Abstract
The titanium implant was treated with plasma electrolytic oxidation and subsequent ionic exchange and thermal treatment in order to obtain bioactive layer consisting of titanium oxide, calcium and sodium titanates and hydroxyapatite, as confirmed by X-ray diffraction (XRD). Scanning electron microscopy (SEM) revealed that the given method, besides corresponding phase composition, enables suitable nanotopology for cell attachment and proliferation. Cytotoxicity investigations by MTT, LDH and propidium iodide assays and light microscopy showed that these coatings were not toxic to L929 cells.
Introduction
Titanium and its alloys are widely used as implant materials since they show very good strength-to-weight ratio, corrosion resistance, good mechanical behavior and biocompatibility, but they have also low wear resistance and high friction coefficient which limit their more extensive applications.1, 2 Plasma electrolytic oxidation (PEO) is a very economical and efficient technology for production of stable oxide coatings on the surface of lightweight metals, like titanium, with controllable composition and morphology, excellent bonding strength with the substrate, high micro hardness, and high-quality wear and corrosion resistance.3–5 Also, it enables formation of bioactive oxide coatings with very interesting nanotopography and surface porosity, which is very important since recent tendency in development of clinical oral implants is presumably focused on topographical changes of implant surfaces.6–8 PEO additionally enables specific surface pore configuration through the growth of the oxide film,9, 10 as well as the crystal structures of the titanium oxide.9–11 One of the main advantages of PEO method is production of chemically and biologically active surface of titanium oxide phases, mainly constituted of active phases like anatase, without additional annealing.12, 13
The excellent biocompatibility of thin native oxide films on titanium implants6, 7, 14 makes these coatings interesting for application in oral surgery and orthopedics. Since there is not much information about biological performances of coatings obtained by PEO method, the coatings obtained in this study were subjected to cytotoxicity investigations.
Methods
Sample preparation
For the deposition of oxide coatings, the titanium tiles with dimensions 25 mm × 5 mm × 0.25 mm and 99.5% purity were used. The oxidation process was carried out in an electrolytic cell with flat glass windows. 15 Platinum wires were used as cathodes. As a precursor solution for the PEO of titanium a water solution of Na2SiO3·5H2O (10 g/l) and acetonyl acetonate (10 ml/l) were used. The electrolyte was prepared by using deionized water and pro analysis grade chemical compounds. Anodizing was carried out at current density of 200 mA/cm2. The temperature of the electrolyte was maintained during the PEO process at 21°C. The time of PEO processing was 5 and 15 minutes. After the PEO, the samples were rinsed with distilled water and dried. After drying, samples were firstly immersed in 0.1 M water solution of (NH4)2HPO4 for three days, then rinsed with distilled water and immersed in 0.1 M solution of Ca(NO3)2 for one day, rinsed with distilled water and again immersed in 0.1 M solution of (NH4)2HPO4 for one day. Finally, the samples were rinsed with distilled water, dried at 100ºC for 2 h and thermally treated at 450ºC for 4 h.
Methods of characterization
In order to analyze morphology and particle size distribution of the obtained coatings, a field emission scanning electron microscope (FESEM, FEI Scios 2) was used. The phase analysis of the obtained coatings was done by X-ray diffraction (XRD) (Rigaku SmartLab 3KW). Additional characterization is given in the supplementary material.
Cytotoxicity investigations
In order to investigate potential biological application cytotoxic activity, the PEO treated titanium implant was investigated in direct cytotoxicity assays, in accordance with ISO 10993-5 guidelines. 16 Test samples were tiles of titanium implant treated with PEO, as well as the samples of the initial titanium substrate, without any modification. Before the samples were added to the cells in culture, they were sonicated in an ultrasonic bath for 15 minutes, washed and disinfected in 70% alcohol, dried and finally washed with sterile Roswell Park Memorial Institute (RPMI) medium (Sigma).
Murine fibroblasts cells (L929), obtained from the European Collection of Animal Cell Cultures (Salisbury, UK), were used to test cytotoxicity of the test samples. Forty-eight hours prior to the addition of the samples, the cells were seeded in a number of 3 × 105 in a final volume of 2 ml per well. After 48 h cell confluence was approximately 80%, which was a prerequisite for performing the test. All experiments were done in triplicate in 6-well plates. Cells cultivated alone (without any substrate) served as positive control.
Metabolic activity of the cells was analyzed on the basis of the reduction of a tetrazolium salt of 3-4,5-dimethylthiazol-2-yl-2,5-diphenyltetrazolium bromide (MTT). 17 Ti samples treated with PEO for 5 and 15 minutes and non-treated Ti samples were placed on a monolayer culture. Cells were cultivated for 24 h, after which their metabolic activity was tested by the MTT test.
Cell necrosis was assessed by cell staining with propidium iodide (PI). 18 PEO treated Ti samples and non-treated Ti sample were placed on monolayer culture. Cells were cultivated in triplicates for 24 h, after which the samples were removed and cells were trypsinized to prepare the cell suspension. Cells (1 × 105/100 µL) were stained with PI and number of necrotic (PI) positive sells was assessed using flow cytometry. Cell were analyzed with flow cytometer (CyFow® Cube6, Partec) using blue laser excitation light source (488 nm wavelength) and FL3 interference bandpass filter 675/20 nm for detection of orange/red fluorescence.
The quantification of cell necrosis was performed by assay detecting the release of lactate dehydrogenase (LDH) in the supernatants of the cell cultures. 19 PEO treated Ti samples and non-treated Ti sample were placed on a monolayer culture. In order to detect the maximum amount of LDH released, the cells were cultured without the Ti samples in the presence of 1% solution of Triton X-100. Cells were cultivated for 24 h, after which quantification of LDH in the culture supernatants was done.
Proliferation of L929 cells was studied by using a [3H]-thymidine incorporation assay. 20 The subconfluent cell cultures were exposed to PEO treated Ti samples and non-treated Ti sample for 18 h. After removing the samples, [3H]-thymidine (1 µCi/well) was added for 8 h.
Qualitative verification of the results was performed by light microscopy based on morphological characteristics of the cells and the confluence of the cell growth in the culture. The one-way ANOVA followed by Student–Newman-Keulsposthoc test was used for evaluating the differences between Ti samples and control cells.
Results
SEM observations
Highly developed morphology of the Ti implants surface, after PEO treatment and additional treatment described earlier, can be observed in scanning electron microscopy (SEM) images (Figure 1). There is no significant difference in shape and size of particles and agglomerates between PEO treated samples for 5 and 15 minutes, but the coating is much denser for the sample treated for 15 minutes and much more particle layers can be seen (Figure 1(c)). The size of the smallest particles on the coating surface is from 30 to 150 nm. Bigger particles are actually agglomerates of a large number of smaller particles, and their size varies between 300 and 700 nm (Figure 1(d)), with some bigger agglomerates in deeper layers.

SEM. Appearance of untreated and PEO treated Ti implants (magnification × 5000): (a) untreated implant, (b) PEO treated for 5 minutes, (c) PEO treated for 15 minutes, and (d) typical appearance of the coating surface under higher magnification (× 50,000).
XRD analysis
Figure 2(a) shows the XRD spectrum of untreated Ti implant, where the peaks corresponding to crystal planes of pure Ti can be seen.

XRD pattern of the Ti implant: (a) untreated, (b) PEO treated for 5 minutes, and (c) PEO treated for 15 minutes.
The XRD spectrum of the titanium implant PEO treated for 5 minutes, additionally treated with (NH4)2HPO4 and Ca(NO3)2 and finally thermally treated (Figure 2(b)) revealed the presence of the following phases: NaTiO2 phase (signed as NaT), Na2TiO3 (signed as Na2T), Na4TiO4 (signed as Na4T), anatase (signed as A), rutile (signed as R), CaTiO3 (signed as CaT) and calcium hydroxyapatite (signed as C). The peaks on the XRD spectrum of the titanium implant after PEO treatment for 15 minutes and additional treatment (Figure 2(c)) revealed the presence of: NaTiO2, Na2TiO3, Na2Ti9O19 (signed as Na2Ti9), anatase, rutile, CaTiO3 and hydroxyapatite phase.
Results of cytotoxicity investigations
To investigate whether PEO treated Ti samples influence cell viability in comparison to untreated samples, metabolic activity of L929 cells was firstly analyzed. Almost unchanged values of MTT reduction were detected in cultures with PEO treated samples regardless of plasma process duration. However, values of MTT reduction were lower in culture with untreated Ti samples, indicating a lower metabolic activity of L929 cells. Observed inhibition was around 40% but statistically insignificant (Figure 3).
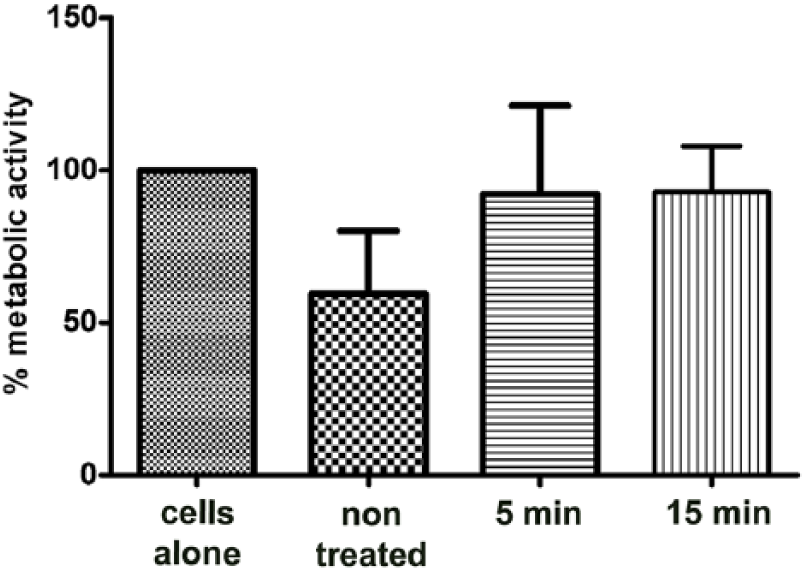
Effect of PEO treated Ti implants on metabolic activity of L929 cells. Ti samples treated with PEO for 5 and 15 minutes and non-treated Ti samples were placed on a monolayer culture. Cells were cultivated for 24 h and metabolic activity was tested by MTT assay. The results are shown as relative values (mean values ± SD of triplicates) of metabolic activity in comparison with control as 100% (cells cultured without dental alloy). The one-way ANOVA followed by Student–Newman-Keulsposthoc test was used for evaluating the differences between Ti samples and control cells. Analyzed values were statistically not significant.
In order to assess whether decrease in metabolic activity was a consequence of cell death, cell necrosis was analyzed. To measure loss of cell membrane integrity, cells were stained with PI and release of LDH in cell supernatants was measured. Using flow cytometry, a slightly enhanced number of necrotic cells was detected only in culture with untreated Ti- samples but not with PEO coated samples (Figure 4(a)). However, concentration of LDH was almost the same in all tested samples and without any difference in comparison with control cells (Figure 4(b)). As a positive control of total cell necrosis, L929 cells were treated with Triton X-100. Observed LDH values in cultures with all tested samples including untreated cells were around 6% of LDH values in culture with total cell necrosis, suggesting basal level of cell death in the presence of tested samples.
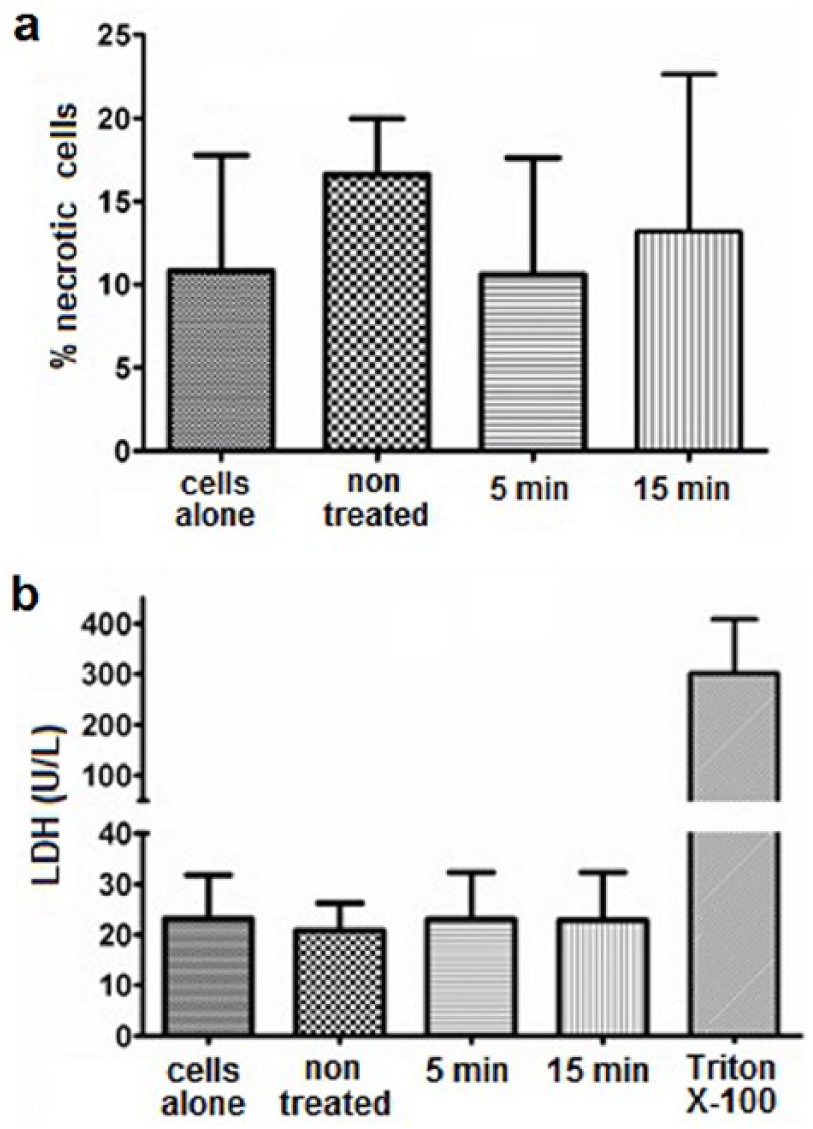
Effect of the PEO treated Ti implants on necrosis of L929 cells. Ti samples treated with PEO for 5 and 15 minutes and non-treated Ti samples were placed on a monolayer of L929 cells and cultivated for 24 h. (a) Cells were trypsinized to prepare a cell suspension and stained with PI. Cell necrosis was assessed based on quantification of PI positive cells. Results are presented as a percentage of PI stained cells (mean values ± SD of triplicates). (b) Supernatants form cell cultures were analyzed for the level of LDH. In order to measure LDH in culture with 100% necrotic cells, L929 cells were cultured without alloys in the presence of 1% Triton X-100. Results are shown as absolute values of LDH concentration (mean values ± SD of triplicates). The one-way ANOVA followed by Student–Newman-Keulsposthoc test was used for evaluating the differences between Ti samples and control cells. Analyzed values were statistically not significant.
These results were confirmed by morphological analysis of cell growth. No differences between tested samples were observed (Figure 5). No signs of cytopathic effect could be seen in proximity to Ti samples or PEO samples. However, when samples were removed, some impaired confluence of cell growth could be seen on the plate surface beneath all samples.

Morphological aspect of cytotoxic effects of PEO treated Ti implant on L929 cells. Ti samples treated with PEO for 5 and 15 minutes and non-treated Ti samples were placed on a monolayer of L929 cells and cultivated for 24 h. Morphological characteristics of cells and confluence of their growth were analyzed by phase contrast microscopy. Magnification × 10.
Finally, cell proliferation ability in the presence of Ti and PEO modified samples was assessed. Using [3H]-thymidine uptake assay, slight but significant (in comparison to control cells) elevation of proliferative activity of L929 cells in culture with all tested samples was observed. No difference in cell proliferation was observed between cultures with different samples (Figure 6).

Effect of the PEO treated Ti implants on proliferative activity of L929 cells. Sub confluent cell cultures were exposed to Ti samples treated with PEO for 5 and 15 minutes and non-treated Ti sample for 18 h. After samples removing, [3H]-thymidine (1 µCi/well) was added for additional 8 h. Results are expressed as relative values (mean ± SD) of triplicates using corresponding controls as 100% (cells cultivated alone without samples). The one-way ANOVA followed by Student–Newman-Keulsposthoc test was used for evaluating the differences between Ti samples and control cells. ***P <0.001, compared to control untreated cells.
Discussion
Oxide films particles on the surface of Ti implants, obtained after PEO treatment, provide nanotopology convenient for cell adhesion and growth, which makes them promising from the aspect of potential biological application (Figure 1). Also, the films showed super-hydrophilic properties, which is very desirable from the aspect of cell adhesion (see supplementary material). Beside the presence of sodium and calcium titanates and titanium oxides, XRD analysis revealed the presence of calcium hydroxyapatite, which was formed during the additional treatment of PEO obtained coatings with (NH4)2HPO4 and Ca(NO3)2. Since hydroxyapatite is the main component of the natural bone, it thus provides good compatibility with surrounding bone tissue upon implantation (Figure 2). Fourier transform infrared spectroscopy (FTIR) analysis, given in the supplementary material, confirmed formation of these phases. The coating thicknesses were 1–1.2 µm, and Auger electron spectroscopy revealed that Na+ ions reached the depth of ~375 nm, while Ca2+ ions were present only in top layers, to the depth of 125 nm (see supplementary material).
The analysis of the metabolic activity and necrosis of L929 cells in the presence of PEO modified Ti implant showed that the PEO process slightly improved biocompatibility of Ti materials (Figures 3 and 4(a)). PEO treated implants did not modulate metabolic activity and death of tested cells, whereas untreated implants may negatively affect these cell properties. Even though a higher number of necrotic cells was observed in the culture with untreated implants, no difference in levels of LDH were detected (Figure 4(b)). Although both used methods are based on integrity of cell membrane, detected difference in number of necrotic cells (less than 6%) obviously was not sufficient in order to reflect quantity of released LDH.
Monitoring cell growth using phase contrast microscopy showed reduced cell growth only under the samples, but not in their immediate vicinity (Figure 5), and therefore the reduction of cell growth beneath the samples cannot be associated with cytotoxicity of the tested materials but is a consequence of mechanical effect of the samples (samples occupied less than 10% of growth surfaces, which is in agreement with ISO guidelines 16 ). Considering the similar number of necrotic cells, almost the same LDH values as in control cultures, it is unlikely that reduced cell confluence under the Ti samples is a consequence of their cytotoxicity. Additionally, following regulative of ISO 10993-5 guidelines, 16 reactivity grade for tested samples can be determined as grade 2 (mild) and considered as non-cytotoxic.
Finally, non-toxicity of PEO treated and untreated implants have been confirmed by proliferation assay with L929 cells. Their proliferative response was slightly increased (but significant in comparison to control cells) in culture with all tested samples and could reflect cell reactivity to soluble factors released by Ti samples. However, there was one unexpected finding in regard to a higher proliferation of L929 cells in the presence of untreated samples regardless of their lower metabolic activity observed in comparison to control cells and PEO treated samples, as shown in Figure 6 and Figure 3, respectively. A possible explanation could be specificity of cell growth in higher density cell population and assumption that cellular metabolic activity varies greatly throughout the life cycle of cells. MTT assay is one of the most used assays for measuring cell metabolic activity and is an end-point assay that actually measures enzyme activities more than cell growth in a culture. 21 On the other side, cellular DNA is independent of metabolic changes in cells and quantification of DNA synthesis is more sensitive for measuring mitogenic response. 22 In our study this has been done by [3H]-thymidine assay, where [3H]-thymidine uptake into new strands of chromosomal DNA during mitotic cell division is directly proportional to cell proliferation (Figure 6). 20 Considering our findings, we can assume that decrease in metabolic activity cannot be always followed by impairment in proliferative response of cells. Lower metabolic activity and preserved proliferative potential of cells treated with Ti samples should be clarified in further studies.
Conclusion
The PEO method was used to obtain coatings on the surface of Ti implants, with phase composition and nanotopology convenient for cell adhesion and proliferation. The obtained results of biological behavior of PEO treated Ti implants in vitro did not show significant modulation of metabolic activity or cell death in the tested cell line. Slightly increased proliferative response of L929 cells suggests preserved proliferative capacity of fibroblast cells in the presence of these materials. All these findings showed that the obtained coatings were non-toxic and thus support the idea of their in vivo possible application.
Supplemental Material
Supplementary_material – Supplemental material for Nanodesigned coatings obtained by plasma electrolytic oxidation of titanium implant and their cytotoxicity
Supplemental material, Supplementary_material for Nanodesigned coatings obtained by plasma electrolytic oxidation of titanium implant and their cytotoxicity by Marko Magić, Božana Čolović, Saša Vasilijić, Nenad Tadić, Stevan Stojadinović and Vukoman Jokanović in Journal of Applied Biomaterials & Functional Materials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (project number 172026) and Medical Faculty of the Military Medical Academy, University of Defence in Belgrade (project MFVMA 10/13-15).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
