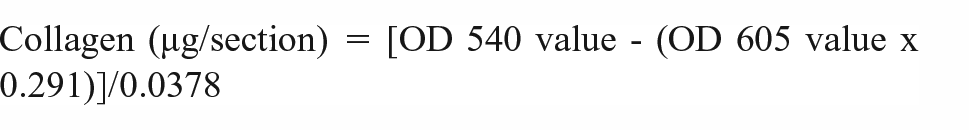Abstract
There are scarce published data suggesting, that collagen extracted from fish skin may be an attractive alternative to mammalian-derived collagen for the in vitro cell cultures. In this study, we investigated proliferation potential and differentiation capability into osteogenic and adipogenic lineages of rat adipose-derived mesenchymal stem cells (rASCs) and human adipose-derived mesenchymal stem cells (hASCs) cultured on collagen extracted from silver carp and African sharptooth catfish skins, compared with commercially available mammalian collagen and collagen-free culture dishes. Our results revealed no significant differences between fish collagen and mammalian collagen in supporting cell viability and proliferation capacity. Fish-derived collagen is a cheap material derived from production waste, does not contain transmissible pathogens of mammalian origin, supports human cell cultures at comparable level to conventional collagen sources, and may be considered as the product of choice for the in vitro cell cultures.
Keywords
Introduction
Collagen ranks as one of the most widely studied naturally occurring polymers and as a major component of the extracellular matrix of connective tissues such as bones, cartilage, ligaments and skin. 1 As the most ubiquitous protein in the mammalian body, collagen plays a key role in the formation of organs and tissues and is considered to be an appropriate scaffold for maintaining cell culture proliferation. Other aspects of cell behavior affected by collagen are cell adhesion, differentiation, proliferation, and cellular migration, so it makes it an ideal material for cell culture research.2–6
Commercially available mammalian collagen is generally derived from rat tail, and bovine or porcine sources. However, safety concerns aroused due to the risk of animal transmissible diseases, such as bovine spongiform encephalopathy or foot-and-mouth disease, and ethical implications in a social setting.7,8 This indicates the search for safer alternatives, that have been found in marine sources. In the fishing industry, large amounts of fish material are discarded, and with the increasing consumption, more fish waste is being produced each year. Discarded parts are a rich source of collagen. 9 Marine collagen, in terms of amino acid composition and biocompatibility, has been proven to be similar to conventional porcine and bovine collagens. Additionally, marine sources are attractive, because collagen can be easily obtained in high yield and at low costs. 10
As mentioned above, collagen extracted from various fish by-products is commonly used not only in industry, but also in research. Fish collagen have good bioactive properties, like high biodegradability, good biocompatibility, and low immunogenicity.11,12 According to the latest research data, collagen is able to mimic the components of the stem cell niche, promoting stem cell self-renewal and controlled differentiation ex vivo.13,14 Additionally, fish collagen has potential to promote endothelial and osteogenic differentiation of the mesenchymal stem cells (MSCs), but on the other hand data shows, that marine collagen inhibits the expression of adipogenic and chondrogenic markers.5,10,15 Moreover, fish collagen-based scaffolds with MSCs in the injured site seem to improve the process of wound healing in skin regeneration. MSCs are also found to proliferate better on fish collagen scaffolds versus on regular collagen, and research results indicated that MSCs-loading fish collagen scaffolds are promising for the in vivo experiments. 16 We hypothesized that these properties of fish collagen would be highly advantageous for proliferation of adipose-derived mesenchymal stem cells (ASCs) and differentiation into osteogenic and adipogenic lineages.
In our study, we used collagen extracted from skins of African sharptooth catfish and silver carp. We evaluated the suitability of fish collagen for the regenerative potential of adipose-derived mesenchymal stem cell (ASC) cultures. In addition, we used two different donors of adipose stem cells, a human and a rat. Zolocinska et al., 17 in their study, indicated rat as an optimal research model, among mouse and rabbit, for cell therapy experiments with ASCs. Different concentrations of fish collagen (0.01% (w/v), 0.002% (w/v), 0.001% (w/v)) were also applied, to find an appropriate one for the cell culture. We investigated growth rate, clonogenic potential, and differentiation capability into osteogenic and adipogenic lineages of rat adipose-derived mesenchymal stem cells (rASCs) and human adipose-derived mesenchymal stem cells (hASCs), cultured on collagen extracted from skins of both fish species, compared with commercially available collagen-coated surfaces and non-coated dishes. The findings contribute to a growing body of evidence, that marine collagen is a novel biomaterial suitable not only for in vitro applications, but also as a scaffolding material for cell cultures.
Materials and methods
Raw materials
Silver carp and African sharptooth catfish skins were processed by Inventia Technologies Sp. z o.o. (Zulawka, Poland) for use in this study. Fish materials were obtained from qualified suppliers, and the raw material were stored at −18 °C. After thawing, the fish material was processed by repeated washing with water to discard adherent tissues. Then it was washed repeatedly with chilled distilled water for 10 min. Purified skins (0.3 kg) were extracted with 4 dm3 of 0.5% lactic acid for 18 h. The obtained extract was passed through a series of polyester filters with decreasing pore size. After filtration, an odorless and transparent gel formulation was obtained. Within 24 h of the filtration step, collagen was lyophilized. Afterward, the material was fragmented, placed in polyethylene bags, and was stored at −25 °C until used.
Preparation of collagen
Freeze-dried collagen was dissolved in acetic acid (0.1 mol L−1) by the way of continuous stirring for 5 h at room temperature (RT). Collagen stock solution was prepared by adding chloroform to the mixture in proportion 1:9, the resulting solution was stored overnight at +4 °C. The working solutions of collagen were prepared by final dilution with an adequate volume of sterile water, to obtain 0.01% (w/v), 0.002% (w/v) and 0.001% (w/v) concentrations.
Preparation of collagen-coated culture plates
For dish coating, 0.01% (w/v), 0.002% (w/v), or 0.001% (w/v) collagen solutions from silver carp or African sharptooth catfish were added into 6-well culture plates (Falcon®, USA) and kept overnight below 4 °C. The collagen solutions were removed from the dishes, and they were air-dried overnight at room temperature (RT). Commercially purchased Rat Tail collagen I-coated plates (Life Technologies™, USA) were used as a positive control, and uncoated culture plates were used as negative control.
Sirius Red/Fast Green collagen staining
Collagen fibers on plastic surfaces were detected using Sirius Red/Fast Green Collagen Staining Kit (Chondrex, Inc, USA), as described in the manual of the staining kit. Sirius Red binds to the fibrillary collagens type I to V, whereas Fast Green binds to non-collagenous proteins. The OD values of the extracted dyes were read with a spectrophotometer at 540 and 605 nm and used to determine amount of collagen according to the following formula:
The OD 540 value was corrected by deducting the contribution of Fast Green at 540 nm, which is 29.1% of the OD 605 value. Color equivalency for collagen is 0.0378 at OD 540 and 605 (OD values/µg protein). Additionally, collagen-coated surfaces stained with Sirius Red/Fast Green were observed using an optical microscope.
Isolation of ASCs from human adipose tissue
Human adipose tissue was harvested from superficial abdominal regions of healthy donors by liposuction. To remove red blood cells, the adipose tissue was mixed in a 2:1 (v/v) ratio with phosphate-buffered saline (PBS) and was shaken up at 15-minute intervals. Following phase separation, PBS containing red blood cells was discarded. The purification process was repeated three times. Afterward, 0.075% (w/v) collagenase solution from Clostridium histolyticum (Sigma-Aldrich, USA) in PBS was added to adipose tissue (1:2), shaken, and incubated at 37 °C for 1.5 h, to digest the tissue. The fat and collagenase mixture were shaken every 15 min. After obtaining a homogenous suspension, human albumin with a final concentration of 2% (v/v) was added to stop the digestion process. The mixture was centrifuged at 400g for 10 min, at room temperature (RT). The liquid fat and salt interphases were discarded and the cell pellet was suspended in PBS. The cell suspension was filtered through a 100 µm nylon mesh, washed in PBS, and centrifuged (400g) for 10 min, RT. The amount and viability of the cells were determined, and cells were seeded into plastic flasks at a density of 8 × 104 cells/cm2 in DMEM medium (Sigma-Aldrich, USA) supplemented with 10% of fetal bovine serum (FBS; Life Technologies™, USA) and 1% Penicillin-Streptomycin (Pen-Strep; Gibco™, USA) for further experiments.
Isolation of adipose stem cells from human adipose tissue was approved by Bioethics Committee at the Maria Sklodowska-Curie National Research Institute of Oncology in Warsaw (Approval number: 33/2021).
Isolation of ASCs from rat adipose tissue
Eighteen male Wistar Albino Glaxo inbred rats aged 3–4 months and weighing 275 g to 312 g were used throughout experiment. After euthanasia of animals (isoflurane over-dose), white adipose tissue was collected from inguinal, perigonadal, and perirenal regions using method described in our previous study. 18 After mechanical fragmentation, rat adipose tissue was digested with 0.075% (w/v) collagenase solution from Clostridium histolyticum in PBS at 37 °C for 30–40 min. The mixture of fat and collagenase solution was shaken every 15 min. Following the incubation period, fetal bovine serum (FBS) was added, and the cell suspension was filtered through a 100 µm nylon mesh, washed in PBS, and centrifuged (400g) for 10 min, RT. After discarding the supernatant, the cell suspension was washed 2× in PBS. Cells number and viability were determined, and cells were seeded into plastic flasks at a density of 8x104 cells/cm2 in DMEM medium supplemented with 10% FBS and 1% Pen-Strep for further experiments.
All animal experiments were approved by the II Local Bioethical Committee in Warsaw, Poland (approval number: 31/2011), and were performed according to the Guidelines for the Regulation of Animal Experiments.
Cell culture
Human (hASCs) or rat (rASCs) adipose derived stem cells were cultured in DMEM (Dulbecco′s Modified Eagle′s Medium) with 10% FBS (Gibco) and 1% Pen-Strep (Gibco), at 37 °C with 5% CO2 and 95% humidity. When primary cultures reached sub-confluency, cells were detached with 0.25% trypsin-EDTA (Gibco™, USA) and re-plated at a density of 5 × 103 cells/ml for subsequent passage. All experiments were performed on hASCs and rASCs from the 3 to 5 passages. For further experiments, cells were seeded on prepared collagen-coated 6-well plates (Figure 1).

Morphology of ASCs from rat and human donors cultured on collagen from the skin of African sharptooth catfish. Magnification ×10. hASCs – human adipose-derived mesenchymal stem cells; rASCs – rat adipose-derived mesenchymal stem cells.
Clonogenic potential
The clonogenic potential of hASCs and rASCs was determined using the CFU-F test. Cells were seeded with a density of 260 cells per well and cultured for 8 days in DMEM supplemented by 20% FBS and 1% Pen-Strep, in standard conditions (37 °C, 5% CO2, 95% humidity). Afterward, cells were fixed by May Grunwald/Giemza staining, and colonies of over 50 cells were counted as 1 colony forming unit under an inverted microscope.
Growth rate and doubling time
ASCs from human and rat donors were seeded into 6-well plates, 1 × 104 cells per well. Daily cell counts were performed under Nikon Ti microscope (recording mode) until reaching the monolayer. Growth rate and doubling time were calculated using the free program Doubling Time Online Calculator (https://www.doublingtime.com/compute_more.php).
In vitro osteogenic differentiation
hASCs and rASCs were seeded in prepared collagen-coated dishes from silver carp or African sharptooth catfish, uncoated plate (negative control), and commercially purchased collagen-coated plate (positive control), at the density of 28.8 × 103 cells per well. hASC and rASC were stimulated into osteogenic differentiation using commercially available Human Mesenchymal Stem Cell (hMSC) Osteogenic Differentiation Medium BulletKit™ (LONZA, Switzerland). Cells were cultured in hMSC medium for 3 weeks, with the medium changed every 3 days. After 3 weeks, cells were fixed for 30 min with 10% (v/v) formaldehyde (Sigma-Aldrich, USA) and stained with Alizarin Red S (Sigma-Aldrich, USA). Osteogenic differentiation was evaluated under a standard light microscope.
In vitro adipogenic differentiation
ASCs from human and rat donors were seeded at a density of 100 × 103 cells per well, into prepared collagen-coated dishes from silver carp or African sharptooth catfish, uncoated plate, and commercially purchased rat tail collagen-coated plate. Adipogenic differentiation was stimulated by using Human Mesenchymal Stem Cell (hMSC) Adipogenic Differentiation Medium BulletKit™ (LONZA, Switzerland). When plated cells reached 98%–100% confluence, three cycles of induction were performed. Each cycle consists of feeding cells with an adipogenesis induction medium for 3 days (37 °C, 5% CO2, 95% humidity), followed by 2 days of culture in an adipogenic maintenance medium. Then, cells were cultured in an adipogenic maintenance medium for 7 days. The production of lipid droplets was quantified by Oil Red O staining (Sigma-Aldrich, USA). Prior to staining, cells were fixed for 45 min with 10% (v/v) formaldehyde and then for 5 min with 60% (v/v) isopropanol (Sigma-Aldrich, USA). After fixation and staining, wells were rinsed with distilled water and visualized by standard light microscopy.
Statistical analysis
The data were analyzed using Statistica 13 PL computer program. All experiments were repeated three times, and statistical comparisons were performed using the Kruskal-Wallis one-way analysis of variance test. The data were considered statistically significant if p values <0.05.
Results
Sirius Red/Fast Green collagen staining for quantitative analysis of collagen from African sharptooth catfish and silver carp
The Sirius Red/Fast Green method was used to determine the content of extracted collagen from African sharptooth catfish (Figure 2(a)) and silver carp (Figure 2(b)) skins on 6-well culture plates, commercially purchased collagen-coated plates, and uncoated dishes. The results of the spectrophotometric evaluation of collagen content from both fish species, as well as commercially purchased collagen plate, are shown in Table 1. The amount of collagen is similar for 0.01% (w/v), 0.002% (w/v), and 0.001% (w/v) collagen concentrations, with a slight difference between the fish species – the content of collagen from silver carp was a little higher (⩾0.30 µg for 0.01% (w/v); ⩾1.18 µg for 0.002% (w/v), ⩾0.55 µg for 0.001% (w/v)). The amount of collagen from the commercially purchased plate was at the lowest (1.07 µg/per well), compared to dishes coated with both fish collagens. Results confirmed the positive coating of the 6-well culture plates with collagen from the skins of African sharptooth catfish and silver carp.

Microscopic evaluation of the coating effect with collagen from African sharptooth catfish (a) and silver carp (b) skins. Well plates coated with different concentrations of fish collagen and commercially purchased plate with mammalian collagen (positive control), stained with Sirius Red/Fast Green staining kit. Magnification ×10.
Spectrophotometric evaluation of collagen on 6-well plates.

Functional characteristics of rASCs and hASCs on collagen from African sharptooth catfish and silver carp
Clonogenic potential, measured by the CFU-F test was not influenced by the concentration of collagen, and the differences between the ASCs donors were statistically insignificant. (Figure 3(a) and (b)). However, the highest number of colonies for rASCs were observed on 0.002% (w/v) collagen concentration from African sharptooth catfish (3.67 ± 0.67) or 0.01% (w/v) collagen concentration from silver carp (4.67 ± 1.67), and for hASCs on 0.001% (w/v) collagen concentration from both type of fishes (5.33 ± 1.33 African sharptooth catfish; 4.67 ± 0.33 silver carp).
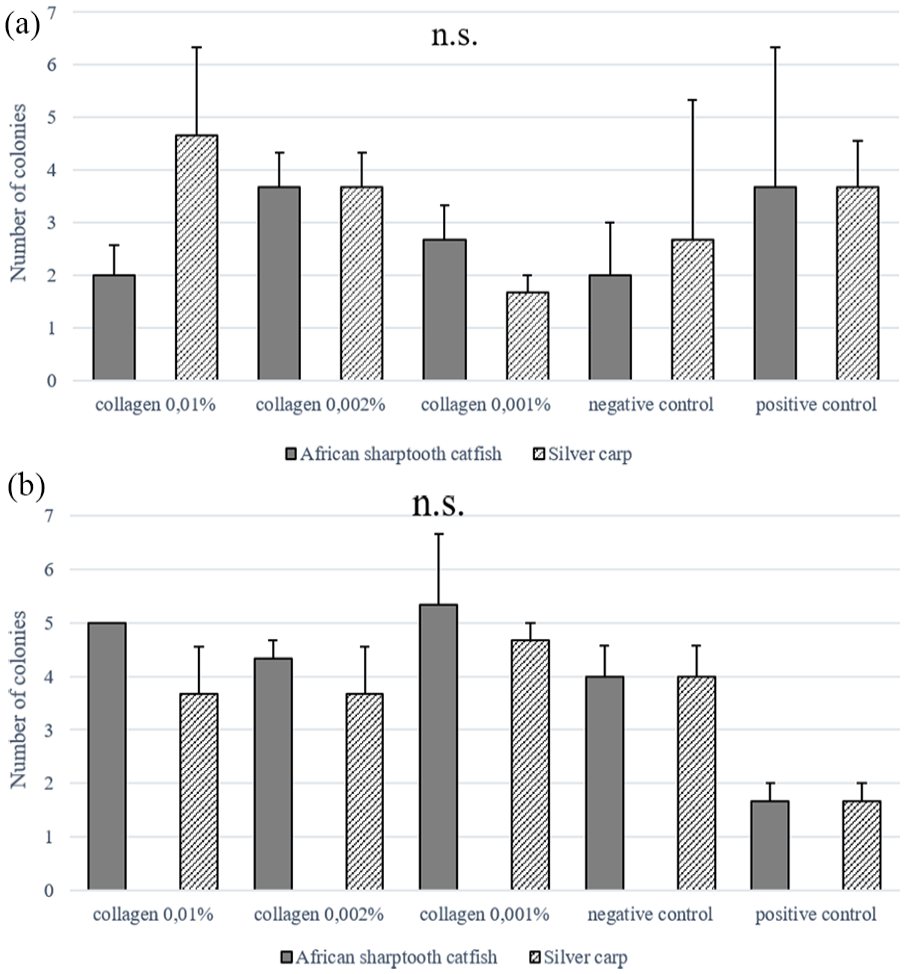
Clonogenic potential of rat adipose-derived mesenchymal stem cells (rASCs) in African sharptooth catfish and silver carp: (a) collagen-coated dishes, and human adipose-derived mesenchymal stem cells (hASCs) in African sharptooth catfish and silver carp and (b) collagen-coated dishes, measured by CFU-F test. Commercially purchased mammalian collagen-coated plate (positive control) and non-coated plate (negative control) were used as controls.
Analysis of the growth rate of rASCs on the collagen extracted from African sharptooth catfish (Figure 4(a)) and silver carp (Figure 4(b)) showed time-dependent changes in response to different collagen concentrations. In 7-day cell culture, the growth rate increased with culture time in all tested groups. At days 1–2 there were no differences among the fish collagen and the control groups, at the later time point the highest growth rate of rASCs on both fish collagens (on the seventh day) was observed for 0.002% (w/v) collagen concentration, followed by 0.01% (w/v) concentration or 0.001% (w/v) collagen concentration.
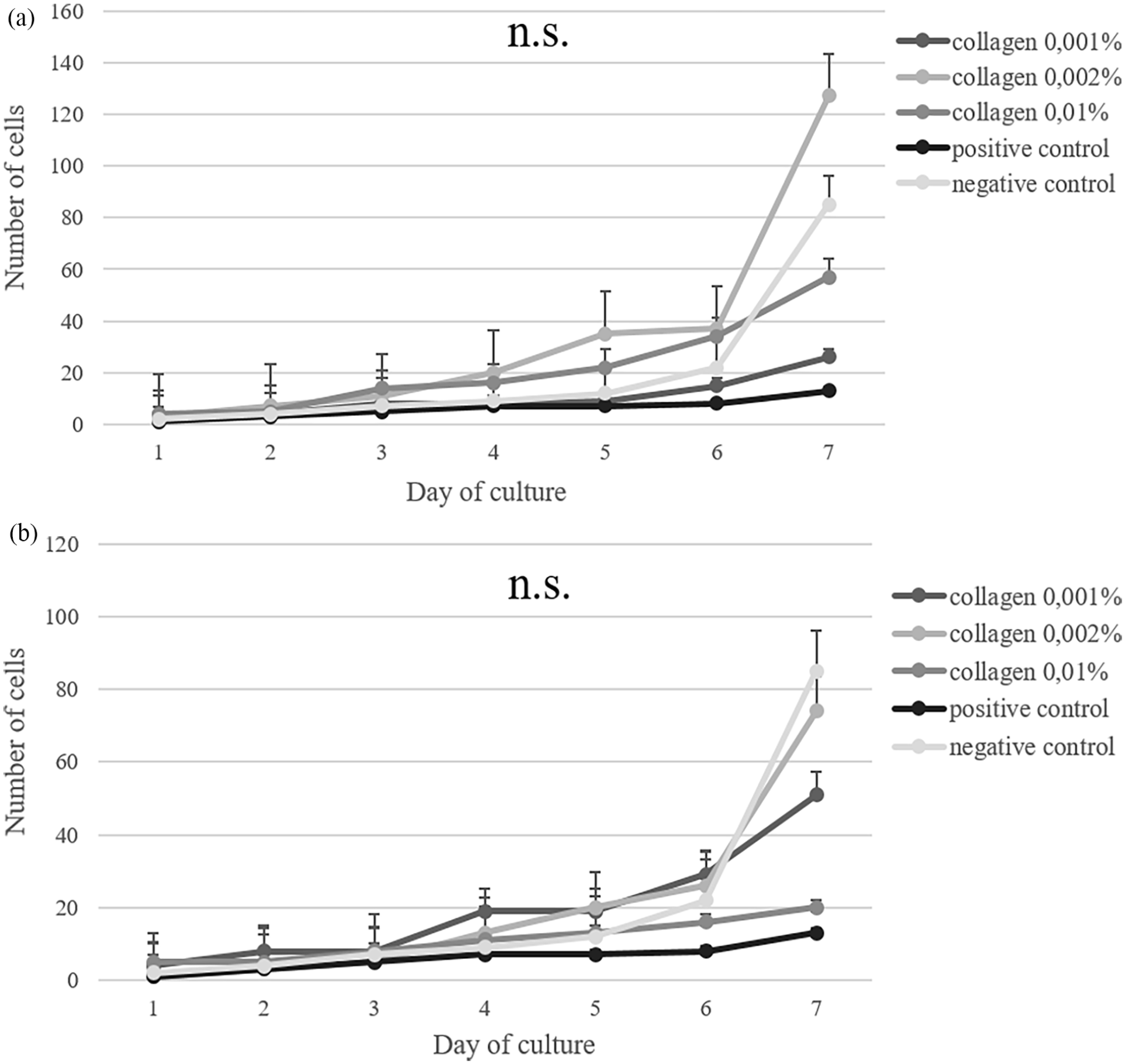
The growth rate of rat adipose-derived mesenchymal stem cells (rASCs) cultured on African shraptooth catfish (a) and silver carp (b) collagen. Commercially purchased plate with mammalian collagen as positive control, and uncoated plate as negative control.
Analysis of the doubling time of rASCs cultured on collagen from skins of African sharptooth catfish and silver carp is shown in Table 2. The lowest rate of doubling time of rASCs seeded on African sharptooth catfish collagen was reported for 0.002% (w/v) concentration (27.45 h), on silver carp collagen for 0.002% (w/v) concentration (25.99 h).
The doubling time of rASCs cultured on collagen from African sharptooth catfish and silver carp skins (Doubling Time Online Calculator).

Analysis of the growth rate of hASCs cultured on collagen from the skin of African sharptooth catfish (Figure 5(a)) and silver carp (Figure 5(b)) showed time-dependent differences corresponding to collagen concentrations. During 20-day cell cultures, the growth rate increased with culture time in all tested groups, on collagen from both fish species. However, the growth rate among all groups did not show any significant differences. The highest growth rate of hASCs was observed for 0.002% (w/v) collagen concentration from African sharptooth catfish and silver carp, followed by 0.01% (w/v) collagen concentration also from both fish species.

The growth rate of rat adipose-derived mesenchymal stem cells (hASCs) cultured on African sharptooth catfish (a) and silver carp (b) collagen. Accordingly, commercially purchased plate with mammalian collagen as positive control, and uncoated plate as negative control.
Analysis of the doubling time of human ASCs cultured on collagen from skins of African sharptooth catfish and silver carp is shown in Table 3. The lowest rate of doubling time was reported on 0.002% (w/v) African sharptooth catfish collagen concentration (86.8 h) and on 0.001% (w/v) silver carp collagen concentration (133.69 h).
The doubling time of hASCs cultured on collagen from African sharptooth catfish and silver carp skins (Doubling Time Online Calculator).

These results indicated that the ASCs from human and rat donors cultured on both types of fish collagens may provide similar growth compared to the commercially purchased mammalian collagen.
Differentiation potential of ASCs from rat and human donors
Osteogenic differentiation of ASCs from human and rat donors was evidenced by Alizarin Red S staining. Cells cultured on collagen from African sharptooth catfish and silver carp for 3 weeks in an osteo-inductive medium did not show any significant differences compared to their controls in terms of osteogenic potential (Figures 6 and 7).

Evaluation of the osteogenic differentiation of hASCs on collagen from African sharptooth catfish (a) and silver carp (b) skins. Magnification ×10.

Evaluation of the osteogenic differentiation of rASCs on collagen from African sharptooth catfish (a) and silver carp (b) skins. Magnification ×10.
Both hASCs and rASCs were able to differentiate toward the adipogenic lineage when cultured in an adipogenesis induction medium and an adipogenic maintenance medium. Results showed the presence of oil-red lipid vesicles inside the cells and loss of the fibroblastoid-like shape for more rotund, as evidenced by Oil Red O Staining. Adipogenic differentiation of hASCs and rASC on collagen from African sharptooth catfish and silver carp, did not differ significantly from controls (Figures 8 and 9). rASC adipogenic differentiation was best supported by both fish collagens.

Evaluation of the adipogenic differentiation of hASCs on collagen from African sharptooth catfish (a) and silver carp (b) skins. Magnification ×10.

Evaluation of the adipogenic differentiation of rASCs on collagen from African sharptooth catfish (a) and silver carp (b) skins. Magnification ×10.
ASCs isolated from both donors – a human and a rat, differentiated into osteogenic, as well as adipogenic lineages. The intensity of differentiation was confirmed by specific staining, and results were very similar not only between ASCs donors, but also for different fish collagen concentrations. However, mammalian collagen (commercially purchased) seemed to be less supportive in terms of adipogenic potential compared to fish collagen extracted from both fish species.
Discussion
Collagen is an essential component that provides structural support, regeneration, and tissue development. In recent years, fish collagen has drawn considerable attention among researchers, as an increasingly valuable biomaterial and an alternative to commonly used mammalian collagen. Fish collagen has higher bioavailability and more efficient absorption (up to 1.5 times) into the body over bovine and porcine collagens.19–21
In our study, we confirmed the suitability of fish collagen as a good biomaterial for adipose-derived mesenchymal stem cell (ASC) cultures. We examined the growth rate and clonogenic potential of human ASCs and rat ASCs on dish surfaces coated with African sharptooth catfish or silver carp collagens, and with rat tail collagen and uncoated dishes for comparison. In terms of clonogenic potential, there were not any significant differences in colony forming between the fish and mammalian collagen. The growth rate of both hASCs and rASCs increased with culture time, but with no significant differences among the tested groups. Additionally, the potential of hASCs and rASCs to differentiate into osteogenic and adipogenic lineages on fish collagens was positively assessed. Results confirmed osteogenic and adipogenic differentiation of ASCs from human and rat donors on collagen from African sharptooth catfish and silver carp. Adipogenic differentiation of ASCs seemed to be better supported on both fish collagens than osteogenic differentiation.
Collagen has a major structural feature in the architecture of bone and cartilage. For bone formation, an essential feature is osteogenic differentiation. Scaffolds formed by collagen are crucial for cell adhesion and anchoring of macromolecules.22,23 Recently, Zhang et al., 24 demonstrated the influence of collagen hydrolyzates from silver carp skin on combating osteoporosis in chronologically aged mice. Moreover, Guillerminet et al., 25 compared collagens from bovine, porcine, and fish origins in in vitro study, and their results similarly to ours did not show any significant differences in the growth rate of cells with collagens from different sources. At the same time, the activity of a well-known marker of osteogenesis – ALP, was increased in the primary culture of bone cells in the presence of collagen hydrolyzates from bovine, porcine, and fish sources. Similar results were reported by Liu et al., 26 collagen hydrolyzates from silver carp skin were effective in promoting osteogenic differentiation, with no significant effect on cell proliferation after treatment with different collagen concentrations. In our study, we used mesenchymal stem cells, precisely adipose-derived mesenchymal stem cells from human and rat donors to assess the biocompatibility of collagen. Adipose stem cells are multipotent cells, which can differentiate into various lineages such as adipogenic, osteogenic, and chondrogenic. 27 Several reports have shown that fish collagen promotes the multidirectional differentiation of various stem cells types, such as chondrogenic differentiation of adipose stem cells or osteogenic differentiation of human periodontal ligament cells.27,28 In our study, we confirm the positive effect of collagens from African sharptooth catfish and silver carp on the osteogenic differentiation of ASCs. A similar observation was reached by Liu and Sun 29 , their results demonstrated that hydrolyzed fish collagen promotes osteogenic and endothelial differentiation of rat bone marrow mesenchymal stem cells. However, contrary to their findings we did not find fish collagen inhibited adipogenic differentiation of our cells. Collagen from both fish species in our study seemed to be supportive in adipogenic differentiation, especially with rASCs. Moreover, according to Mazur et al., 30 differentiation into adipose tissue is an advantage for breast reconstruction in oncological patients. The inhibition effect of fish collagen on adipogenic differentiation was also reported by others. 31 However, there is not much literature data about the effect of fish collagen on the regulation of adipocyte differentiation. Debski et al., 18 in their research showed the role of ASCs and collagen-based scaffold in damage recovery, precisely in regeneration of an injured nerve. Other results also indicate potential of ASCs in tissue engineering products, for example ASC-seeded scaffolds showed improved scaffold vasculogenesis. 32
Important characteristic features for designing collagen-based biomaterials are morphology and biocompatibility. 33 Collagen extracted from African sharptooth catfish and silver carp fish by-products is a good resource for biomedical applications, due to its biocompatibility, efficient absorption into the body, and no risk of zoonosis transmission. The physicochemical properties, morphology, and amino acid composition of African sharptooth catfish and silver carp have been studied and published in the literature.33–35 Faralizadeh et al., 33 conducted a study that confirmed biocompatibility and suitability of silver carp skin collagen for cell growth. Another study by Rodziewicz-Motowidlo et al., 36 demonstrated the applicability of silver carp skin waste as a potentially abundant source of collagen. Given its relatively high thermal stability, silver carp skin collagen is an attractive alternative for the food and pharmaceutical industries.37,38 Furthermore, some literature sources reported the suitability of African sharptooth catfish collagen in the acceleration of wound healing, and as a scaffold for tissue engineering.39,40
The results of our research imply that collagen extracted from African sharptooth catfish and silver carp skins is biocompatible, in terms of ASCs growth, clonogenic potential, and differentiation in osteogenic and adipogenic lineages. Furthermore, fish collagen due to its anti-inflammatory and immune-modulatory properties, ability to stimulate cell adhesion, can improve wound resolution and closure, and has potential in bone and cartilage tissue engineering.20,41,42 Despite its numerous applications, collagen derived from fish by-products is a promising biomaterial for use in cell and tissue culture as an alternative to mammalian collagen, for example for both osteogenic and chondrogenic differentiation of cells.21,26 In vivo, cells are surrounded by the extracellular matrix (ECM), which is their natural embedding, and one of its components is collagen. Research data shows, that collagen can be used as a component of jellified suspension of ECM and with polycaprolactone (PCL) scaffolds for 3D cell culture.21,43 Moreover, collagen can be a promising biomaterial as a component of bioinks in the era of 3D bioprinting.
The generality of the current findings needs to be established by further research, but the present study has clearly demonstrated that fish collagen could be a good alternative to mammalian collagen and has potential as a biomedical biomaterial.
Conclusions
Fish collagen extracted from African sharptooth catfish and silver carp skins, in comparison with mammalian collagen, shows no difference in supporting cell viability and proliferation capacity.
Footnotes
Acknowledgements
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Contributorship
Conceptualization, K.R., Z.P.,T.D.; methodology, K.R., K.S., A.Z.; software, K.R., K.S.; validation, K.R., A.S., K.S., A.Z.; investigation, K.S., A.Z., T.D.; resources, T.D, K.S., A.Z., J. M-J., A.A; data curation, K.S., A.Z., T.D.; writing—original draft preparation, K.R.; writing—review and editing, Z.P., K.S., A.S., T.D.; visualization, K.R., A.S.; supervision, Z.P.; project administration, Z.P.; funding acquisition, Z.P. All authors have read and agreed to the published version of the manuscript.