Abstract
Objectives:
Dentine hypersensitivity (DH) is a common concern in dentistry that has the potential to restrict daily activities and harm a person’s quality of life. In this study, the remineralization characteristics of nano-hydroxyapatite (nHAp) extracted from waste eggshells and fish scales were comparatively assessed.
Materials and methods:
The extraction methods used to obtain nHAp from both fish scales and eggshells are also described. The effect of the extraction process and bio-waste source on the physicochemical characteristics of the nHAp such as Ca/P ratio, functional groups, crystallinity and phase change, and surface morphology are presented in the study. The remineralization properties were evaluated using dentine models (n = 15). A field scanning electron microscope was used to evaluate the effectiveness of the dentine tubules occlusion. The percentage occluded area for all the specimens was evaluated statistically using a one-way analysis of variance (α = 0.05).
Results:
The results showed that there were variations in the physicochemical characteristics of the nHAp extracted, including the crystallinity, particle size, and surface morphology, and buffering effects against citric acids. The EnHAp extracted from eggshells had higher crystallinity, superior buffering effects, and smaller particle size compared to the nHAp extracted from fish scales, making it a more favourable material for remineralization of teeth. The statistical evidence showed that there were statistically significant differences in the dentine occluding properties measured in the nHAp (p < 0.001). The highest mean % occluded area was measured with the EnHAp group.
Conclusions:
The findings of this study provide insights into the use of bio-waste materials for the development of sustainable and effective products for oral health care.
Highlights
Dentine hypersensitivity (DH) has been a common concern.
Remineralization characteristics of nHAp extracted from eggshells and fish scales.
Physicochemical characteristics of the nHAp extraction.
EnHAp extracted from eggshells showed higher crystallinity.
Superior buffering effects and smaller particle size.
EnHAp shows more favourable material for remineralization of teeth.
Introduction
Recently, the use of bio-based materials has gained popularity and has thus attracted many researchers who aim to explore their potential for biomedical applications.1,2 The unique properties of these materials such as their non-toxicity, abundant availability, biocompatibility, and biodegradability make them favourable for use in various applications including antimicrobial coatings, 3 drug carriers 4 and adsorbents. 5 Particularly, the healthcare sector has been revolutionized by the use of bio-based materials due to the growing use in a variety of multidisciplinary fields including dentistry. 6 From a dentistry context, the use of bio-based materials has brought about tremendous growth and advantage in dental practice. For example, bio-based materials such as hydroxyapatite have been widely used in preventive and conservative dentistry,7,8 restorative and periodontal treatment. 9 This makes hydroxyapatite bio-based materials a game-changer in the treatment and management of dental and oral-facial conditions.
In the era of preventive dentistry, the literature suggests periodontal therapy and improved home care practices may result in individuals retaining their teeth for longer, potentially leading to an increase in the number of exposed dentin surfaces. 10 This could result in a condition known as dentine hypersensitivity (DH). DH, a common oral health problem, is defined by the Canadian Association of Dentistry as a ‘short, sharp pain arising from exposed dentin in response to stimuli typically thermal, evaporative, tactile, osmotic or chemical, and which cannot be attributed to any other form of dental defect or pathology’. 11
In an attempt to manage DH, a variety of topical desensitizing toothpastes, which are dentin tubule occluding materials containing materials like potassium oxalates, 12 strontium salts 13 and sodium fluoride 14 among others. The use of hydroxyapatite particles, particularly in the form of nanohydroxyapatite (nHAp), has emerged as a promising material for remineralizing dentin. This is supported by clinical evidence, which suggests that nHAp-based desensitizing agents have shown promising results in improving DH.7,8 It is important to note that nHAp is a natural component of the human tooth and bones, composing about ~90% of enamel and ~70% of dentine, 15 which thus makes nano-hydroxyapatite ideal for remineralization of damaged dentine tubules.
In our previous studies, we demonstrated the in vitro extraction of nHAp using eggshell waste as substrate and its use in occluding dentin tubules.16,17 Fish scales can also be a source of nHAp. Fish scales, which are often considered waste, have been shown to contain valuable inorganic and organic components, including collagen (40%–55%) and hydroxyapatite (15%), making them a useful alternative source for hydroxyapatite extraction.18,19 Thus, fish scale waste has tremendous unexploited potential as a biomaterial that could potentially add value to waste.
The fish market generates a significant amount of waste, which is either discarded or allowed to rot and represents a major environmental threat by acting as bacterial incubators. 20 These wastes generate large amounts of methane and other harmful gases 21 that pose a significant challenge to the environment. Thus, the creation of valuable, sustainable products from fish waste would serve as a dual-purpose strategy, enabling the extraction of nHAp from a sustainable source while also producing a valuable product. Furthermore, using the fish scale in the extraction of nHAp is important in addressing the environmental problem of waste management and towards a circular economy that supports a cleaner and greener environment. The objective of this study was to evaluate the effectiveness of nHAp derived from fish scales in occluding dentinal tubules.
In this study, we compared the characteristics of nHAp extracted from fish scales with that of eggshells. Fish scale nHAp (FnHAp) was extracted from fish scales in two separate batches. In the first batch, we use common chemicals such as sodium hydroxide (NaOH) and acetic acid in the extraction process. Thereafter, the extracted FnHAp were milled using a planetary ball-milling process. We assess the effectiveness of these materials in the remineralization of dentine tubules using the agitation process proposed by Onwubu et al. 22 To the best of our knowledge, there is little evidence in research that has comparatively compared the effectiveness of natural nHAp extracted from eggshell and fish scale wastes in occluding dentine tubules. The study hypothesized that there will be no significant difference in the occluding abilities of the nHAp extracted from eggshells (EnHAp) and fish scales (FnHAp).
Materials and method
Extraction of nHAp from eggshell and fish scale waste
Collected eggshells and fish scale waste from local outlets in South Africa were processed following the recommendations given by Onwubu et al. 23 These were heated in a furnace for 1 h at 300°C, then heated for 3 h at 900°C at a rate of 3°C min−1 to produce a white powder. This was a result of the calcium carbonate component of the eggshell waste breaking down to yield a calcium oxide powder. 16 Using the stoichiometric ratio of 1:5, 20 g of the white powder and 13.4 g of sodium triphosphate were wet-milled together in a 250 mL bowl containing 100 mL deionized water at 500 rpm for 5 h. The mixture was then centrifuged for 30 min at a speed of 1000 rpm and dried in an oven for 5 days at 40℃. Thirty stainless steel balls with a diameter of 10 mm were used in a planetary ball mill (Retsch® PM 100) during the milling process.
The extraction of nHAp from fish scales was carried out in two batches. In the first batch, the scales were immersed in 1M hydrochloric acid (HCl) for 24 h. After 24 h, they were thoroughly rinsed with reverse osmosis water and then soaked in 1M sodium hydroxide (NaOH) for an additional 24 h to remove proteins. The fish scales were then rinsed and placed in a beaker with water, which was boiled for 20 min on a hot plate at 80°C. The fish scales were dried in an oven at 80°C for 2 h, then placed in crucibles and heated in a furnace at 700°C for 2 h. In the second batch, the obtained power (FnHAp) was dry ball-milled at 500 rpm for 2 h using the same milling parameters as explained above to obtain milled fish scale nHAp (mFnHAp).
Characterization
Fourier transform infrared spectroscopy analysis
The functional groups that were present in the prepared powders were determined using a Perkin Elmer Universal ATR. A background investigation was initially conducted before scanning. A small quantity of the prepared sample powders was then placed in a sample holder and scanned between 400 and 4500 cm−1 with a resolution of 4 cm−1.
X-ray diffraction analysis
To track any potential changes in the crystallinity of the prepared powders, an X-ray diffraction (XRD) analysis was conducted. The patterns were captured using a diffractometer (D8 Advance BRUKER AXS instrument Germany instrument); Cu-Ka radiation (lKa1 = 1.5406) the patterns were analyzed between 10 and 80 (2 theta). The parameters used were 40 kV of voltage, 40 mA of current and 0.5 s of pass time.
High-resolution transmission electron microscopic analysis
In a high-resolution transmission electron microscope (HRTEM); (model: Philips CM 120) operating at 120 kV, the size, shape, and distribution of the prepared powders were investigated. Before HRTEM observation, tiny amounts of the powders were each dispersed in 10 mL of ethanol and sonicated at 10 kHz for 10 min. Finally, using a Leica microtome (South Africa), thin cross-sections of cryo-microtomed specimens were created and placed on carbon copper grids measuring 100 mm × 100 mm.
Elemental analysis of samples
The calcium to phosphate ratio of the samples was evaluated using the Energy Dispersive X-ray (EDX) technique. The findings were presented as a percentage weight of all the elements. Due to the interest in estimating the calcium to phosphate ratio that exhibits the component of the tooth’s hydroxyapatite, data for carbon, oxygen, phosphorus and calcium are presented.
pH test
A pH metre (BOECO, BT-675, Germany) with a temperature sensor was used to monitor the pH value. About 1.5 g of each of the sample powders (EnHAp, FnHAp and mFnHAp) was put into a beaker containing 50 mL of deionized water. This mixture was stirred continuously for 10 min at a speed of 600 r min−1, with pH readings taken every minute.
Dentin tubule occlusion test
Recently extracted anterior bovine-enamel teeth (15) were acquired at a slaughterhouse in Durban, KwaZulu Natal, South Africa. The bovine teeth were washed thoroughly and sterilized in a solution of 10% chloroxylenol. Using a low-speed water cooling diamond saw, dentin discs measuring 5 mm × 5 mm × 1 mm were cut below the dentin-enamel junction. The generated dentin disc was then wet-processed for 60 s using silicon carbide polishing sheets (600–1000 grits). Before creating the sensitive tooth model, the dentin discs were placed in a resin (AMT composite, South Africa). Afterwards, to open the dentine tubules, the specimens were submerged for 5 min in a citric acid solution with a concentration of 8 wt%. As shown in Table 1, the specimens were randomly allocated into four groups (n = 5) and treated using agitation techniques described by Onwubu et al. 16
Sample groups.

n = number of bovine teeth.
SEM evaluation of the occluded specimen
The occluded dentin before and after treatment was evaluated under controlled air conditions using an SEM (field emission, Carl Zeiss) operating at 20 kV. A thin, electrically conductive gold coating was applied to the surface to prevent an electrostatic charge build-up before SEM observation. With the aid of the ImageJ software (National Institute of Health, USA, http://imagej.nih.gov/ij) the ratios of both the occluded and the opened tubules were calculated. The ratios were determined by dividing the area of blocked tubules by the area of all tubules using 10,000 magnification photos (n = 5).
Analysis of the data
To study the mean values of the occluded area ratio in the SEM investigation, a one-way analysis of variance (ANOVA) was conducted using statistical software (IBM SPSS Statistics v28; IBM Corp). Afterwards, a multi-comparison test with Bonferroni correction (=0.05) was performed. To determine the statistical significance of differences between the groups. The results were considered statistically significant if the p-value was less than 0.05. The findings of the SEM investigation were presented as the mean ± standard deviation of the occluded area ratio.
Results
Characterization of the extracted hydroxyapatite samples
The image in Figure 1(a)–(c) shows the characteristic absorption peaks of the extracted nHAp powders as revealed by the FTIR analyses. The absorption bands typical of hydroxyapatite characteristics were observed around ~1000 and ~560 cm−1 for all the samples, respectively. This is due to the asymmetrical stretching and asymmetric bending vibrations of the PO43− group, as reported in literature.16,24 While the carbonate bands at 1450 cm−1 were evident in all the samples (Figure 1(a)–(c)), the intensity was much more prominent in the EnHAp (Figure 1(c)). The presence of the carbonate group in all the samples is attributable to out-of-plane bending mode and asymmetric stretching of CO32− ions in the structure of the nHAp produced. 17 The reason for the carbonate peak found in all the samples may be associated with the substitution of the PO43− ions in the structure of nHAp.24,25 The hydroxyl group (O–H) bending was observed at around ~3750 cm−1, identified by a small absorption peak in samples shown in Figure 1(a) and (b). In contrast, the absorption band for EnHAp (Figure 1(c)) was very visible around 3750 cm−1 and it is attributable to the OH stretching. 17 Furthermore, the broadband around ~3450 and 3000 cm−1, and also the sharp band at 1600 cm−1 associated with water molecules 16 were evident in EnHAp samples alone (Figure 1(c)). The probable cause for the absence of water molecules in Figure 1(a) and (b) could be related to the alkaline hydrolysis and mechanochemical processes involved in the extraction of nHAp from fish scale.

FTIR spectra of: (a) FnHAp, (b) mFnHAp and (c) EnHAp.
The image in Figure 2(a)–(c) shows the XRD patterns of the extracted nHAp. The diffraction angles marked at 25.8° (002), 31.8° (121), 32.2° (112), 32.9° (030), 39.8° (310), 46.7° (222) and 49.4° (123), observed in all samples, are indicative of nHAp, according to the international standard (JCP2-76-0694). The peak observed at 29.4° corresponds to calcite, while the peak around 34.1° suggests the presence of Portlandite (20). The crystallinity value of EnHAp (82.19%) was found to be higher when compared to FnHAp (67.48%) and mFHAp (67.22%). The close values in crystallinity between FnHAp and mFnHAp suggest that milling did not impact the crystallinity of the nHAp extracted from fish scales. The Gaussian plot was used to determine the peak area, and the formula below was used to determine the crystallinity.


XRD pattern of: (a) FnHAp, (b) mFnHAp and (c) EnHAp.
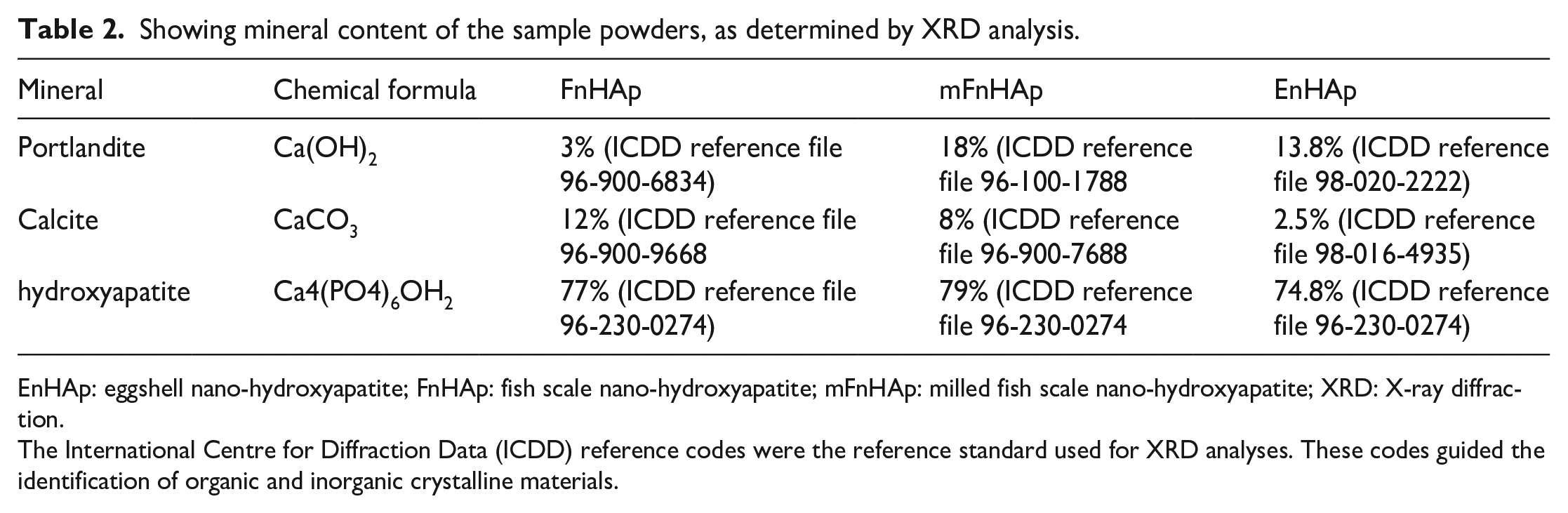
The mineralogical content in the different extracted nHAp (FnHAp, mFnHAp and EnHAP) obtained from the XRD analysis is further represented in Table 2. The analysis revealed differences in the mineralogical composition of the key minerals identified by the XRD analysis. The highest composition of hydroxyapatite (79%) and portlandite (18%) were measured in mFnHAp, respectively. By contrast, calcite content was higher in FnHAp (12%) compared to mFnHAp (8%) and FnHAp (2.5%).
Showing mineral content of the sample powders, as determined by XRD analysis.
EnHAp: eggshell nano-hydroxyapatite; FnHAp: fish scale nano-hydroxyapatite; mFnHAp: milled fish scale nano-hydroxyapatite; XRD: X-ray diffraction.
The International Centre for Diffraction Data (ICDD) reference codes were the reference standard used for XRD analyses. These codes guided the identification of organic and inorganic crystalline materials.
The highest concentration of hydroxyapatite in mFnHAp indicates that milling the fish scales improved the mineral content compared to the initial extracted FnHAp. The presence of portlandite in mFnHAp suggests that the milling process could have led to the formation of new phases from the reaction of Ca(OH)2 with CO2 from the atmosphere. The difference in calcite content between FnHAp and mFnHAp highlights that the milling process might have caused the loss of some of the calcite content during the process. EnHAp showed the lowest content of hydroxyapatite (70%) compared to the other two samples, which suggests that the extraction process using an alkaline solution might have resulted in the loss of some of the hydroxyapatite content. The presence of calcite (2.5%) and portlandite (26%) in EnHAp indicates that the extraction process using an alkaline solution might have led to the formation of new minerals in the sample.
The elements present in the sample groups (FnHAp, mFnHAp and EnHAp) were analyzed by EDX and the results are shown in Table 3. The elements found in the samples were carbon (C), calcium (Ca), oxygen (O), phosphorus (P) and sodium (Na) with the presence of sodium being only evident in mFnHAp and EnHAp. All sample groups had high levels of calcium and phosphorus in the form of carbonate and phosphate. The ratio of calcium to phosphorus (Ca/P) was found to be different for each sample, with mFnHAp having a ratio of 1.9, slightly higher than the 1.67 ratio found in human bones and teeth. The ratios for FnHAp and EnHAp were slightly different from the stoichiometric ratio of 1.67, which is likely due to the presence of trace elements like sodium and magnesium in natural nHAp.26,27
Elemental analysis of the samples.

The HRTEM image in Figure 3(a)–(c) shows the surface morphology of the sample powders. There were visible differences in the surface morphology of the nHAp extracted from eggshells and fish scales. The image in Figure 3(a) and (b) shows that the nano-hydroxyapatite from fish scales (FnHAp and mFnHAp) particles presented an irregular morphology. After milling at 500 rpm for 2 h, the mFnHAP (Figure 3(b)) showed an increase in surface smoothness in comparison with FnHAp (Figure 3(a)). By contrast, an irregular-rod-like structure was observed for the eggshell nano-hydroxyapatite (EnHAp). The particles also appeared to assemble into short chains that stuck together into clusters (Figure 3(c)). In addition, the particle size of the sample powder measured using Image J software showed that the EnHAp had a mean diameter of 11.6 ± 2.7 nm. By contrast, the mean diameter measured for FnHAp and mFnHAp were 190 ± 81.8 and 74.6 ± 33.7, respectively.

HRTEM micrograph of: (a) FnHAp, (b) mFnHAp and (c) EnHAp.
Figure 4 shows the pH of the sample powders in deionized water and citric acid solution with initial pH of 2.06. The findings indicated that EnHAp had the highest mean pH value in deionized water (13.09), whereas the mFnHAp had the lowest value (10.77). All the sample powder had alkaline characteristics that may be attributable to the elevated calcium content. EnHAp had the best buffering characteristics when compared to both FnHAp and mFnHAp from fish scales in the sample powder study using the citric acid solution at pH 2.06 since it effectively neutralized the pH of the citric acids (Figure 4).

Buffering characteristics of samples.
Additionally, it was also noted that the EnHAp showed the greatest buffering capacity as the slope of the EnHAp was the smallest among the sample groups. The results showed that EnHAp was more effective in maintaining the stability of the pH in the citric acid solution compared to FnHAp and mFnHAp. The ability to buffer pH is important in several biological processes and the results suggest that EnHAp may have potential applications in the biomedical field.
Dentine tubule occluding characteristics
FESEM was used to examine the morphological alterations of the occlusion of the dentine tubule before and after treatment (agitation). The samples treated with EnHAp (Figure 5c2) and mFnHAp (Figure 5b2) had more occluded areas than the samples treated with FnHAp (Figure 5a2). All samples show visible evidence of dentine tubule occlusion after agitation. The nanoparticles can be seen on the surface of the dentine specimens, and it can be inferred that the nanoparticles have entered the dentine tubules and have caused the occlusion. The results indicate that the EnHAp and mFnHAp were more effective in occluding the dentine tubules compared to FnHAp, which could be due to the difference in size and surface morphology of the particles. The smaller size and the rod-like structure of the EnHAp particles might have allowed for easier penetration into the dentine tubules, leading to more effective occlusion. On the other hand, the irregular shape of the FnHAp particles might have hindered their penetration into the tubules, resulting in less effective occlusion.

Remineralization test: (a) FnHAp, (b) mFnHAp and (c) EnHAp (1 represents after agitation).
The mean % occluded area of the dentine tubules for the treated specimens is given in Table 4. The ANOVA value shows that the total mean % ratio of the dentine tubules occluded area for the specimens treated with sample powders (EnHAp, FnHAp and mFnHAp) was statistically significant (p < 0.001). The results indicate that EnHAp had the highest % mean occluded area (78.2 ± 1.5%) while the lowest was measured for the FnHAp group (59.7 ± 1.7%). According to the results of the Bonferroni test, the percentage of tubules occluded for the EnHAp group was statistically higher than that of the FnHAp group and the mFnHAp group (P = 0.002 and 0.001, respectively). In comparison to the FnHAp group, the % occluded area measured for the mFnHAp group was higher (p = 0.001).
ANOVA test comparison of the occluded area.
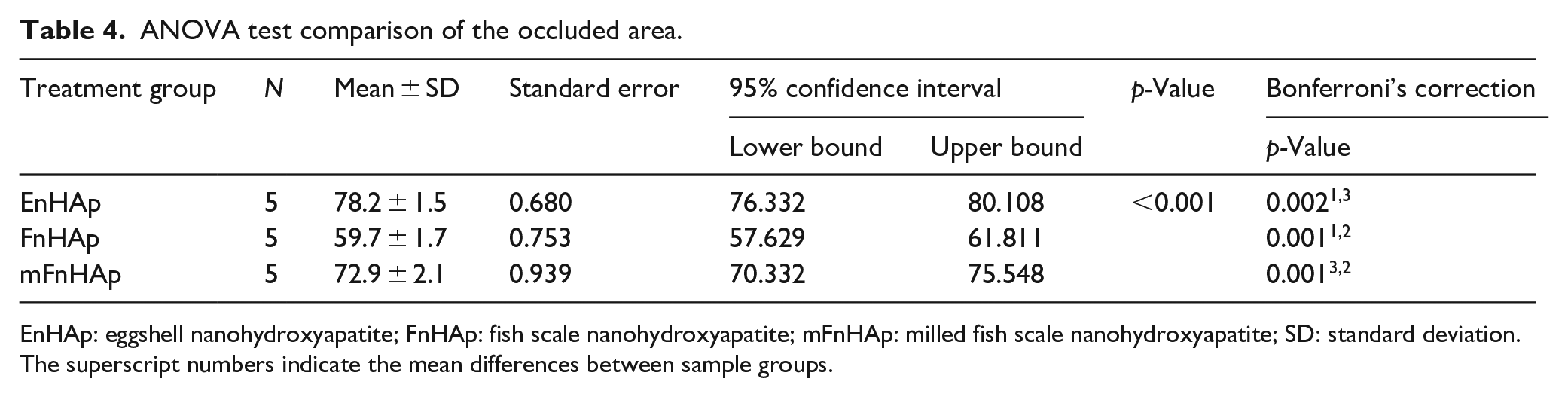
EnHAp: eggshell nanohydroxyapatite; FnHAp: fish scale nanohydroxyapatite; mFnHAp: milled fish scale nanohydroxyapatite; SD: standard deviation.
The superscript numbers indicate the mean differences between sample groups.
Discussion
The clinical condition of DH has the potential to impede and restrict daily activities, which negatively impacts the person’s quality of life. 28 According to Balhuc et al, 29 the use of more complex products and combinations between nano-hydroxyapatite (nHAp) and various compounds is an innovative method for significantly reducing DH and remineralizing dentinal tubules. While synthetic nHAp is commonly used in biomaterial applications, the need for environmental sustainability products has created a market for bio-waste materials. In this present study, we comparatively assessed the remineralization characteristics of nHAp extracted from waste eggshells and fish scales. The results showed slight variations in the physicochemical characteristics of the nHAp extracted (Figure 1). While the nHAp from eggshells had higher crystallinity, the mineralogical content of hydroxyapatite was higher in the fish scale nHAp (Table 2).
The surface morphology also revealed variations between the nHAp extracted from eggshells and fish scales (Figure 3). The study results showed that milling the nHAp extracted from the fish scale does not negatively impact the physicochemical characteristics, but the milled fish scale nHAp (mFnHAp) had a smoother surface morphology and reduced particle size after milling (Figure 3). This may be due to the effect of the mechanical energy during milling, which could have triggered a transition from aggregated FnHAp to a smoother mFnHAp (Figure 3). Another study reported that milling stimulates transitions of substances due to the impact of mechanical energy. 30 Based on the study results, it can reasonably be assumed that the physicochemical characteristics of the nHAp such as the crystallinity, particle size and surface morphology vary due to the source of bio-waste, method of extraction, pH and temperatures. 31
Furthermore, the results indicate that the buffering characteristics of eggshell nHAp (EnHAp) against citric acids were superior to those measured for nHAp extracted from fish scales (Figure 4). The reason for EnHAp having higher buffering effects compared to FnHAp and mFnHAp could be due to the differences in the crystal structure and particle size. The irregular rod-like structure and smaller particle size of EnHAp may provide a larger surface area for reaction with the citric acid, allowing it to effectively neutralize the pH of the solution. The higher surface area could lead to a greater number of surface-active sites, resulting in more efficient buffering effects. It is also possible that differences in the chemical composition and the presence of trace elements in each sample group could play a role in the buffering characteristics of the materials. This indicates that EnHAp provides better protection against enamel demineralization from acidic substances by effectively neutralizing citric acid. The implication for oral health are that EnHAp provides high bioavailable calcium that is crucial for tooth remineralization.17,32 This is supported by the FESEM image in Figure 5, which shows that EnHAp had the most increase in dentine tubules occlusion. In agreement with Tschoppe et al, 33 this means that the higher pH values of the nHAp slurries favour tooth remineralization. Statistical results indicate a statistically significant difference in the treated dentine specimens (p < 0.001), and the formulated study hypothesis was rejected based on the results obtained.
Overall, the EnHAp group had the highest mean % occluded area of the dentine tubules (Table 4). The plausible difference in the dentine tubules observed may be due to the physicochemical characteristics of the nHAp extracted. This aligns with Kong et al 34 who suggest that the performance of nHAp is influenced by its crystallinity, particle size, and surface morphology. This means that the smaller particle size of EnHAp and the higher pH values favour its remineralization characteristics over the fish scale nHAp. Additionally, the particle sizes measured for the nHAp extracted are smaller than the size of tubules close to the dentine-enamel junction, which is around 3–5 μm. 35 This indicates that smaller nHAp particles can easily be attached to dentinal surfaces and occlude exposed dentine tubules. This may explain the dentine tubule occlusion in all the tested nHAp from both fish scales and eggshells (Figure 5). Another possible reason is the polarity and the large surface proportion of the atomic number of nHAp, which allows the particles to bind to collagen and hydroxyapatite in dentine. 36
Conclusions
The study we have compared the remineralization characteristics of nano-hydroxyapatite (nHAp) extracted from waste eggshells and fish scales. The results showed that there were variations in the physicochemical characteristics of the nHAp extracted from the two sources. The EnHAp extracted from eggshells had higher crystallinity and superior buffering effects against citric acids compared to the nHAp extracted from fish scales. The smaller particle size and higher pH values of EnHAp favoured its remineralization characteristics. The results showed that EnHAp had superior buffering effects against citric acids and higher mean % occlusion of dentine tubules compared to FnHAp and milled FnHAp (mFnHAp). The ANOVA results also indicated that there was a statistically significant difference (p < 0.001) in the dentine tubule occlusion between EnHAp and FnHAp. These findings highlight the potential of nHAp from natural bio-waste as a low-cost alternative for dental applications and warrant further research to optimize its use in clinical practice.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The support offered by the National Research Foundation of South Africa (Grant Number: 129492) is acknowledged.
Data availability statement
On reasonable request, the corresponding author will provide the datasets generated during and/or analysed during the current study.
