisted histological image analysis
At 24 weeks follow-up
At 24 weeks, histopathology showed a moderate decomposition in both implant types (Figure 3). Both materials exhibited vascular ingrowth as well as osteogenesis and woven bone advancing from the thickened dural (inner) periosteum and from the drilled bone walls toward the center of the defect. In the composite scaffold group, the implant material was present mainly in variable-sized ragged pieces, whereas in the chronOS Strip group, the implant deposits were in general smooth-surfaced and larger. Both materials induced a histiocytic reaction around the implant material consisting of macrophages and large multinucleated giant cells. In addition, focal lymphocyte aggregates, a variable number of neutrophils, and single eosinophils were present. The reactive tissue was subjectively more abundant in the composite scaffold than in the chronOS Strip, whereas the chronOS Strip material appeared to show slightly more bone and osteogenesis than the composite scaffold (Figure 3).
In the AI-assisted histological image analysis at 24 weeks, the amount of combined bone and osteogenesis areas and the amount of remaining implant material exhibited no statistically significant difference when comparing material groups (Figure 4). The median percentages (IQR) of new bone and osteogenic areas were 9.5% (IQR 11.78%) in the composite scaffold group and 12.0% (IQR 8.43%) in the chronOS Strip group. Fibrous tissue was more prevalent in the chronOS Strip than in the composite scaffold (p = 0.043), whereas the composite scaffold exhibited a more intense histiocytic (p = 0.043) and lymphocytic reaction (p = 0.043) in comparison with the chronOS Strip.
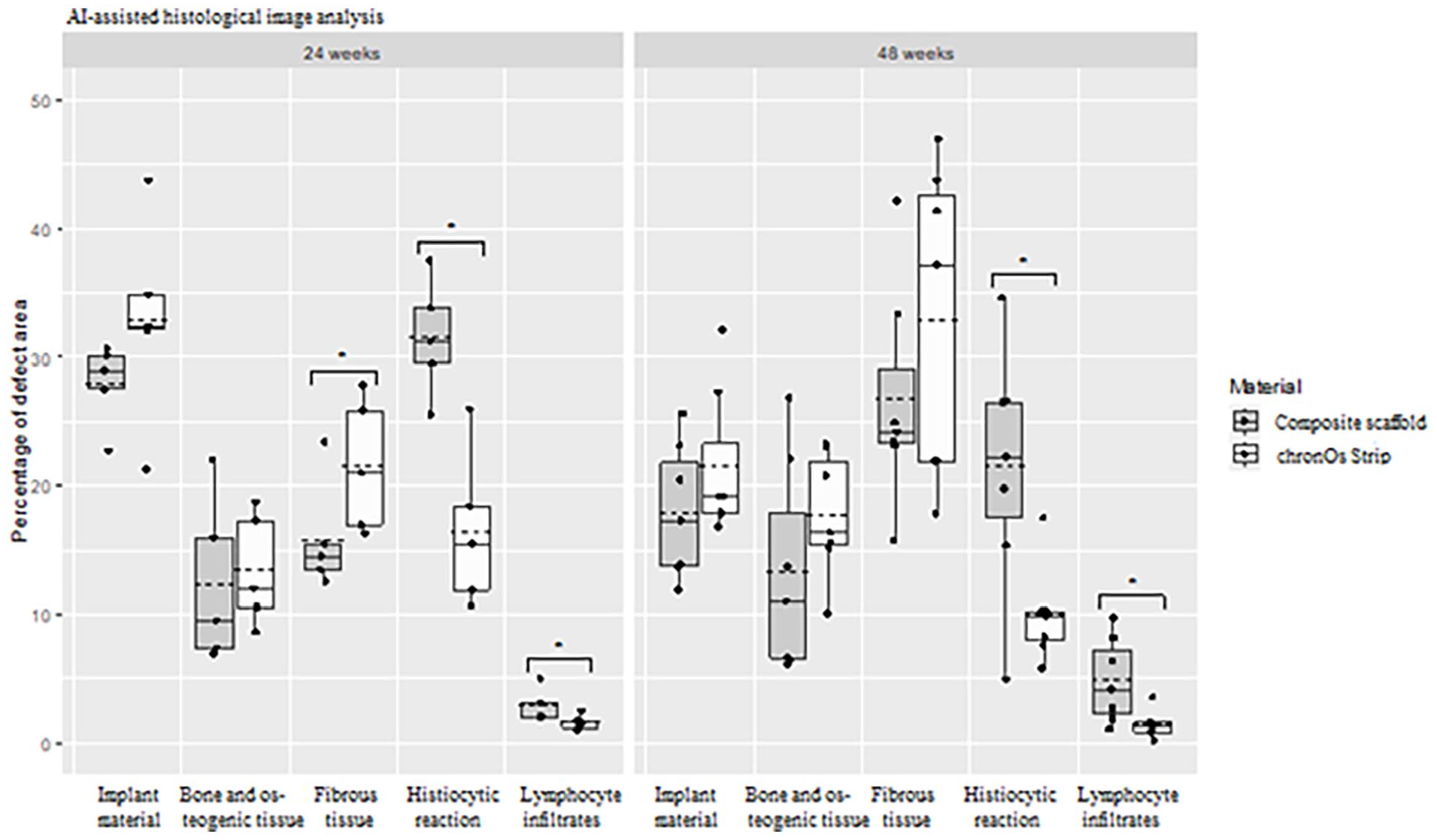
Percentage of implant material and different tissue types in the composite scaffold- and chronOS Strip-filled defects in AI-assisted histological image analyses at 24 weeks (n = 5) and at 48 weeks (n = 7).
The median percentages of bone bridging were 74% (IQR 24%) in the composite scaffold and 73% (IQR 49%) in the chronOS Strip.
At 48 weeks follow-up
In the histopathology at 48 weeks, osteogenesis appeared to advance mostly along the dura in both materials and variably from the drill hole walls (Figure 3). Both materials showed advanced decomposition of the implant material as well as a reduction in the histiocytic reaction and an increased amount of fibrotic tissue in comparison to 24 weeks. In addition, fibrosis appeared to be somewhat more pronounced in the chronOS Strip than in the composite scaffold group. Both groups exhibited blood vessel ingrowth to the implant.
In the AI-assisted histological image analysis at 48 weeks, the composite scaffold exhibited more pronounced histiocytic (p = 0.028) and lymphocytic reaction (p = 0.043) than the chronOS Strip (Figure 4). However, the materials did not show a significant difference in the abundance of the remaining implant material, or in the combined bone and osteogenesis areas or fibrosis.
The median percentages of bone bridging were 19% (IQR 52%) in the composite scaffold and 59% (IQR 18%) in the chronOS Strip.
Comparison between 24 and 48 weeks in the AI-assisted analysis
Both the composite scaffold (p = 0.012) and chronOS Strip (p = 0.019) demonstrated a significant loss of implant material between 24 and 48 weeks. However, the amount of bone was not significantly altered in either material. The histiocytic reaction decreased significantly (p = 0.019) in the chronOS Strip and fibrotic tissue increased significantly (p = 0.012) in the composite scaffold between 24 and 48 weeks.
Comparison between descriptive histopathology and AI-assisted analysis
In all the cases at 24 and 48 weeks, the descriptive text written by a pathologist correlate well with the quantitative data collected from the AI-analysis and there was no inconsistency between the results.
Discussion
This study compared the tissue ingrowth, bone formation, tissue reactions, and usability of a porous and bioabsorbable β-TCP/PLCL composite scaffold bone-graft substitute with a commercially available bone-graft substitute, the chronOS Strip, at 24- and 48-weeks follow-up.
The usability of the material is an important feature for surgeons. Although the chronOS Strip is advertised as being flexible and cuttable, the new composite scaffold was clearly more resilient and easily moldable during surgery. Due to its composition, the composite scaffold becomes more elastic and softer, while it heats up near the body temperature during the handling and compressing the material in a surgery. Implantation of the elastic composite scaffold into a convex and slightly irregular defect was easier compared with that using the chronOS Strip. The composite scaffold sealed the void space accurately and the contact with the bone was more consistent than with the chronOS Strip. Implants with poor adaptability of the defect edges due to the stiffness of the material, might cause more fibrous tissue formation.25,26 ChronOs Strip caused significantly more fibrous tissue at 24 week time point. Finally, particles from the implant crumbled when handling the chronOS Strip but were not observed during the handling of the composite scaffold.
Both materials showed similar, but quite modest, 10%–16% new bone formation in the defect total area. Even though, the overall bone formation was quite modest, bone bridging percentages were quite high, little bit over 70%, in both materials at 24-weeks follow-up, meaning that new bone formation advanced throughout the material as a thin layer and covered the calvarial defect. However, the coverage of the bone decreased at 48-weeks follow-up.
Calvarial model is commonly used model in bone healing studies 27 even though the healing of a calvarial defect is an overall complex process and shows some unique features compared with long-bone healing. Calvarial defect healing takes place via intramembranous ossification, where the osteoblasts form weak woven bone, which is later replaced by lamellar bone. 28 Dural osteogenesis is a common finding in calvarial defect healing.24,29,30 A 12 mm calvarial defect has been proved to be a critical size defect at least 24 weeks, 24 but to our knowledge, this is the first calvarial defect study extending over 24 weeks. In our study, the halt of new bone formation was observed between 24 and 48 weeks in both materials. It has been shown that ossification following trauma in the calvarial bones is not as complete as in the weight-bearing long bones, and the development of new bone will cease before complete healing occurs.31,32 Also, fibrous tissue formation in bone defects might restrain or halt new bone formation. 33
Moreover, the porosity of the implant is essential for vascularization and tissue ingrowth. 34 The average pore size of the composite scaffold was 380 µm, supporting the data received from earlier studies, whereby pore size of between 200 and 400 µm is ideal for osteoblast migration and proliferation and vascularization.23,35–37 The porosity was created by using supercritical CO2 and this method enabled high porosity (65%) but also quite high ceramic concentration (50 w%). 16 The exact pore size of the chronOS Strip is not reported but is stated to feature an optimal pore size for tissue ingrowth. In our study, the new bone primarily advanced to the implant from the dura, while there were also scattered bony islands not connected to the dura. Thus, both materials appeared to show appropriate pore size for vascularization, tissue ingrowth, and bone formation throughout the implanted material.
The composite scaffold was previously studied in the rabbit calvarial defect model using an empty defect as a control. 23 The histopathological findings of the composite scaffold were similar to those we previously reported at 4-, 12-, and 24-weeks follow-up. In this study, both materials induced expected tissue reaction, that was observed on the surface of both materials; histiocytic and lymphocytic reactions were more pronounced in the composite scaffold whereas chronOs Strip induced more fibrosis. 24 In previous studies, both polylactide-co-ε-caprolactone and β-TCP induced inflammatory reactions with multinucleated giant cells and macrophages.10,14,23 Lymphocytic reaction is less studied part of a foreign body reaction, but it is seen with different biomaterials including β-TCP.38,39 They interact with magrophages and multinucleated giant cells, and they also have a role in biomaterial induced fibrous tissue formation.40,41 None of the animals had clinical signs, like swelling, fluid accumulation, sinus tract formation or abnormal scar tissue formation, related to the histologically observed foreign body reaction.
The degradation time of the implant material should mimic the healing time of the repaired tissue and should allow the desired tissue to replace the substitute.27,42,43 Soft tissue, especially adipocyte, intrusion can negatively interfere with the healing of a calvarial defect.23,44 In our study, both materials yielded sufficient structural support for the tissue ingrowth and prevented soft-tissue intrusion into the defect. The total amount of implant material and the rate of degradation were quite similar in both materials, but the composite scaffold appeared to degrade into multiple smaller pieces, whereas the chronOS Strip degraded into fewer and in larger pieces. In previous study, the compression and handling of the composite scaffold caused tear of the PLCL surface, creating new holes in the pores and causing β-TCP granules protrude out from the PLCL coating. 15 This results in a large total surface area of the implant material and might be the principal reason for the long term and pronounced histiocytic and lymphocytic reaction at both 24 and 48 weeks in composite scaffold. In addition, the even distribution of small β-TCP particles and significantly larger proportional volume in the composite scaffold in contrast to the chronOS Strip material may intensify the histiocytic reaction.
The deep convolutional neural network-based AI platform, Aiforia Create, is approved for clinical use in EU and there are multiple publications about various clinical and pathological conditions.45 –48 In addition, it can be employed to analyze any image data like concrete porosity and composition were the images analyzed are very similar than micro-CT images. With AI-based image analysis we were able to analyze large amount of images with complicated structures and multiple components in reasonable time and with repeatable method. With large quantity of analyzed images in the micro-CT analysis abled us to create overview of the 3D distribution of the β-TCP and bone throughout the whole defects. From the histopathological images precise and objective morphometric data enabled a detailed quantitative evaluation of the tissue types inside the implant and implant degradation, thus allowing for credible statistical comparisons between implant material groups and time points. In a visual assessment, the descriptive histopathological findings and the AI-assisted image analysis results showed a good association while we cross-checked the results. Wide interindividual variation was also observed in quantitative analysis. Further studies with comparison of quantitative data between traditional histomorphometric studies including all the tissue types and AI-based technology, are needed. In the future, parallel AI-assisted image analysis of both multiple defined-level histological sections and serial micro-CT images covering the entire investigated defect can be analyzed with a reasonable amount of work.
Limitations of this study were the use of calvarial model instead of long bone or spine fusion model and lack of randomization and blinding. Even though the calvarial model is widely used in bone formation study, the unique bone formation and healing in calvarial bone might have had effects on the fact that both materials showed quite humble amount of new bone formation. Since both materials have very characteristic appearance in every method we used, blinding was not feasible. Randomization was not performed due to a problem to get adult animals and welfare problems that it would have caused during long follow-up time. In addition, the collection of bone marrow aspirate was difficult in the first rabbits, which all belonged to the 48-week follow-up cohort. Without bone marrow aspirate or a bone graft, composite scaffold carries only osteoconductive properties. These might have affected the amount of new bone formation.
The composite scaffold showed its potential as a bone-graft substitute because of its elastic structure and usability during the surgery. It was biodegradable and enabled 3D bone and other tissue formation into the defect. The modest new bone formation requires changes in the composition of the material to increase the new bone formation. Because of the downsides in calvarial model, further studies with this material in a long-bone model is needed.
Supplemental Material
sj-docx-1-jbf-10.1177_22808000241299587 – Supplemental material for Long-term comparison of two β-TCP/PLCL composite scaffolds in rabbit calvarial defects
Supplemental material, sj-docx-1-jbf-10.1177_22808000241299587 for Long-term comparison of two β-TCP/PLCL composite scaffolds in rabbit calvarial defects by Hanna Pihlman, Jere Linden, Kaarlo Paakinaho, Markus Hannula, Mikael Morelius, Mikko Manninen, Outi Laitinen-Vapaavuori and Pauli Keränen in Journal of Applied Biomaterials & Functional Materials
Footnotes
Acknowledgements
The authors extend their thanks to Kati Hagman for technical assistance during the surgical operations, Anna Knuuttila, DVM, PhD, from Aiforia Technologies, for her expert guidance on the Aiforia platform use and AI training, and Docent Petteri Muukkonen, from the Department of Geosciences and Geography, for his invaluable help with processing the geospatial data shape files.
Authors contribution
All the authors designed the experiments. HP, JL, KP, MMa, OL-V, and PK contributed reagents, materials, analysis tools, and funding. HP, KP, MMo, MMa, and PK performed the animal experiments. HP and JL performed histopathological analysis and AI-analysis. MH performed micro-CT imaging. HP, JL, KP, MH, OL-V, and PK analyzed the data. HP, JL, KP, MH, MMo, MMa, OV-L, and PK wrote the article. All the authors have read and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Finnish Funding Agency for Technology and Innovation and through grants from the Finnish Foundation of Veterinary Research. Open access funded by Helsinki University Library.
Ethical approval
The Animal Experiment Board in Finland (ESAVI/5398/04.10.07/2014) approved this study and complied with Finnish legislation on animal experimentation and European Union Directive 2010/63/EU.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
