Abstract
Hydroxyapatite, renowned for its biocompatibility and osteoconductive properties, plays a fundamental role in bone regeneration owing to its resemblance to natural bone mineral, thus offering considerable potential for advancing tissue engineering strategies. In this article, the innovative integration of silicon ions into biogenic (bovine-derived) hydroxyapatite (SiBHA) via a tailored sol-gel process is reported. The resultant SiBHA scaffolds exhibited an interconnected microporous structure with a total porosity of 70% and pore dimensions ranging from 120 to 650 µm. Fourier-transform infrared spectroscopy and X-ray diffraction studies validated the effective incorporation of silicon ions into the BHA lattice, with energy-dispersive X-ray and inductively-coupled plasma mass spectrometry further confirming a Ca/P molar ratio for SiBHA between 1.63 and 1.74. Moreover, SiBHA scaffolds demonstrated commendable chemical and thermal stability. Of note, SiBHA scaffolds were found to display significantly enhanced mechanical properties, including compressive strength and Young’s modulus, compared to the control BHA scaffolds. In vitro assessments highlighted the capacity of SiBHA scaffolds to foster cell viability, proliferation, and osteogenic differentiation of Saos-2 cells. Immunohistochemical analysis revealed a significant increase in osteonectin expression, a key bone matrix protein, after 14 days of incubation under osteogenic conditions. These findings highlight the biocompatibility and therapeutic potential of SiBHA scaffolds, suggesting their suitability as biomaterials for dental bone regeneration applications.
Introduction
In recent years, the use of bone grafts and substitutes in dentistry has significantly risen, driven by progress in dental implantology and a growing demand for craniofacial bone repair. 1 These skeletal defects can result from trauma, periodontal disease, surgical removal, cranioplasty, infections, congenital abnormalities, or oral cancer. A common issue in dentistry is the lack of sufficient bone following tooth loss, as alveolar bone quickly resorbs due to the lack of intraosseous stimulation, which periodontal ligament fibres would typically provide. 2 It has been estimated that up to 50% of all dental implant procedures currently performed will involve the use of bone grafts due to bone loss after tooth loss, inadequate bone quantity and to support implant integration. 3 At present, Geistlich Bio-Oss® a hydroxyapatite based ceramic derived from bovine bone is regarded as the state-of-the-art bone substitute in dental bone regeneration.1,3
The biocompatibility and osteoconductive properties of hydroxyapatite (Ca10(PO4)6(OH)2) (HA) and its similarity to the mineral composition of bone and teeth have rendered it a popular choice of bioceramic material for dental regeneration applications.4,5 The undeniable advantages of synthetic HA products, including their non-toxic nature, minimal inflammatory response, and osteoconductivity, underscore their potential for broad utilisation in medical fields. However, their classically poor mechanical properties, elevated dissolution rates in physiological environments and the associated high production expenses leading to increased production costs, have spurred researchers on to exploring alternative avenues for deriving HA. 6
Naturally occurring HA has been successfully extracted from various biogenic sources, for example, mammalian bones of bovine, equine and porcine origin, to name a few; marine sources such as fish bones, scales, and seashells; poultry sources such as eggshells; mineral sources such as limestone and even some unconventional sources such as wood or algae etc.7 –9 Naturally occurring hydroxyapatite (HA) exhibits non-stoichiometric characteristics, as the molar ratio of calcium (Ca) to phosphate (P) may deviate slightly from the ideal value of 1.67. This variation arises from the natural presence of trace elements such as sodium, iron, carbonate, magnesium, and fluoride, amongst others, which substitute the calcium and hydroxyl groups within the HA structure in varying proportions. These elemental substitutions exert a positive influence on the physical, chemical, mechanical, and biological properties of HA, thereby contributing significantly to its role in regulating bone metabolism. 10 Bovine HA prepared using repurposed waste bovine bones from the food industry is a prominent source of natural HA preparation due to its abundant availability. Such repurposing of bone waste for potential biomedical applications also minimises the environmental impact of a material which is otherwise disposed of via landfills or incinerated causing air pollution and contributing to climate change. 11 Bovine HA has been proven to present better physicochemical, mechanical, and biological properties when compared to synthetic HA, and bovine HA sourced from countries such as New Zealand offers the added advantage of being free from risks of prion-based diseases such as bovine spongiform encephalopathy. 12
Ionic substitutions in HA, where one or more anions or cations partially or entirely replace HA’s calcium, phosphate and hydroxyl groups, are seen as the solution to address some of the demerits of pure, synthetic HA. Substitutions in the HA lattice have a substantial impact on the physical properties, including crystal lattice parameters, morphology, solubility, and thermal stability. Numerous studies have been conducted on synthetic HAs replaced with silicon, copper, magnesium, strontium, zinc, magnesium, fluoride, and carbonate. Currently, several of these substituted HAs are accessible for bone augmentation, bone repair, and as covering/lining materials for orthopaedic implants.13,14
Silicon is an important trace elements found in the human body essential for bone growth and development. Silicon deficiency has been linked to abnormal skeletal growth and defects in skull development in mice and rats.14,15 silicon substitution is known to enhance the bioactivity of hydroxyapatite. Several studies have documented the advantageous effects of various ionic substitutions on the properties of synthetic hydroxyapatite (HA).
Until recently, the exploration of integrating beneficial ions into naturally derived HA to augment their potential remained largely unexplored, thereby representing a notable knowledge gap in the field. 16 This underscores the imperative to investigate and address this area to advance our understanding of natural HA and harness its full therapeutic capabilities. The present study is the first of a kind which details the ionic substitution of silicon ions into biogenic HA (SiBHA) prepared from New Zealand-sourced bovine cancellous bones. This study aims to incorporate silicon ions into bovine-derived hydroxyapatite and evaluate the silicon ion-substituted bovine-derived hydroxyapatite’s physical, chemical, mechanical and biological properties, thus investigating their potential for dental bone regenerative applications.
Materials and methods
Preparation of silicon ion-substituted bovine hydroxyapatite
The defatting and deproteinising procedure produced a bovine-derived hydroxyapatite (BHA) scaffold from the condyle portion (epiphyseal region) of the bovine femur bone, which has been detailed elsewhere.12,17 The prepared BHA which is similar to Geistlich Bio-Oss® was used as a control. Silicon ions were incorporated into the BHA ionic structure to produce silicon ion-substituted bovine hydroxyapatite (SiBHA) scaffolds using a modified sol-gel procedure, as detailed below.16,18 Hydroxyapatite has a (Ca)/(P) mole ratio of 1.67. 12 To substitute silicon ions, the mole ratio of (Ca)/(P) was adjusted to 1.73, whereas the mole ratio of (Ca)/(P + Si) was maintained at 1.67. 19 The quantities for each precursor were calculated accordingly (Table 1) to prepare the SiBHA scaffolds using the modified sol-gel method.
Constituents required to prepare Si sol-gel.

Aqueous ammonia = 60 mL
De-ionized water = 50 mL
Ammonium hydrogen phosphate ((NH4)2HPO4) (Sigma Aldrich, NZ) and silicon tetraacetate (Si(CH3COO)4) (Sigma Aldrich, NZ) were first dissolved in 50 mL of de-ionized water with vigorous stirring for 15 min at room temperature and pressure. The phosphorous-silicon (P-Si) containing solution was transferred into a custom-made beaker while stirring, and the temperature was maintained at 85°C throughout the process. Calcium nitrate-tetra hydrate Ca(NO3)2.4H2O was dissolved in 50 mL of de-ionized water. The Ca salt-containing solution was added dropwise using a glass dropping funnel into the P-Si-containing solution. As the solution turned into a chalky white colour, 5–6 cubes of BHA scaffolds were placed on top of the wire mesh, and the remaining Ca-containing solution was added slowly to the P-Si-containing solution. Aqueous ammonia was added to maintain a pH above ten throughout the stirring process. The solution was stirred for a further 4 h until gel formation occurred. The gel was allowed to settle overnight, after which the bone cubes were removed, and the sol-gel was transferred into a separate beaker. The supernatant was discarded using a syringe after the settlement of the gel. The bone cubes were then mildly agitated in the saturated sol-gel for 4 h using an orbital shaker and then transferred to an oven for further drying at 60°C for 24 h. The bone cubes were finally sintered in a hot air furnace at 600°C for 4 h (Figure 1).

Flow chart of the sol-gel technique used to incorporate silicon ions into the BHA scaffold.
Characterization
The functional groups of SiBHA and BHA scaffolds were determined using an ATR-IR spectrometer (Bruker, Darmstadt, Germany), which operates in the mid-infrared frequency range of 400–4000 cm−1. The crystal phase composition of SiBHA and BHA samples was evaluated using an X-ray diffractometer (XRD; PANalytical X'Pert PRO MPD System (PW3040/60)) in the region 20° < 2θ < 70° with Cu Kα radiation (
Chemical stability and degradation in simulated body fluid
Using a simulated body fluid (SBF) solution (pH 7.4) made in accordance with the Kokubo protocol
20
was used to evaluate the in vitro chemical stability and degradation profile of the SiBHA scaffolds (
Mechanical properties
The mechanical properties of the SiBHA (
Cell culture
Human osteoblast-like cells (Saos-2) (ATCC® HTB-85™, USA) were used to assess the in vitro biocompatibility of the SiBHA and BHA scaffolds. Minimal essential medium alpha (MEM-α; Invitrogen, New Zealand) supplemented with 10% foetal bovine serum (FBS; Thermo Fisher Scientific, NZ) and 1% penicillin–streptomycin antibiotics (10,000 units/10 mL penicillin, 10 µg/10 mL streptomycin, 25 µg/mL amphotericin B) (Life Technologies, Auckland, NZ, USA) was used to culture the cells in a humidified incubator operating at 37°C with 5% CO2. 10 nM dexamethasone, 5 mM β-glycerophosphate (β-GP) and 100 μM L-ascorbic acid 2 – phosphate supplements were added to the Saos-2 growth medium (S-GM) to prepare the differentiation medium (DM) for the ALP assay and immunohistochemistry. The SiBHA and BHA scaffolds were prepared using an 8 mm biopsy punch. Before cell seeding, the scaffolds were sterilised in 70% ethanol under UV radiation, followed by equilibrating in 1 mL of cell culture media for 24 h and washing with phosphate-buffered saline (PBS) for 2 min. Cells from the 3rd to 8th passage were used for the experiments. Once the cells were 70%–80% confluent, 6 × 103 cells/scaffold were directly seeded onto each scaffold and incubated for 1 h to allow the cells to attach to the scaffold before adding 1 mL of growth media.
Cell viability/cytotoxicity assay
The LIVE/DEAD® test (Invitrogen, New Zealand) was used to assess the scaffolds’ cell viability (
Cell proliferation assay
To determine the cell proliferation on the SiBHA and BHA scaffolds (
Alkaline phosphatase activity
Cells were seeded at a density of 20 × 103 cells/scaffold (
Immunohistochemical analysis
Statistical analysis
PRISM software (GraphPad Prism 6, version 9.0, Boston, MA, USA) was used for the statistical analysis. The error bars displayed the standard error of the mean (S.E.M.) as ±. A one-way analysis of variance (ANOVA) was employed to identify statistically significant differences. If differences were detected, multiple comparisons were made using Tukey’s multiple comparison tests at a confidence level of 95% (
Results
The as-prepared silicon ion-substituted bovine hydroxyapatite (SiBHA) scaffolds presented a pure white color in appearance and were subjected to various characterisation techniques to confirm that silicon ions had become substituted into the hydroxyapatite lattice of the bovine bone. A detailed characterisation of the SiBHA is given below.
Characterization
Fourier transform infrared spectroscopy
Figure 2 illustrates the superimposed spectra of the BHA and SiBHA. The FTIR spectra illustrated hydroxyapatite’s characteristic vibrational peaks, including the OH- band at 3571 cm−1 and the ν3 and ν1 phosphate bands at 1100–960 cm−1 (1089, 1032, and 962 cm−1) and the ν4 bands 660–520 cm−1 and 473 cm−1 respectively. Carbonate was retained in the hydroxyapatite structure, confirmed by the carbonate ν3 and ν2 stretching modes in the region 1455–1418 cm−1. However, the intensity of these groups was considerably lower than that of BHA. The two peaks observed at 490 and 890 cm−1 corresponded to the silicate groups in the apatite structure.23,24
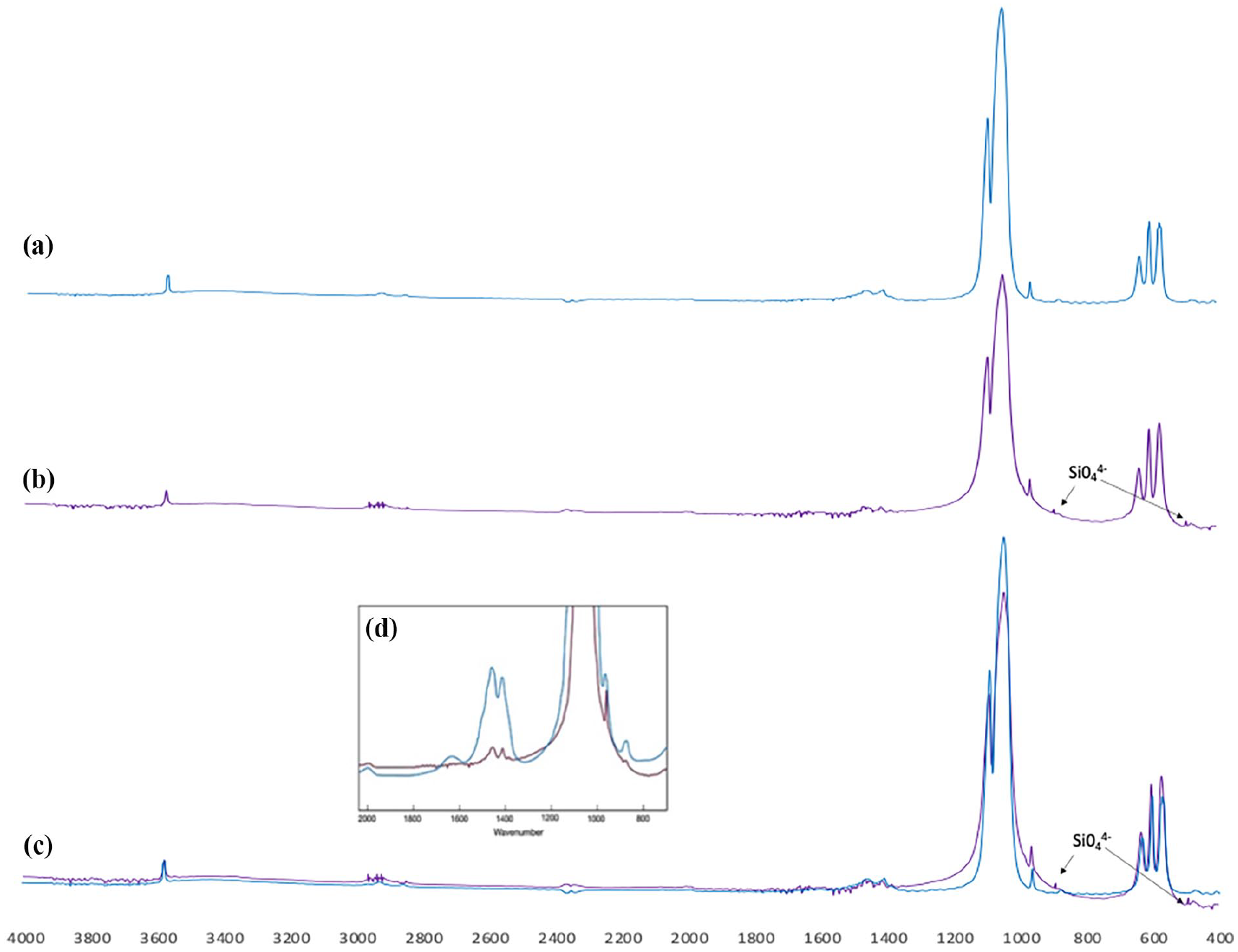
(a) FTIR spectra of BHA. (b) FTIR spectra of SiBHA. (c) Superimposed spectra of BHA (blue) and SiBHA (purple). (d) Superimposed spectra of the carbonate groups of BHA (blue) and SiBHA (purple).
SiBHA’s XRD patterns were compared with those of BHA. SiBHA’s XRD patterns showed almost exact alignment with BHA’s. However, in contrast to BHA, the diffraction peaks of SiBHA had a broader spectrum and lower intensity peaks. In addition, compared to BHA, the peaks of SiBHA exhibited a little shift to a lower Bragg’s angle.
Scanning electron microscopy (SEM)
SEM analysis was used to study the pore sizes and surface morphology. SEM analysis showed a microporous structure for both scaffold types with pores ranging from ~120 to 650 μm. As indicated in yellow arrows, agglomerations were observed in the SiBHA scaffold compared to the BHA scaffold.
Energy dispersive X-ray (EDX) and inductively coupled plasma-mass spectrometry (ICP-MS) analysis
SiBHA’s inorganic constituents included traces of magnesium, sodium with calcium, and phosphorus as the major constituents. The weight percentage of silicon varied from 0.8% to 1%. The (Ca)/(P) mole ratio ranged from 1.63 to 1.71 according to EDX analysis but 1.70 according to ICP-MS analysis.
Micro-CT
The 3D micro-CT image reconstruction of the SiBHA scaffold revealed an interconnected porous architecture with a total porosity of 70.54% ± 1.14%.
Thermogravimetric analysis (TGA)
TGA analysis showed minimal weight loss for the SiBHA scaffold losing ~1.3% of its original weight when heated to 1000°C.
Chemical stability and degradation in simulated body fluid
Chemical stability
The pH values of the SBF solution conditioned with SiBHA scaffolds presented a consistent increasing trend up to day 7, reaching a slightly basic value of 7.65. However, following 14 days of immersion, the pH value stabilized to a neutral value of 7.32 and remained consistent till day 28. One-way ANOVA analysis using Tukey’s multiple comparisons showed no statistically significant difference during the investigated time points (ANOVA,
Degradation
There was an initial weight loss for the SiBHA scaffolds up to day 7, losing ~3.5% of their original weight (
Mechanical properties
The SiBHA scaffold presented a high yield strength, measuring 0.827 MPa compared to the BHA scaffolds at 0.71 (
Biocompatibility
LIVE/DEAD cell viability assay
Cell viability of Saos-2 cells seeded on SiBHA scaffolds at 24 (Figure 11(a)–(c)), 48 (Figure 11(d)–(f)) and 72 (Figure 11(g)–(i)) hours of observation. Cells were seen penetrating the macropores of the SiBHA scaffolds (white arrow) (Figure 11(d)).
MTS cell proliferation assay
Figure 12 compares the cell proliferation on BHA and SiBHA scaffolds. A threefold increase in cell number was observed for both the scaffold types, and the SiBHA scaffold exhibited a higher cell number compared to BHA during the investigated time points. However, no significant differences were observed between the two types of scaffolds at 24, 48 and 72 h (
Alkaline phosphatase (ALP) assay
On day 1, both BHA and SiBHA scaffolds exhibited a lower expression of ALP activity. However, the ALP activity significantly increased from days 1 to 14, peaking at day 14 (
Immunohistochemistry
Immunohistochemistry results showed a positive green immunofluorescence staining for osteonectin on the surface of the SiBHA scaffold after 14 days of cell culture.
Discussion
Studies have focused on substituting silicon into synthetic hydroxyapatite using various synthesis methods such as precipitation, hydrothermal and mechanochemical processing.23,24 Synthetic HA substituted with small amounts of silicon ions has shown superior biological functions in improved bone apposition, bone ingrowth, and cell-mediated degradation.12,15 However, no studies have attempted substituting silicon ions into bovine-derived hydroxyapatite (BHA) due to its highly ordered crystal structure. Therefore, the main premise of this study was to incorporate silicon ions using a modified sol-gel method to produce a silicon ion-substituted bovine hydroxyapatite scaffold (SiBHA) while maintaining its porous microstructure. Sol-gel synthesis provides many advantages including homogenous mixing of the calcium and phosphorus precursors. 25 Other advantages include low operating temperature and the producing phase-pure nano-hydroxyapatite.26,27
The following chemical reaction illustrates the formation of HA from the sol-gel process. 26

In this study, the quantities of the reactants were calculated according to the protocol established by Gibson et al. 28 who successfully substituted silicon ions for phosphate ions in synthetic HA using a sol-gel process. To avoid rapid, uneven precipitation and to improve chemical homogeneity, a slow titration process was used with maintenances of pH above 10.5 using aqueous ammonia (which also acted as the gelling agent). Several studies have shown that maintaining a pH below 9 during the synthesis process promotes the formation of calcium-deficient HA, thus compromising its material properties. 29
The infrared spectral analysis of SiBHA revealed characteristic peaks consistent with hydroxyapatite, including the prominent peak at 3571 cm−1, associated with the hydroxyl group and the phosphate bands at 1100–960 cm−1, 660–520 cm−1 and 473 cm−1. Notably, the intensity of the hydroxyl peak at 3571 cm−1 was considerably lower compared to that in (unsubstituted) BHA. These findings are primarily attributed to the compensatory mechanism addressing the charge imbalance resulting from the additional negative charge introduced by the silicate (SiO44−) groups. Substituting SiO44− ions leads to a diminished intensity of the carbonate-related peaks.30,31 The carbonate peaks (1455–1418 cm−1) for SiBHA exhibited a reduced intensity relative to BHA, indicating that silicon ions substituted for phosphate in the HA structure, with the carbonate ions occupying both the phosphate and hydroxyl sites.31,32 Another notable effect on the silicon substitution is the changes in the phosphate bands between 800 and 1100 cm−1and 500–700 cm−1. Although the SiBHA spectrum exhibited characteristic ν3 and ν1 phosphate bands (1091, 1043, 961 cm−1) and ν4 bands (629,603 and 569 cm−1), the ratio of peak heights, most notably for the peaks at 1091 and 1043 cm−1 were considerably lower compared to BHA suggesting that silicon ions occupied some of the phosphate groups. These results agreed with the study by Gibson et al. 28 when silicon ions was substituted for synthetic hydroxyapatite using a precipitation method. 28
The X-ray diffraction (XRD) patterns obtained for SiBHA exhibited similarities to those of BHA, albeit with reduced intensity and broader spectral lines, indicative of diminished crystallinity (Figure 3). Moreover, a noticeable shift towards lower Bragg’s angles was evident in the XRD patterns of SiBHA compared to BHA (Figure 3(b)). This phenomenon can be attributed to the disparity in bond lengths, where Si-O bonds (1.62 Å) exceed the lengths of P-O bonds (1.51 Å). Additionally, silicon ions (“Si4+”) possess a larger ionic radius (0.042 nm) in contrast to phosphorus ions (“P5+”) (0.035 nm), resulting in distortion of the phosphate tetrahedra within the hydroxyapatite (HA) lattice. 33 These observations corroborate previous studies by Aminian et al. 30 and El Yacoubi et al., 31 wherein silicon substitution in synthetic HA yielded similar outcomes. Furthermore, the absence of undesired secondary phases such as calcium oxide (CaO) and beta-tricalcium phosphate (β-TCP) further underscores the phase purity and integrity of the synthesized SiBHA material.

(a) X-ray diffraction patterns of BHA (blue) superimposed with SiBHA (red). (b) Superimposed XRD patterns of BHA and SiBHA showing peak shift.
The SiBHA scaffolds presented a well-defined interconnected microporous architecture, featuring pore sizes ranging from 120 to 650 μm (Figure 4), indicative of successful defatting and deproteination processes. These observed pore dimensions align with the research of Orlovskii et al.,
34
who underscored the necessity of minimum pore sizes ranging from 100 to 135 μm to facilitate bone ingrowth through cellular activities such as migration, proliferation, and differentiation.
34
Additionally, smaller pores are advantageous for protein adsorption and bone cell adhesion. Micro-CT analysis revealed the porosity of the SiBHA scaffold to be ~70% (Figure 5), demonstrating its suitability for cellular infiltration, whereas the approximate porosity of

(a) Cross-sectional image of the scanning electron micrograph of the BHA. (b) SiBHA scaffold, (c) ×1000 of the SiBHA scaffold illustrating the agglomerations after the sol-gel process. Bar = 100 μm.

3D image of the SiBHA scaffold analysed using micro-CT.
EDX and ICP-MS analysis unveiled trace amounts of sodium, magnesium, and strontium, characteristic of mammalian-sourced hydroxyapatite which are judged to be beneficial to foster bone regeneration. 35 Silicon content ranging from 0.8 to 1 wt%, as analysed by EDX (Figure 6), aligns with findings by Šupová et al. 36 recommending 1 wt% silicon as optimal for enhancing biological activities. Comparative analysis with previous studies revealed a slightly lower (Ca)/(P) mole ratio for SiBHA due to the dissociation of carbonate groups. According to Table 2, toxic elements such as arsenic, cadmium and lead remained below ASTM standard limits, affirming the scaffold’s safety for biomaterial applications.

EDX analysis of the SiBHA scaffold.
Chemical composition of the SiBHA scaffold according to ICP-MS and SEM-EDX analysis.

Thermogravimetric analysis (TGA) demonstrated the stability of the SiBHA scaffold, exhibiting only a marginal loss (~1.3%) of its original weight (Figure 7). The initial weight loss (endothermic) observed between 50°C and 200°C resulted from the evaporation of absorbed water, while the subsequent exothermic weight loss (~0.5 wt%) between 250°C and 600°C corresponded to the decomposition of residual carbonates. The theory of the carbonate weight loss aligns with the findings of FTIR spectra (Figure 2(b)), which indicated low-intensity carbonate peaks in the range of 1455–1418 cm−1.

The thermogravimetric curve for SiBHA showing weight loss over the tem-perature range (0–1000°C).
There was an initial increase in the pH value by day 7, reaching 7.65. It is attributed to releasing calcium (Ca2+) and silicate ions (SiO44−) from the SiBHA lattice (Figure 8). Bang et al. 33 observed a similar finding when synthetic silicon ion-substituted hydroxyapatite (SiBHA) was soaked in SBF. Furthermore, several studies have reported that substituting small amounts of silicon ions is advantageous as it increases solubility, resulting in an early bone remodelling process.14,37 During the 28 day incubation period, the SiBHA scaffold was structurally stable. The initial weight loss of approximately 3% wt could have resulted from the dissolution of calcium (Ca2+), phosphate (PO43−) and silicon ions from the SiBHA lattice (Figure 9). The dissolution of these ions is beneficial in vivo as these ions occupy the interface between the scaffold and host bone. 38 Furthermore, LeGeros et al. 39 showed that a scaffold exhibiting an interconnected porous structure and the presence of carbonate and trace elements enhances the biodegradation of a scaffold.14,16 The SiBHA scaffold presented all the above-mentioned features, suggesting that the scaffold would degrade with time.

Chemical stability of the SiBHA scaffolds in terms of change in pH during the investigated time points (

In vitro biodegradation of SiBHA as a function of time in simulated body fluid. (
The SiBHA resulted in notable enhancements in mechanical properties, particularly compressive strength and Young’s modulus compared to BHA (Figure 10). It is widely recognized that highly porous structures often display inferior mechanical properties. While the sol-gel process employed in this study effectively reduced pore sizes and bolstered mechanical performance, forthcoming research endeavours may necessitate in vivo trials involving wetting of the SiBHA material with biological fluids to ascertain its clinical viability.

Yield strength and Young’s modulus of the BHA and SiBHA scaffolds were analyzed using student
Human osteosarcoma cells (Saos-2) are a cell line derived from an 11-year-old Caucasian female in 1973 and were initially isolated and characterized by J Fogh and G Tempe and is commonly used to assess the biocompatibility of materials related to bone tissue engineering due to its osteoblast-like properties. 40 The SiBHA and the control BHA scaffolds, similar to Geistlich Bio-Oss® were subjected to the LIVE/DEAD cytotoxicity assay. The assay showed that the SiBHA was non-toxic, allowing Saos-2 cells to adhere and populate on the surface of the SiBHA scaffold. In addition, the cells penetrated the pores (Figure 11(d)) of the SiBHA scaffold. Cellular penetration is essential for cell migration and attachment into the interior pores, where cells extend cellular protrusions such as filopodia to reach internal pores to strengthen the attachment between the cells and scaffold surface, which is integral for osseointegration. 10 Both the SiBHA and BHA scaffolds showed increased cell proliferation from 24 to 72 h, with a higher cell number for the SiBHA scaffold at each time point than BHA (Figure 12) which could be be attributed to the bioactivity of silicon ions. 41 A study conducted by Camaioni et al., 35 Aminian et al., 30 and Hond et al., 42 showed that silicon ion-substituted synthetic hydroxyapatite (SiHA) stimulated human osteoblasts compared to synthetic hydroxyapatite. Furthermore, both Gibson et al. 19 and Kim et al. 43 showed that osteoblast-like cell activity was significantly enhanced on silicon-substituted synthetic hydroxyapatite (Si-HA) when compared against synthetic HA. The ALP activity, indicating osteoblastic new bone formation, increased for both the scaffolds during the incubated time, with the SiBHA scaffold exhibiting a significant increase in the ALP activity at day 14 (Figure 13). Rodrigues et al. 44 found that silicon ions enhanced biomarkers of new bone formation such as alkaline phosphatase expression and osteocalcin expression, which indicate the accelerated osteoblastic activity. Furthermore, Botelho et and Kim et al. 43 showed that 0.8 wt% Si-HA increases ALP production compared to synthetic HA. The trace elements found in SiBHA (Figure 5 and Table 2), which influence various biomechanical reactions, could have played an essential role in the increased ALP activity leading to cellular differentiation. The differentiation of Saos-2 cells and laying down of bone matrix on the SiBHA scaffold was further confirmed by immunohistochemical analysis after revealing a strong expression for “osteonectin” after 14 days of culture (Figure 14).

Fluorescent images showing the cell viability of the Saos-2 cells seeded directly on the SiBHA scaffolds at 24 h (a–c), 48 h (d–f) and 72 h (g–i). Green = live cells (calcein), Red = dead cells (ethidium homodimer-1). Bar = 50 μm.

Cell proliferation of Saos-2 cells as a function of time. (

The alkaline phosphatase activity of Saos-2 cells seeded on BHA (in black) and SiBHA (in white) scaffolds at 1, 3, 7, and 14 days. (
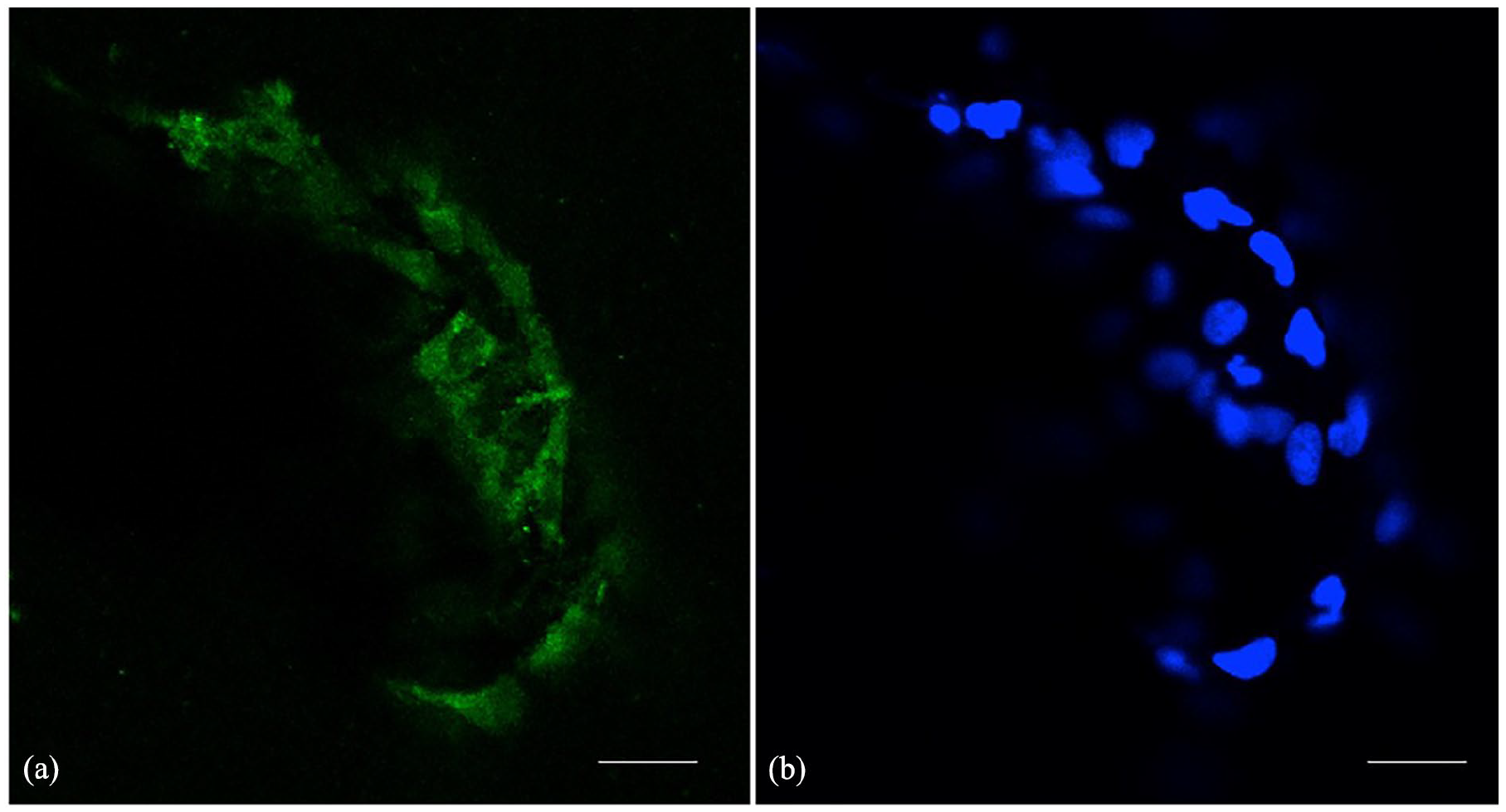
Expression of osteonectin (in green) on the SiBHA scaffold. Nuclear stain-ing/DAPI staining (in blue) of the Saos-2 cells. Bar = 100 μm. (a) Expression of osteonectin (in green) on the SiBHA scaffold. (b) Nuclear stain-ing/DAPI staining (in blue) of the Saos-2 cells. Bar = 100 μm.
The study has several limitations. For example, the scalability of the sol-gel process to produce the SiBHA scaffolds should be further investigated. The sol-gel process used in this study requires precise control over reaction conditions, requires expensive starting materials and is time-consuming. 45 Furthermore, an in vivo study using a critical size defect is essential to investigate osteointegration and the clinical feasibility of SiBHA as a dental bone graft substitute. There would be variability from the source of the bovine bone used as age, gender and breed of the animal could have resulted in variable results.
Conclusion
Silicon ions were successfully substituted to bovine-derived hydroxyapatite using a modified sol-gel method which produced a bone graft material with a well-defined porous structure conducive to cellular infiltration, adhesion, and nutrient diffusion, crucial for facilitating bone regeneration. Chemical characterization confirmed the successful integration of silicon into the BHA lattice, while mechanical assessments revealed enhanced compressive strength and Young’s modulus compared to BHA controls. In-vitro cell culture investigations showed that the SiBHA facilitated new bone formation. Overall, SiBHA scaffolds present a promising and cost-effective alternative to synthetic hydroxyapatite-based products or commercially available Geistlich Bio-Oss®, holding considerable potential for dental bone regenerative and augmentative applications such as filling defects in the periodontium after root resection, apicoectomy, or cystectomy, extraction socket management, minor and major bone augmentation, and sinus floor elevation.
Footnotes
Acknowledgements
We acknowledge the technical assistance of the Otago Micro and Nano Imaging (OMNI) for scanning electron microscopy and David Barr, Department of Chemistry for his assistance with ICP-MS analysis and University of Otago Doctoral scholarship.
CRediT authorship contribution statement
Jithendra Ratnayake: Conceptualization, Methodology, Formal analysis, Investigation, Data curation, Writing – original draft. Maree L Gould: Methodology, Investigation, Data curation, Writing – original draft, Writing – review and editing. Michael R Mucalo: Conceptualization, Methodology, Validation, Investigation, Writing – review and editing. Niranjan Ramesh: Methodology, Writing – review and editing. George J Dias: Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Project administration, Writing – review and editing, Funding acquisition.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional review board statement
Not applicable.
