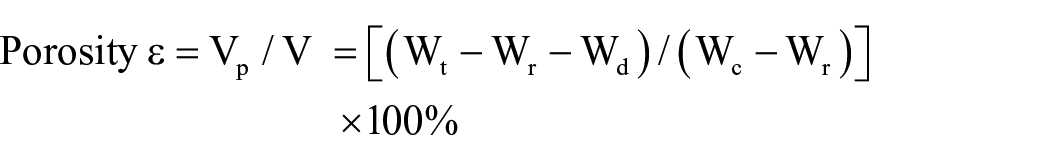Abstract
Both collagen fibres and nanohydroxyapatite crystals have anisotropic magnetisation, which allows them to be oriented by a high magnetic field. Highly oriented nanohydroxyapatite/collagen composites were prepared using a high magnetic field combined with in situ synthesis. These highly oriented composites were investigated and compared with conventional composites. The results showed that the collagen fibres in the magnetically induced highly oriented nanohydroxyapatite/collagen composites had a preferred orientation and smaller molecular spacing, while the nanohydroxyapatite crystals were tightly adhered along the collagen fibre surface. The magnetically induced composites exhibited superior resistance to swelling and degradation along with high compressive strength. This artificial composite, with a structure and composition similar to natural bone, represents a new idea for improving materials for vertical or horizontal bone augmentation.
Introduction
It is common to see alveolar ridge defects caused by inflammation, tumours, trauma, congenital malformations and many other causes, and these defects are a difficult problem for doctors who specialise in clinical dental implant restoration. 1 With the principle of ‘restoration-guided surgery’ and the theoretical system of implant aesthetics, the amount of bone in the alveolar ridge of the upper anterior region of the patients becomes increasingly critical. 2 Therefore, the development of block bone materials that can maintain a certain spatial shape while having good biocompatibility is a topic of increased research interest. Although a wide variety of bone grafting materials has been applied to alveolar ridge bone defects, there are limitations in the clinical application of autologous, allogeneic and xenogeneic bone3,4; moreover, synthetic alloplastic block grafts have their own unique advantages.
Natural bone is a biomineralized substance consisting mainly of hydroxyapatite (HAp) and type I collagen, with a complex hierarchical structure. Currently, synthetic alloplastic grafts are based on hydroxyapatite/collagen, which are mostly fabricated by the coprecipitation method, 5 comixing method 6 and in situ synthesis. 7 However, regardless of the chosen method, the composites suffer from low mechanical strength.
It has been shown that nanohydroxyapatite (nHAp) crystals attach to collagen fibres and that collagen fibres and the C-axis of nHAp have a preferred orientation, which is parallel to the maximum load direction of natural bone. 8 Therefore, if the natural bone structure can be simulated and a composite with a preferred orientation can be developed, the mechanical properties of the nHAp/collagen composite could be greatly improved. In recent years, researchers have studied the behaviour of some nonmagnetic materials in high magnetic fields more extensively 9 ; for example, polymer networks,10–12 colloids 13 and the crystalline regions of molecular chains 14 can be oriented by high magnetic fields. It has been shown that both collagen fibres and nHAp crystals have anisotropic magnetisation, with the smallest magnetisation in the long-axis direction of the collagen fibres and along the C-axis of nHAp crystals.15,16 Based on these theories, Wu et al. 17 prepared a unidirectionally oriented nHAp/Col composite using a high magnetic field combined with a comixing method under slow rotation conditions. Chen et al. 18 also prepared a highly oriented tilapia fish-scale collagen hydrogel with a high magnetic field and subsequently crosslinked it with ethyl-dimethylcarbodiimide (EDC). It was found that the viscoelasticity of the magnetically induced collagen hydrogel significantly improved.
In this study, a magnetically induced highly oriented nHAp/collagen composite (MI-NHAC) from bovine type I collagen inspired by the structure of natural bone and previous studies was prepared using the self-assembly technique in a high magnetic field. Then, its microstructure, mechanical properties and biocompatibility were compared with those of the nHAp/collagen composite (NHAC) prepared without a magnetic field.
Materials and methods
Materials
Bovine Achilles tendon type I collagen was kindly supplied by Sannie Bioengineering Technology Co., Ltd. (Tianjin, China). CaCl2∙2H2O and anhydrous ethanol were purchased from Chemical Reagent Corp. (Beijing, China). Phosphate-buffered saline, 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride, MEM alpha modification [HyClone], foetal bovine serum [HyClone], penicillin-streptomycin solution [HyClone], 0.25% trypsin solution [HyClone], type I collagenase, 2.5% glutaraldehyde solution, a CCK-8 kit and a live/dead cell double staining kit were purchased from Xintai Biotechnology Development Co., Ltd. (Shaanxi, China).
Fabrication of MI-NHAC and NHAC
Bovine Achilles tendon type I collagen was dissolved in phosphate-buffered saline (PBS) solution to form a preassembled collagen phosphate macromolecule template with a collagen content of 6.7 mg/mL and a phosphate concentration of 0.06 mol/L. A certain proportion of calcium chloride dihydrate was dissolved in ultrapure water to form a soluble calcium ion solution with a Ca2+ concentration of 0.1 mol/L. A 3000 mL beaker was used as the reactor for the self-assembly system. First, 300 mL of ultrapure water was added, and the temperature of the liquid was maintained at 25 ± 2°C. Second, the two prepared solutions were sequentially added to the reactor. The pH of the mixed solution was 8 ± 0.1. The samples were then maintained at 25°C for 12 h with or without a 10 T high magnetic field. In the experiment with the magnetic field, the reactor was placed on a slowly rotating platform, with the rotation axis perpendicular to the magnetic field direction. 15 In the experiment without the magnetic field, the reactor was also mechanically rotated under the same condition. The supernatant was filtered off, and the resulting precipitate was washed three times with ultrapure water and stored at 4°C for 12 h. The samples were then crosslinked in 0.8% 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC) solution at a temperature of 25°C for 4 h. After dialysis, the samples were lyophilised and isostatically pressed at 200 MPa. The specific preparation process is shown in Figure 1.

Schematic diagram showing the preparation of materials.
Characterisation
Microscopic investigation
Scanning electron microscopy (SEM) was used to examine the micromorphology of the scaffolds before the other studies. The composites were made in the form of blocks with flat surfaces. These blocks were mounted on an aluminium plate, coated with platinum and then observed under a scanning electron microscope (Hitachi S-4800, Japan).
Fourier transform infrared (FTIR) spectroscopy
The composites were prepared as cylindrical blocks with a diameter of 4 mm, a height of 2 mm and a flat surface. Additionally, a pure collagen scaffold (Col) was prepared as a control test group. The samples were tested in attenuated total reflection mode using an FTIR spectroscopy instrument (Nicolet iS 50, USA). The FTIR spectra were collected from 4000 to 500 cm−1 with a detection bandwidth of 2 cm−1.
X-ray diffraction (XRD)
The aforementioned samples were fixed on the observation platform and tested using an XRD instrument (D8 Advance, Germany) at a working voltage of 40 kV, an angle range of 10°–80° and a rate of 10°/min. The phases were investigated by comparing the experimental results with the standard diffraction card of hydroxyapatite (PDF#09-0432).
X-ray photoelectron spectroscopy (XPS)
The two samples were separately placed in an X-ray photoelectron spectrometer (Kratos AXIS UltraDLD, UK) to analyse the type and content of elements contained in the composites and calculate the calcium to phosphorus ratio.
Thermogravimetry-derivative thermogravimetry (TG-DTG)
In this analysis, 10 mg of each sample was placed into a simultaneous thermal analyser (STA 449 F3 Jupiter®, Germany) and heated from 25°C to 800°C at a rate of 20°C/min under the protection of N2.
Determination of porosity
We measured the porosities of the two samples by Archimedes’ principle. 19 Three parallel experiments were set up for each group. The porosity of the two composites was measured at 25°C and was determined using the following formula:
where Wc is the weight of the pycnometer after the composite was filled with ethanol, Wd is the dry weight of the composite, Wt is the total weight of the pycnometer after the composite was immersed in ethanol and Wr is the residual mass after the composite was removed.
Physical performance
Swelling properties
The original mass of the composite W0 was recorded using an analytical balance after immersion in PBS solution and placement in a 37°C incubator. The samples were tested at various time intervals (0, 0.5, 2, 6, 12, 24, 48 and 72 h) after the water was removed from the surface of the composites using clean filter paper. The mass of the composite at the selected times was recorded as Wt. The swelling ratio was determined as (Wt − W0)/W0.
Mechanical properties
The mechanical properties of the composites were evaluated according to ISO standards (ISO3386-1-2010). The composites were prepared as cylindrical blocks with a diameter of 8 mm and a height of 4 mm and prewetted with PBS solution in a 37°C incubator for 24 h. An electronic universal material testing machine (MTS CMT6103, USA) was used to test the uniaxial compression performance and the isostrain cyclic compression performance of the composite sites, and the compression speed was set at 0.3 mm/min.
Biological properties
In vitro degradation
We used type I collagenase (100 U/mL) to study the degradation properties of the composites in vitro. After sterilisation with Co-60 irradiation, the samples were weighed, added to tubes containing 2 mL of enzyme solution, and placed in a 37.0°C incubator. The samples were removed after 1, 2, 3 and 4 days, rinsed gently two times with PBS solution, lyophilised for 24 h, and weighed. Wr = W1/W0, where W0 and W1 are the weights of the composites before and after enzymolysis, respectively, was used to compute the residual weight ratio. Three parallel experiments were set up for each group.
Cytotoxicity assay of the composites
The cytotoxicities of the composite extracts were evaluated according to ISO standards (ISO10993.12-2012). After Co-60 irradiation sterilisation, the composites were placed into tubes with fresh culture medium at a ratio of 100 mg/mL and placed in a 37°C incubator for 24 h. Then, the composites were removed from the tubes, the leaching solution was reserved, and the samples were stored at 4°C in the refrigerator until use. Prior to the other steps, MC3T3-E1 cells were cultured on 96-well plates (100 μL/well) at a density of 3.5 × 104 cells/mL and incubated at 37°C for 12 h. After that, the culture medium was removed, and the composite extracts and fresh culture medium (blank group) were applied to various wells (100 μL/well) for incubation for 1, 3 and 5 days. Another cell-free group was set up with the addition of fresh culture solution as zeroing wells. After incubation, the cells were incubated for 2 h in a 37°C incubator with extracts containing 10% CCK-8 reagent. A microplate reader was used to measure absorbance at 450 nm. For each group, three parallel experiments were set up. The relative growth rate (RGR) was determined using the following formula:
where As is the absorbance of the composite extract group, Ab is the absorbance of the cell-free group and Ac is the absorbance of the blank group.
Cell attachment and live/dead cell staining analysis
A pure collagen scaffold group was also introduced as a control group to investigate the impact of the composites on the development of MC3T3-E1 cells. In this part of the experiment, the scaffolds were cut into cylindrical blocks with diameters of 8 mm and heights of 2 mm after Co-60 irradiation sterilisation. Following that, these cylindrical blocks were inserted into 48-well culture plates. To prewet the samples, fresh culture media (1 mL/well) was added to the wells, which were then incubated in a 37°C incubator for 24 h. Afterwards, the culture medium was removed, and MC3T3-E1 cells were evenly inoculated at a density of 1 × 105 cells/mL onto each scaffold (50 μL/well). After 4 h of cell adhesion, more media (450 μL/well) was added, and the cells were cultured on scaffolds for 1, 3 and 5 days. After 3 days of incubation, portions of each sample were fixed in 2.5% glutaraldehyde, dehydrated in various concentrations of ethanol (30%, 50%, 70%, 80%, 90%, 95% and 100%), dried under vacuum and investigated by SEM.
To assess the cell growth on the samples, the remaining portions of the samples were stained with a live/dead cell double staining kit. A laser confocal microscope (Olympus FV1000, Japan) was used to observe the stained live and dead cells. Three parallel experiments were set up for each group.
Quantitative analysis of cell proliferation
Cell proliferation on the composites was analysed quantitatively via the CCK-8 assay. MC3T3-E1 cells were evenly inoculated on the MI-NHAC, NHAC and Col scaffolds according to a method described in previous work. After the cells were cultured on the scaffolds for 1, 3 and 5 days, all the cell-seeded scaffolds were transferred to new 48-well plates. Then, the samples were incubated with fresh culture medium (500 μL/well) containing 10% CCK-8 reagent for 2 h in a 37°C incubator. The culture medium from each sample was partially transferred to 96-well plates (100 μL/well), and the optical density (OD) was measured at 450 nm using a microplate reader. The experiment was replicated for three parallel samples.
Statistical analysis
All statistical analyses were carried out using SPSS software (version 26), and p < 0.05 was considered statistically significant. Data were processed and graphed using GraphPad Prism 8 software as well as Origin 2021 software.
Results and discussion
Characterisation of the composites
In previous studies, the changes in collagen fibres under a high magnetic field have been verified by a number of traditional experiments.17,18 In this study, the precursor state of clinical application of the composites after isostatic pressing was observed by SEM to explore the difference in their micromorphology. The SEM images showing the microscopic morphology of NHAC and MI-NHAC are shown in Figure 2. SEM revealed that there were significantly more pores on the surface of the NHAC than there were on the MI-NHAC. The pores in NHAC materials were dense and uniform with a pore size range of 50–200 μm, while the pores of the MI-NHAC materials varied in size from 50 to 800 μm. Thus, the pore sizes of the composites were appropriate for bone tissue engineering applications; this finding was in agreement with prior relevant investigations.20,21 The performance of the two materials was compared at 50×, 300×, 5000× and 30,000×. By comparing the field of view at 300×, it was found that the collagen fibres were arranged in a disordered manner in NHAC, while they show anisotropy in MI-NHAC. On the other hand, by comparing the field of view at 30,000×, it was observed that the nHAp crystals were arranged haphazardly on the collagen fibres of the NHAC, while they were more uniformly and densely arranged on the collagen fibres in the MI-NHAC. By combining the differences in the microscopic performance of collagen fibres and nHAp crystals in both materials, the effect of a high magnetic field on the microstructure of the materials was clearly observed.

SEM images showing the microscopic morphology of NHAC and MI-NHAC. (a–d) Microscopic representation of NHAC at magnifications of ×50, ×300, ×5000 and ×30,000. (e–h) Microscopic representation of MI-NHAC at magnifications of ×50, ×300, ×5000 and ×30,000.
FTIR spectroscopy is an important means of characterising the structure of a substance because it reflects the characteristic chemical bonds of substances. Figure 3(a) shows the spectra of NHAC, MI-NHAC and the Col scaffold tested under the same conditions. The characteristic peak of amide A at 3306 cm−1 represents the N-H stretching vibration. By comparing the three materials, it was found that the NHAC and MI-NHAC materials were relatively close to the low wavenumber region at this peak, indicating that crosslinking reactions have occurred within the collagen, increasing the hydrogen bonding content and resulting in a more ordered collagen molecular structure. 22 The peaks at 1639, 1538 and 1236 cm−1 belong to amide I, amide II and amide III, respectively, and these peaks are mainly generated by the C=O stretching vibrations, N-H bending vibrations and C-N stretching vibrations. 23 The amide A, amide I and amide III peaks are highly sensitive to the triple helix configuration of collagen molecules and are generally used as a basis for determining the presence or absence of the triple helix structure in collagen molecules. 24 The figure shows that the amide A, I and III peaks of the NHAC and MI-NHAC composites have significantly lower absorption than those of the uncrosslinked Col scaffold materials. This result indicates that the two composites retain the triple helix structure of the collagen molecules to some extent, while the nanohydroxyapatite crystals are bound to the collagen fibres. MI-NHAC and NHAC show two characteristic peaks corresponding to PO43− at wavelengths of 1020 and 554 cm−1 in the figure, which are generated by P-O(ν3) bending vibrations and P-O(ν4) asymmetric stretching vibrations, respectively; this result also indicates the presence of hydroxyapatite components. 25 The amide I/II peak area ratio of MI-NHAC is lower than that of the NHAC composites because the amide I/II peak area ratio of the composites is influenced by the collagen alignment direction,26,27 resulting in a corresponding change in the IR spectrum.

Characterisation of MI-NHAC and NHAC: (a) FTIR spectra, (b) XRD patterns, (c) XPS spectra and (d and e) TG-DTG curves of MI-NHAC and NHAC.
XRD is a common tool used primarily to analyse the crystal structure of materials. Figure 3(b) shows the XRD patterns of the two composites, which were compared to the standard diffraction card of HAp (PDF#09-0432). The results show that the diffraction patterns of MI-NHAC and NHAC correspond to multiple crystalline diffraction peak positions in the standard card, indicating that the inorganic phase of both composites is mainly hydroxyapatite. 28 In addition, both composites show a ‘bun’ diffraction peak near 2θ = 19.8°, which is caused by diffuse reflections and represents multiple structural layers within the collagen molecules. 24 The XRD patterns of the two composites have broadened peaks and partially overlapping diffraction peaks, which are similar to the diffraction peak shapes of natural bone crystalline surfaces, indicating the low crystallinity of nHAp in both composites. In comparison, MI-NHAC has narrower diffraction peak shapes and higher peak intensities for the (211) and (112) crystal planes, which may be attributed to the directional alignment and adhered nHAp crystals that result from being in a high magnetic field; thus its inorganic phase exhibits higher crystallinity.
XPS techniques can be used to measure the elemental composition of materials and to evaluate the chemical and electronic states of the elements. As shown in Figure 3(c), both MI-NHAC and NHAC contain C, O, N, P and Ca. The calculated Ca/P ratios of NHAC and MI-NHAC are 1.60 and 1.57, respectively; both of these values are relatively close to the Ca/P ratio of natural bone. The slightly lower Ca/P ratio of MI-NHAC may be due to the reduced spacing of the collagen fibres after directional alignment, resulting in a slight decrease in the amount of Ca2+ that binds to the collagen phosphate macromolecular template for mineralisation in the same amount of time.
The TG-DTG results of the NHAC and MI-NHAC composites are shown in Figure 3(d) and (e). After treatment at 800°C under a N2 atmosphere, the specific gravity values of the residues of the two composites were found to be 54.63% and 56.13%, respectively; these values are close to the inorganic content in natural bone. 8 Two heat absorption decomposition peaks were observed near 83°C and 346°C for both composites. The former corresponds to the decomposition vaporisation of adsorbed water in the collagen fibre spacer, and the latter corresponds to the collapse, decomposition of type I collagen and mass loss of crystalline water from the composite. 29 The comparison reveals no significant difference between the TG and DTG curves of NHAC and MI-NHAC, indicating that there is no significant difference in the ratio of inorganic and organic contents or in the ratio of the adsorbed and crystalline water contents of the two composites.
The mechanical properties and various biological properties of a material are affected by its porosity; thus, the porosity of implant materials is an important parameter. The porosities of NHAC and MI-NHAC are 76.98 ± 2.54% and 47.09 ± 4.25%, respectively. The NHAC composites are more porous than the MI-NHAC composites, which is similar to the microscopic morphologies observed by SEM. Thus, NHAC may have a looser internal structure, while MI-NHAC has a denser and more solid structure. Many studies have confirmed that higher porosity is more conducive to bone tissue formation, probably due to the greater contact surface area, increased ion exchange, and easier adsorption of osteoinductive factors.30,31 However, other factors, such as the material degradation rate and mechanical properties, should be considered when assessing the material porosity.
Physical performance of the composites
The swelling properties reflect the diffusion of liquid within the material, which reflects the hydrophilic properties of the material and relates to the ability of the composite to absorb nutrients and excrete waste toxins in the body. 32 As shown in Figure 4, both MI-NHAC and NHAC exhibited swelling with increasing immersion time in PBS solution, and the swelling rate was higher during the initial 6 h. While NHAC reached swelling equilibrium after 12 h in PBS solution, the MI-NHAC maintained a low rate of swelling for the next 3 days without reaching equilibrium. Throughout the experiment, MI-NHAC exhibited antiswelling properties that were superior to those of NHAC. This difference may have been due to the smaller molecular spacing and greater intermolecular forces of the collagen fibres in the MI-NHAC composite, which stems from the preferred orientation.

Swelling properties of MI-NHAC and NHAC.
The mechanical properties of bone grafts are vital for osteogenic action, particularly in tissues that need initial strength during the regeneration process. 33 In this part of the experiment, the wet mechanical properties of the composites were evaluated considering the possible practical application of the material in a clinical setting. The stress-strain curves for the uniaxial compression experiments of the two composites are shown in Figure 5(a). Macroscopically, none of the samples showed structural collapse at 70% strain, which indicates their good flexibility and suggests that the mineralised crystals formed on the collagen scaffold in the mineralised solution are uniformly dispersed. From comparing the stress values at the specific strain values in Figure 5(b), it was found that the mechanical strength of MI-NHAC was significantly greater than that of NHAC, possibly because the collagen fibres are preferentially oriented in the direction perpendicular to the magnetic field direction, which is the axial direction of the cylindrical material; thus, MI-NHAC exhibits superior mechanical properties in response to the external forces applied in the axial direction. In addition, the stress values of MI-NHAC and NHAC increased sharply after the strains exceeded 50% and 60%, respectively, due to the compression of the internal space of the material, the pores and the molecular spacing decrease, resulting in an increase in intermolecular repulsion. 34

Mechanical properties of MI-NHAC and NHAC. (a) Stress-strain curves for the uniaxial compression experiments of MI-NHAC and NHAC. (b) Stress values of MI-NHAC and NHAC at 20%, 40% and 60% strain; *p < 0.05. (c and d) Cyclic stress-strain curves and ultimate stress values of NHAC. (e and f) Cyclic stress-strain curves and ultimate stress values of MI-NHAC.
The cyclic stress-strain curves and the ultimate stress values for a single cycle for both materials are shown in Figure 5(c) to (f). Excluding the curve of the first cycle, the last two curves of the cycles overlap each other well because the material still contains some residual liquid inside in the first compression cycle. After reaching 40% strain, the material cannot completely absorb the extruded liquid back from the test bench during the rebound process, so the difference between the curves of subsequent cycles and the curve of the first cycle appears. The good overlap of the latter two curves of the cycles indicates that the two composites have good elasticity. The stress values at 40% strain for both materials are in general agreement with the uniaxial compression experimental results. From the cyclic stress-strain curves, it was found that both composites showed significant hysteresis, especially MI-NHAC. This hysteresis is because there is energy dissipation during the cyclic compression of the composite. After the external force is removed, the molecular chains need to overcome the inherent friction in the process of returning to the original state, and the closer the molecular chains are, the greater the frictional force that needs to be overcome; thus, more energy is dissipated. 35
By comparing the physical performance of the two composites, it was found that MI-NHAC was more resistant to swelling and had a higher compressive strength. These characteristics are important for enhancing the spatial maintenance of bone grafting materials and to better restore the contour of the bone defect profile during the bone defect repair process.
Biocompatibility assessment
The degradation properties of bone grafting materials are an important assessment metric. Although it is accepted that the degradation rate of an ideal bone graft material should match the rate of new bone formation, the expected performance of bone graft material varies slightly for different areas of bone defects in clinical practice. 36 The difference in degradation properties of the two composites was assessed under collagenase conditions. After 4 days of degradation, both MI-NHAC and NHAC largely maintained their three-dimensional structural integrity, and no significant morphological structural collapse was observed. Figure 6 shows that the degradation of MI-NHAC was slow and smooth, while the NHAC composites degraded at a faster rate and had greater variation in their residual weight ratio in the first 2 days. After 4 days of degradation in collagenase solution, the residual weight ratios of NHAC and MI-NHAC were 47.67 ± 1.81% and 90.92 ± 1.96%, respectively; these values were significantly different. The results show that both materials largely meet the scaffold performance requirements of the bone tissue engineering field. 37 MI-NHAC has a more stable structure, and it maintains its mechanical strength significantly better than NHAC.

Degradation properties of MI-NHAC and NHAC.
Good cytocompatibility is one of the most essential properties of bone grafting materials. There is a broad consensus on the method of culturing cells with extracts from the materials to evaluate the cytotoxicity of biological implant materials.37,38 In this study, the cytotoxicities of the MI-NHAC and NHAC scaffold extracts on MC3T3-E1 cells were assessed using the CCK-8 assay. As shown in Table 1, the relative growth rate remained above 80% in both the NHAC group and the MI-NHAC group after 1, 3 and 5 days, and the toxicity ratings were all grade 1, compounding the ISO standard. Although the values in the MI-NHAC group were lower than those in the NHAC group, the toxicity evaluation results of the two groups met the implant material standards. This result may be due to incomplete dialysis, which was caused by the low porosity of the dense MI-NHAC and the small molecular spacing of the oriented collagen fibres.
Results of the NHAC and MI-NHAC cytotoxicity experiments.

It is known that the ability of cells to adhere to scaffolds highly influences the formation of new bone tissue. 6 To investigate the growth of MC3T3-E1 cells on MI-NHAC and NHAC, cell attachment and live/dead cell staining experiments were performed. The cell morphologies were observed using SEM after 3 days of cell culture on the NHAC, MI-NHAC and Col scaffolds. As shown in Figure 7(a), at ×300 and ×1000 magnification, the cells attached to the surface of the material, and extended pseudopods that were in good condition, which indicated that all the materials had good biocompatibility. The results of the live/dead cell staining experiments further illustrate the proliferation of MC3T3-E1 cells on the three materials. The staining kit uses esterified calcein to fluorescently label the live cells with strong green fluorescence, while the dead cells were stained with pyridinium iodide, which is a red fluorescent dye that does not penetrate the cell membrane. As shown in Figure 7(b), the number of fluorescently labelled live cells (green) on the three scaffold materials was comparable 1 day after inoculation of the cells. With increasing incubation time, the number of viable cells on the three materials also gradually increased, and differences in the numbers gradually became apparent. After 5 days of incubation, the number of live cells on the material was in the order of Col > NHAC > MI-NHAC, while the number of dead cells on the material was in the order of MI-NHAC > NHAC > Col, which is mostly consistent with the results of previous cytotoxicity experiments.

Biological properties of MI-NHAC, NHAC and Col. (a) SEM micrographs showing MC3T3-E1 cell adhesion to the three scaffolds after 3 days of incubation. (b) After 1, 3 and 5 days, live/dead staining was conducted; the scale bar represents 100 μm. (c) MC3T3-E1 cells cultivated on the three scaffolds after 1, 3 and 5 days of incubation. The CCK-8 test was used to determine the OD values at 450 nm; *p < 0.05.
In addition, the CCK-8 method was used to quantify the proliferation of cell adhesion on the materials. With increasing incubation time, the absorbance at 450 nm increased for the NHAC, MI-NHAC and Col groups, indicating that live cells were present throughout the culture and that cells on all three materials proliferated. After 5 days of incubation, the OD values at 450 nm for each group were in the order of Col > NHAC > MI-NHAC, and the differences were statistically significant. This result indicates that the three materials had different rates of cell proliferation, which is generally consistent with the previous cytotoxicity results and the results of the live/dead cell staining analysis.
Various properties of the scaffold, such as the surface morphology, porosity, and surface treatment, affect cell adhesion, were observed. 39 Collectively, the above results indicate that although there were some numerical differences between MI-NHAC and NHAC, they showed acceptable biocompatibility. However, the mechanical performance of MI-NHAC was significantly different from that of NHAC. In response to the specific clinical problem of severe bone defects in the upper anterior region encountered in oral implant restorations, the substantially improved degradation resistance, swelling resistance and compressive strength of MI-NHAC make it a promising candidate for research applications.
Conclusion
The ideal bone graft material can be used for all clinical applications; although desirable, this is often difficult to achieve. The vast majority of bone graft materials need to remain stable after implantation into the host, and the graft area needs to have an adequate blood supply and a sufficient source of osteogenic precursor cells. 40 Therefore, the choice of which bone grafting material to use depends heavily on the biological and mechanical properties requirements. 41
This study explores a new idea for preparing synthetic alloplastic block grafts for horizontal and vertical bone augmentation needs in oral implant restorations, aiming to substantially improve the mechanical strength of conventional nanohydroxyapatite/collagen composites and thus enhance the maintenance of osteogenic space during in vivo application. A magnetically induced highly oriented nanohydroxyapatite/collagen composite was developed based on a self-assembly technique using the properties of collagen fibres and nanohydroxyapatite crystals in a high magnetic field, and its physicochemical and biological properties were evaluated. Although the mechanical properties of MI-NHAC are not as good as those of autologous bone blocks, MI-NHAC showed significant improvements in terms of its degradation resistance, swelling resistance and mechanical strength compared with NHAC. Therefore, it has certain research and application prospects. In addition, combining MI-NHAC with conventional NHAC composites to form a bilayer composite that is similar to the structure of cortical-cancellous bone will be a focus of future studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Key Research and Development Program of Shaanxi, China (2020SF⁃014).