Abstract
The pro-inflammatory/anti-inflammatory polarized phenotypes of macrophages (M1/M2) can be used to predict the success of implant integration. Hence, activating and inducing the transformation of immunocytes that promote tissue repair appears to be a highly promising strategy for facilitating osteo-anagenesis. In a previous study, titanium implants were coated with a graphene oxide-hydroxyapatite (GO-HA) nanocomposite via electrophoretic deposition, and the osteogenic differentiation of bone marrow mesenchymal stem cells (BMSCs) was found to be significantly enhanced when the GO content was 2wt%. However, the effectiveness of the GO-HA nanocomposite coating in modifying the in vivo immune microenvironment still remains unclear. In this study, the effects of GO-HA coatings on osteogenesis were investigated based on the GO-HA-mediated immune regulation of macrophages. The HA-2wt%GO nanocomposite coatings exhibited good biocompatibility and favored M2 macrophage polarization. Meanwhile, they could also significantly upregulate IL-10 (anti-inflammatory factor) expression and downregulate TNF-α (pro-inflammatory factor) expression. Additionally, the microenvironment, which was established by M2 macrophages, favored the osteogenesis of BMSCs both in vivo and in vitro. These findings show that the GO-HA nanocomposite coating is a promising surface-modification material. Hence, this study provides a reference for the development of next-generation osteoimmunomodulatory biomaterials.
Introduction
Titanium (Ti) and its alloys are used to manufacture most commercially available implants 1 owing to their good biocompatibility, biological inertness, high specific strength, low elastic modulus, and excellent corrosion resistance. 2 However, there are inherent drawbacks associated with the use of these materials. For instance, Ti ions released from the implant can affect microbial cytokine and receptor expression. 3 Moreover, given its immunogenicity, Ti can cause a type IV hypersensitivity reaction in some patients.4,5 Therefore, the surface modification of Ti implants has attracted immense attention. Accordingly, over the past few decades, several surface treatments (e.g. plasma spraying, 6 acid etching treatment 1 ), as well as bio-active coatings (e.g. hydroxyapatite, 7 bio-active glass 3 ), have been applied to Ti implants.
Unfortunately, while most implant coating materials show excellent results in vitro, experiments in pre-clinical models often yield the opposite results, likely due to the complexity of the in vivo environment. Implant insertion post-surgery is swiftly followed by immune system activation. Immunocytes such as macrophages recognize the foreign body and respond by secreting a variety of cytokines, generating a new microenvironment around the implant. The consequent interaction of the immunocytes with the implant material plays a crucial role in inflammations, immune responses, 8 and tissue reconstruction, 9 eventually determining the initial stability and longevity of the implant.10–13 Among the different types of immunocytes, macrophages play crucial roles in osteoimmunological interactions. Macrophages can respond to various stimuli and switch between the pro-inflammatory M1 and anti-inflammatory M2 phenotypes.11,14 Through such phenotypic switching, macrophages can regulate osteoblast activity by secreting different cytokines and molecules. Furthermore, M1 and M2 macrophages also have different functional characteristics.15,16 M1 macrophages release chemoattractants and cytokines that stimulate osteoclastogenesis.17,18 Moreover, their prolonged presence leads to chronic inflammation, manifesting as the expression of fibrotic factors and granuloma formation, and ultimately leads to implant encapsulation and failure. 19 In contrast, M2 macrophages secrete anti-inflammatory cytokines that induce angiogenesis in vivo, stabilize the newly formed vessels, and promote osteo-anagenesis. So It is a promising strategy to rescue titanium implant infection through immune regulation. 20
In vitro studies have demonstrated the dynamics of M1 and M2 macrophages under specific immune microenvironments. 20 These studies have shown that a timely shift from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype can lead to active bone tissue regeneration. For instance, anti-inflammatory cytokines secreted by M2 macrophages (e.g. interleukin [IL]-10) are essential in the osteogenic environment 21 and can efficiently regulate osteoblast differentiation.22,23 In turn, macrophage polarization itself is vulnerable to the effects of chemical elements on the surface of biomaterials24–27 and their surface morphology.28–30 Hence, biomaterials that can modulate macrophage polarization have attracted increasing attention. Therefore, in addition to considering factors such as mechanical properties, surface structure, and osteogenic activity, it is also important to focus on the role of immunocytes in bone repair while designing or identifying materials for implant surface modification. Specifically, whether the surface modification material can induce immunocyte transformation to facilitate tissue repair must also be considered.
Hydroxyapatite (HA, Ca10(PO4)6(OH)2) is among the more specific bioactive coatings used for implant surfaces. HA is widely used in clinical studies due to the similarity between its chemical composition and that of the main inorganic minerals in human skeletal tissues. 31 Meanwhile, graphene oxide – with its unique structure and high mechanical strength, high surface area, high electrical conductivity, high thermal conductivity, low density, and other excellent physical properties 32 – is often used to prepare composites with unique functions. Previously, graphene oxide (GO)-coated surfaces have been found to influence macrophage polarization and inflammatory cytokine expression via the Toll-like receptor pathway. 33
Implant-associated infection remains a challenging clinical issue and traditional treatments rely on the use of antibiotics or physical surgery to remove the implants and the infected tissue. 20 In previous studies, GO-HA nanocomposite coatings have been successfully applied on the surface of Ti implants via electrophoretic deposition (EPD) to increase their roughness and hydrophilicity. In these studies, electrochemical impedance experiments demonstrated that the corrosion resistance of the GO-doped implants (GO group, HA-2wt%GO group, and HA-5wt%GO) was significantly higher than that of Ti implants and HA-coated implants, and the GO coatings showed superior barrier properties. Moreover, compared with Ti, HA, and GO coatings, the GO-HA composite coating facilitated cell adhesion and spreading, significantly improved the proliferation and osteogenic differentiation of bone mesenchymal stem cells (BMSCs) cultured in vitro, upregulated the expression of osteogenesis-related genes, and also enhanced the alkaline phosphatase (ALP) activity of BMSCs. Additionally, the osteo-anagenesis capacity of each modified implant was examined after implantation into rat femurs, and the HA-2wt%GO group showed significantly more peri-implant neoplastic bone than the other experimental groups. These findings confirmed that the GO-HA nanocomposite coating could promote osseointegration and in vivo osteogenesis. 34
However, the osteoimmune regulation induced by GO-HA composite coatings and their effects on osteogenic differentiation or bone fusion have not been studied so far. Therefore, in this study, we added GO-HA nano-composite coatings on the surface of pure Ti using the EPD method and cultured macrophages on the surface of these implants to study the osteoimmunoregulatory properties of the composite coating. Further, we investigated how the conditioned medium of coating-treated macrophages affects the osteogenic differentiation of BMSCs. The results highlight the potential of GO-HA composite coatings in immunoregulation and their application as biomaterials for osteo-anagenesis.
Methods
Preparation and characterization of the GO-HA coating
Samples were prepared using a previously reported method 34 (Figure 1). Specifically, commercial pure Ti was cut into square plates (10 mm × 10 mm × 1 mm), sandpapered, and polished step-by-step to 1200#. The Ti plates were subjected to ultrasonic cleaning in anhydrous acetone, anhydrous ethanol, and deionized water. A graphite electrode was used as the anode, and the Ti substrate was used as the cathode; both electrodes were placed into the electrolyte (see Table 1 for electrolyte parameters). The voltage of the DC power supply was set to 30 V, the distance between the cathode and anode was 2 cm, and the duration of EPD was 2 min. Magnetic stirring was used to keep the particles uniformly dispersed throughout the EPD process. After EPD was complete, the particles were removed from the electrolysis cell and dried at room temperature for a sufficient amount of time. Then, the samples were sintered in a vacuum sintering furnace at 600°C for 1 h. The coatings were divided into the HA group, GO group, HA-2wt%GO group, HA-5wt%GO group, and pure Ti group (blank control group) according to the corresponding electrolyte used.
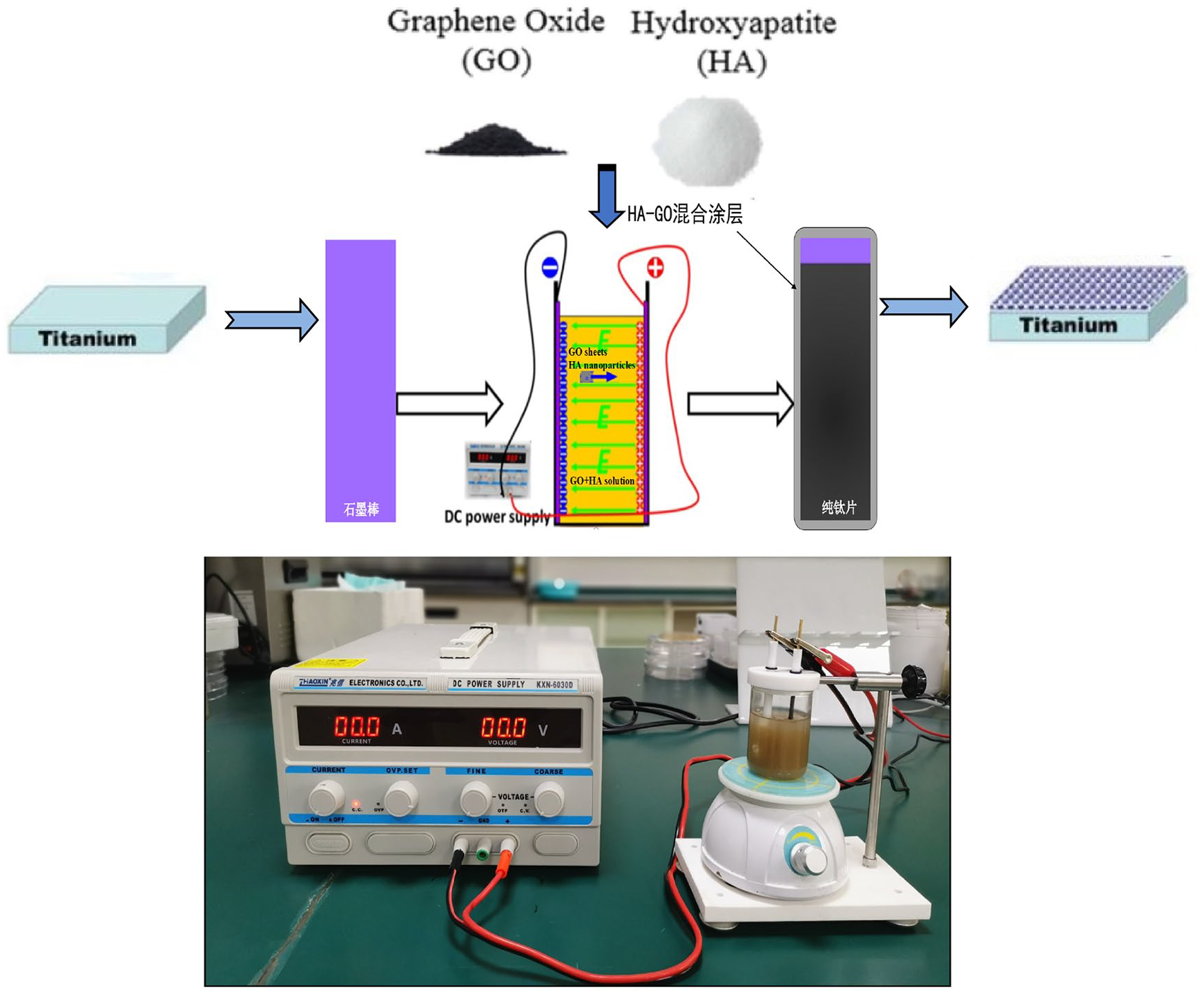
Schematic representation of the electrodeposition set-up.
Electrolyte compositions used for the preparation of coatings via the electrophoresis method.

Characterization: First, the morphology of the samples was observed using scanning electron microscopy (SEM; JEOL JSM-6700F, Japan) at an accelerating voltage of 5.0 KV. Meanwhile, the surface morphology and roughness of the samples were determined using atomic force microscopy (AFM; NT-MDT Solver Pro), and the phase patterns of the coatings were evaluated by X-ray diffraction spectroscopy (XRD, Shimadzu, Japan). Raman spectra were obtained from 1000 to 2000 cm−1 using a Raman Spectroscopy System (Raman, HORIBA Scientific Company, France). Finally, a contact angle meter (SL200B, Solon, China) was used to record the image of a stationary drop of deionized water at the initial time point to evaluate the surface hydrophilicity of the coatings.
Cytocompatibility tests
RAW264.7 cells and BMSCs culture
The mouse RAW264.7 cell line used for in vitro tests was purchased from the Chinese Academy of Sciences (Shanghai, China). RAW264.7 cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM; HyClone, USA) supplemented with 10% heat-activated fetal bovine serum (FBS; Gibco, USA) and 1% penicillin/streptomycin (HyClone). RAW264.7 cells (1 × 105) were seeded onto the surfaces of the samples in six-well culture plates containing DMEM and incubated at 37°C in 5% CO2, unless indicated otherwise. The medium was refreshed every 2 days.
Meanwhile, mouse BMSCs were isolated from the femoral bone marrow of 4-week-old BALB/c mice, which were purchased from the Laboratory Animal Center of Xinjiang Medical University (Xinjiang, China). The cells were cultured for 4 days in DMEM containing 10% FBS and 1% penicillin/streptomycin at 37°C and 5% CO2 under aseptic conditions. Unaffixed cells were removed through rinsing, and the remaining affixed primary BMSCs were designated as P0. Subsequent experiments were performed using P3 BMSCs.
Proliferation of RAW264.7 cells
To evaluate the growth of RAW267.4 cells on different coating surfaces, the Cell Counting Kit-8 (CCK-8) assay was used. RAW267.4 cells (1 × 105/mL) were seeded on the samples for 4 and 7 days. Subsequently, the samples were incubated in fresh complete medium and CCK-8 reagent (10:1 v/v) for 4 h at 37°C. After incubation, the effect of the materials on cell proliferation was evaluated by measuring the absorbance at 450 nm using an enzyme labeling instrument (Tecan Infinite M200, Switzerland).
In order to observe the adhesion of RAW267.4 cells on each type of sample, RAW267.4 cells (1 × 105 cells/mL) were inoculated on the samples for 4 days, washed twice with phosphate-buffered saline (PBS), and fixed with paraformaldehyde (PFA, 4%). Then, they were rinsed twice with PBS. The cells were stained using 4′,6-diamidino-2-phenylindole (DAPI) (Sigma, USA), and fluorescence images were obtained using an immunofluorescence microscope (Leica, Germany). In another set of experiments, the cells were co-cultured for 4 days, fixed in 4% PFA, and dehydrated using a gradient of ethanol solutions. The cells were dried and sprayed with gold, and their morphology on the surface of each group of samples was visualized and photographed using SEM.
Detection of RAW264.7 cellular phenotype on different coating surfaces
Flow cytometry
The expression of M1 and M2 cell-surface markers in RAW264.7 cells (C-C chemokine receptor type 7[CCR7] and cluster of differentiation 206 [CD206], respectively) was determined using flow cytometry. Specifically, the five different coating materials were sterilized and placed in 24-well plates. Cultured cells were removed from the incubator for observation under a microscope, and second- or third-generation cells in the peak growth phase were selected for inoculation. Subsequently, RAW264.7 cell suspensions (1 × 105 cells/mL) were slowly added dropwise onto the surface of each coating material in the 24-well plates. After 30 min, the plates containing the coating material and cells were replenished with fresh medium (total volume, 1 mL) and placed in the incubator. On the 4th day, the culture medium was removed, and the surface of the coating material was rinsed gently with PBS. The cell suspension was trypsinized, and the cells were centrifuged at 1200 rpm and 4°C for 5 min. Specific binding sites were blocked using PBS (containing 1% bovine serum albumin [BSA]) for 30 min at room temperature, and antibodies against CD206 and CCR7 (Abcam, UK) were added to the cell suspension. The cells were incubated with the antibodies for 1 h at room temperature under dark conditions. Subsequently, they were washed twice with PBS (containing 1% BSA), resuspended, and added to new 96-well plates for flow cytometry (Beckman, Beckman). The cell count was set to 5000 cells per sample. The results were analyzed using Kaluza software.
Immunofluorescence staining
The expression levels of inducible nitric oxide synthase (iNOS; surface marker of M1 macrophages) and CD206 (surface marker of M2 macrophages) in RAW264.7 cells cultured on different coating surfaces were examined with immunofluorescence staining. Each coating material was inoculated with RAW264.7 cells (1 × 105 cells/mL) in 24-well plates, as described in the previous section. On the 4th day, the culture medium was removed, and the cells on the coating surface were gently rinsed with PBS. The cell suspension was trypsinized and centrifuged at 1200 rpm and 4°C for 5 min. Specific binding sites were blocked with PBS (containing 1% BSA) for 30 min at room temperature. The macrophages were incubated with primary antibodies against CD206 and iNOS (Abcam, UK) at 4°C overnight. On the following day, the cells were washed 1–2 times with PBS, and the surface of the material was incubated with the appropriate amount of diluted (1:200) secondary antibody for 1 h at room temperature in the dark. The cells were washed with PBS 2–3 times and then incubated with a DAPI staining solution for 5 min at room temperature. After several gentle washes, the cells were observed and photographed using an inverted fluorescence microscope (Olympus, Japan).
Enzyme-linked immunosorbent assay (ELISA)
Each material was placed in 24-well plates and inoculated with 1 × 105 RAW267.4 cells. After 4 days of incubation, the cell culture medium was collected, and the concentrations of various factors (IL-6, iNOS, tumor necrosis factor [TNF]-α, and CD206) were calculated based on standard curves using ELISA kits (Anogen, Canada) and an enzyme-labeling instrument (Thermo Fisher Scientific, USA).
Real-time polymerase chain reaction (RT-PCR) analysis
RT-PCR was performed to detect the expression of immune-related genes. Each surface coating was placed in a 24-well plate and inoculated with RAW267.4 cells (1 × 105 cells per) well for 4 days. Subsequently, the cells on the surface of the material were lysed using TRIzol reagent (Roche, Switzerland) for 30 min at 4°C, and the lysate was collected in enzyme-free EP tubes. RNA was extracted with isopropanol, and complementary DNA (cDNA) was synthesized from the total RNA using the Transcriber First Strand cDNA Synthesis Kit (Takara, Japan) according to the manufacturer’s protocol. PCR was performed using the FastStart Universal SYBR Green Master Kit (Roche, Switzerland) and a thermal cycler (LightCycler 96, Roche, Switzerland). The expression of the target genes CD206 and TNF-α was consistent with that of the reference gene GAPDH. RT-PCR experiments were performed at least twice, and each sample was analyzed in triplicate. Relative expression was quantified based on the comparison threshold method.
Interactions between mouse BMSCs and the immune system
Preparation of macrophage conditioned medium
RAW267.4 cells were inoculated onto the surface of the coating materials in a 6-well plate for 4 days. Then, the medium was collected and centrifuged at 1000 rpm for 5 min. The supernatant was mixed with DMEM at a ratio of 1:2 to prepare the macrophage conditioned medium. Subsequently, a 1 × 105/mL suspension of BMSCs was prepared, inoculated into 24-well plates, and cultured in macrophage conditioned medium. After 7 and 14 days of culture, the cells were removed, washed thrice with PBS, and fixed with 4% PFA for 30 min. The fixed cells were stained with an ALP staining reagent under dark conditions for 30 min. Then, the ALP staining solution was aspirated, and the cells were washed thrice with PBS before being observed using an optical microscope (Leica) (Figure 2).
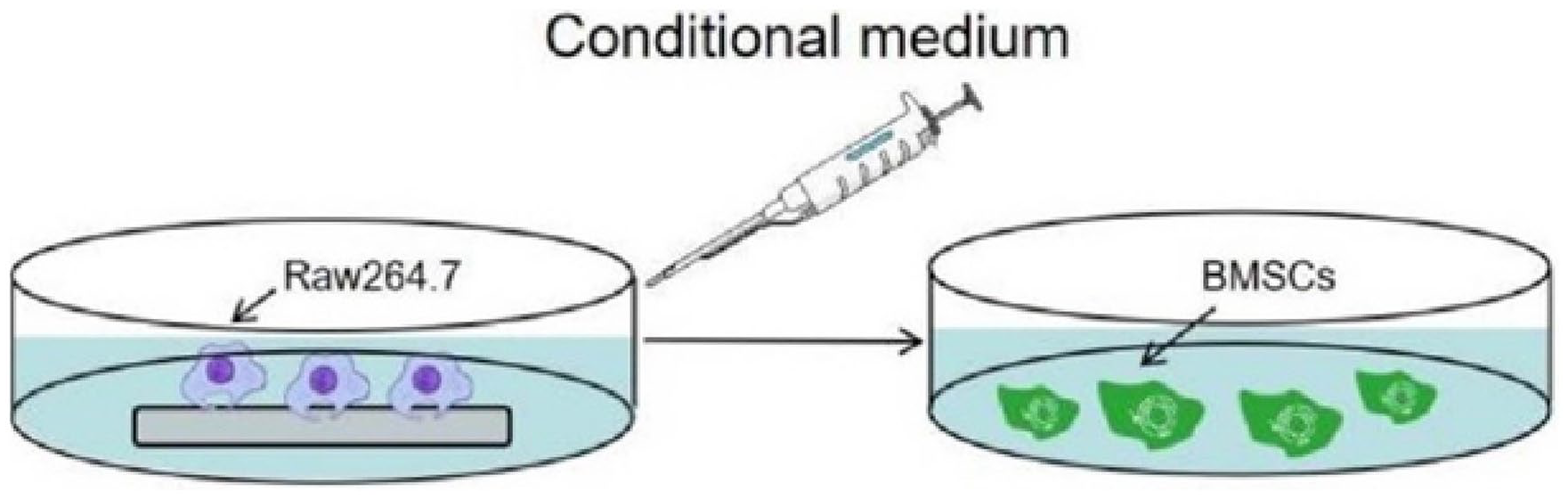
Schematic illustration of the effect of the conditional medium from coatings induced RAW264.7 cells on BMSCs.
Expression of osteogenesis-related genes in BMSCs cultured in macrophage conditioned medium
The expression of osteogenesis-related genes (BMP-2 and RUNX-2) was examined in BMSCs cultured in macrophage conditioned medium using RT-PCR. Specifically, BMSCs were inoculated in 24-well plates at a concentration of 1 × 105 cells/mL and co-cultured with different groups of macrophage conditioned medium in a cell culture incubator. The culture medium was replaced regularly, and after 7 days, the cells were lysed with TRIzol for 30 min at 4°C. The lysate was collected in enzyme-free EP tubes; RNA was extracted with isopropanol and reverse transcribed into cDNA using the Transcriber First Strand cDNA Synthesis Kit (Takara, Japan). PCR was performed using the SYBR® Fast qPCR Mix (Takara) kit. The expression of the target genes BMP-2 and RUNX-2 was consistent with that of the reference gene GAPDH.
ALP activity
First, 1 mL of a 1 × 105 cells/mL BMSC suspension was inoculated into a 24-well plate and treated with different types of macrophage conditioned medium. The cells were subsequently cultured in a cell culture incubator. For ALP staining, the medium was discarded, and the cells were washed 2–3 times with PBS before fixing with 4% paraformaldehyde for 30 min. The BCIP/NBT working solution was prepared according to the instructions of the ALP staining kit, and the cells were incubated with the BCIP/NBT working solution for 1 h at room temperature under dark conditions. Subsequently, the BCIP/NBT working solution was discarded, and the chromogenic reaction was terminated by washing. The cells were observed and photographed under a light microscope (Leica, Germany). Moreover, the ALP activity was calculated based on the absorbance at 405 nm using a microplate reader.
In vivo immunoregulation
Mouse air-pouch model
Thirty male C57BL/6 mice, 6–8 weeks old and weighing about 22–25 g, were selected for this experiment. The mice were divided into groups of five per group. The air pouch model was established as follows. In a standard sterile environment, 4 mL of sterile air was injected into the subcutaneous region on the backs of the mice using a 5 mL syringe to create a dorsal air sac. After 4 days, 4 mL sterile air was again injected into the dorsal air sac to prevent it from shrinking or flattening. Twenty-four hours later, the mice were anesthetized via the intraperitoneal injection of 1% pentobarbital sodium. The hair around the air sacs was removed using a razor. After disinfecting the surgical site, the mice were placed on sterile bedding, covered with a sterile hole towel, and positioned on a sterile operating table. A transverse incision of appropriate size was made on the dorsal air sac, and the experimental coating samples were slowly implanted into the air sac from the open incision using tweezers. The incision was sutured and disinfected, and the mice were returned to the rearing cage and provided with adequate water and feed. All experiments involving animals were approved by the Animal Ethics Committee of Xinjiang Medical University and were performed in strict accordance with the requirements and regulations of animal ethics.
Sampling and preservation of lavage fluid and air sac tissue from mice
Four days after the dorsal air sac implantation, the lavage fluid and skin tissue were obtained. Specifically, the mice were anesthetized using an intraperitoneal injection of 1% pentobarbital sodium. The hair on the surface around the air sac was shaved off using an animal razor and the remaining hair was removed using a hair removal cream. The surgical site was disinfected, and the mice were placed on sterile bedding, covered with a sterile hole towel, and positioned on the sterile operating table. The upper edge of the air sac was incised, and the gap between the material and the tissue in the air sac was repeatedly washed with 3 mL of PBS using a syringe. The lavage fluid was subsequently filtered with a nylon filter to remove any tissue mass. The obtained lavage fluid was centrifuged at 1200 rpm and 4°C for 5 min, and the supernatant was stored at −80°C. Additionally, the tissue in contact with the coating materials was excised and fixed in 4%formaldehyde, and the mice were sacrificed via cervical dislocation.
ELISA for the cytokines in the air sac lavage fluid
The contents of key factors (TNF-α and IL-10) in the air sac lavage fluid were analyzed and studied using ELISA. Specifically, the lavage fluid was filtered and centrifuged, and the supernatant was collected. The ELISA kit reagents were allowed to reach room temperature. Meanwhile, sample wells, standard wells, and blank wells were set up: 100 μL of the sample diluent was added to the blank wells, and 100 μL of standard and samples were added to the standard and blank wells, respectively. Then, 100 μL of working solution A was added to each well and incubated at room temperature for 1 h. The supernatant was discarded, and the wells were washed several times. Then, 100 μL of working solution B was added to each well and incubated at room temperature for 1 h. The supernatant was discarded, and the wells were washed several times. Then, 90 μL of substrate was added to each well and incubated at room temperature for 30 min. Subsequently, 50 μL of termination solution was added, and the absorbance of the solution in each well was examined at 450 nm. A standard curve was plotted according to the absorbance values of the standard solutions.
Histologic analysis of air sacs
The mouse air sac tissue that was in contact with the coating materials was excised and subjected to hematoxylin and eosin (H&E) staining and Masson staining to observe the effect of the material on the inflammatory infiltration in the surrounding tissues.
Statistical analysis
All in vitro experiments were repeated at least three times. Data are presented as the mean and standard deviation. The results of in vivo and in vitro experiments were analyzed using one-way analysis of variance, and differences were considered statistically significant at p-values of <0.05.
Results
Material characterization
Figure 3(A: a-1, b-1, c-1, d-1, e-1) shows the SEM images of the different coating materials. The Ti and GO groups exhibited a flat surface, while the HA group (Ac-1), HA-2wt%GO group (Ad-1), and HA-5wt%GO group (Ae-1) showed a porous surface. Nevertheless, the HA (Ac-2) group and the HA-5wt%GO group (Ae-2) showed more cracks on their surface, whereas the HA-2wt%GO group (Ad-2) showed a homogeneous and crack-free surface. AFM results (Figure 3(A: a-3, b-3, c-3, d-3, e-3)) demonstrated that the GO group had the lowest roughness, followed by the Ti group. Meanwhile, the HA group had the highest roughness, and in the HA-2wt%GO and HA-5wt%GO groups, the roughness increased in a GO concentration-dependent manner.
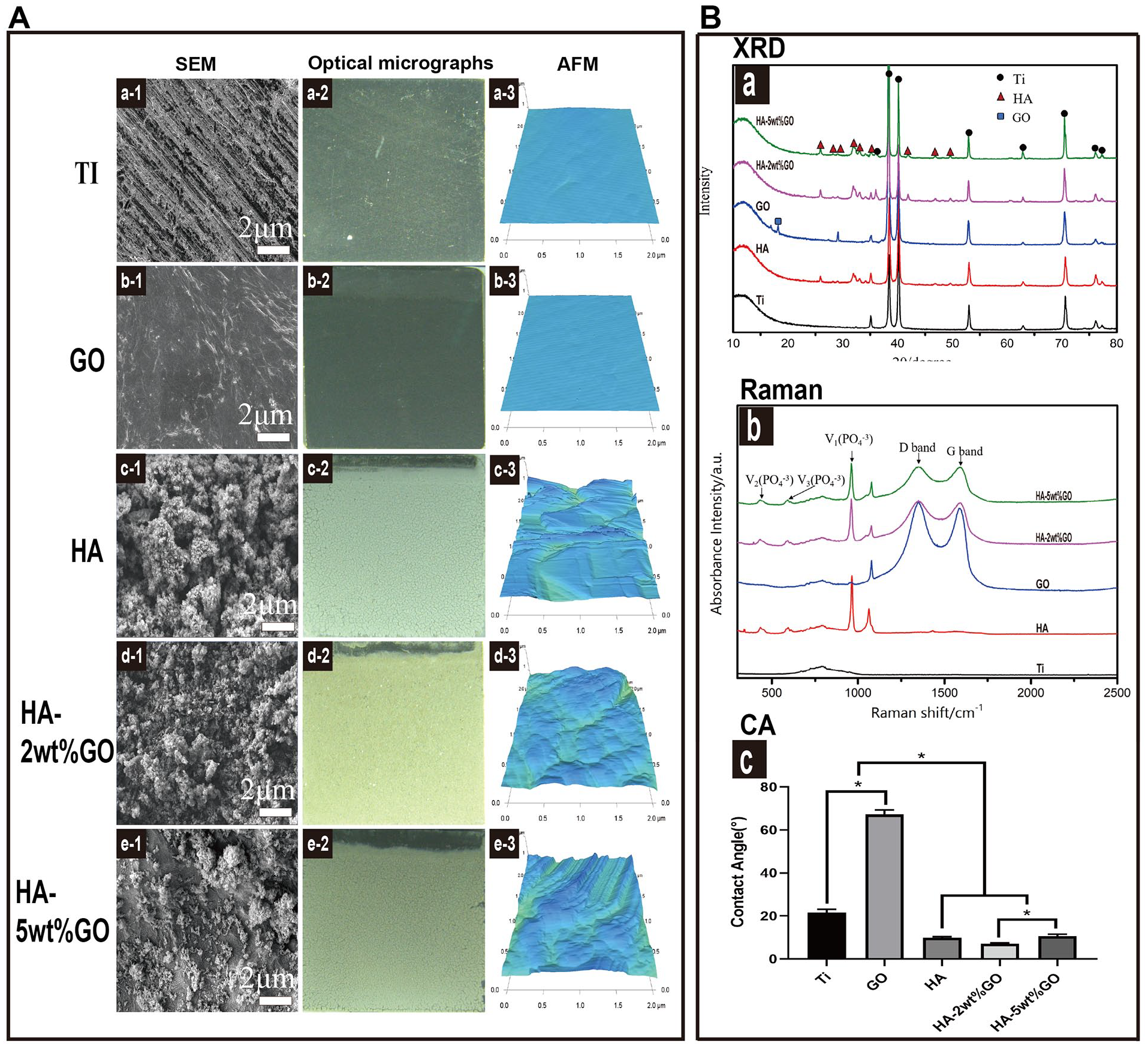
(A) SEM images of the Ti (a-1), GO (b-1), HA (c-1), HA-2wt%GO (d-1), and HA-5wt%GO (e-1). Optical micrographs of the Ti (a-2), GO (b-2), HA (c-2), HA-2wt%GO (d-2), and HA-5wt%GO (e-2). Surface morphology and roughness of the Ti (a-3), GO (b-3), HA (c-3), and HA-2wt%GO (d-3) measured by AFM. (B) (a) XRD pattern of the samples, (b) Raman spectrum of the samples, and (c) contact angle measurements showing the wettability of the samples.
In the XRD patterns (Figure 3B(a)), the presence of HA and Ti phases in the matrix indicated that HA, HA-2wt%GO, and HA-5wt%GO were successfully deposited on the surface of Ti by EPD. The GO phase could be observed in the GO group, but not in the HA-2wt%GO and HA-5wt%GO groups, likely due to the destructive order of the crystal pattern and the irregular atomic arrangement. 35 Therefore, Raman spectroscopy was utilized to confirm the presence and nature of GO nano-fillers in the composite coatings. As shown in Figure 3B(b), the spectrum of the Ti samples contained a straight line without any peaks. In contrast, the GO, HA-2wt%GO, and HA-5wt%GO samples showed a typical D-band (1355 cm−1) and G-band (1593 cm−1) due to the presence of GO. The representative signals of the D-band and G-band demonstrated that GO had been successfully conjugated onto the coating surface. 36 The surface wettability characteristics of Ti, HA, GO, HA-2wt%GO, and HA-5wt%GO are shown in Figure 3B(c). The experiment showed that the surface of the HA-2wt%GO group and the HA-5wt%GO group exhibited extraordinary hydrophilicity.
In vitro cell behavior
Attachment and proliferation of RAW264.7 cells
The CCK-8 assay was employed to detect the proliferation of RAW264.7 cells inoculated on the surface of each coating material after 4 and 7 days of culture (Figure 4). The relative cell proliferation rates in all groups gradually increased at 4 and 7 days of culture. On day 4, the proliferation of RAW264.7 on different coating surfaces showed the following trends: the HA-2wt%GO and HA-5wt%GO groups had the highest cell proliferation rate, and these rates were significantly higher than those in the Ti, HA, and GO groups. Similarly, on day 7, the RAW264.7 cell proliferation rate was the highest in the HA-2wt%GO and HA-5wt%GO groups, that is, significantly higher than that in the Ti, HA, and GO groups (p < 0.05). Meanwhile, there was no significant difference in the cell proliferation rate among the Ti, HA, and GO groups. DAPI staining showed that after the inoculation of the RAW264.7 cells on the surface of the five coating materials for 4 and 7 days (Figure 5), the HA-2wt%GO and HA-5wt%GO groups had more densely packed cells on the material surface and a higher number of proliferating cells. Consistent with the CCK-8 results, these results showed that HA-2wt%GO has better cytocompatibility and can maintain a higher cell number.
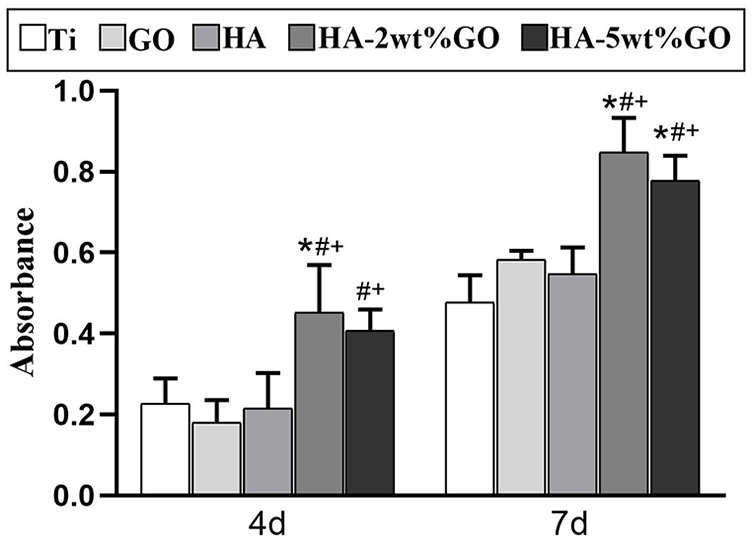
CCK-8 was used to detect the activity of RAW264.7 cells cultured on Ti, GO, HA, HA-2wt%GO, and HA-5wt%GO groups for 4 and 7 days respectively.

Flow cytometry analyses of cell-surface markers on RAW264.7: (A) Scatter diagram for RAW264.7 cells expressing CD206 (M2 maker), (B) Scatter diagram for RAW264.7 cells expressing CCR7 (M1 maker), (C) quantitative analysis for RAW264.7 cells expressing CD206 (M2 maker), and (D) quantitative analysis for RAW264.7 cells expressing CCR7 (M1 maker).
Field emission scanning electron microscopy (FESEM) was employed to observe the spreading of the macrophages on the surface of the five coating materials after 4 days of incubation. As shown in Figure 5, RAW264.7 cells could tightly adhere to the different coating surfaces. However, the morphology of the adhered macrophages differed according to the coating surface. The cell spreading area of the macrophages was higher in the HA-2wt%GO and HA-5wt%GO groups and lower in the Ti, HA, and GO groups. In particular, in the HA-2wt%GO group, the RAW264.7 cells flattened significantly to form sheet-like structures. These cells could superimpose atop each other and showed a wide distribution. Moreover, they were interconnected, and their spreading area was higher than that of the macrophages present on the surfaces of the other materials. In the Ti and GO groups, few clusters of cells adhering to the surface were observed. Additionally, the cells on the HA-2wt%GO surface were largely oval, while those on the Ti, GO, and HA surfaces tended to be spindle-shaped.
RAW264.7 cell polarization on different coating surfaces
The effect of different coating surfaces on the polarization of RAW264.7 cells was investigated via flow cytometry. M1 macrophages were labeled with CCR7, and M2 macrophages were labeled with CD206. Accordingly, flow cytometry was used to detect the effect of coating materials on RAW264.7 cell polarization in each group. The proportion of macrophages expressing the M2 marker CD206 was the highest in the HA-2wt%GO and HA-5wt%GO groups, followed by the HA and GO groups and the Ti group (Figure 6(A)). In contrast, the proportion of macrophages expressing the M1 marker CCR7 was the highest in the Ti group, followed by GO and HA-5wt%GO groups and the HA and HA-2wt%GO groups (Figure 6(B)).
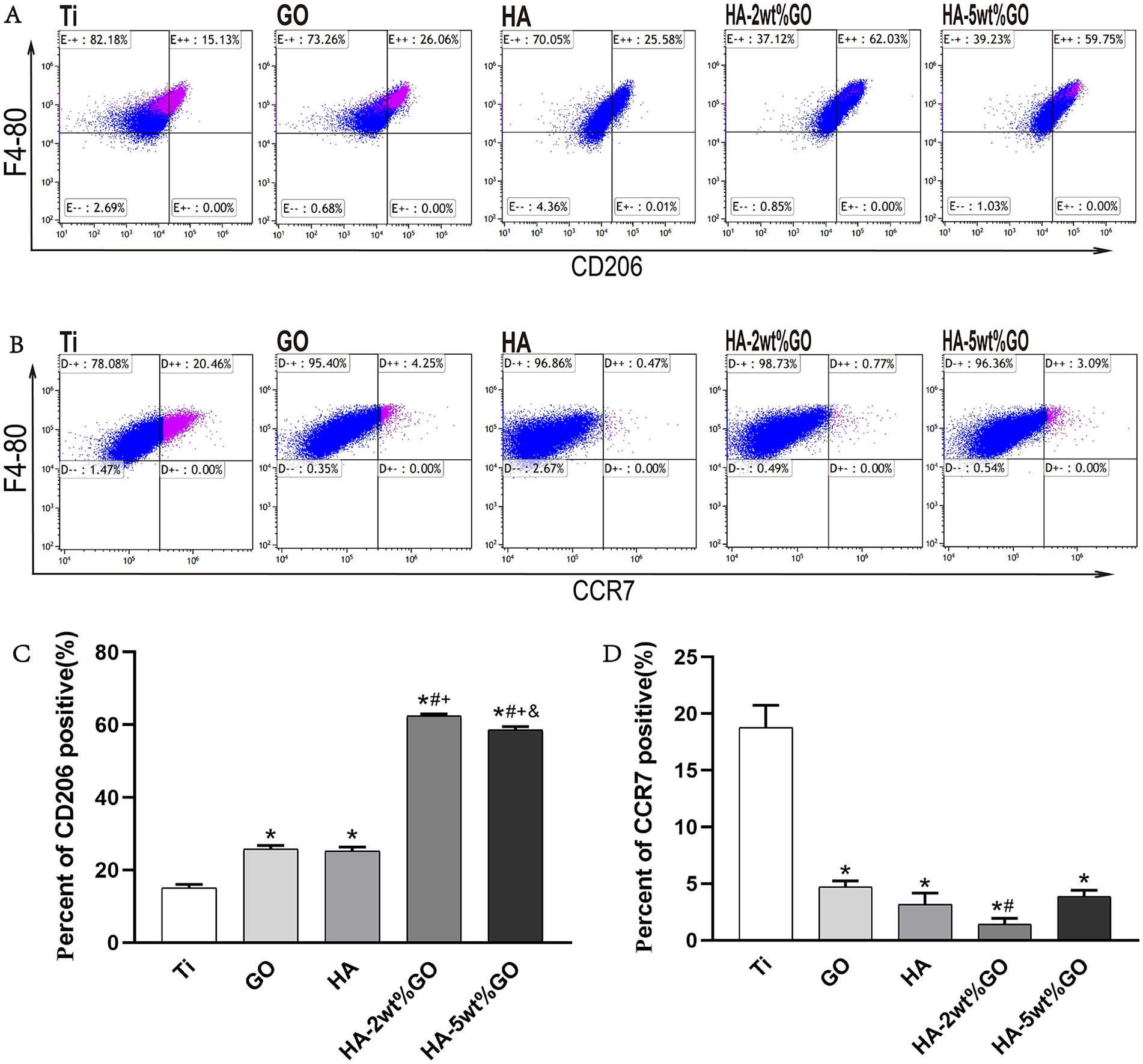
Flow cytometry analyses of cell-surface markers on RAW264.7: (A) Scatter diagram for RAW264.7 cells expressing CD206 (M2 maker), (B) Scatter diagram for RAW264.7 cells expressing CCR7 (M1 maker ), (C) quantitative analysis for RAW264.7 cells expressing CD206 (M2 maker), and (D) Quantitative analysis for RAW264.7 cells expressing CCR7 (M1 maker).
The expression of M1 and M2 surface markers was also determined using immunofluorescence staining to assess the effect of different coating materials on RAW264.7 polarization. The M2 marker CD206 was labeled using green fluorescence, while the M1 marker iNOS was labeled using red fluorescence. As shown in Figure 7, the Ti group and the GO group showed a small number of macrophages, but most of them were positive for iNOS (strong red fluorescence), indicating that they were M1-type macrophages. In the HA group, HA-2wt%GO group, and HA-5wt%GO group, red fluorescence was limited, and strong green fluorescence indicative of CD206 expression was observed. This indicated that most of the macrophages in these groups were of the M2 phenotype.
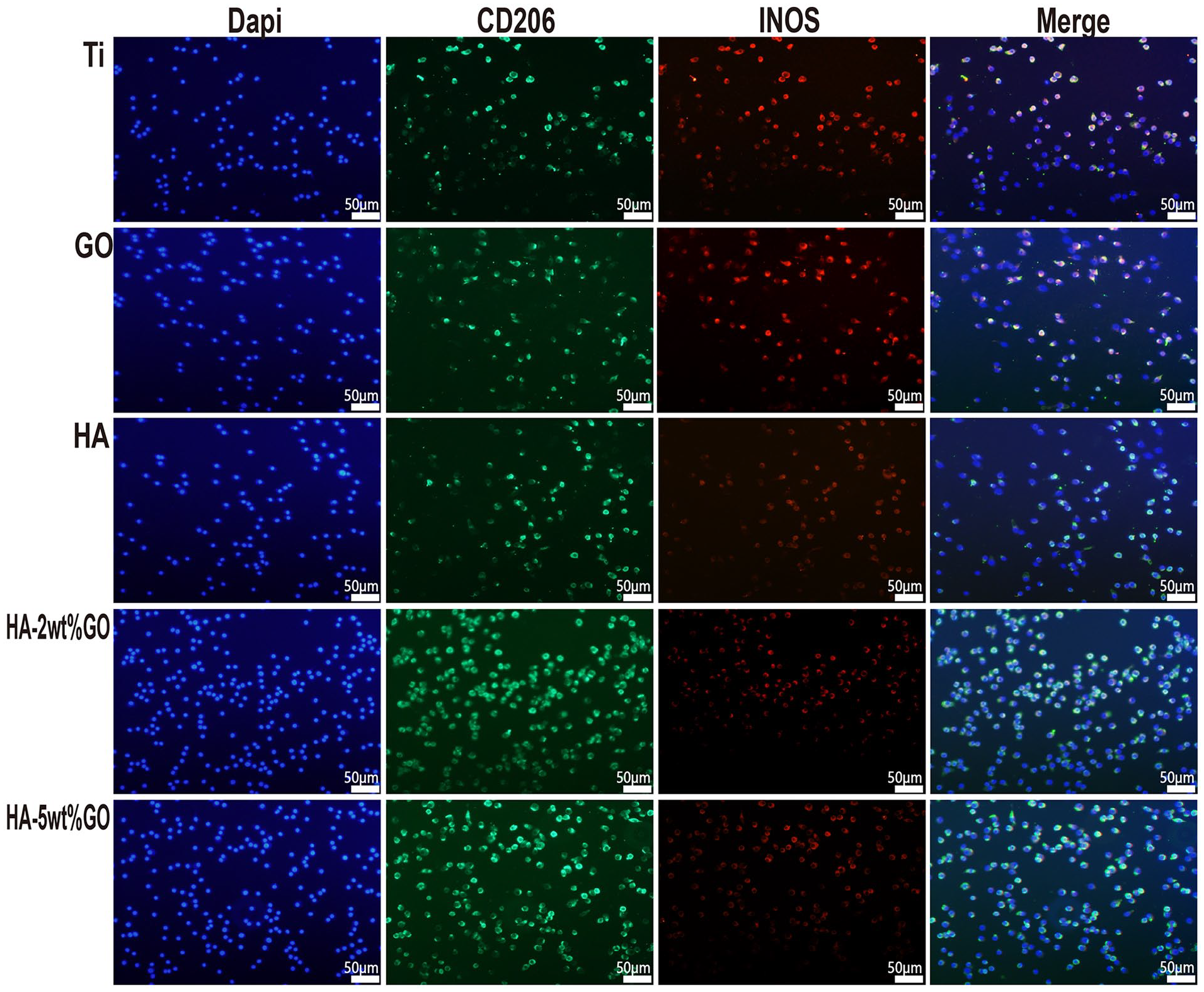
Immunofluorescent staining of RAW264.7 cultured on each groups.
Detection by ELISA
The effect of different coating materials on the expression of pro-inflammatory cytokines (IL-6, iNOS, TNF-α) and anti-inflammatory cytokines (CD206) in RAW264.7 cells was examined (Figure 8). RAW264.7 cells were inoculated on different coating surfaces for 4 days. The number of RAW264.7 cells expressing the pro-inflammatory cytokines IL-6, iNOS, and TNF-α was significantly lower in the HA-2wt%GO and HA-5wt%GO groups than in the Ti, HA, and GO groups (p < 0.05). Specifically, the trend of IL-6 and iNOS expression was consistent across all groups, that is, the levels were significantly lower in the HA-2wt%GO and HA-5wt%GO groups than in the Ti, HA, and GO groups but showed no significant difference between the HA-2wt%GO group and the HA-5wt%GO group. Meanwhile, the levels of anti-inflammatory cytokines were the highest in the HA-2wt%GO group, significantly higher than those in the Ti and GO groups (p < 0.05). However, the values in the HA group and the HA-5wt%GO group were not significantly different.
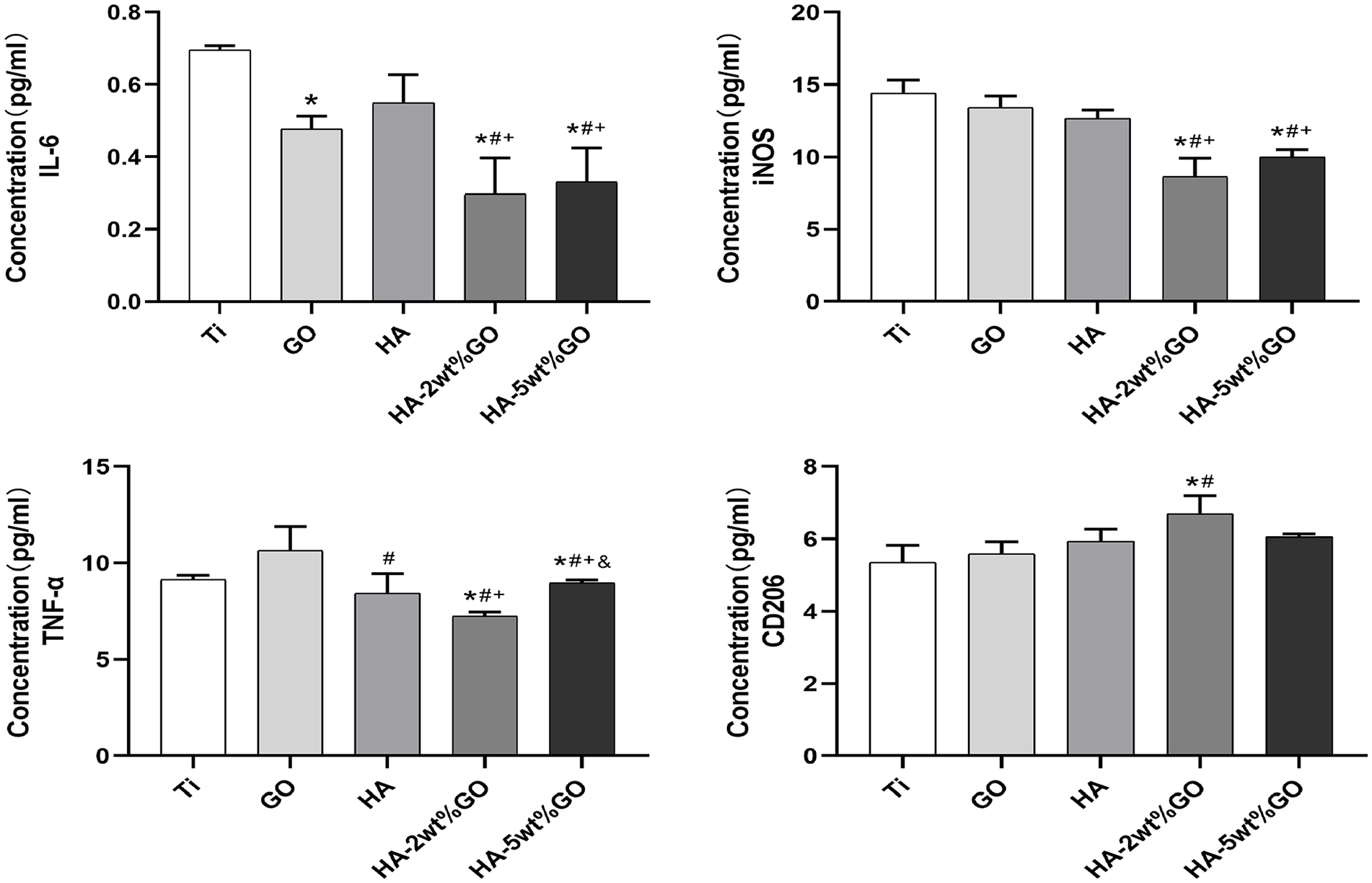
The secretion of IL-6, iNOS, TNF-a, and CD206 of RAW264.7 cultured on each groups for 4 days.
Expression of polarization-related genes
M1 macrophages express the inflammation-related gene TNF-α, while M2 macrophages express anti-inflammatory genes such as CD206. In this study, RAW264.7 cells were inoculated on different coating materials for 4 days, and the expression levels of polarization-related genes were detected (Figure 9). Compared with the Ti, HA, and GO groups, the HA-2wt%GO group showed a significant downregulation of TNF-α expression in RAW264.7 cells. The expression levels of CD206 in the HA-2wt%GO and HA-5wt%GO groups were higher than those in the Ti and GO groups. However, these levels were not significantly different among the HA, HA-2wt%GO, and HA-5wt%GO groups. Overall, surface coating with HA-2wt%GO could effectively inhibit the M1 polarization of macrophages and downregulate the expression of pro-inflammatory genes when compared to Ti alone and GO and HA surface coatings.
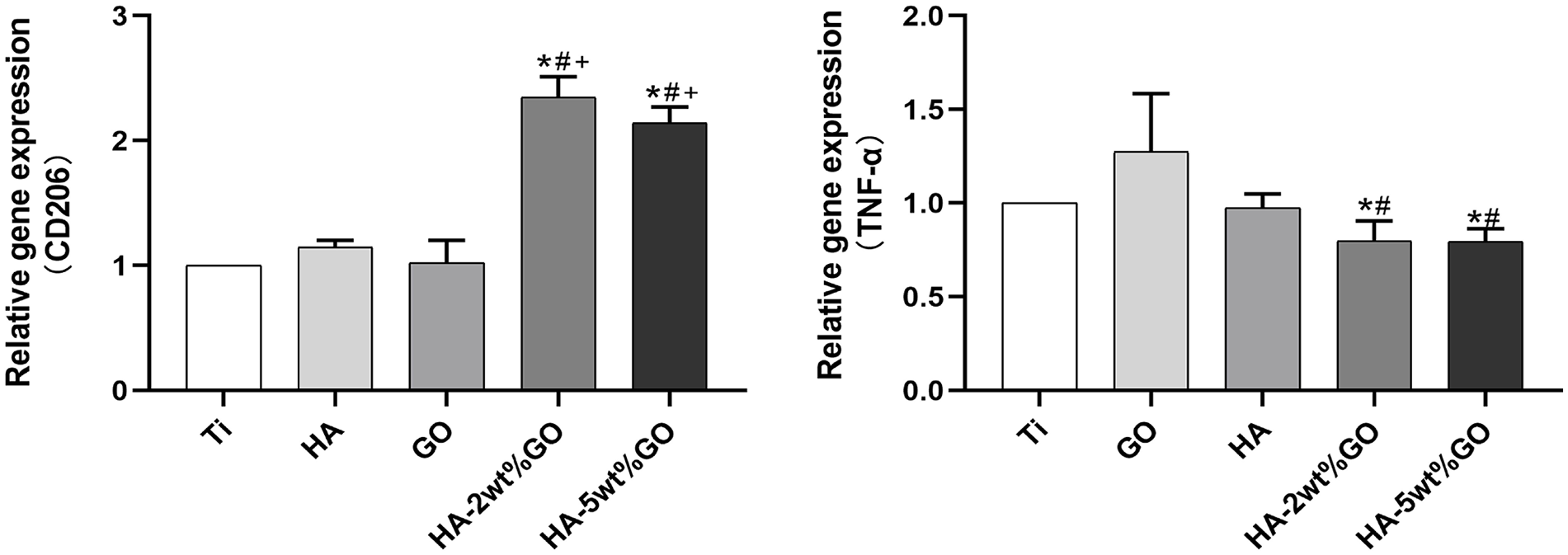
The expression of related genes of RAW264.7 cultured on the surface of Ti For Peer Review, GO, HA, and HA5wt%GO groups for 7 days.
Activity of ALP in BMSCs after culture in macrophage conditioned medium
ALP activity of BMSCs
ALP is a marker enzyme for mature osteoblasts. Thus, to examine osteogenesis, the ALP levels in BMSCs were examined after 7 days of incubation in macrophage conditioned medium. As shown in Figure 10, the area of staining in the HA-2wt%GO and HA-5wt%GO groups was significantly greater than that in the HA, GO, and Ti groups. Quantitative analysis showed that ALP activity in the HA-2wt%GO group was higher than that in the Ti group. The ALP activity in the HA-2wt%GO group was comparable to that in the HA-5wt%GO group and the HA group.
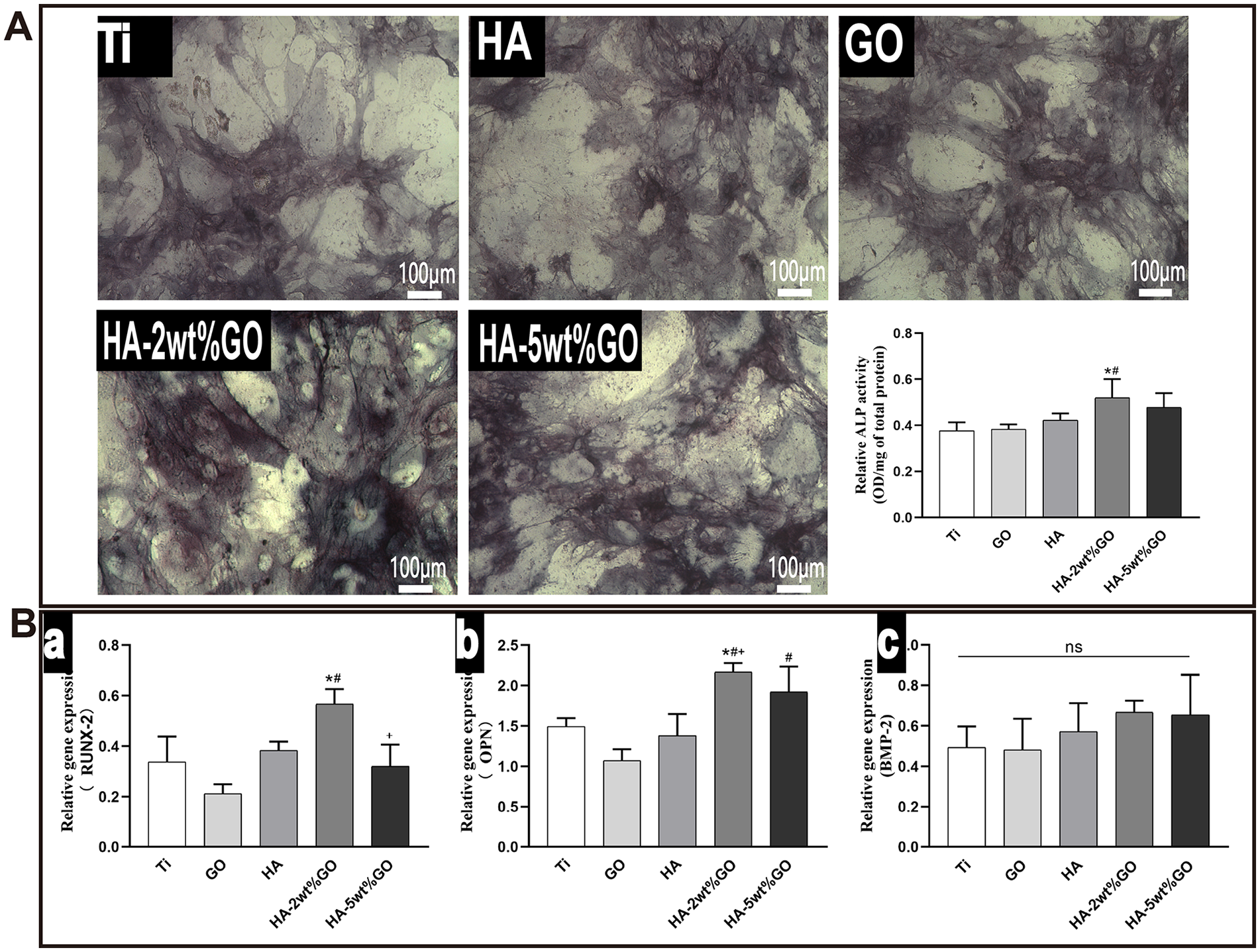
(A) ALP staining and activity detection of BMSCs cultured in RAW264.7 cell conditioned medium induced by different groups and (B) the expression of RUNX-2, OPN, and BMP-2 genes of BMSCs cultured in RAW264.7 conditioned medium of each groups.
In vivo effects of the surface coating materials
The induction of the mouse dorsal air sac model is shown in Figure 11(A). H&E staining and Masson staining were used to stain the mouse dorsal air sac tissues that came into contact with each type of implant after 4 days of implantation (Figure 11(B)). In the Ti, GO, and HA groups, the peripheral fibrous layer of the air sac tissue appeared to be thickened, and the thickening was the most obvious in the GO group. In contrast, relatively thin fibrous layers of the air sac tissue were observed in the HA-2wt%GO group and the HA-5wt%GO group. Specifically, the thickness was the lowest in the HA-2wt%GO group, followed by the HA-5wt%GO group, the HA group, and the Ti and GO groups (Figure 11(C)).
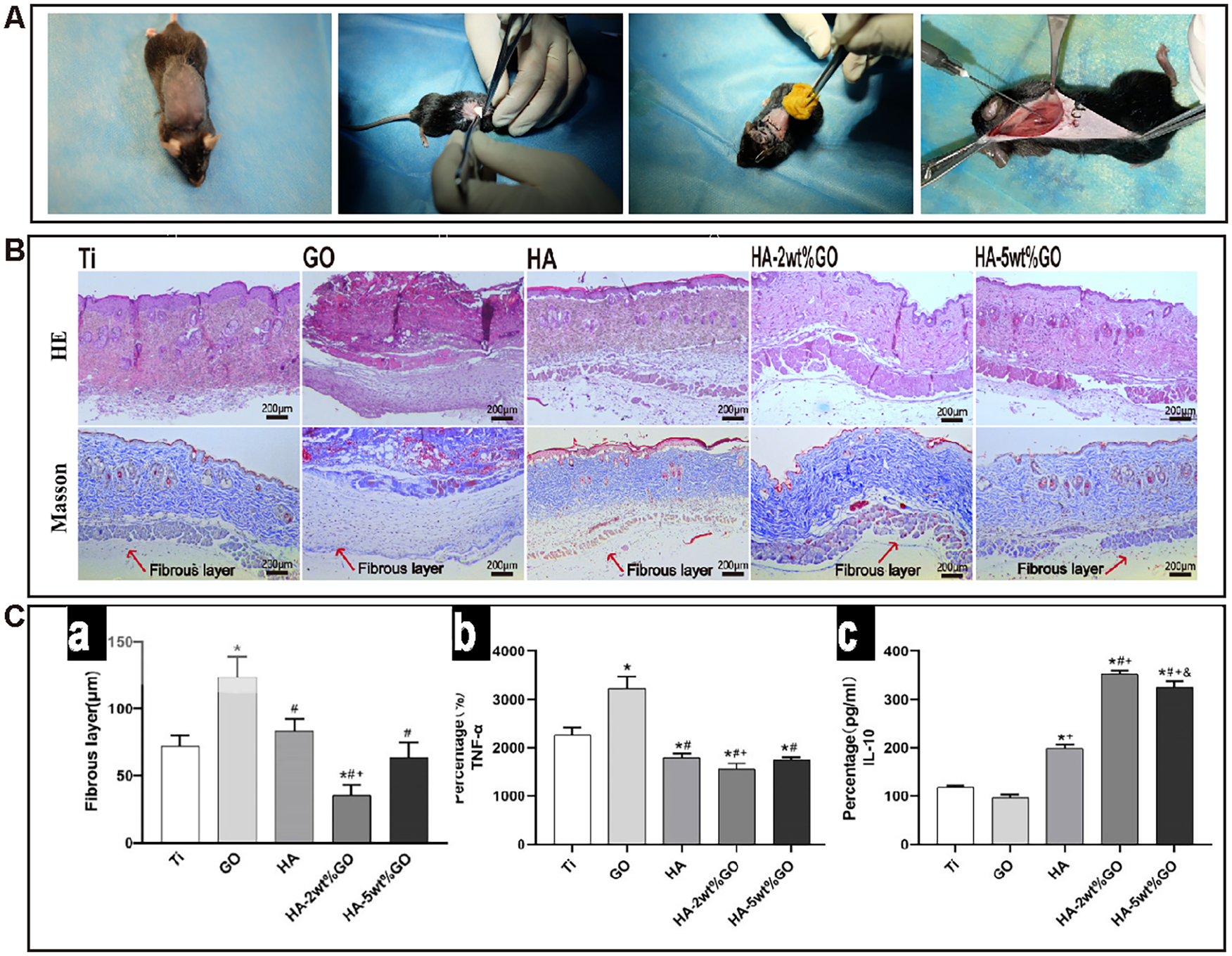
(A) Construction of mouse back air-pouch model, (B) images of H-E staining and Masson’s trichrome staining of the air-pouch tissues in each group, and (C) thickness of fibrous tissue formation around air-pouch tissues and ELISA determination of cytokines from the mouse air-pouch exudates in each group: (a) thickness of fibrous tissue formation of air-pouch tissues, (b) the production of cytokines TNF-α, and (c) the production of cytokines IL-10.
Subsequently, ELISA was employed to detect secreted cytokine levels in the lavage fluid of each group (Figure 11(C-b, c)). Compared with the Ti, HA, and GO groups, the HA-2wt%GO, and HA-5wt%GO groups showed significantly decreased levels of TNF-α in RAW264.7 cells. Moreover, IL-10 expression was the highest in the HA-2wt%GO group, and the difference was significant in comparison to the other groups.
Discussion
During the fabrication of orthopedic biomaterials, the composition and structure of the base material are modified to enhance its osteogenic activity and biocompatibility in order to achieve optimal biological characteristics and bioactivity.37,38 Nevertheless, recent studies on the interactions between the immune system and orthopedic biomaterials have reported that the immune system and related cells can directly influence the in vivo osteogenic effects of biomaterials. 39 Orthopedic implants and biomaterials are recognized as foreign bodies by host immune cells and can induce immunoreactivity. The first immunocytes to be activated during an immune response are intrinsic immunocytes,14,40 mainly neutrophils and macrophages. Among them, macrophages play a dominant role in immune responses. Artificial implants can activate macrophages, causing them to secrete a series of signaling molecules and cytokines that are closely related to bone metabolism and angiogenesis. These molecules can also influence the bone repair and regeneration process around the implant. Macrophages – as effector cells of the immune system – typically attempt to phagocytose implant biomaterials and participate in the formation of inflammatory granulomas around the implant, which are detrimental to osseointegration between the implant and host tissue. However, under certain circumstances, macrophages can also produce extracellular signaling molecules that promote the osteogenic differentiation of BMSCs and secrete matrix metalloproteinases that facilitate angiogenesis. 41 Overall, macrophages can influence the differentiation of tissue cells through paracrine secretion in special environments. 42 Based on their functional characteristics, macrophages can be divided into pro-inflammatory M1 macrophages and repair-promoting M2 macrophages, both of which have completely different effects on tissue repair and healing. In most cases of implant failure, significant inflammatory infiltration is observed around the implant, with M1 macrophages accounting for the majority of infiltrating cells. In contrast, normal implants do not show significant inflammatory tissue infiltration, and the implant is surrounded by M2 macrophages. 43
As one of the most diverse types of cells in the body, macrophages show a certain degree of plasticity. Although M1 and M2 macrophages have different roles and functions, normal macrophages are at the junction between the M1/M2 transition. Under the influence of external stimuli, such as lipopolysaccharide or IL-4, macrophages undergo M1 or M2 polarization, 44 promoting the development of a pro-or anti-inflammatory immune microenvironment. As foreign bodies, implants also serve as an external stimulus, causing macrophage polarization. The activated macrophages not only accumulate around the material but also release specific signals to regulate a series of biological behaviors.45,46 Thus, it is theoretically feasible to prevent implant rejection by modulating the macrophage response, and this can be achieved by modifying the surface structure or composition of the implanted biomaterials. Therefore, novel biomaterials should not only contribute to osteogenesis but also have immunoregulatory functions. Specifically, they must enable the M2 polarization of macrophages, thus promoting an immune microenvironment that favors tissue repair. 47
To this end, in this study, a GO-HA composite coating was prepared by combining HA (that has osteogenic effects) with GO (that has immunoregulatory effects) on the surface of Ti implants. This amalgamation of “osteogenesis” and “immunoregulation” was expected to promote bone tissue repair. In this study, in vitro experiments were performed using mouse mononuclear macrophages (RAW264.7 cells), which were inoculated on different coating surfaces to test the effects of these materials on the functional state of the macrophages.
Notably, RAW264.7 cells showed different degrees of proliferation on the surfaces of the five biomaterials (Figure 4), consistent with the findings from BMSCs inoculated on these biomaterials. Additionally, the macrophages from the HA-2wt%GO group showed especially high proliferation rates. DAPI staining after 4 and 7 days of inoculation on biomaterial surfaces (Figure 5) showed that the RAW264.7 cells in the HA-2wt%GO and HA-5wt%GO groups were more densely packed, and the number of proliferating cells was higher in these groups. Overall, the HA-2wt%GO biomaterial had higher cytocompatibility, exerted no cytotoxicity against RAW264.7 cells, and supported the accumulation of more cells on its surface.
It has been demonstrated that the surface properties of biomaterials can alter macrophage-related biological behaviors. 48 For example, hydrophilic coating surfaces are more favorable for macrophage adhesion, 49 while corrosive metallic materials such as nickel or amine-based materials can accelerate the infiltration of lymphocytes and macrophages, resulting in more intense inflammation. Specifically, implants with micro- and nano-pore structures support the surface accumulation of more M2 macrophages and promote vascularization, enabling successful clinical treatment. 50 Additionally, macrophage inflammation is lower on the surface of micro- and nanoscale-modified materials, 51 and the amount of pro-inflammatory substances released by macrophages and the levels of related inflammatory factors are reduced.52,53 Through experiments in animal models, Wisniewski et al. 54 reported that nanomaterials can effectively mitigate adverse host immune reactions. Moreover, studies have shown that immune cells are highly sensitive to the nano- and micro-scale morphology of the implant and can respond differently depending on the nanostructure of the implant’s surface. Specifically, greater surface roughness reduces biocompatibility, promotes inflammation, creates localized microenvironment instability, and increases the risk of immune rejection. Indeed, the surface roughness of Ti is within the narrow optimal range (Ra = 0.51–1.36 μm) that promotes the upregulation of anti-inflammatory factor-related gene expression in macrophages. 55
In this study, flow cytometry and immunofluorescence experiments revealed that the GO-HA composite coating has good macrophage polarization-inducing functions. In addition to modifying the structure of the implant at the nano and micro scale, HA also releases Ca2+ from the composite coating. This Ca can act as an agonist and regulator of Ca-sensitive receptors (CaSR) 56 in osteocytes to facilitate the osteogenesis, proliferation, and mineralization of BMSCs.57,58 Meanwhile, modification of the Ti implant surface with 10% Ca can increase the M2 polarization of RAW264.7 cells. This effect can be attributed to the strong hydrophilicity of the Ca coating surface, which enhances protein adsorption and cell adhesion.59,60 Meanwhile, GO can also exert immunoregulatory effects, although its poor degradation in vivo has remained a pressing concern. However, new evidence shows that GO can activate neutrophils in vivo, which leads to the release of lysosomal molecules for the degradation of GO.61,62 The use of GO as an implant surface coating causes inherent immunoreactions in vivo, especially macrophage activation, which affects osteogenesis. 63 However, at low concentrations, GO can inhibit the generation of reactive oxygen species, thus inhibiting M1 polarization and promoting M2 polarization. Moreover, given its high surface area, GO has excellent slow-release properties. 64 Hence, the GO-HA composite coating can slow-release Ca2+ from HA and continuously stimulate CaSR. Previous studies have demonstrated that GO implants in the peritoneal cavity of mice do not stimulate the M1 polarization of macrophages. Instead, M2 macrophages are more abundant under these conditions. Furthermore, low concentrations of GO can significantly promote M2 polarization in vitro and enhance BMSC osteogenesis via OSM and the NF-κB pathway. 65 Hence, the combination of GO and HA at an optimal ratio can produce a synergistic effect, promoting the M2 polarization of macrophages.
In order to further evaluate the functional status of macrophages under the direct action of different biomaterial coating surfaces, the relevant functional properties of macrophages were investigated at the gene and protein levels in this study. On day 4, the secreted levels of pro-inflammatory cytokines (IL-6, iNOS, and TNF-α) were significantly lower in RAW264.7 cells inoculated on HA-2wt%GO and HA-5wt%GO implants than in those inoculated on Ti, HA, and GO implants. Meanwhile, the levels of these factors were not significantly different between the HA-2wt%GO group and the HA-5wt%GO group. Overall, the HA-2wt%GO group had the lowest expression of TNF-α, followed by the HA-5wt%GO group. Further, the levels in the HA group were higher than those in the GO group.
Changes in the levels of macrophage-related genes are indicative of changes in the functional status of macrophages. Compared with the Ti, HA, and GO groups, the HA-2wt%GO group showed significantly reduced expression of the inflammation-related factor TNF-α in RAW264.7 cells. TNF-α can affect the bone repair process and inhibit the expression of bone matrix proteins, ALP and RUNX-2. 66 Notably, in this study, the expression level of CD206 was significantly upregulated in the HA-2wt%GO and HA-5wt%GO groups. Notably, the level of this gene was significantly higher in these groups than in the Ti and GO groups. Overall, the HA-2wt%GO implant surface was more effective at downregulating pro-inflammatory gene expression in macrophages and inhibiting M1 polarization than the Ti, GO, and HA implant surfaces. Owing to the synergistic effects of Ca2+ from and GO, the HA-2wt%GO implant could regulate the function of macrophages during in vitro co-culture. In addition, this implant coating could inhibit the M1 polarization of macrophages in vitro while simultaneously promoting M2 polarization, creating an immune microenvironment more conducive to the formation of bone tissue.
Owing to the continuous integration of immunology and bone biology, the relationship between the immune system and the skeletal system has been gradually understood. As a result, osteoimmunology has emerged as an important area of research. Previous studies have demonstrated that after bone biomaterials are implanted into the human body, immune-associated cells accumulate at the interface between the bone tissue and the material. Importantly, the chemical composition and microstructure of the material’s surface can induce differential biological behaviors in macrophages.67,68 Our study examined macrophage adhesion and proliferation by inoculating macrophages directly onto the coating surface. The findings also showed that GO-HA coating surface can induce M2 polarization in vitro. However, these in vitro conditions did not fully recapitulate the in vivo environment of bone tissue and stem cells. Hence, we investigated the effect of the immune microenvironment induced by different coatings on the osteogenesis of BMSCs.
Stem cell osteogenesis plays a key role in bone repair after implantation. In order to confirm that the GO-HA composite coating could regulate macrophage responses during osteogenesis, macrophages were inoculated on the surface of the biomaterials, and the supernatant was collected after 4 days of incubation to obtain macrophage conditioned medium. The BMSCs were cultured in the macrophage conditioned medium and tested for multiple osteogenesis markers to evaluate the osteogenic effect of the cytokines secreted by different groups of macrophages forming different immune microenvironments. The BMSCs of the HA-2wt%GO group were found to have the highest ALP activity and the strongest ALP staining. Meanwhile, RT-PCR showed that although there were no statistically significant differences in the expression of individual osteogenesis-related genes among the groups, BMP-2 expression was the highest in the HA-2wt%GO group. Overall, the proportion of M2 macrophages was the highest in the HA-2wt%GO group, leading to the increased secretion of anti-inflammatory cytokines and osteoprotegerin. Therefore, the findings showed that macrophages co-cultured on the HA-2wt%GO surface induce an immune microenvironment that is favorable for BMSC osteogenesis.
Biomaterials are widely used in orthopedics and other medical fields, and their application depends on their physical, chemical, and biochemical properties. When biomaterials are inserted into the body, they can activate the host immune response. Poorer biomaterials can induce a stronger, more harmful inflammatory reaction. Thus, they cannot maintain their function in the body over a prolonged period. However, when biomaterials have better physicochemical and biological properties and can regulate inflammatory cells and promote osteocyte proliferation, they can coexist with host tissues and play their functional role for a long duration. Our in vitro tests revealed that the HA-2wt%GO coating has certain advantages with respect to immune regulation, and conditioned medium from macrophages grown on this substrate can promote the osteogenic differentiation of BMSCs. Hence, the in vivo effects of the implants with HA-2wt%GO coating were examined after implantation in a dorsal air sac mouse model. Subsequently, lavage fluid was collected from the dorsal air sacs to examine cytokine secretion by macrophages. In particular, the HA-2wt%GO group showed a higher expression of the anti-inflammatory cytokine IL-10 and low levels of the pro-inflammatory cytokine TNF-α. These results were consistent with the results of in vitro tests and also supported by the H&E staining and Masson staining analysis of air sac tissue. In the HA-2wt%GO group, the inflammatory infiltration in the air sac tissue was low, and fibrous layer hyperplasia was not obvious. In contrast, the other experimental groups showed fine inflammatory infiltration and obvious fibrous layer hyperplasia. Notably, these histological changes promote the aseptic loosening of bio-implants and lead to osteolysis in the vicinity of the bio-implants, reducing their service life. 69 Our findings showed that the HA-2wt%GO coating can promote M2 macrophage polarization, alleviate inflammation, reduce inflammatory cell infiltration, and inhibit fibrous layer hyperplasia. Hence, they suggest that this coating may improve the service life of bio-implants. It is undeniable that current interfacial reconstruction methods are still in the infancy stage of development, the structural hierarchy of the interface makes tissue engineering a great challenge. 70
Conclusions
Using the EPD method, a GO-HA composite coating with excellent physical and chemical properties was successfully added onto the surface of Ti implants. Both in vivo and in vitro tests verified that the HA-2wt%GO coating could induce the M2 polarization of macrophages, exert immunoregulatory effects, and promote the osteogenic differentiation of BMSCs, creating an immune microenvironment favoring bone repair. These findings lay a solid theoretical foundation for the clinical application of HA and GO composite coatings.
Footnotes
Acknowledgements
We would like to thank the Scientific and Technological Assistance Programs of Xinjiang province for their assistance and guidance in this research.
Contributorship
Wufanbieke Baheti and Huiyu He searched the literature and conceived the study. Xiaotao Chen, Mila were involved in protocol development, obtaining ethical approval, patient recruitment, and data analysis. Wufanbieke Baheti wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Department of Xinjiang Uygur Autonomous Region [Grant Number 2023D01C78] and National Natural Science Foundation of China [Grant Number 82260195].
Guarantor
Huiyu He
