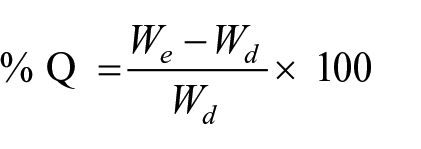Abstract
Purpose:
Bleeding is a leading cause of mortality and morbidity in the trauma and surgery field, using effective hemostatic agents can help us reduce bleeding especially in parenchymal hemorrhage. Nowadays polyvinyl alcohol (PVA) is known as a safe candidate for wound dressing and maybe a hemostatic agent. PVA-based hydrogel is a popular biocompatible material in the biomedical field especially when it has high water absorption. In this study, we investigated the PVA hydrogel’s mechanical and biological properties as well as its hemostatic potential in parenchymal bleeding.
Methods:
PVA hydrogel had made by the freeze-thawing approach, we used PVA hydrogel in comparison to standard treatment to investigate hemostatic potency. Also, we performed MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) tests to survey PVA cellular toxicity. After an acute liver injury, two groups of 12 rats were treated with PVA hydrogel or standard treatment with sterile gauze. The results including the time and volume of bleeding, and the time and survival rate of the rats were measured and compared.
Results:
We saw that PVA hydrogel was safe with no cellular toxicity in the MTT assay. Regarding efficacy, PVA hydrogel increased rats’ survival after bleeding from 75% to 91.7%, and decreased bleeding time (p: 0.015), and bleeding volume (p: 0.03) compared to the control group.
Conclusion:
Polyvinyl alcohol is safe. It has good biological properties with no cellular toxicity and has a significant hemostatic effect and can be regarded in control of parenchymal hemorrhage.
Introduction
Bleeding is one of the important causes of death in the trauma field and surgical procedures.1 –3 Failure to control bleeding at the scene of trauma, including cases of war, conflict, and traffic accidents; causes serious and sometimes irreversible complications, including acidosis, hypothermia, and ischemic damage to vital organs,4,5 which reduces the efficiency of surgery after the patient arrives at the hospital and reduces survival.3,6 Using a tourniquet and packing with gauze can significantly reduce bleeding in trauma patients at pre-hospital levels. Despite this, uncontrollable and unpackable bleeding in the parenchyma, including internal organs, is known as the main cause of early death following trauma to the central region of the body. On the other hand, bleeding in areas such as the axilla and the groin cannot be treated by using the tourniquet. 7 All these cases indicate the necessity of using hemostatic agents and dressings that are more effective than current treatments in controlling bleeding. An ideal hemostatic dressing should be available and cheap, easy to use, without side effects, stable in different environmental conditions, and has a long shelf life. 3
Hydrogels are hydrophilic polymer-based three-dimensional structures with high water-absorbing potential exhibiting a parallel similarity with biological tissues. 8 Polyvinyl alcohol has an intensive application in the biomedical field thanks to good hydrophilicity, gel-forming characteristics, ease of processibility, and high chemical resistance despite its synthetic nature.9 –11 Numerous cross-linking approaches have been utilized to fabricate PVA-based hydrogels; however, chemical gelation methods potentially would increase the cytotoxicity and dwindle the flexibility and water uptaking prospect.8,12 Thus, physical gelation techniques are taken into consideration. The freeze-thawing approach is a unique mechanism incorporating phase separation into a PVA-rich and a water-rich phase that alters the PVA matrix’s crystalline structure.13 –15 Therefore, a viscoelastic hydrogel with high water absorption is achieved, widening its application in pharmaceutical and biomedical applications.16,17
Polyvinyl alcohol, which is already well known in medicine for its use as a dressing with tissue healing function, has also shown good hemostatic performance in some studies, although there is not enough evidence of its potency, and more evidence and studies are needed. In this study, we decided to investigate the mechanical characteristics and In Vitro biocompatibility of PVA hydrogel and compare its effect on controlling liver parenchymal bleeding in an animal model in comparison to standard treatment.
Methods and materials
PVA with
Characterizations
Swelling behavior
The water uptake potential of freeze-thawed hydrogel was investigated to evaluate the changes in PVA structure during freezing and structural densification during the formation of the semicrystalline structures and its impact on the water uptake prospect. The freeze-thawed sample was first weighed
In vitro degradation
The degradability rate can be examined experimentally by immersing the desired sample in PBS. The sample was immersed in PBS at 37°C. On 1,3 and 7 days, the remained substrate was weighed
Rheometric analysis
The viscoelastic properties of the freeze-thawed hydrogel were measured using the Paar Physica MCR 300 (SN634038) parallel disk rheometer. Samples with a dimension of 30 × 30 mm and a thickness of 800 microns were prepared and loaded onto the device. Frequency sweeps (0.1–100 Hz with the rate of 1 Hz at a strain amplitude of 0.1%) at 37°C were performed to determine the storage moduli (G′) and loss moduli (G″).
Compression analysis
Compression was performed via a Universal materials testing system TB30T model with a 50 N load cell to investigate the compression properties of the freeze-thawed sample. The test was performed via a strain rate of 100% per minute on the samples with 20 mm diameter.
MTT assay
In vitro cell viability on the PVA hydrogel was analyzed by MTT colorimetric assay after 24, 48, and 72 h of incubation. Human skin fibroblast cell line (HFSF-PI3) was seeded on the hydrogel in 96 well plates at the density of 50 × 103 cells per hydrogel. Then, the cell-seeded PVA hydrogel was incubated in a serum-free media and MTT reagent at 37°C for 3 h. The absorbance of formazan crystals at 570 nm was detected after soaking the PVA hydrogel. In the following step, a solution containing 0.5 mg/ml of MTT dye and 1 mL of staining solution was included in the PVA sample after removing the solution. In the last stage, the staining solutions were washed out using PBS. The sample was transferred to the well plate to investigate by fluorescent microscopy (Olympus inverted fluorescence imaging system).
In vivo application
Twenty-four healthy male Wistar rats weighing 150–200 g were divided into two groups of 12 rats. Anesthesia was performed with the combination of Ketamine with a dose of 60 mg/kg and Xylazine with a dose of 7 mg/kg. After anesthesia, a 3 cm long median incision was made in the abdomen and 1.5 cm was cut from the lower part of the left lobe of the liver. After the start of bleeding, the mice were treated by applying a 1 ml polyvinyl alcohol gel (Figure 1) or packing with sterile gauze in the control group. In the following, the mice were examined and compared in terms of the amount of bleeding in milliliters, bleeding time until reaching hemostasis, survival rate, and survival time in three groups.

Applying PVA gel on the bleeding site.
Up to 10 min after the start of bleeding, the rats were monitored for survival, time, and volume of bleeding. After the end of the bleeding, the amount of bleeding in the mice was measured and recorded by suction with an insulin syringe with an accuracy of one-tenth of a milliliter. Bleeding time was calculated from the start of bleeding to the end of bleeding or the death of the mouse. Euthanasia was performed through CO2 gas inhalation. The animals were placed one by one in a special chamber and the flow of CO2 gas was established from the cylinder into the chamber. The animals remained in the chamber for at least 6 min and at least 1 min after complete respiratory arrest, and after leaving the chamber, they were checked for possible vital signs.
Research data were analyzed using SPSS version 26 software. The normal distribution of the data was checked with the Kolmogorov-Smirnov statistical test. Independent sample t-test statistical analysis was used to compare the bleeding time and volume between the two groups. The Kaplan-Meier curve was used to compare the two groups’ survival after bleeding. This study was performed in compliance with the guidelines of the Research Ethics Committee of Laboratory Animals, Tehran University of Medical Sciences, Tehran; approval No. IR.TUMS.AEC.1400.051.
Result
Swelling ratio and weight loss
As Figure 2(a) illustrates, an increasing trend in imbibing water by freeze-thawed PVA can be observed. However, after 24 h, a decrease in the amount of absorbed water is detected. This phenomenon results from the dissolution of the number of amorphous chains of PVA not participating in crystallization during freeze-thawing at 37°C. Additionally, Figure 2(b)) represents that during 3 days, more than 65% of the total hydrogel was dissolved in the PBS due to the physical cross-likings among the structure.
(a) The water uptake potential of 5-cycle freeze-thawed PVA hydrogel during 24 h and (b) the weight loss percentage after 4 days of immersing in PBS solution at 37°C.
Mechanical properties
Compression test and rheometric analysis
Figure 3 and Table 1 exhibit the dynamical and compression properties of the 5-cycle freeze-thawed PVA, (a) showing the storage, (b) loss modulus, and (c) compressive stress-strain curve of freeze-thawed PVA hydrogel.

(a) and (b) demonstrate the storage and loss modulus of the final PVA substrate. The storage and loss modulus at the frequency of 1 Hz was measured respectively at about 6.13 and 2.43 KPa. (c) shows compressive stress-strain curve of PVA hydrogel with a good compressive properties.
The rheological properties.

G': Storage moduli (KPa); G": Loss moduli (KPa); Frequency (Hz).
Moreover, the compression modulus of the PVA-based hydrogel was obtained at 0.2 MPa, and good compressive properties were observed in Figure 3(c)).
Cell viability
As illustrated in Figure 4, representing the cytotoxicity of the PVA platform, less cell viability is observed on the freeze-thawed PVA hydrogel compared to the control group. Although a dwindling trend in cell survival is detectable, after 72 h, the fibroblast survival percentage reached 80% ± 10 on the PVA platform indicating non-toxic and biocompatible potential.

Cell viability assay histogram measured by MTT assay until 72 h.
Hemostasis effect
Bleeding time in the PVA hydrogel group (107.67 s) was significantly (p: 0.015) lower than in the control group (186.58 s) (Table 2).
Surgery outcomes.

Bleeding Time (sec); Bleeding Volume (milliliter).
Also, PVA gel significantly decreased bleeding volume (p: 0.03); the volume loss was 1.84 and 2.98 ml respectively for the gel and control group. Figure 5 shows PVA hydrogel controlling liver bleeding.
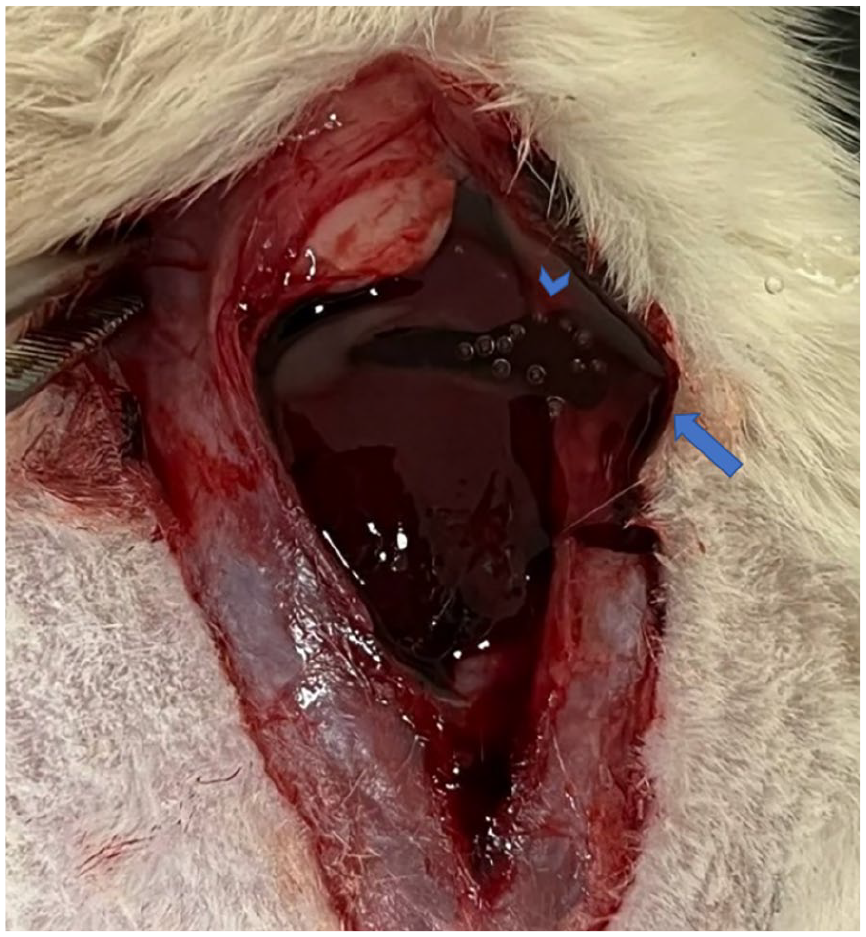
PVA gel (clear liquid marked with an arrow) surrounded and packed bleeding (dark liquid marked with an arrowhead.).
Survival analysis
The Kaplan-Meier diagram below (Figure 6) shows the survival status of rats after bleeding and treatment in two different groups, as it is evident in the control group, 9 out of 12 rats survived and three rats died due to bleeding. In the treatment group with PVA hydrogel, only one rat died due to bleeding. Indeed, treatment with hydrogel increased rat survival rate after liver hemorrhage.

Survival analysis after bleeding in control and PVA gel group.
Discussion
The freeze-thawing method is mainly utilized to form physical cross-linking among a PVA-based hydrogel structure.14,17 During freezing to −20°C, small crystallites or crystals from small crystallites are formed in the PVA structure. 18 More precisely, nucleation in the thawing process occurs during water evaporation. Crystal formation leads to the development of a stable physical network in the entire structure.15,18
Increasing the number of cycles improves the degree of crystallinity in the PVA polymeric chains and involves more amorphous PVA chains in the crystallization process.8,14 Previous studies reported that performing 5–7 freeze-thawing cycles contributes to a more substantial crystalline structure.13,18 Also, it is proved that low to intermediate molecular weight PVA represents better performance when undergoing physical gelation via the freeze-thawing procedure. Thus, we put intermediate molecular weight PVA undergoing five freeze-thawing cycles under investigation in this study.8,10,15 Our research revealed an excellent mechanical property whose compressive modulus and viscoelastic exhibit logical conformity with the previous studies indicating its ideal application in biomedicine applications.
Holloway et al. represented a direct correlation between compressive modulus and the number of freeze-thawing cycles for pure PVA hydrogel. So, the final compression modulus of the hydrogel based on PVA with89,000-96,000 molecular weight with the hydrolysis degree of 99% after two, four, and five freeze-thawing cycles were 0.1, 1.8, and 0.28 MPa respectively. 18
Shauna R and co-workers analyzed the compressive strain of freeze-thawed PVA hydrogels made of PVA with Mw = 79200 that underwent different freeze-drying cycles whose frozen temperature was −20°C for 1–24 h. The results revealed that the compressive strain after two, three, four, and five cycles was 0.18, 0.16, 0.1, and 0.08 respectively. 11
In another research conducted by Xie et al., the compressive properties of freeze-thawed pure PVA hydrogels based on PVA with a molecular weight of70,000 were frozen at −18°C for 22 h during several cycles. The results indicated a range of 16-24 ± 2 KPa for the compressive modulus of final hydrogels. 19
Huiyang Bian and co-workers reported that the compressive modulus for PVA-based hydrogel after one cycle of freeze-thawing with −18°C of frozen time during 24 was 70 KPa. Additionally, the storage and loss modulus (G' and G") at the frequency of 1 Hz was 200 and 18 Pa. 19
Wersindo et al. also reported that a compressive modulus of 0.25 ± 2 KPa by applying 50-69% strain can be obtained from a freeze-thawed PVA hydrogel based on PVA with 89000–98000 Da after six cycles whose frozen temperature was −25°C for 24 h. 20
Following the study carried out by Yang et al. the freeze-thawed pure PVA hydrogel after three cycles represented G' and G" of 5000 and 800 Pa at 1 Hz. It is worth mentioning that the PVA had a degree of polymerization of 1750±50 and a 98% of hydrolysis degree. 21
Bleeding is the most important cause of death in trauma. The prognostic role of some hemostatic factors in survival after bleeding has been shown,22,23 but the role of PVA is not clear. Our results show that the use of PVA gel in the rat experimental bleeding model reduced mortality.
In a study by Zhao et al., the authors investigated the role of hemostatic factors in the control of arterial bleeding in both rat and pig models. In the rat model, treatment with PVA caused faster hemostasis and decreased bleeding. In the pig’s model, the gauze could not stop the bleeding, but PVA created hemostasis. Mean arterial blood pressure decreased significantly in the gauze group but remained stable in the PVA group. 24 Our findings suggest similar results that PVA gel could shorten bleeding time and make better hemostasis than the standard treatment, Contrary to us the study by Akkan et al. in 2019, which was conducted to compare the effectiveness of treatment protocols to prevent anterior epitaxy, the group treated with tranexamic acid was superior in terms of reducing the rate of rebleeding in comparison to PVA group. 25 In another study by Li et al. in 2011, on 43 patients with known cesarean scar pregnancy, similar to our results, it was observed that the combination of PVA compared to the standard treatment with methotrexate controls bleeding more effectively in patients treated with transcatheter arterial chemoembolization. 26
Our result suggests that PVA can decrease bleeding volume more than the standard treatment. Contrary results in different studies about PVA hemostatic power may be because of their different fabrications or utilizing different control groups or standard treatments in different studies. For example, also some studies like us reported the superiority of PVA to standard treatment or control groups like packing with sterile gauze,24,27 but some others reported that PVA is less effective compared to chemical hemostatic agents such as tranexamic acid and chitosan.24,25 Perhaps the sum of these reports is trying to reveal a medium hemostatic role of PVA, which is more powerful than packing with gauze but less effective than powerful hemostatics. So, we think this idea needs to be more investigated.
Our MTT test showed that PVA hydrogel is biocompatible. The reason for less cell viability on the PVA substrate than the control group may be rooted in either the inefficient pore size or porosity content formed during the freeze-thawing process. Moreover, during the cell culture process, a remarkable number of cells would be washed away by the culture media or PBS, affecting the MTT results. 28 Although there are not enough studies to confirm the powerful hemostatic efficacy of PVA compared to other hemostatic agents, PVA safety has been shown in many studies and it seems consequential and undeniable.29,30
So far, it has been proven that many hemostatic agents, despite their good efficacy, have disadvantages such as systemic and local toxicity, heat damage, expensiveness, and tissue inflammation and adhesions, which limit their use.3,31 Contrary to this, various studies demonstrated the positive role of PVA in biomedical uses, benefits of PVA such as biocompatibility, anti-inflammatory and antimicrobial properties are consequential and cannot be ignored even if PVA shows less efficacy than other hemostatic agents.32,33 In our study in the same way with previous studies, no cytotoxic effects of PVA were observed. And it can be a great advantage for using this material despite the unproven powerful effect.
Limitations
Since animal models with larger bodies, such as pigs and sheep, are more similar to human bleeding models in terms of bleeding, a study model in larger animal models could provide us with more detailed information about the functioning of hemostatic factors in controlling human bleeding.
Although the safety of PVA consumption has been observed in previous studies, and our MTT test; but doing more tests like hematological and histological examination could be helpful in terms of the safety check.
Conclusion
In conclusion, PVA hydrogel is safe and biocompatible. It seems to have good hemostatic effects compared to standard treatment on a parenchymal hemorrhage. Although many studies reported the safety of PVA even in human applications, but more research is needed to confirm its exact effect on hemostasis. Considering the nonpromising prehospital treatments for severe bleeding and their potential complications, it seems that more studies on hydrogels with biocompatible and safe materials such as PVA are useful and maybe necessary.
Footnotes
Acknowledgements
N/A
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.