Abstract
Objective:
Chronic bowel disease has the characteristics of high recurrence rate, prolonged and non-healing, and the incidence has increased year by year in recent years. Cannabidiol (CBD) has significant anti-inflammatory and antioxidant activities, but it is limited by its characteristics of fat solubility and low bioavailability. This study aims to treat chronic inflammatory bowel disease by preparing a CBD-loaded hydrogel system (GelMA + CBD) that can deliver CBD in situ and improve its bioavailability through slow release.
Method:
The study designed and constructed GelMA + CBD, and its surface morphology was observed by scanning electron microscopy, and its pore size, swelling rate and release rate were evaluated to evaluate its bioactivity and biosafety. The expression of various inflammatory factors was detected by ELISA, and the expression of protein and reactive oxygen species were observed by laser confocal microscopy to evaluate their anti-inflammatory and antioxidant properties.
Results:
Our study found that GelMA + CBD with biosafety, could make CBD be slowly released, and effectively inhibit the M1-type polarization of macrophages in vitro, and promote the M2-type polarization. In addition, GelMA + CBD can also reduce the expression of pro-inflammatory factors (such as iNOS) in macrophages, and increase the expression of anti-inflammatory factors (such as Arg-1), clear intracellular reactive oxygen species (ROS), and relieve oxidative stress.
Conclusion:
The vitro experiments have confirmed that the CBD-loaded hydrogel system has good biosafety, and can alleviate inflammation by regulating the polarization direction of macrophages, and then inhibiting the secretion of pro-inflammatory factors, laying a strong foundation for the treatment of chronic enteritis.
Graphical abstract

Introduction
Ulcerative colitis (UC) is a chronic non-specific inflammatory bowel disease (IBD) accumulated in the gastrointestinal tract. IBD is a chronic, non-specific inflammatory bowel disease. It is prone to recurrence, persistent and irreversible, and can be transformed into colon cancer. Patients with ulcerative colitis have a 5–8 times greater risk of developing colorectal cancer than the normal population, and there is no clinically effective cure. In recent years, with the in-depth research on the etiology and pathogenesis of UC, macrophages and neutrophils infiltrate, and the abnormal continuous activation of intestinal mucosal immune response, leading to intestinal inflammation and ulcer formation. 1 It is an effective therapeutic strategy to regulate macrophage behavior during the development and treatment of UC.
In many chronic inflammatory microenvironments, macrophages, as plastic cells, can be transformed from one phenotype to another: macrophages activated by Th1 response are called M1 macrophages, that is, classically activated; Macrophages activated by Th2 response are called M2 macrophages. A recent study by Wu et al. 2 have shown that the polarization of macrophages is closely related to the occurrence and development of ulcerative colitis, and that the intestinal inflammatory microenvironment can be significantly improved by adjusting the polarization direction of macrophages. Macrophages regulate the progression of inflammation by responding functionally to the microenvironment and signals encountered in each specific tissue, such as secreting pro-inflammatory and anti-inflammatory factors, and activating different signaling pathways. Studies have shown that there is a close relationship between inflammatory response and mononuclear macrophage polarization.3,4 In the inflammatory microenvironment, macrophages are polarized toward M1, which causes the imbalanced immune regulation process by releasing pro-inflammatory factors and initiating cellular pathways such as NLRP3, thus aggravating tissue inflammatory damage5,6: For example, CD11chi pro-inflammatory monocyte-like cells accumulate in the inflammatory colon of IBD patients, while circulating monocytes excessively secrete pro-inflammatory cytokines, such as IL-23 and TNF-α, resulting in intestinal tissue inflammatory damage that persists. 7 Therefore, precisely regulating the polarization direction of macrophages to improve the inflammatory microenvironment and inhibit the progression of inflammation is the theoretical basis for the future treatment of inflammatory bowel disease.
A study has shown that phenolic compounds show antibacterial and antioxidant activities against a variety of bacteria. 8 Cannabidiol (CBD) is considered as a non-psychoactive compound derived from cannabis plant, which has good anti-inflammatory and antioxidant activities. 9 At present, plant cannabinoids have been used in preclinical models of gastrointestinal inflammation, 10 and some clinical studies have verified that cannabinoids have good efficacy in the treatment of IBD.11,12 Yekhtin et al. 13 found that CBD could regulate the polarization of macrophages toward M2b in inflammatory environment and reduce the secretion of pro-inflammatory factors such as IL-6 and TNF-α. At the same time, it can reduce the release of nitrogen monoxide (NO), improve the oxidative stress response of cells, and give full play to the anti-inflammatory and antioxidant effects. However, due to the fat-soluble characteristics of CBD, direct use will face many drawbacks such as rapid clearance and low bioavailability, which will significantly reduce the efficacy of the drug, so its clinical application in the treatment of inflammation is obviously limited.
In recent years, studies have shown that novel drug delivery systems can be used for the targeted therapy of chronic inflammation based on the changes of different factors under chronic inflammation.14,15 Since UC is a physiological property of inflammation located in the colon, the delivery system of drugs to treat UC has gradually become a new research focus. Pu et al. 16 built polymer-derived drug delivery systems against IBD, opening up the possibility of a laboratory-to-bedside drug delivery polymer system for IBD treatment. The accurate and effective drug delivery system can reduce the side effects of drugs on normal tissues, increase the concentration of drugs in pathological tissues, and can be designed to form a phased drug release mode according to the pathological characteristics of the inflammatory microenvironment, which has unique advantages in the treatment of chronic inflammation. GelMA (Methacrylate Gelatin) is a kind of gelatin-derived photopolymeric biomaterials, and also a natural biopolymer with good cell adhesion. 17 Because it can support a variety of anti-inflammatory drugs and has certain targeting and slow release effects, it has gradually become a new research focus. The advantage of GelMA hydrogel loading anti-inflammatory drugs is that it can deliver drugs directly to the inflammation and release them slowly, thus ensuring local effective drug concentration and avoiding systemic adverse reactions caused by oral or intravenous injection.18,19 Therefore, through the development of a new strategy to build a hydrogel system loaded with CBD anti-inflammatory drugs, the biological functions of immune cells are regulated, such as inhibiting M1 polarization of macrophages, promoting M2 polarization, reducing the secretion of pro-inflammatory factors, alleviating inflammation, rebuilding the cell regeneration microenvironment, and providing new ideas for the treatment of chronic inflammation.
Materials and methods
Chemicals and materials
CBD power (purity 99%) was purchased from Zhongsheng Biotechnology Co., Ltd., (Heilongjiang, China). GelMA was obtained from Engineering for Life Co., Ltd., (Zhejing, China). Lipopolysaccharides (LPS, surface of Gram-negative bacteria) were purchased from Med Chem Express (New Jersey, USA). Rabbit anti mouse iNOS/Arg-1 primary antibody were obtained from abcam system (US). Enzyme-linked immunosorbent assays (ELISA) for iNOS and Arg-1 were purchased from R&D Systems (Minneapolis, MN, USA). RAW 264.7 cells, penicillin/streptomycin and α-MEM medium were purchased from Shandong Sparkjade Biotechnology Co. Ltd., (Shandong, China). Cell counting kit (CCK-8) was purchased from Beyotime Biotechnology Co. Ltd., (Beijing, China). Fetal bovine serum (FBS) was obtained from Gibco, (Thermo Fisher Scientific, Waltham, MA, USA). The Live/Dead cell viability assay kit was obtained from US EverBright Inc., (Jiangsu, China). The goat anti- rabbit IgG was obtained from Bioss (Beijing, China).
Preparation of GelMA + CBD hydrogel system
The 5 g of GelMA solid was dissolved in 10 mL of PBS solution to prepare a GelMA solution with a concentration of 5% w/v. Then, the 0.1% w/v photoinitiator lithium phenyl-2,4,6-trimethylbenzoyl phosphate (LAP) and 10 μL of CBD solution (a solution formed by dissolving CBD powder in dimethyl sulfoxide with a concentration of 125 μM) were added to the above solution with stirring in a constant temperature water bath at 50°C until completely dissolved. The GelMA + CBD hydrogel was obtained by irradiating the above solution with 365 nm wavelength ultraviolet light (UV) for 10–20 s. To confirm the characteristic peak of the aldehyde group in OSA, a Fourier transform infrared (FTIR) spectroscope (TENSOR II, Bruker, Germany) was used and the measurement was performed at room temperature and recorded in the 4000–400 cm−1 range.
Characterization of physical and chemical properties of injectable hydrogels containing CBD
Microstructure characterization of injectable hydrogels containing CBD
The microstructure of the hydrogel was observed by scanning electron microscopy (SEM, QUANTA 200, Czech). Firstly, hydrogels with 5% g/mL according to manufacturer’s suggestion were prepared and lyophilized. After that, the hydrogel sample was cut into small pieces, placed on a platform, and coated with a gold layer. Then, the image of cross section morphology was observed by SEM. Using Image J software version 1.54, the aperture of different SEM images of four groups of hydrogels was determined.
Swelling property of injectable hydrogel containing CBD
The freeze-dried hydrogels of each group were weighed and immersed in PBS, swelling at 37°C for 24 h.
20
After complete swelling, the hydrogels were removed from PBS, absorbent paper absorbed excess water, and then weighed. The swelling properties of each group were measured according to the formula of swelling rate (%)=
Release behavior of CBD loaded injectable hydrogel in vitro
To analyze the release kinetics of CBD from GelMA hydrogel, the hydrogels were incubated in 2 mL PBS at 37°C in the incubator. At regular intervals, 1.5 mL release liquid was taken out to determine the concentration of CBD, and another 1.5 mL PBS was added to maintain the total volume at 2 mL. The samples of CBD release solutions were stored at −20°C and analyzed by ultraviolet spectrophotometer (UV-2700, Shimadzu, Japan). Then, the cumulative release content of CBD was calculated according to the concentration of CBD at day 1, 3, and 5 time points and the standard curve set by the concentration of 0–100 mΜ. 21
Cell culture
Raw264.7 macrophages were purchased from Shanghai Cell Bank. Raw264.7 cells were cultured with added high sugar. The cells were cultured in a 5% CO2 humidified incubator at 37°C.
In vitro cytotoxicity evaluation of injectable hydrogels containing CBD
Grouping
The study was divided into blank control-LPS group (without LPS stimulation), LPS group (adding 1 μg/mL LPS to stimulate M0 macrophages), and GelMA group (adding hydrogel to M0 macrophages after 1μg/mL LPS stimulation). CBD control group (M0 macrophages were stimulated by 1μg/mL LPS and then added 1.25 μmol/L CBD), GelMA + CBD group (M0 macrophages were stimulated by 1 μg/mL LPS and then added 1.25 μmol/L CBD loaded hydrogel system).
Evaluation of cytotoxicity
Cell Counting Kit-8 (CCK-8) was used to evaluate the cytotoxicity of hydrogels with CBD in different concentrations and determine the safety of the materials. Cell Counting Kit 8 assay (CCK-8, Biosharp, Shanghai) was used to determine the biocompatibility of RAW 264.7 cells to GelMA according to the methods described in relevant literature. 22 and the standard of ISO 10993-5: 2009. Cell viability greater than 70% is considered non cytotoxic. The cells were treated with water gel extract for 1 and 5 days and then incubated in a medium containing 10% CCK-8 in the dark at 37°C for 2 h. Finally, absorbance was obtained at 450 nm using an enzymoleter (BioTek, USA). Normal growth medium was set as control group.
Cells were stained dead or alive and cell activity was observed by confocal microscope (Zeiss. LSM710, Germany): The biocompatibility of RAW 264.7 cells to hydrogel extracts was determined using a live/dead cell staining kit (Invitrogen, USA). The cells were treated with water gel extract for 1 and 5 days. The cells were then washed twice with PBS, labeled at 37°C with 2 mM calcein-AM (fluorescein green) and 4 mM propyl iodide (PI, fluorescein red) in the detection buffer for 30 min, and gently rinsed again with PBS. Finally, a laser scanning confocal microscope was used for observing the samples.
A freeze-dried hydrogel scaffold was placed into a 24-well plate using a Scanning Electron Microscope (SEM, QUANTA 200, Czech). RAW 264.7 cells with good growth condition were selected and inoculated on the scaffold at a density of 106 cells per well. After 4 h, 500 μL medium was added to the well. After 24 h of culture, the medium was removed, 1 mL 2.5% glutaraldehyde was added to each well and fixed at 4°C overnight. The scaffold was dehydrated by gradient ethanol (soaked in 70%, 80%, 90%, and 95% ethanol for 5 min, and soaked twice in 100% ethanol). After 48 h drying, the adhesion of cells on scaffolds was observed by SEM.
Evaluation of in vitro antioxidant properties of injectable hydrogels containing CBD
In order to study the intracellular ROS clearance ability of hydrogels, RAW 264.7 cells were first inoculated in confocal dishes with a density of 105 cells per dish, cultured in complete medium for 12 h, and then stimulated with LPS according to the group: LPS group (control group), LPS group, GelMA group, CBD group and GelMA + CBD group. After 12 h of treatment, the cells were washed three times with PBS, and then DCFH-DA probe (20 μM) was added. Then, fluorescence in RAW 264.7 cells was observed by confocal microscope (Zeiss. LSM710, Germany).
Evaluation of in vitro regulation of macrophage polarization behavior by CBD-loaded injectable hydrogel
Immunofluorescence staining was first applied to evaluate the effect of CBD on M0 macrophages by surface markers of M1 phenotype (iNOS) and M2 phenotype (Arg 1). RAW264.7 cells were inoculated in 48 well plates at a density of 105 cells per well, cultured in complete medium for 12 h, and then stimulated with LPS according to the groups: LPS group (control group), LPS group, GelMA group, CBD group, and GelMA + CBD group. After 12 h of treatment, the cells were washed with PBS and fixed with 4% paraformaldehyde at 4°C for 10 min. 0.1%TritonX-100 was broken at room temperature for 10 min and 10% goat serum was closed at room temperature for 45 min. Rabbit anti-mouse iNOS/Arg-1 primary antibody (dilution ratio: 1:200) was incubated with cells at 4°C overnight, and rewarmed at room temperature for 1 h, then washed with PBS 8 times for 5 min each time. Goat anti-rabbit secondary antibody (dilution ratio: 1:200) was incubated with the cells for 2 h and washed with PBS 8 times for 5 min each time. Finally, the water-soluble DAPI seals were dried away from light and then photographed under a confocal microscope (Zeiss. LSM710, Germany).
After culturing with LPS for 12 h, RAW264.7 was stimulated with different solution from groups for 12 h. Then, the proteins are extracted from cells for the ELISA detection according to operation instruction to evaluate the effect of on iNOS and Arg-1 secretion.
Statistical analysis
The SPSS 26 (IBM SPSS, USA) was used for statistical analysis, and all experimental results were shown as mean ± Standard Deviation (SD, Standard Deviation). The calculation part of the statistics is performed by T-test or one-way Analysis of variance (ANOVA) and Tukey test. The statistical significance was *p < 0.05, **p < 0.01, and ***p < 0.001. All experiments were repeated at least three times.
Results
Physicochemical characterization of GelMA + CBD hydrogel
The morphology of GelMA + CBD was observed by SEM. Figure 1(a) shows the hydrogel without CBD, and Figure 1(b) shows the hydrogel with CBD. As shown in Figure 1(a) and (b), the results show that both groups of hydrogels have dense and interpenetrating three-dimensional porous structures, and the structures are smooth without roughness. Among them, with the addition of CBD, it did not destroy the internal structure of the hydrogel, and the porous structure of the hydrogel became more rounded and the microporous structure increased, which also contributed to the increase of cell mechanical properties, cell adhesion and intracellular signal transduction. In short, the addition of CBD did not have a great effect on the microstructure of hydrogels. Appropriate hydrogel pore size plays an important role in cell adhesion, proliferation and migration. As shown in Figure 1(c), the aperture of GelMA group was 228.76 ± 66.78 μm. Even with the addition of CBD to GelMA, there are no significant changes in pore size (p > 0.05).

Performance characterization of GelMA + CBD hydrogel system. (a and b) GelMA + CBD scanning electron microscope analysis; (c) Pore size analysis; (d) swelling rate analysis; (e) The average cumulative release of CBD in CBD-GelMA hydrogel (different lowercase letters represented significant differences between groups, p < 0.05); (f) CBD and hydrogels (GelMA, GelMA + CBD group) analysis by FTIR.
Figure 1(d) shows the swelling property of hydrogel materials. The results show that the swelling rate of GelMA group is larger, reaching 847.6% ± 64.8% on average, while the swelling rate of GelMA + CBD group is smaller, but it can also reach about 799.8% ± 83.3%. With the addition of CBD, swelling decreased, but there was no significant difference between the two groups (p > 0.05). Figure 1(e) shows the in vitro release of GelMA + CBD hydrogel material. The results showed that the release of CBD of GelMA + CBD hydrogel material gradually increased over time, and reached a stable release on the fourth day (p > 0.05). In addition, the FTIR results of hydrogel was shown in Figure 1(f). The characteristic chemical group “C=O” of OSA at 1733 cm−1 was detected. The above results confirmed that the OSA had been successfully synthesized. In addition, the characteristic chemical group of CBD at 3518 and 3406 cm−1 from hydroxyl stretching vibration 1580 and 1441 cm−1 and 3406 cm−1 from benzene ring was detected in GelMA + CBD group, which proved GelMA + CBD hydrogel was successfully prepared.
In vitro cell biological evaluation
Cell biosafety evaluation of GelMA + CBD hydrogel system in vitro
In order to further determine the optimal drug concentration, CCK-8 assay was used to verify the cytotoxicity of CBD at different concentrations in hydrogel, as shown in Figure 2(a). CCK-8 assay of RAW264.7 cells showed that the drug at 1.25 μmol/L concentration had no significant difference from the normal group, that is, it had no toxic effect on cells. Later, this concentration of CBD was taken for further study.
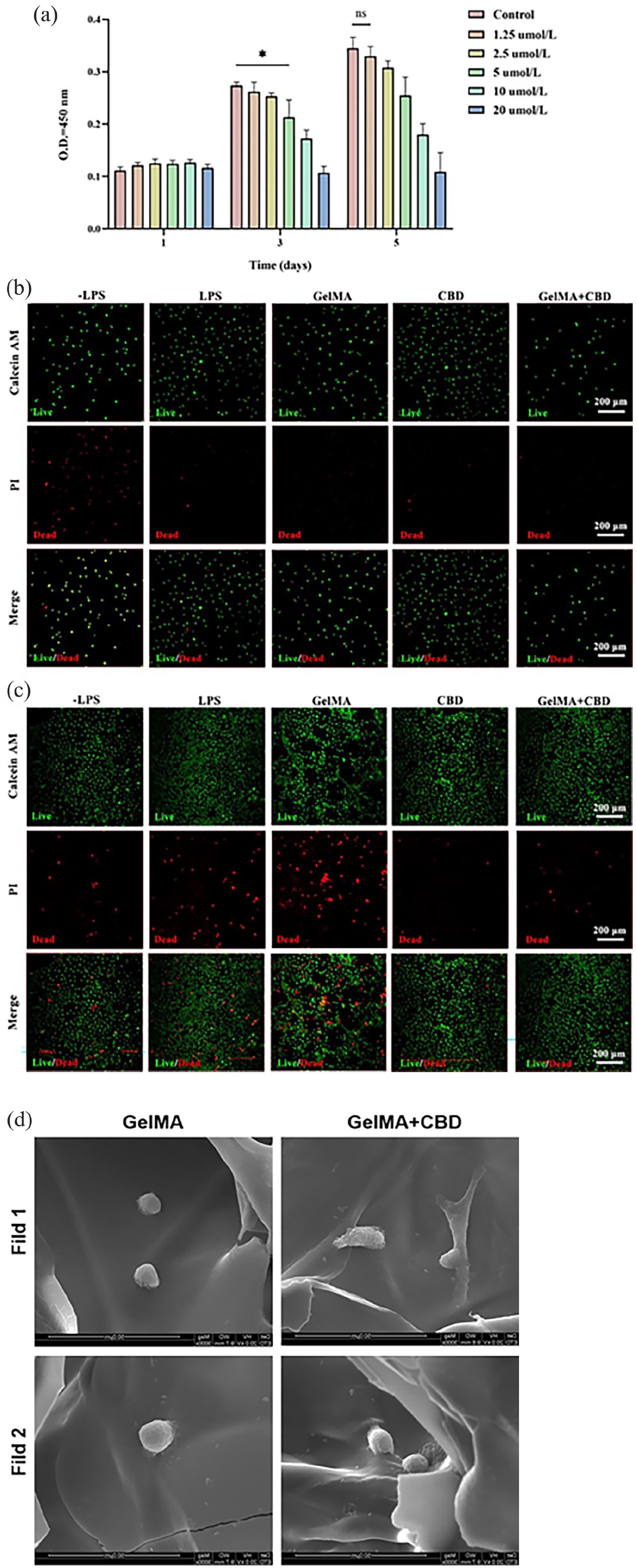
In vitro cell biosafety evaluation of GelMA + CBD hydrogel system. (a) CCK-8 results of different CBD concentrations in hydrogel; (b) The death and living experiment of RAW264.7 cells on hydrogel at day 1, green and red fluorescence indicate living and dead cells, respectively. (c) The death and living experiment of RAW264.7 cells on hydrogel at day 5, green and red fluorescence indicate living and dead cells, respectively. (d) Cell adhesion on hydrogel (*p < 0.05).
The biocompatibility of the material was further analyzed through the death and living experiment of RAW264.7 cells, as shown in Figure 2(b) (day 1) and Figure 2(c) (day 5). Confocal microscopy results showed that macrophages grew well after LPS stimulation compared with the first day, but the death of macrophages in LPS group was significantly increased, as was the case in CBD group. Compared with the first day, the number of macrophage death in the GelMA group and the GelMA + CBD group did not increase significantly on the fifth day. The experimental results indicate that CBD may have certain cytotoxicity, and the addition of GelMA can reduce the cytotoxicity of CBD to a certain extent, and the GelMA + CBD hydrogel system has good biosafety.
The adhesion behavior of RAW264.7 cells was observed after co-culture with two groups of hydrogels. As shown in Figure 2(d), both in the GelMA group and in the drug-loaded group, the cells in the each group exhibited good adhesion and normal stereospread morphology, accompanied by the extension of pseudopods, indicating adhesion between cells and hydrogels. The adhesion of hydrogels was not affected by the addition of drugs. The experimental results show that the hydrogel has cell adhesion, which is conducive to the subsequent migration and adhesion of macrophages and the full play of the drug effect of CBD, so as to play the role of immune regulation.
GelMA + CBD hydrogel system in vitro cellular immune regulation
Evaluation of cellular antioxidant properties of GelMA + CBD hydrogel in vitro
It was documented that under oxidative stress, excessive ROS production will destroy cellular proteins, lipids, and DNA, leading to fatal cell damage, which will lead to many diseases. The inhibition effect of hydrogel on ROS was explored, and the results are shown in Figure 3. Compared with the blank control group, ROS levels (green fluorescence signal) of activated macrophages were significantly increased after LPS treatment, indicating that LPS treatment can significantly damage the oxidative stress state of activated macrophages. Compared with the ROS levels in the CBD group and LPS group, it was found that CBD could significantly improve the oxidative stress state of activated macrophages damaged. Compared with LPS group, GelMA group still expressed higher ROS levels, indicating that GelMA did not significantly activate oxidative stress in macrophages. After LPS stimulation, ROS levels of activated macrophages in the GelMA + CBD group were significantly reduced compared with those in the GelMA group, indicating that gelatin-loaded CBD can significantly restore the oxidative stress state of activated macrophages.

Confocal fluorescence microscopy results of ROS products from macrophages.
Evaluation of anti-inflammatory activity of GelMA + CBD hydrogel system in vitro
The protein expression of RAW264.7 cells was shown in the Figure 4(a) and (b) after stimulation by LPS (1 μg/mL) and regulation by all groups. ELISA results showed that: First of all, the expression of Arg-1, a protein secreted by M2 macrophages, was significantly increased after LPS stimulation, and the expression of this protein was significantly up-regulated in both CBD group and GelMA + CBD group. Among them, CBD group, GelMA + CBD group and GelMA group all had significant differences (p < 0.05). CBD group has the most obvious effect, which may be due to the fact that CBD is stimulated by the target concentration, while GelMA + CBD group cannot achieve the target concentration in the short term, so it is lower than CBD group, but compared with GelMA group, it can also significantly up-regulate the expression of M2-related proteins. According to the above results, it can be seen that drug loading in hydrogel can well regulate the immunological behavior of macrophages, increase the polarization of macrophages to M2 type, promote the secretion of anti-inflammatory factors, and then exert anti-inflammatory effects. In addition, immunofluorescence staining as shown in Figure 4(c) and (d) was used to further confirm the advantage of CBD group and GelMA + CBD group in promoting M2 polarization of macrophages. The expression of iNOS protein secreted by M1-type macrophages was significantly increased after LPS stimulation, while the expression of inos protein in CBD group and GelMA + CBD group was significantly down-regulated. There were significant differences in CBD group, GelMA + CBD group and GH group, respectively. The results showed that hydrogels loaded with CBD drugs could regulate and inhibit the polarization of macrophages to M1 type, thereby reducing the secretion of pro-inflammatory factors. Confocal microscopy and quantitative results showed that hydrogels loaded with CBD drugs could increase the M2 polarization of macrophages, regulate and inhibit their M1 polarization, and give full play to the anti-inflammatory effect.

Evaluation of anti-inflammatory activity of GelMA + CBD hydrogel system in vitro; (a and b) The expression of Arg-1 and iNOS protein in macrophages was detected by ELISA. (c and d) Expression of Arg-1 and iNOS in macrophages under confocal laser microscopy.
Discussion
UC is a typical chronic inflammatory bowel disease characterized by intestinal inflammation accompanied by watery diarrhea, rectal bleeding, weight loss, edematous shortened colon, and inflammatory mucosal infiltration. Bacterial infection is often one of the main pathogenic factors of gastrointestinal inflammation, and the main pathogenic substance is LPS in the cell wall. LPS can induce the polarization imbalance of macrophages and produce inflammatory factors. At the same time, it accelerates the accumulation of ROS and destroys the antioxidant defense mechanism, causing oxidative stress and gradually aggravating chronic inflammation. CBD, as a typical cannabinoid, can regulate macrophage polarization and reduce inflammatory response, and has been used in preclinical models of gastrointestinal inflammation. 23 In this study, LPS-induced cellular oxidative damage model was established, and the GelMA + CBD hydrogel system was constructed to ensure the biosafety, slow release and stability of the system, while making full use of CBD’s anti-inflammatory and antioxidant properties to achieve the purpose of treating chronic inflammation.
GelMA hydrogel, as a multifunctional biomaterial, has been widely used in biomedicine. 17 GelMA-based hydrogel is biocompatible, biodegradable, non-cytotoxic and non-immunogenic, and CBD has anti-inflammatory and antioxidant effects, so we choose to construct GelMA + CBD hydrogel system. The results show that the GelMA + CBD hydrogel system has a compact, interpenetrating and stable three-dimensional porous structure, and can fully load CBD without affecting its pore size, swelling rate and structural stability. The stable hydrogel structure can play a certain supporting role to facilitate subsequent drug release and cell infiltration. In addition to showing the basic characterization of the GelMA + CBD hydrogel system, this study fully demonstrated the biosafety characteristics of the GelMA + CBD hydrogel system by exploring the results of CCK-8 experiment and dead/alive staining, which laid the possibility of its future clinical application. Studies have shown that different cells adhere to and grow on the surface of GelMA matrix, and can be wrapped in GelMA hydrogel matrix, with good survival ability.24,25 The SEM results of this study also showed cell adhesion characteristics of GelMA + CBD hydrogel system. Controlled drug release is another area where the use of GelMA hydrogels is being explored. Serafim et al. 26 evaluated the controlled release mechanism of the drug naacillin sodium by constructing a GelMA-polyacrylamide (PAA) mixed hydrogel family as a model, and they adjusted the possibility of antibiotic release profile by changing the composition of the mixture (i.e., increasing the proportion of PAA). The GelMA + CBD hydrogel system in this study showed stable in vitro release characteristics, reaching a high release rate on day 4, ensuring the drug concentration of CBD.
When the production rate of ROS in the body is greater than the removal rate of antioxidants, excess ROS will attack intracellular DNA and proteins, thereby causing cellular oxidative damage and aggravating the progression of chronic inflammation. 27 In this study, intracellular ROS content was used as an important index to measure the degree of oxidative damage. The results showed that ROS expression in CBD group was significantly decreased compared with LPS group, and the low expression of green fluorescence signal in GelMA + CBD group also showed better antioxidant activity. Prevent the formation of superoxide free radicals to reduce oxidation conditions; Chelating transition metal ions involved in the Fenton reaction can form highly reactive hydroxyl radicals to reduce the production of reactive oxygen species (ROS). 28 In addition to the above direct antioxidant effects, CBD also regulates the activity of the endocannabinoid system, thus affecting its interaction with promoter regions such as superoxide dismutase. Thus, it exerts an indirect antioxidant effect.29,30 This study demonstrated that GelMA + CBD hydrogel system can fully guarantee the antioxidant activity of CBD and alleviate the LPS-induced oxidative damage of cells by removing excessive ROS.
Previous studies have shown that CBD has anti-inflammatory and immunomodulatory effects in addition to significant antioxidant activity.31,32 In most pathological processes, regulation of macrophage activity and function is essential for balancing tissue immune homeostasis and driving or resolving inflammation. 33 Given the close relationship between inflammation and macrophages, CBD has shown significant advantages in inhibiting inflammation by regulating the direction of macrophage polarization. 13 The results of this study showed that macrophages were polarized to M1 type after LPS stimulation, and a large amount of iNOS protein was expressed, and the large production of iNOS could lead to the production of high concentration of nitric oxide (NO), and then the oxidative phosphorylation function of mitochondria was destroyed, resulting in oxidative stress damage of cells. Our results showed that GelMA + CBD had the anti-inflammatory ability as expected. Under LPS stimulation, GelMA + CBD could regulate the inhibition of macrophages towards M1 type polarization and reduce the expression of pro-inflammatory factors. Compared with LPS group, iNOS protein in CBD group and GelMA + CBD group was significantly decreased, while Arg-1 protein was significantly expressed in both groups. This indicates that CBD can regulate and inhibit the M1-type polarization of macrophages and promote the M2-type polarization. M2 macrophages have a strong ability to release anti-inflammatory cytokines. By participating in tissue remodeling and repair after injury, M2 macrophages can remove debris and promote angiogenesis, reduce inflammatory damage, and maintain a normal regenerative microenvironment. 34 The GelMA + CBD group also showed anti-inflammatory properties, with no significant difference compared to the CBD group. This indicates that the addition of GelMA ensures the drug release rate without affecting the efficacy of CBD.
To sum up, this study prepared GelMA + CBD hydrogel system by loading CBD with GelMA hydrogel, making full use of its slow-release, biosafety and cell adhesion properties, so as to avoid many drawbacks brought by CBD due to its fat solubility, such as low drug concentration and low bioavailability. However, this study also has certain limitations. For example, the experiment in vivo has not been further improved. In the subsequent research, we can further explore the therapeutic effect in vivo of the GelMA + CBD hydrogel system by building a mouse model of inflammatory bowel disease, and explore the deep mechanism of the system regulating the macrophage polarization using molecular biological technology. It is believed that with the further development and research of medical biomaterials in the future, hydrogels as medical carriers may provide opportunities for precise regulation of the inflammatory recovery process. At the same time, new treatment strategies are provided to alleviate patient pain, reduce treatment costs, improve patient quality of life and prognosis.
Conclusion
In this study, GELMA + CBD hydrogel system was constructed to make full use of its slow release, biosafety and cell adhesion characteristics, and CBD regulated macrophages to M2 type polarization, reducing the expression of pro-inflammatory factors and other anti-inflammatory and antioxidant activities, so as to increase drug concentration in local tissues, improve efficacy and reduce side effects. With the continuous development of tissue engineering technology and the continuous maturity of transplantation technology, GelMA hydrogel combined with anti-inflammatory drugs to treat chronic inflammatory bowel disease will become a reality.
Footnotes
Correction (January 2025):
In figure 4, the ***p value was incorrect “≤ 0.01” and has been corrected to ***p ≤ 0.001.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
