Abstract
Background
In this study, we fabricated prototype bicomponent polyester/silk fibroin small-diameter arterial prostheses using a specially designed narrow ribbon shuttle loom.
Methods
The 2-layered flat fabrics were then heatset on a mandrel to form tubes with a round cross section.
Results
The woven samples had a wall thickness between 0.23 mm and 0.29 mm and an inner diameter between 3.53 mm and 3.95 mm, depending on the yarn type and the weave structure.
Conclusions
The bicomponent polyester/silk fibroin samples had superior bursting strength, circumferential strength and suture retention strength compared with a commercial small-diameter arterial prosthesis made from ePTFE. In addition, these prototype samples had greater suture retention strengths than a dog femoral artery, which indicates that they have adequate biostability for clinical use. While their amount of radial compliance was superior to that of the ePTFE commercial graft control, it did not match that of a natural artery. So there is still a need for future improvement in compliance. All of the woven prototypes had water permeability values between 26 and 180 ml/(cm2*min), which confirms that none of these arterial prostheses needs to be preclotted at the time of implantation. The biocompatibility of the woven prototypes was evaluated using porcine endothelial cells and an MTT assay. Their cytocompatibility was found to be superior to the ePTFE commercial control, and the level of cell attachment was observed to increase on these prototypes woven with a higher silk fibroin content.
Introduction
At present, an increasing number of people are affected by cardiovascular disease. It is reported that in 2003 about 1 million people died from cardiovascular disease, and nearly 84 million people were affected by this disease, with another 13.2 million people suffering from coronary artery disease (1). Transplantation is an effective way to treat this condition, but it requires autologous vessels to avoid immunological complications and rejection. Because of the limited availability of healthy autologous vessels from patients suffering from cardiovascular disease, and because of the small number of donor vessels, there is an immediate need for the development of a small-diameter arterial prosthesis. There are several different ways to fabricate a small-caliber prosthesis, such as electrospinning. Electrospun grafts have some degree of porosity through the thickness of the wall, but the structure is not uniform or easily controlled, and depends on the conditions used in the electrospinning process (2). Cell-based tissue engineered-grafts have superior biocompatibility, yet their strength is inferior to that of native arteries (3). Among the textile technologies, weaving is an efficient method to fabricate arterial prostheses that have both a uniform wall structure and excellent mechanical properties (4, 5).
An arterial prosthesis should have both good biocompatibility and superior mechanical properties. It is evident that silk fibroin has good biocompatibility, it has been widely used as a biocompatible material in the application of anterior cruciate ligament (6), as a template for hydroxyapatite crystal growth (7) and for skin scaffolds (8). Furthermore, the small-diameter vascular grafts made from fibroin have been found to have significantly higher patency rates than the expanded polytetrafluoroethylene (ePTFE) grafts after 1 year of implantation in male Sprague-Dawley rats (9). However, when used as woven material, sericin, which is contained in silk yarns, is not biocompatible and is known to contribute to a severe inflammatory response. Consequently, a degumming process is required after weaving to remove the sericin from the silk yarns. In addition, polyethylene terephthalate (PET) was widely used in the field of large diameter (>6 mm) arterial prostheses (10, 11). It was evident that Dacron was more effective in binding antibiotics than PTFE when applied in aortic grafts (12).
So by selecting both silk yarns and polyester yarns to weave a small-diameter bicomponent arterial prosthesis, it was hypothesized that an arterial prosthesis with both superior mechanical properties and excellent biocompatibility would be obtained. To avoid yarn sizing (the process of coating and protecting the yarns from abrasion during the weaving process), the warp yarns were braided to improve their abrasion resistance. In addition the surface coating of sericin was left on the silk yarns to protect them and avoid breakage during weaving.
Much research has been reported investigating the mechanical properties of homogenous arterial prostheses (13, 14). However, there is a lack of information about the mechanical properties of the bicomponent polyester/silk fibroin arterial grafts. The purpose of this study was to weave bicomponent small-diameter arterial prostheses from silk warp yarns and polyester weft yarns so as to fabricate a uniform wall structure and a controlled inner diameter. The mechanical properties of the different woven structures were investigated and compared with a natural artery and a commercial ePTFE prosthesis. Furthermore, the relationship between the silk fibroin content and the cytocompatibility was also studied.
Materials and methods
Materials
Biomedical grade multifilament yarns were selected for this study. Polyester weft yarns and silk warp yarns were obtained from Suzhou Suture Needle Company, Jiangsu, and Soho International Silk Co. Ltd., Jiangsu, respectively. To prepare a warp yarn with appropriate size and strength without the need for applying size, the original silk yarns were braided. Table I shows the yarns used in the study.
Mechanical properties of the yarns
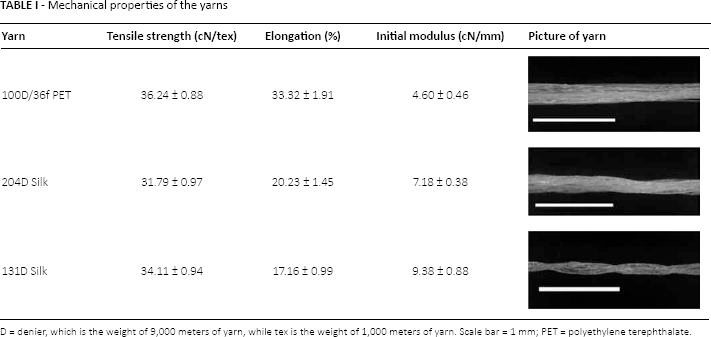
D = denier, which is the weight of 9,000 meters of yarn, while tex is the weight of 1,000 meters of yarn. Scale bar = 1 mm
PET = polyethylene terephthalate.
The 204 denier (204D) and 131D silk yarns were braided from 3 ends of 63D and 42D silk yarn, respectively. Some of the twist in the original yarn was lost during the braiding process, which caused the 204D silk yarn to be slightly weaker than the 131D yarn.
Fabrication, degumming and heat setting
Six small-diameter vascular prostheses were woven on a specially designed prototyping narrow ribbon shuttle loom at Donghua University, Shanghai, China. Details of the warp and weft yarns as well as the weaving parameters are listed in Table II. Seamless samples measuring from 1.0- to 80.0-cm long were woven on the loom as a double cloth with the layers joined by the weft yarns at the 2 selvages.
Weaving parameters of the samples
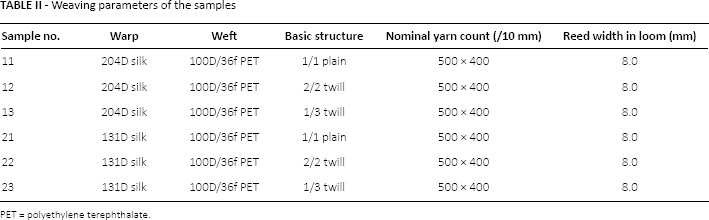
PET = polyethylene terephthalate.
From Table II we can see that samples 11, 12 and 13 contained the same yarns, but had different woven structures – namely, a 1/1 plain, 2/2 twill and 1/3 twill weave, respectively. Likewise samples 21, 22 and 23 contained the same yarns, but had the same 3 woven structures as described above. The difference between these 2 series was that samples 11, 12 and 13 contained coarser braided silk yarns in the warp direction than in the 20 series (see Tab. II).
After weaving, the 6 double cloth samples were degummed to remove the surface sericin by soaking in a sodium carbonate solution (0.05 wt%) overnight and then heating in the same solution at 90°C for 90 minutes (15).
After degumming, the woven bicomponent polyester/silk fibroin samples were heat set by mounting the double cloth samples on stainless steel mandrels and placing them in a hot-air oven for 10 minutes at a temperature of 120°C.
Dimensional measurements
After conditioning in a standard atmosphere for more than 24 hours, the thickness and flat width of the degummed prostheses were measured. The outer diameter was calculated as follows:

where OD (mm) is the outer diameter, FW (mm) is the flat width.
The thickness of the prototype structures was measured using a thickness gauge (Model: CH-10-A/AT), according to Standard Method ISO 7198: 1998 (16). The inner diameter was calculated from the known outer diameter and the wall thickness using Equation 2:

where ID (mm) is the inner diameter, OD (mm) is the outer diameter and T (mm) is the thickness. Figure 1 shows a schematic of the dimensions of the prosthesis.
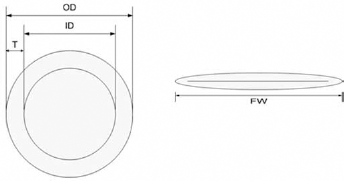
Schematic of the dimensions of the prosthesis. FW = flat width (mm); ID = inner diameter (mm); OD = outer diameter (mm); T = thickness (mm).
Circumferential strength
Twenty-centimeter long specimens were measured for circumferential strength on a universal mechanical tester (Model: YG-B 026H) following the standard method described in ISO 7198: 1998 (16). Each sample was measured in triplicate. The breaking strength was recorded, and the circumferential strength was calculated using Equation 3:

where F (N/mm) is the circumferential strength, T (N) is the breaking force and L (mm) is the length of the specimen (20 mm).
Probe bursting strength
To measure the probe bursting strength, the double cloth samples were cut open, and 1 × 1 cm2 square specimens were prepared. A probe measuring 1.5 mm in diameter was forced through the mounted specimen at a speed of 50 mm/min. The maximum breaking force was recorded, and the probe bursting strength (17) was calculated as follows:

where F (N/mm2) is the probe bursting strength, T (N) is the maximum breaking force and D (mm) is the diameter of the probe (1.5 mm).
Suture retention strength
The suture retention strength was measured on a universal mechanical tester (Model: YG-B 026H) following the standard method described in ISO 7198: 1998 (16). The double cloth specimen measuring 20 mm in length was clamped in the bottom jaws and opened at the top with a metal mandrel. We performed the suture retention test using several sizes of sutures: to make sure the suture would not break down before the samples were broken, a 2-0 braided polyester suture (Jinhuan Medical Supplies Co. Ltd, Shanghai, China) was selected. The suture was passed through the woven fabric exactly 2 mm from the cut end. Then the suture was pulled out at a speed of 50 mm/min until the specimen failed. The maximum force when the suture pulled out of the fabric was recorded as the suture retention strength. The test was repeated 3 times for each sample. Dog femoral artery (14) and the Gore-Tex standard wall (18) commercial graft were used as controls.
Radial compliance
A Test Bench Biodynamic mechanical simulation system (Bose Corporation) was used to measure the radial compliance of the samples. A sinusoidal wave form at a 1-Hz frequency was selected to measure the radial compliance over the following 3 pressure ranges: 50-90 mm Hg, 80-120 mm Hg and 110-150 mm Hg, in accordance with the ISO 7198: 1998 (16) standard.
The radial compliance of the samples was calculated using Equation 5:
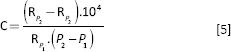
where C (%/100 mm Hg) is the radial compliance, P1 (mm Hg) and P2 (mm Hg) are the lower and higher pressures, and RP1 (mm) and RP2(mm) are the radii corresponding to pressures P1 and P2, respectively. The results were compared with the radial compliance of pig carotid artery and the ePTFE commercial graft (19).
Water permeability
The water permeability of the samples was measured on a water permeability tester for vascular prostheses as described in the standard method in ISO 7198: 1998 (9). The specimens were exposed to distilled water for 10 minutes at a pressure of 120 mm Hg, and the amount of water was collected after each minute for the 10 minute duration of the test. The average water permeability was calculated as follows:

where W [ml/(cm2*min)] is the water permeability, Q (mL) is the volume of water collected, A (cm2) is the exposed area of the specimen and t (minutes) is the duration of the test.

where D (cm) is the diameter, and L (cm) is the length of the specimen.
Evaluation of cytocompatibility
In addition to the double cloth samples, a series of 3-cm wide flat single-layer fabrics were woven using the same materials and weaving designs. Porcine iliac endothelial cells (PIECs; No. GN 015; Cell Bank of the Chinese Academy of Sciences, Shanghai, China) (20) were seeded at a concentration of 1.4 × 104 cells per well on 14-mm diameter fabric specimens as well as on an ePTFE commercial sample in 24-well tissue culture plates (TCPs). They were incubated at 37°C for 24, 72, 120 and 168 hours. Cells were also seeded on empty TCPs without fabrics, which served as controls. Every sample was cultured in triplicated. After 24 hours and 3, 5 and 7 days of incubation, cell viability was checked using an MTT assay, in which the fabrics and cells were incubated with 100 μL of 5 mg/mL 3-(4, 5)-dimethylthiahiazo(-z-y1)-3,5-diphenytetrazoliumromide (MTT) and 300 μL medium (per well) for 4 hours. The medium was then extracted, and 100 μL of dimethyl sulfoxide (DMSO) was added, followed by dissolution of the crystals with 15 minutes of shaking in 37°C. Then 100 μL of the solution was pipetted into the wells, and the absorbance was measured at 492 nm using a spectrophotometer (MK3; Thermo Fisher Scientific, Waltham, MA, USA). Cell viability was determined by expressing the absorbance of the experimental wells as a percentage of that of the control wells (21).
Confocal microscopy was used to observe the migration of cells through the fabrics. The samples cultured for 3 days were first washed twice with phosphate-buffered saline (PBS), and then 4% paraformaldehyde was added to the wells and cooled at 4°C for 20 minutes. After washing twice again with PBS, the specimens were stained with rhodamine phalloidin (5 μg/mL; Cytoskeleton Inc., USA) at room temperature for 30 minutes. After rinsing twice with PBS, the specimens were stained with DAPI (0.1 mg/mL; SIGMA, USA) for 10 minutes at room temperature in the dark.
Statistics analysis
Statistics analysis was performed using Origin 8.0 software (Origin Lab Inc., USA). The experimental values were averaged and expressed as means ± standard deviation (SD). Statistical differences were determined by a 1-way analysis of variance (ANOVA), and the means were considered significantly different at a p value ≤0.05.
Results and discussion
Prosthesis dimensions
The samples can be woven in different lengths. They were double-layered cloth with round cross section (See Fig. 2).

Photo of woven sample (sample 12).
The wall thickness of the bicomponent polyester/silk fibroin prostheses was measured after degumming. The results are shown in Figure 3. From Figure 3 we can see that sample 11 had the thickest wall (0.293 mm), while sample 21 had the thinnest wall (0.228 mm). Figure 3 shows that the samples with the same woven structure but different warp yarns, such as samples 11 and 21, samples 12 and 22 and samples 13 and 23, had different wall thicknesses. This was likely due to the 204D silk warp yarn being thicker than the 131D yarn. In addition, the wall thickness of the different samples appeared to be influenced by the type of woven structure.
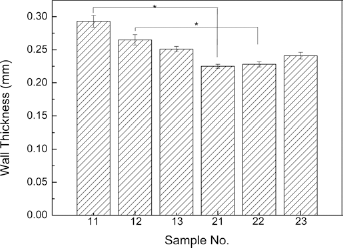
Wall thickness of the bicomponent polyester/silk fibroin small-diameter arterial prostheses after degumming. *p≤0.05.
The inner diameters of the degummed bicomponent polyester/silk fibroin arterial prostheses were calculated using Equation 2, and the average results are presented in Figure 4. From Figure 4 we can see that there were no significant differences in inner diameter between the samples. The reason for this was that all 6 samples were woven from the same size and type of weft yarns. After finishing, the shrinkage of the same weft yarns was the same, which resulted in the similar inner diameter of different samples.
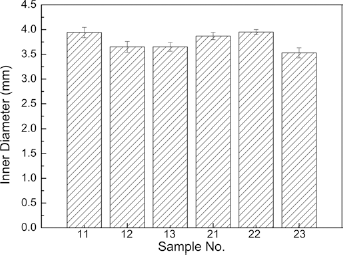
Inner diameters of the bicomponent polyester/silk fibroin small diameter arterial prostheses after degumming.
Probe bursting strength
Figure 5 shows the probe bursting strength of the bicomponent polyester/silk fibroin samples as well as that of the Gore-Tex ePTFE standard wall (SW, inner diameter = 6 mm) control sample (19). The figure shows that the probe bursting strength of samples 11, 12, 13 and 21 was significantly greater than that of the commercial control (p≤0.05), while samples 22 and 23 were equivalent to the commercial control. Sample 21, with a 131D silk warp yarn, a 100D polyester weft yarn and a 1/1 plain weave had the highest bursting strength value, which was almost twice the strength of the ePTFE control. The results in Figure 5 confirm that the bursting strength of a woven sample is influenced not only by the woven structure, but also by the combined warp and weft yarns.
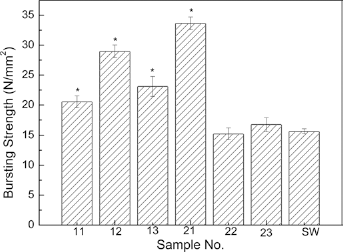
Comparison of probe bursting strength of the bicomponent polyester/silk fibroin arterial prostheses and Gore-Tex ePTFE standard wall (SW) control. *p≤0.05.
Circumferential strength
As described in the section “Circumferential Strength” above, each sample was measured in triplicate. Figure 6 gives the representative strength curves of the samples. From the figures we can see that the elongation of the samples ranged from 91.47% to 148.77%. Figure 7 shows the results of the circumferential tests on the 6 prototype samples as well as the Gore-Tex ePTFE standard wall (19) control sample. From Figure 7 we can see that all of the woven samples were significantly superior in circumferential strength compared with the ePTFE small-diameter commercial graft (p≤0.05). Moreover, the circumferential strength of all of the woven samples was more than 10 times greater compared with that of the Gore-Tex ePTFE standard wall control.
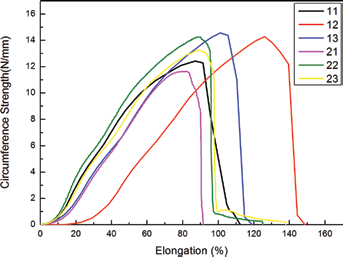
Representative circumferential strength curves of the bicomponent polyester/silk fibroin arterial prostheses.
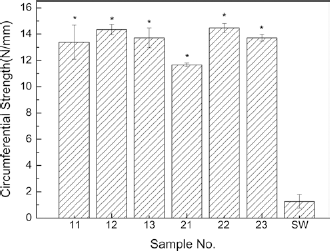
Comparison of circumferential strength of the bicomponent polyester/silk fibroin arterial prostheses and Gore-Tex ePTFE standard wall (SW) control. *p≤0.05.
Among the 6 woven bicomponent polyester/silk fibroin samples, sample 21 with its 1/1 plain weave, had the lowest circumferential strength. Apart from sample 21, there were no significant differences between the woven samples. This is most likely due to the fact that all of the woven samples contained the same 100D polyester weft yarns.
Suture retention strength
Figure 8 shows that the highest value of suture retention strength was achieved by sample 12, with its 204D silk warp and 100D polyester weft yarns, and its 2/2 twill weave. In fact the samples woven in a 2/2 twill structure had a superior suture retention strength to the 1/1 plain or 1/3 twill weave samples. Furthermore, all of the woven bicomponent polyester/silk fibroin samples had a higher suture retention strength than either the commercial ePTFE small-diameter prosthesis and or the dog femoral artery. Furthermore, the suture retention strength of the woven samples amounted to more than 300% of that of the natural artery (dog femoral artery). This confirms that woven bicomponent polyester/silk fibroin prostheses have superior suture retention properties compared with those of natural artery.
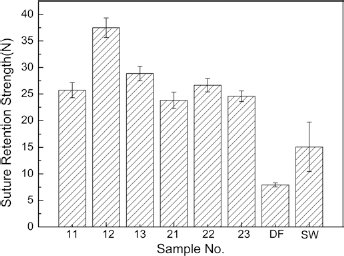
Comparison of the suture strength force of the bicomponent polyester/silk fibroin arterial prostheses, natural blood vessel (DF) and Gore-Tex ePTFE standard wall (SW) controls.
Radial compliance
Figure 9 shows that the radial compliance of the bicomponent polyester/silk fibroin samples when tested at low, medium and high blood pressure lay in a narrow range between that of the natural artery and the ePTFE commercial control (12). The reason the compliance values of the woven samples were similar is that all 6 samples contained the same 100D polyester weft yarn. The mechanical properties of weft yarn play an important role in the radial compliance, so having the same weft yarn led to the similar radial compliance of the woven samples.
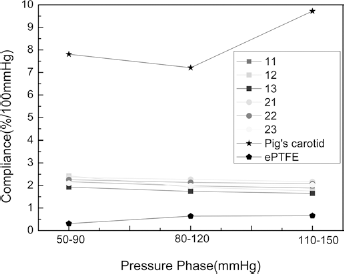
Radial compliance of the bicomponent polyester/silk fibroin woven arterial prostheses compared with pig carotid artery and expanded polytetrafluoroethylene (ePTFE) commercial prosthesis.
Water permeability
From Figure 10 we can see that all of the woven samples had a water permeability value under 180 ml/(cm2*min), which confirms that the woven prostheses can be implanted without preclotting. All of the measured water permeability values were above 0, which indicates that there were open pores in the wall of the samples which could allow nutrients and vapor to pass through. In addition, Figure 10 indicates that samples with the coarser 204D silk warp yarns had lower water permeability values than the samples with 131D warp yarns. This confirms that the size of the yarn can influence the water permeability of the woven samples. Among samples 11, 12 and 13, sample 11 with a 1/1 plain weave had the lowest average water permeability at 26 ml/(cm2*min). Again, among samples 21, 22 and 23, sample 21 had the lowest water permeability. This indicates that the 1/1 plain weave provides a more hemostatic structure than is possible with either a 2/2 twill or a 1/3 twill weave. Above all, we can conclude that the water permeability was affected both by the size of the yarns and the woven structure.
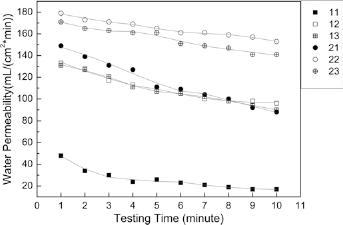
Water permeability of the prostheses during the 10-minute test.
Cytocompatibility
Figure 11 exhibits the viability of the cells seeded on the woven samples, the ePTFE commercial prosthesis and the tissue culture plates, after 24 hours (Fig. 11A), 72 hours (Fig. 11B), 120 hours (Fig. 11C) and 168 hours (Fig. 11D) of culture, as measured by the MTT assay. It is interesting to note that cell proliferation was superior on the woven fabrics and control surface to that on the ePTFE graft.
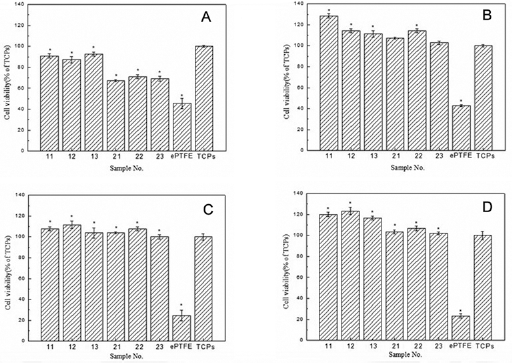
Viability values of porcine iliac endothelial cells (PIECs) seeded on the bicomponent polyester/silk fibroin fabrics and the expanded polytetrafluoroethylene (ePTFE) commercial graft after 24 hours
Comparing the level of cell viability in Figure 11, we can see that after 7 days (168 hours), the cell viability of the woven samples was greater than that of the tissue culture plates, while that of the ePTFE sample was reduced. From this phenomenon, we can conclude that the woven bicomponent polyester/silk fibroin arterial prostheses have better cell compatibility compared with the tissue culture plates and the ePTFE commercial prosthesis.
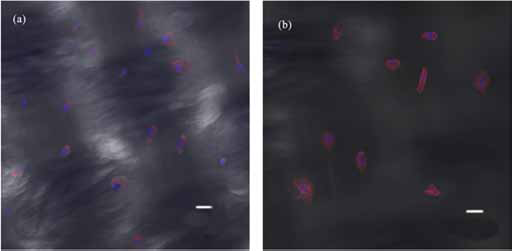
Figure 12 shows the confocal images of the cells attached to 2 of the bicomponent woven samples (samples 12 and 22) after 3 days of culture. Given that sample 12 exhibits a higher cell density than sample 22, we can conclude that a higher silk fibroin content leads to greater cell attachment to the wall of the bicomponent samples.
Conclusion
In this study, bicomponent small-diameter (≤4 mm) arterial prostheses were fabricated from braided silk yarns and polyester multifilament yarns. Seamless double cloth samples were woven in a 1/1 plain, 2/2 twill and 1/3 twill weave on a special narrow ribbon shuttle loom with an 8-mm reed width. After degumming to remove the sericin from the silk warp yarns, the samples were found to have a wall thickness between 0.193 and 0.228 mm. Their inner diameter measured between 3.53 and 3.95 mm.
The mechanical properties of the woven and degummed prostheses were evaluated by measuring the probe bursting strength, circumferential strength and suture retention strength, and comparing the results with the reported Gore-Tex ePTFE standard wall prosthesis and dog femoral arterial as controls. Sample 21 was found to have the highest value of probe bursting strength, and all the woven samples had an equal or higher value of bursting strength compared with the ePTFE commercial control. All of the woven prostheses had significantly higher circumferential strength and suture retention strength compared with the ePTFE commercial control. Of particular interest was the fact that all of the woven prototype prostheses had a higher suture retention strength than the dog femoral artery and a greater radial compliance than the ePTFE commercial control. This indicates that the suture retention strength of the woven samples was superior to that of the native artery, and the radial compliance of the woven samples was better than that of the ePTFE commercial samples. However, the radial compliance of the woven samples was lower than that of the native artery (pig carotid artery) which implied that efforts are still needed to improve the radial compliance of the woven samples. The water permeability of all 6 prototypes was less than 180 ml/(cm2*min) which demonstrates that they can be implanted without the need for preclotting. This indicates that there are open pores in the wall of the woven samples which may allow the penetration of the cells after implantation. Results from the MTT cell viability assay and from confocal microscopy indicated that by increasing the silk fibroin content, the bicomponent prostheses showed improved cytocompatibility with more cell attachment.
Footnotes
Financial support: This work was funded by National Natural Science Foundation of China (NSFC) (No. 51003014, No. 31100682), Doctoral Fund of Ministry of Education of China (RFDP) (No. 20100075110001), Programme of Introducing Talents of Discipline to Universities (111 project) (B07024) and the Fundamental Research Funds of Central Universities (No. 12D10147).
Conflict of interest: The authors do not have any conflict of interest to declare.
