Abstract
The primary goal of bone tissue engineering is to fabricate scaffolds that can provide a microenvironment similar to that of natural bone. Therefore, various scaffolds have been designed to replicate the bone structure. Although most tissues exhibit complicated structures, their basic structural unit includes stiff platelets arranged in a staggered micro-array. Therefore, many researchers have designed scaffolds with staggered patterns. However, relatively few studies have comprehensively analyzed this type of scaffold. In this review, we have analyzed scientific research pertaining to staggered scaffold designs and summarized their effects on the physical and biological properties of scaffolds. Compression tests or finite element analysis are typically used to evaluate the mechanical properties of scaffolds, and most studies have performed experiments in cell cultures. Staggered scaffolds improve mechanical strength and are beneficial for cell attachment, proliferation, and differentiation in comparison with conventional designs. However, very few have been studied in vivo experiments. Additionally, studies on the effect of staggered structures on angiogenesis or bone regeneration in vivo, particularly in large animals, are required. Currently, with the prevalence of artificial intelligence (AI)-based technologies, highly optimized models can be developed, resulting in better discoveries. In the future, AI can be used to deepen our understanding on the staggered structure, promoting its use in clinical applications.
Introduction
Recently, the segmental repairing of bone defects has garnered significant attention for orthopedic and plastic surgeries. Currently, autologous and allogeneic transplantations are used in clinical practice for bone repair and regeneration. 1 Autografts are regarded as the “gold standard,” achieving varying degrees of success in restoring bone function, owing to their high histocompatibility and low immunogenicity. 2 However, harvesting autogenous bone from the iliac crest or other sites may come with surgical risks such as scarring, bleeding, inflammation, and chronic pain.3,4 These limitations hamper their extensive use in clinical applications.
To address this challenge, scaffold-based tissue engineering provides a new approach toward repairing bone defects. Scaffolds are significant as they can provide an ideal environment for cell growth and differentiation, contributing to the development of new bone tissue. 5 Apart from scaffold porosity and pore size, 6 new tissue formation is significantly influenced by the 3D structure of the scaffold. 7 Scaffold architecture can also influence nutrient transport and cell–matrix interaction. Therefore, based on the natural bone hierarchical structure, researchers have designed various scaffold architectures at the macroscale, microscale, and nanoscale. 5 Additionally, recent studies have demonstrated new design patterns for staggered structures. However, relatively few studies have comprehensively analyzed this type of scaffold. The functionality of scaffolds is determined by their permeability, and mechanical and biological properties. 8 Therefore, the aim of this review is to summarize current scientific research regarding the effects of the staggered structure on the physical as well as biological properties of scaffolds.
Literature survey
In this review, systematic literature search was carried out on “PubMed.” The keywords used to collect relevant literature were “staggered, tissue engineering, scaffold,” “shift, tissue regeneration, scaffold,” “offset, tissue engineering, scaffold,” and “compacted, tissue engineering, scaffold.” Only original research studies evaluating the impact of the staggered, shift, offset, or compacted designs on the physical and chemical properties or cell response were selected (Figure 1). Review articles, studies with insufficient data, and research articles that did not aim to regenerate the bone for clinical applications were excluded. The scaffold materials, fabrication methods, experiment content, and design details of the related studies are summarized in Table 1. A narrative review regarding the detailed methodology, physical properties, cell response, and bone regeneration is also provided.
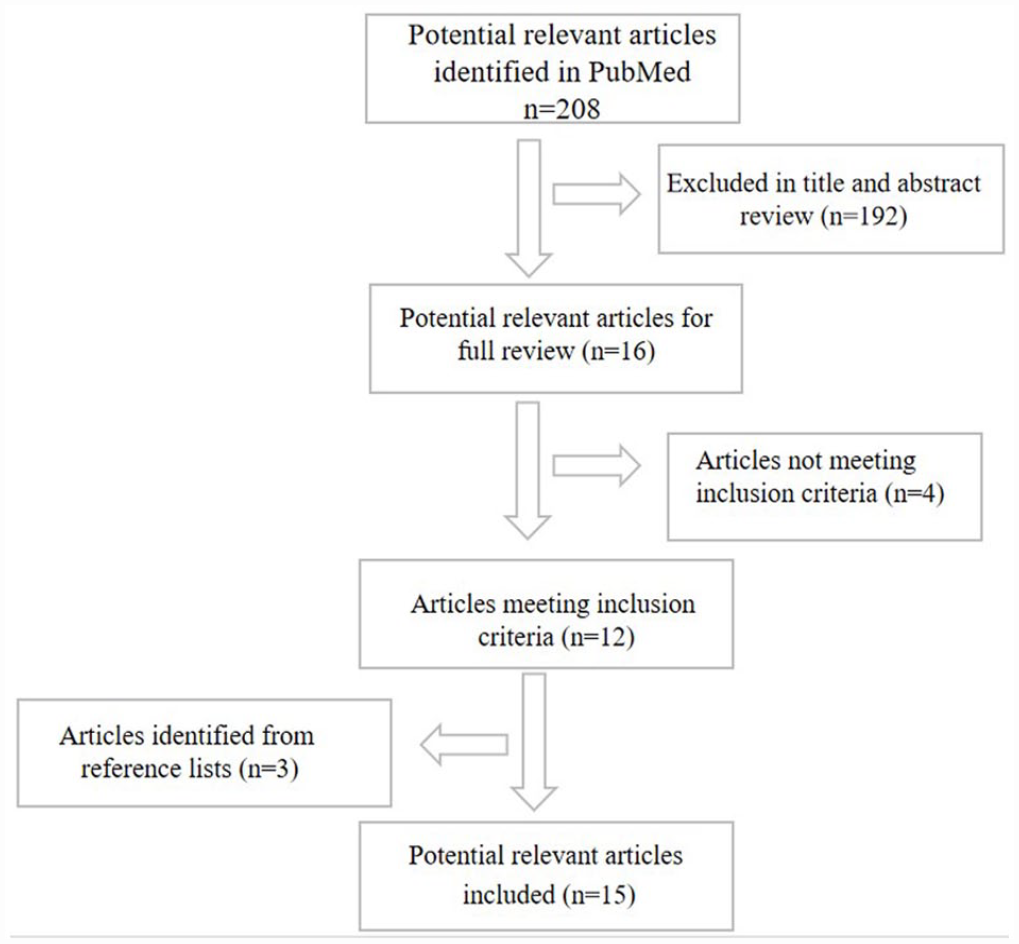
The flow chart of literature selection for the review.
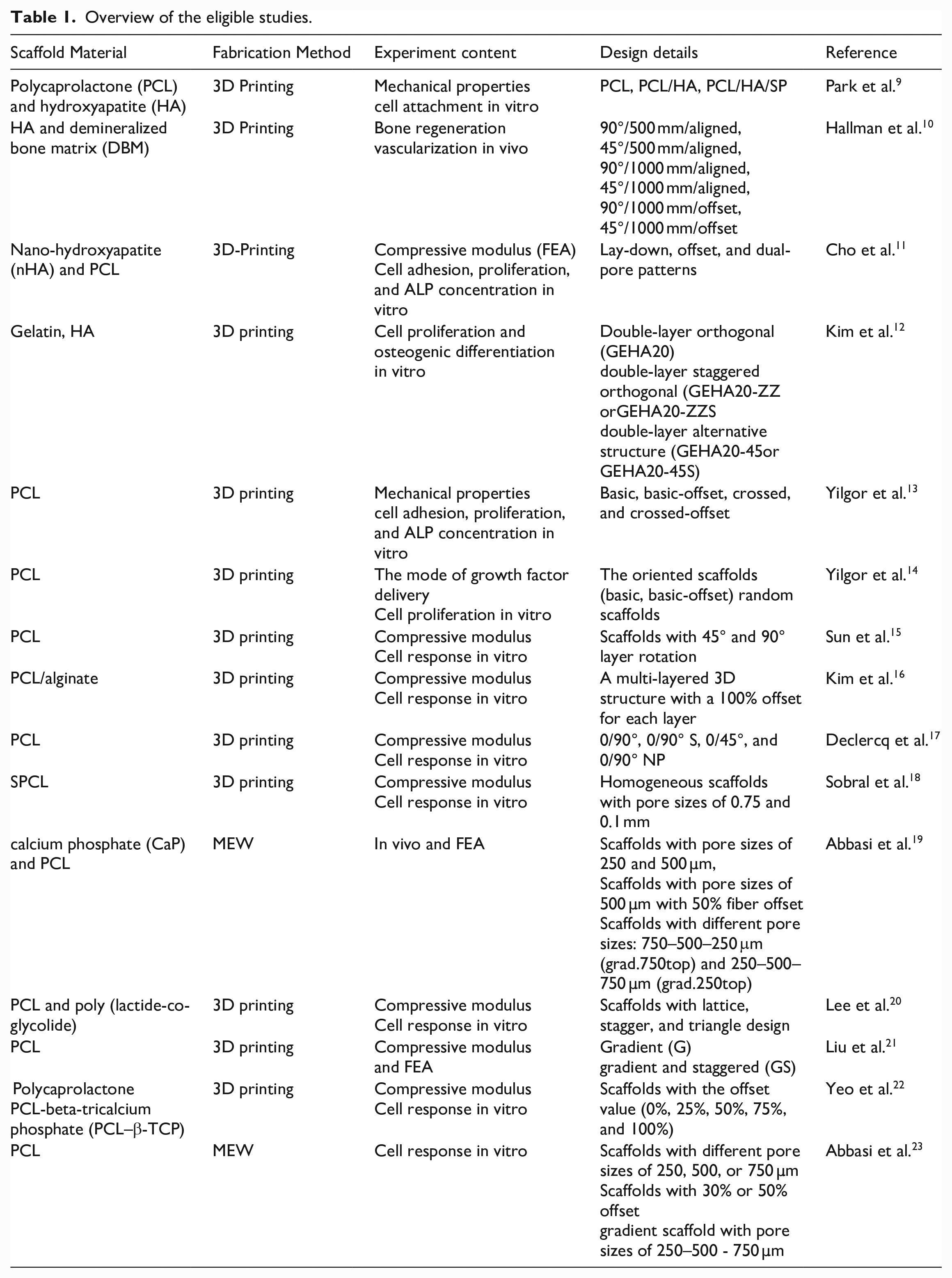
Overview of the eligible studies.
Staggered structures in porous scaffolds
Evaluation methods
Only 15 studies sufficiently met the inclusion criteria. A compression test and finite element analysis (FEA) are two methods typically used to evaluate the mechanical properties of printed scaffolds. Further, FEA predictions and the actual compression moduli of the fabricated scaffolds exhibited similar trends. Cho et al. 11 designed and fabricated scaffolds with lay-down, offset, and dual-pore kagome-structure design, named Conv 1, Conv 2, Offset 1, Offset 2, and dual-pore scaffolds, and evaluated their compression moduli via numerical and experimental analyses. Compared with the results of numerical analysis, experimental analysis yielded compression moduli showed the similar results (numerical and experimental analyses yielded compression moduli of 65 and 62.5 ± 1.8 MPa, respectively, for Conv 1). Additionally, most studies performed in vitro experimental studies to evaluate their biological properties, whereas only two studies performed in vivo experiments.
Fabrication methods
In these designs, two typical scaffold fabrication methods are three-dimensional (3D) printing and melt electrowriting (MEW), although there are many methods that could be used for scaffold fabrication.3D printing, an additive manufacturing (AM) process, prepares scaffolds via a layer-by-layer process and has been widely used to design and fabricate porous scaffolds.25,26 It consists of five main components, as shown in Figure 2(a). 7 It is advantageous as it can fabricate scaffolds that imitate the extracellular matrix (ECM). In addition, it can control porosity, pore size, and pore distribution, 24 and produce complex 3D structures. However, the scale of the fibers that form the scaffolds ranges from hundreds of microns to millimeters, 25 which is considerably larger than the scale of the ECM or cells (10–20 µm). Therefore, it cannot create a favorable microenvironment for cells. Two studies fabricated scaffolds using MEW, a high-resolution AM technology capable of producing small fibers from 800 nm to 50 µm in diameter.26,27 MEW 25 consists of four components: collector, high voltage source, pneumatic system, and electrically-heated system (Figure 2(b)) . 19 In particular, when the collector speeds of MEW are not synchronized with the electrified jet speed, buckling jetting is likely to occur, 28 thereby producing an array of different modeled and predicted patterns. 29 Hochleitner et al. 30 fabricated fibrous scaffolds with sinusoidal patterns using MEW.

Primary components of 3D deposition and MEW. (a) 3D printing device consisting of five main parts: (1) jacket, (2) molten unit, (3) force-controlled plunger, (4) XYZ table, and (5) X-Y-Z motor driver. 7 Copyright © 2003 Elsevier Ltd. (figure reused with permission). (b) MEW consisting of four parts: collector, high voltage source, pneumatic system, and electrically-heated system. 19 Copyright ©2020 by the Naghmeh Abbasi (figure reused with permission).
Staggered scaffold structures
Different staggered scaffold structures
The varied structuring over multiple lengths and material properties of hard biological tissues are significant in biomechanical research. 31 Most of these tissues have complex shapes, but their basic building units typically consist of stiff platelets arranged in a staggered micro-array inside a flexible matrix. 32 These basic building blocks are characterized by a periodic unit. As shown in Figure 3, 31 they consist of two regions: overlap and gap. The former comprises of two overlapping platelets while the latter is the space between two platelet ends.
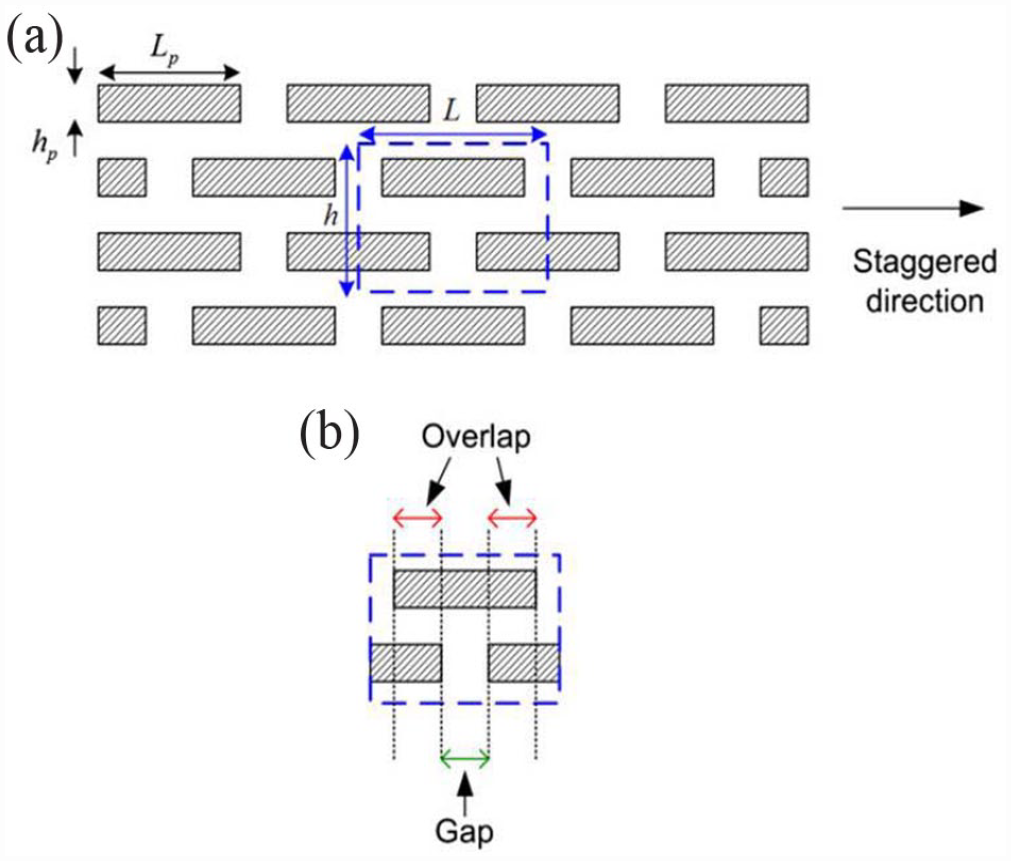
(a) Schematic of a staggered scaffold (L represents length, H represents height) (b) The two regions in a staggered microstructure: Overlap and Gap regions. The former consists of two overlapping platelets and the latter is the space between two platelet ends. 31 Copyright ©2011 Elsevier Ltd. (figure reused with permission).
Based on the geometric properties of the tissue, the researchers have arranged scaffolds in a staggered micro-array. Five types of arrays, designed based on differences between the overlapped sections, are shown in Figure 4. 22 The first layer is printed, and the second layer is printed with different offset values (0%–100%) relative to the first layer. The third and fourth layers are plotted with geometries identical to the first and second layers, respectively. 22

Schematic illustration of the design of different structured Polycaprolactone scaffold. (b–f) Cross-sectional scanning electron microscopy images of various scaffolds with various offset values. (g) Optical images of scaffolds. 22 Copyright ©2012 Royal Society of Chemistry (figure reused with permission).
Effect of the staggered structure on physical properties
In scaffolds used for bone tissue regeneration, appropriate mechanical properties that resist physiological loading and are similar to those of bone tissue, are highly desirable.33,34 Therefore, designing various scaffolds to achieve satisfactory mechanical properties is crucial. Recently, some studies have focused on characterizing the mechanical properties of the staggered structure (Table 2).9–13
Mechanical and biological properties of the analyzed scaffolds.

First, researchers discovered that the compressive strength or modulus of materials can be varied when the scaffold arrays are staggered. Yilgor et al. 13 designed four different poly(ε-caprolactone) (PCL) scaffolds as follows: basic (B), basic-offset (BO), crossed (C), and crossed-offset (CO). All the scaffolds were fabricated via the consecutive deposition of the 2D layers. The B architecture of the second layer is plotted orthogonal to the first layer. While the BO architecture showed a similar structure to B, the second layer is printed with an offset relative to the first layer. The first layer of the C architecture is plotted diagonal to the second layer. Further, the second layer of the CO architecture is printed offset relative to the first layer. The compression moduli of the BO and CO scaffolds are different from that of the B and C scaffolds, respectively. Sobral et al. designed four types of scaffolds, including those with fiber distances of 0.75 (Homog 1) and 0.1 mm (Homog 2), and two scaffolds whose pore size varied with depth, which were named Grad 1 and Grad 2. They observed that the Young’s modulus of the scaffolds with an offset (Grad 1 and Grad 2) are in between Homog 1 and Homog 2. This phenomenon might be attributed to the different porosities of those scaffolds. However, the stress-strain curve plotted for all the samples showed a non-linear correlation, indicating that compression strength modulation was also affected by the orientation and relative location of the fibers along the scaffold.18,35,36 Park et al. 9 used PCL and hydroxyapatite ( HA ) to design three types of scaffolds, including PCL (fabricated without a shifted pattern), PCL/HA (fabricated without a shifted pattern using PCL and HA), and PCL/HA/SP scaffolds (fabricated in a shifted pattern) using a bio-plotting system. They had similar porosities as follows: PCL (91.15), PCL/HA (92.01), and PCL/HA/SP (92.55). Further, compared with the PCL/HA/SP scaffold, compressive testing showed that the compression moduli of the no-shift scaffold (PCL and PCL/HA) were significantly higher. This is due to the juxtaposition of consecutive filaments of the PCL and PCL/HA scaffolds along the z-axis. Similarly, Cho et al. 11 designed PCL/nHA layer-down (Conv 1 and Conv 2), offset (Offset 1 and Offset 2), and a dual-pore Kagome scaffolds. With the same pore size and porosity, the compression moduli of scaffolds with the layer-down patterns were higher compared to those of scaffolds with offset patterns. Sun et al. 15 designed 0/45 (45° layer rotation) and 0/90 scaffolds (90° layer rotation with layer offset of 0.7 mm) with similar porosities and identical sizes. The compression test showed that the 0/90 scaffold was softer than the 0/45 scaffold. Liu et al. 21 demonstrated that the scaffold stiffness was 1.07 MPa under compression and 0.97 MPa under tension when the scaffold was designed with a gradient and staggered geometry.
Furthermore, for different offset scaffold patterns, their stress and strain curves demonstrate different trends. Cho et al. 11 prepared PCL/nHA scaffolds with offset patterns (Offset 1 and Offset 2) and experimentally evaluated their compression moduli to be 41.3 ± 3.9 and 12.0 ± 1.1 MPa, respectively. A similar finding was also reported in another similar study. 22 Yeo et al. 22 designed scaffolds with 0%, 25%, 50%, 75%, and 100% offset values using Polycaprolactone-beta-tricalcium phosphate (PCL-β-TCP ) and calculated their elastic moduli (E) for bending. For similar porosity, the elastic modulus of PT-100 (scaffolds with 100% offset values) was 7.4% higher than that of PT-0 (scaffolds with 0% offset values).
More importantly, researchers observed that the compressive strength of scaffolds were 4–12 MPa and the compressive moduli were 67–445 MPa, comparable to the human trabecular bone. 37 Lee et al. 20 designed scaffolds with identical porosity, strand width, and space, but different structures (lattice, staggered, and triangular) using PCL and poly (lactide-co-glycolide) with compression strengths of 6.05, 7.43, and 9.81 MPa, respectively. and compression moduli of 120.2, 122.3, and 178 MPa, respectively.
In addition to its mechanical properties, the staggered structure can also influence the water-uptake ability and flow velocity of the scaffold. Surface hydrophilicity plays an important role in cell attachment. Dowling et al. 38 reported that the optimum water contact angle for cell adhesion was approximately 64°. Yeo et al. 22 reported that the water uptake of the offset scaffolds was higher compared with that of no-offset scaffolds (PT-0). Moreover, PT-0 had the highest flow speed, and flow velocity gradually decreased with increasing offset value.
Effect of the staggered structure on biological properties
Scaffolds with staggered structures fabricated via 3D printing, which affect the cellular response during tissue regeneration, have been extensively studied in vitro (Table 2).9,11–13,17,18,22 Firstly, the offset patterns in the staggered structures cause a spatial-pore-size gradient to some extent. Therefore, the cell seeding efficiency and distribution are improved. Declercq et al. 17 prepared scaffolds with 0/90, 0/45, 0/90 S (0/90 with shifted patterns), and 0/90 NP (0/90 with pore size of 200 µm). Mouse calvaria pre-osteoblast cells (MC3T3-E1) were seeded into the scaffolds, and the seeding efficiency was tested after 1 day. For the conventional 0/90 design, the cell seeding efficiency was 52.2 ± 2.3%, and those of the 0/90 S, 0/45, and 0/90 NP were 66.4 ± 3.4%, 69.5 ± 3.7%, and 78.02 ± 5.1%, respectively. Thus, an obstructed architecture improved seeding efficiency. Yeo et al. 22 reported that scaffolds with offset patterns (particularly ones with 100% and 50% offset values) exhibited higher efficiencies than no-offset scaffolds as the scaffolds with offset patterns had suitable pore size and slightly complex microstructure. Moreover, 3D printed scaffolds with different offset allow homogenous cell growth. Sobral et al. 18 reported that a more even distribution of cells in Grad 1 scaffolds compared to Grad 2 scaffolds.
Secondly, staggered patterns influence cell proliferation. Yilgor et al. 13 seeded a rat bone marrow mesenchymal stem cell on PCL scaffolds. They reported the cell proliferation numbers on the B, BO, C, and CO scaffolds as235,000,262,000,222,000, and28,700, respectively, demonstrating higher cell proliferation in scaffolds with an offset. Park et al. 9 reported that human osteosarcoma (MG 63) cells on PCL/HA/SP scaffolds had a higher cell proliferation rate compared to PCL and PCL/HA scaffolds. Additionally, researchers measured the total protein content to evaluate cell proliferation after 21 days. The total protein content in the scaffolds with compacted designs was remarkably higher compared to the scaffolds with the typical 0/90 pattern. These results could be attributed to an increased total surface area for cell adhesion in scaffolds with compacted designs.
The impact of different offset patterns on cell differentiation has also been evaluated previously. Yilgor et al. 13 measured alkaline phosphatase (ALP) activities, which are early osteoblastic differentiation markers, and the scaffolds with offset designs had a high expression of ALP. Cho et al. 11 demonstrated that the value of the ALP concentration of Offset 2 was superior to that of Offset 1 at day 7, owing to the preferable pore size. Yeo et al. 22 reported that scaffolds with 100% and 50% offset values showed greater ALP activities compared with any other offset scaffold. They also exhibited increased calcium mineralization in comparison with the scaffold without offset patterns (PT-0) at day 14. Moreover, when cultured in an osteogenic medium for more than 14 days, studies showed that more compacted structure (0/45 and 0/ 90 NP) promoted ALP expression. In addition to higher ALP activities, Col1a1, the osteoclasts marker, was at its peak after 7–14 days on the scaffolds with offset designs compared to the conventional scaffold design (0/90). Further, Bglap, a late osteogenic marker, also up-regulated at day 14 or 21 in all the scaffolds with compacted architectures. 17
Effect of the staggered structure on tissue engineering
Staggered structures also affect expression levels of angiogenesis-related factors and bone regeneration. Abbasi et al. 19 designed five different scaffolds coated with PCL and calcium phosphate (CaP) using MEW, including scaffolds with pore sizes of 250 µm or 500 µm, offset.50.50 (scaffolds with pore size of 500 µm and 50% offset), and two gradient scaffolds. Subsequently, bone repair and vascularization were evaluated using a critical-size calvarial defect model. Immunohistochemistry staining showed the highest staining of vascular endothelial growth factor and a higher expression of CD105 in the offset.50.50 structures after 8 weeks. Newly formed bone growing into the scaffolds was observed via Micro-CT, which showed that new bone distribution was more on the periphery of the offset.50.50 structures rather than the central region.
Discussion
Researchers have investigated the relationship between scaffolds with staggered patterns and their mechanical properties, water-uptake ability, cell activity, and bone regeneration.10–23 Our analysis suggests that the incorporation of staggered designs can help in tuning their mechanical properties and water-uptake ability of scaffolds. Most studies have demonstrated that staggered designs have lower moduli compared to the non-staggered designs. This is because of the juxtaposition of continuous fiber along the z-axis direction in different non-staggered designs of the scaffold. As for biological properties, scaffolds with different offsets are beneficial for homogenous cell distribution, increased cell adhesion, and cell proliferation. They also promote cell differentiation compared to the no-offset structure. In particular, for cell differentiation, a more open geometry (no offset) delays differentiation in comparison with more irregular and denser internal architectures. On the one hand, scaffolds with different staggered designs have a complex inner geometry, thus increasing the number of anchorage points and contact time between the cells and scaffolds 18 along with the uptake ability in different staggered scaffold designs. 39 Sussman et al. 40 reported that a spiral-structured 3D matrix increased cell attachment due to an improved surface to volume ratio and a more complex structure. On the other hand, the flow rate, which is the speed of the cell suspension flows within the scaffolds, might be conditioned by the scaffold with different staggered designs. A low flow rate is beneficial to cell adhesion at the strand surface and connection sites, whereas cell deposition tended to take place at the bottom of the well when the flow rate was high. This result was consistent with that reported by Marín et al. 41 They identified the effect of the suspension flow rate on cell adhesion inside scaffolds using a computational model. The results indicated that static seeding had higher efficiency than dynamic perfusion. More importantly, scaffolds with different offsets offered more anchoring sites for cell extensions and created different angles, affecting the biological aspects in living cells through mechano-transduction. 39
However, other scientific studies provided conflicting results. Several studies have demonstrated that scaffolds with offset structures have no impact on cell distribution, proliferation, and differentiation. Sun et al. 15 reported difficulties in observing any apparent differences between the 0/45 and 0/90 patterns in cell distribution and proliferation. Lee et al. 20 used MC3T3-E1 cells to evaluate the cell response. After seeding on the three different scaffolds for 7 days, the optical density (OD) value was measured using the CCK-8 kit to assess cell adhesion and proliferation. Although a continuous increase in OD levels was observed in every scaffold, no significant difference was observed. Cho et al. 11 demonstrated that there was no difference in the value of the ALP concentration of Offset 2 and Offset 1 scaffolds.
As for the methods to evaluate the properties of scaffolds, FEA may be a more convenient way to assess the physical properties and compare the stress and strain that occur when cells seeded on scaffolds with different internal architectures are subjected to a mechanical load 24 apart from the actual compression test. Egan et al. 42 used FEA to quantify the relationship between elastic modulus, shear modulus, permeability, and porosity for each topology. In addition, the FEA predictions and the actual compression moduli of the fabricated scaffolds exhibited similar trends. 11 However, linear FEA presents some disadvantages when used to investigate scaffold properties. The nonlinear stress-strain behavior under scaffold compression cannot be simulated using linear FEA. 21 Liu et al. 21 used FEA and compression tests to access the mechanical properties of scaffolds with meso-structures. Based on the results, a relative error of 16.5% between the prediction and experiment was observed for the GS15 scaffolds (gradient and staggered scaffolds with strand orientation of 15) owing to the use of linear FEA.
In conclusion, the modification of the scaffold microstructure can regulate its physical and biological properties. From the standpoint of scaffold architecture, it mainly affects two aspects: surface area and flow rate. A higher surface area promotes cell activity, and flow rate can influence nutritional exchange, which is a vital pathway for cell growth and tissue regeneration. 43 This explains why a high permeability promotes cellular differentiation. 44
Future studies
The above studies suggest that scaffolds with a staggered structure can potentially be extensively applied in tissue engineering. However, most studies are at the in vitro cell-culture experimental stage. To apply staggered scaffolds in clinical therapy, in vivo studies, especially pertaining to staggered structures promoting vessel formation and bone information, are required. Further, immune response, especially macrophage polarization, serves an important regulatory role in the prognosis of surgery or scaffold implantation. 45 Guo et al. 46 designed three types of scaffolds with varying substrate moduli (5–266 MPa). Scaffolds with a substrate modulus of 24 MPa enhanced the regenerative responses. It was associated with activating Wnt/β-catenin signaling and promoting the phenotypic transition of macrophages to the M2 anti-inflammatory macrophage. Therefore, the stimulation of recruiting/polarizing immune cells via the staggered structure needs to be further studied.
With improved software and computing hardware performance, other computational methods can be used to evaluate the mechanical and cellular responses. A mechanobiological model can evaluate the designs in a pre-manufactured design phase and predict tissue growth 47 prior to in vivo and in vitro experiments. Recently, artificial intelligence (AI) has been applied to tissue engineering, and AI can provide useful information for designers in the field of regenerative medicine. 48 Qiu et al. 49 reviewed related research on AI-associated privileged scaffolds. They emphasized on many aspects of research, including updates to the knowledge on privileged scaffolds, means of identifying privileged scaffolds, and new designs to replace conventional strategies. In the future, AI may be used to perform tests on scaffold properties and could help to discover highly optimized models, reduce the workload, achieve better discoveries, and deepen our understanding of scaffolds.
Conclusion
This review summarized the current state of research on scaffolds with staggered patterns for tissue engineering applications. This type of scaffold can control its mechanical properties and affect cell adhesion, colonization, differentiation, and bone regeneration. However, only 15 related studies were truly consistent with the topic of this literature review, whose results were validated primarily via in vitro studies. Additionally, no study had the exact same model and set-up, leading to possibly inconclusive cross-comparisons. Therefore, the performance of scaffolds with offset patterns or staggered structures in vivo should be further studied in the near future. More studies are also required to clarify the effect of the scaffold microstructure on the mechanical properties and cell response. AI may aid the development of new designs for tissue engineering applications. AI can also be used to perform tests on scaffold properties and conduct in vivo and in vitro experiments. This review provides a foundation for future research directions.
Footnotes
Author contributions
X.H. literature review, performed data analysis, and drafted the manuscript. Q.Z., N.Z., and J.W. assistance in searching the literature. Y.X. revised the manuscript. Z.X. and Q.S. is the corresponding author who conceptualized the study, and edited the manuscript. All co-authors have seen the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Fundamental Research Funds for the Central Universities, China; The Innovative Talents Program, CSC (China Scholarship Council) Foundation of School/Hospital of Stomatology, Lanzhou University and Open Subject Foundation of Key Laboratory of Dental Maxillofacial Reconstruction and Biological Intelligence Manufacturing, School of Stomatology, Lanzhou University; the study funds of Stomatology, School of Stomatology, Lanzhou University.
Guarantor
Z.X. and S.Q.
