Abstract
Sol-Gel-based reaction mixture sols have been long used to fabricate dense and uniform bioactive coatings with superior mechanical stability over metallic implants. On account of precise control over synthesis, fabrication, formed and low temperature of processing, this technology is one of the most feasible routes to produce bio-ceramic coatings. The study aims to develop a physical barrier over metal implants in form of bioinert Zirconia coatings, phase-stabilized using Dysprosium. The metallic substrates were cut into 10 mm × 10 mm samples and diamond polished after being polished with a 1000 grade emery sheet. Novel spin-coated zirconia films were fabricated over 316L Stainless steel substrates and were sintered at 600°C to obtain firm and uniform crack-free coatings. The thickness of the coatings was determined by ELCA-D meter thermal analysis was performed using TGA-DTA. Phase determination was performed using X-Ray diffraction followed by morphological investigations using Scanning electron microscopy. The corrosion resistance was evaluated with Polarization studies and electrokinetic data was derived using Tafel extrapolation. Biocompatibility evaluation was performed against MG-63 cell lines and RBCs along with bone-forming ability in vitro in SBF. Stable crack-free 3 Layer coatings fabricated at 2000 rpm for 3 s with a thickness of around 1 μm were found to be optimal for corrosion resistance behavior of steel implants at a low ICorr value of 0.501 µA/cm2 and adhesion strength of 40.93 MPa when untreated falling down to 39.92 MPa when immersed in SBF. The study concludes that medium rpm coatings sustain enough sol to produce crack-free coatings that form a strong physical barrier between body fluid and implant surface thereby reducing the attack of corrosive ions and protecting the implant surface without participating in any form of bioactivity but supporting native bone regeneration capabilities.
Research highlights
Evaluation of Dysprosium applicability in phase stabilization.
Fabrication of Thin films of bioinert ceramics over bare steel implants for minimal but active interaction from Host tissues.
Reduction in direct component leeching from Steel implants leading to toxicity.
Development of corrosion resistance over steel implants for longevity and performance improvement of metal implants.
Bioactivity evaluation of coatings with Dysprosium stabilization.
Introduction
Stainless steel (316L) due to its highly flexible and versatile load-bearing applicability has been a favorite choice for the bone healing process. However, the poor corrosion resistance properties unlike Titanium alloys have been a major setback. 1 The lack of bioactive surface often leads to poor interaction with the host cells and often leads to delays in the bone regeneration process. The major issue with the already reported research of forming a bioactive surface over steel implants is the eventual degradation of the surface coating leading to suspended by-products of coatings. 2 Moreover, accelerated corrosion of steel by body fluids and even osteoclasts after coating degradation poses a serious threat of toxicity and adverse effects over host tissue.3,4 This draws our attention toward highly rugged bioinert ceramics like Zirconia which has been successfully applied to high load-bearing applications in form of prosthetics as well as in dentistry.5,6 The ability to selectively interact with the host tissue and withstand body fluids makes it an ideal candidate for corrosion resistance of steel implants. 7
Because of the superior mechanical properties, wear resistance, fracture toughness, chemical and thermal stability of a bio-inert ceramic Zirconia (ZrO2) is used in orthopedic load-bearing biomedical applications such as dentistry and prosthesis are characterized by high strength properties which are based on the stress-induced phase transformation involves the transformation of metastable tetragonal to monoclinic phase.8,9 Three different types of polymorphs of zirconia exist namely monoclinic ZrO2 (m-ZrO2) which is the stable phase up to 1170°C, where it transforms to tetragonal ZrO2 (t-ZrO2), and then cubic ZrO2 (c-ZrO2) at temperatures above 2370°C.10–12 Tetragonal to Monoclinic phase transformation is accompanied by the volume expansion during the subsequent cooling process which results in poor mechanical stability due to its crack propagation of a material.13,14 For scaling down these issues additives can be added to achieve the stabilization by additions of various metal oxides such as CaO, Y2O3, MgO, La2O3, Sc2O3, and CeO2 at low concentrations results in the stabilization of the tetragonal phase (for mechanical toughness) or cubic phase stabilization at higher additive concentrations at room temperature.15–19 The role of oxygen vacancies in the in situ due to valence and ionic radii mismatch between the Zr4+ and the additives (Ca2+, Mg2+, Y3+, Ce4+, and La3+) which can stabilize the t-ZrO2 phase and this has been attributed to several factors, such as crystallite size, surface and lattice defects, adsorbed species on the surface, and domain boundary stresses.20,21
The applicability of rare earth has a proven record to induce better mechanical strength in zirconia systems. 22 Dysprosium substituted at 10% molar concentration for Zirconia has better mechanical properties as elucidated from Young’s modulus and hardness measurements by Kalaivani and Kannan 23 Moreover, these are the closest as compared to that of 316L steel itself that reduces adhesion failures overall due to mechanical irrelevance. For the first time, authors have attempted novel Dysprosium stabilized tetragonal zirconia with excellent mechanical stability fabricated as coatings over 316L SS to act as a physical barrier thereby limiting the interaction of body fluids to the implant surface and increase its service life as well as reduce the release of toxic components from the alloy.
The sol-gel method on account of high reactivity and favorable reaction temperature has been the first choice of biomaterial synthesis. Ease of control over reaction parameters further adds to the applicability of the same but the use of inorganic and organic components at times is deemed unfavorable by some researchers. 24 Over time, the green route of synthesis was formulated with similar performance and negligible toxicity most common route being the Citrate assisted reaction by Pechini in 1967. 25 In this method, the precursors are mixed and the sol is prepared in excess Citric acid from a favorable source thus forming a highly viscous sol that eventually turns to gel. The sol prepared from the above route was spin-coated over 10 mm × 10 mm × 5 mm steel implants. 26 Three layers of sol were coated over the implants at 2000 rpm for 3 s drying each layer after coating.
Materials and methods
Synthesis of sol
Citrate assisted sol-gel technique was followed to synthesize highly viscous sol from the reaction mixture extracted just before gelation of the same. Analytical grade Zirconyl chloride octahydrate (ZrOCl2·8H2O) was used to prepare a stock solution of metal nitrate in 1M Nitric acid (HNO3). A separate stock solution of Dysprosium(III) nitrate hydrate (Dy(NO3)3·xH2O) 10% to that of Zirconium precursor was prepared in deionized water and added dropwise to the Zirconium stock solution at ambient temperature stirring at 300 rpm. The citric acid solution was prepared separately keeping the ratio of nitrates to acid as 1:3 and the resultant nitrate solution after constant stirring for 20 min was added dropwise to the citric acid solution stirring at 450 rpm. The resultant sol was kept stirring and viscosity was measured at regular intervals until it reached 0.035 Pa s using a KU 3 viscometer (Amtek Brookefield). The sol was harvested to be coated over steel implants using Polos SPIN 200i spin coater.
Preparation of implants and spin coating
Surgical grade 316L steel implants were acquired and laser-cut into dimensions of 10 mm × 10 mm × 5 mm. These implants were polished using emery sheets starting from grade 200 all the way up to 1000 followed by diamond paste polishing to acquire a mirror finish thereby reducing the available surface for corrosion. The polished implants were washed with acetone, dried, and wiped with ethanol to avoid any contamination. A drop or two of the sol was put over a metal implant fixed over the spinneret and spun at 2000 rpm (ZDS-A), 3000 rpm (ZDS-B), and 4000 rpm (ZDS-C) for 3 s with an acceleration time of 2 s in a Nitrogen environment followed by drying the implant in a hot air oven repeated thrice for triple-layer coatings.
Coating characterization
A small portion of the sol was dried to raw powder and subjected to TGA-DSC to determine optimized sintering temperature the principle being lower the better. The raw powder was taken in specified crucibles and measured at the in-built balance of TGA-DSC (TA Instruments) followed by heat treatment and measurements recorded at a scan rate of 2°/s. Based on the DSC curve, the optimal temperature for sintering was determined and the implants were sintered at 600°C for half an hour to produce coated steel implants taken for characterization. The resulting thin film was characterized using diffraction analysis using Bruker D8 Advance diffractometer at Cu Kα radiation (λ = 1.54056 Å) produced at 40 kV and 30 mA. The thickness of the samples was measured using an Elca D meter and surface morphology was examined using SEM (Zeiss GeminiSEM 300). The bonding strength of the coated samples was evaluated following ASTM C 633-79 guidelines. 27
Electrochemical corrosion studies
Reagents from Sigma Aldrich were used to formulate the Simulated Body Fluid (SBF) as described by Kokubo et al. and were used throughout the study for SBF-based evaluations as well as the electrolyte in Electrochemical cell for in vitro corrosion studies. To evaluate the bond strength in vitro, the samples were immersed in SBF for different time periods and the respective bond strength was observed after drying in a hot air oven. Electrochemical corrosion studies were carried out in a three-electrode setup where the Implant served as working electrode, Ag/AgCl electrode as the reference, and Pt wire was used as a counter electrode in 100 mL SBF for 100 mm2 samples. 28 Data was acquired using CH-Instruments Electrochemical workstation with bundled software to process the raw data. Statistical calculations and values were generated and graphs were plot using Origin software. For Tafel experiments applied with respect to OCP (Open Circuit Potential) run for 1 h, Ei value was set to −0.250, and EL was set at +0.250 at a scan rate of 0.100 mV/s.
Biocompatibility evaluation
The biocompatibility studies of the samples were done using hemolytic measurements and Cytotoxicity was measured using MTT assay. Test extracts of the coated samples were prepared by immersing the samples in physiological saline for 24 h at 37°C. For the hemolytic assay, test extracts of the samples were treated with diluted blood and incubated for an hour at 37°C. The hemolytic ratio of the treated sample was measured using a (Shimadzu UV-1800) at 545 nm and the Hemolysis ratio (Z) was calculated using the following formula:
Deionized water was used as positive control and saline as a negative control. Similarly, test extracts of the samples were used to perform MTT assay against MG-63 osteosarcoma cell lines and the percent viability was recorded with respect to OD values of the Control. The cultured MG 63 cell lines were used for the following procedure with the pre-prepared test extracts of the samples. The test extracts were seeded with cell lines at a concentration of 1 × 104 cells in the well plate for 72 h. The cells were washed with serum-free media and further incubated for 24 h following which the culture media was removed from the cells. 0.5 mg/mL MTT prepared in 1x PBS was added to cells and incubated at 37°C for 4 h which was followed by removal of MTT and washing with PBS. The as-formed crystals were dissolved in DMSO and the developed color intensity was evaluated at 570 nm. The percentage of cell viability was elucidated with respect to OD values of control using the following calculation:
Coated steel implants were immersed in the SBF solution with respect to coating weight measured after sintering to maintain release concentration which was changed every 24 h. After completion of the treatment period, samples were gently rinsed with deionized water, followed by drying at room temperature, and visualized using SEM. 29
Results
Thickness measurements
The thickness of all the samples was measured to be averaging around 0.9 μm with a standard deviation of 0.07, found to be reduced as the spin rpm was increased. This range of thickness is considered favorable for biocompatible coatings and proved useful in the corrosion-resistant feature of the implants as evaluated with other types of coatings. 30
Thermal measurements
Thermogravimetric measurements divide the weight loss into three steps of the graph. About 74% of the weight is lost around 200°C with an endothermic reaction. The next 7% is lost around 350°C and the final 7.5% is around 450°C (Figure 1). The first step of the TGA curve corresponds to the removal of residual water and moisture from the coatings. The second step corresponds to debonding of water molecules from Zirconium hydrozide molecules followed by condensation and formation of tetragonal zirconia in an exothermic reaction as seen from the DSC plot.
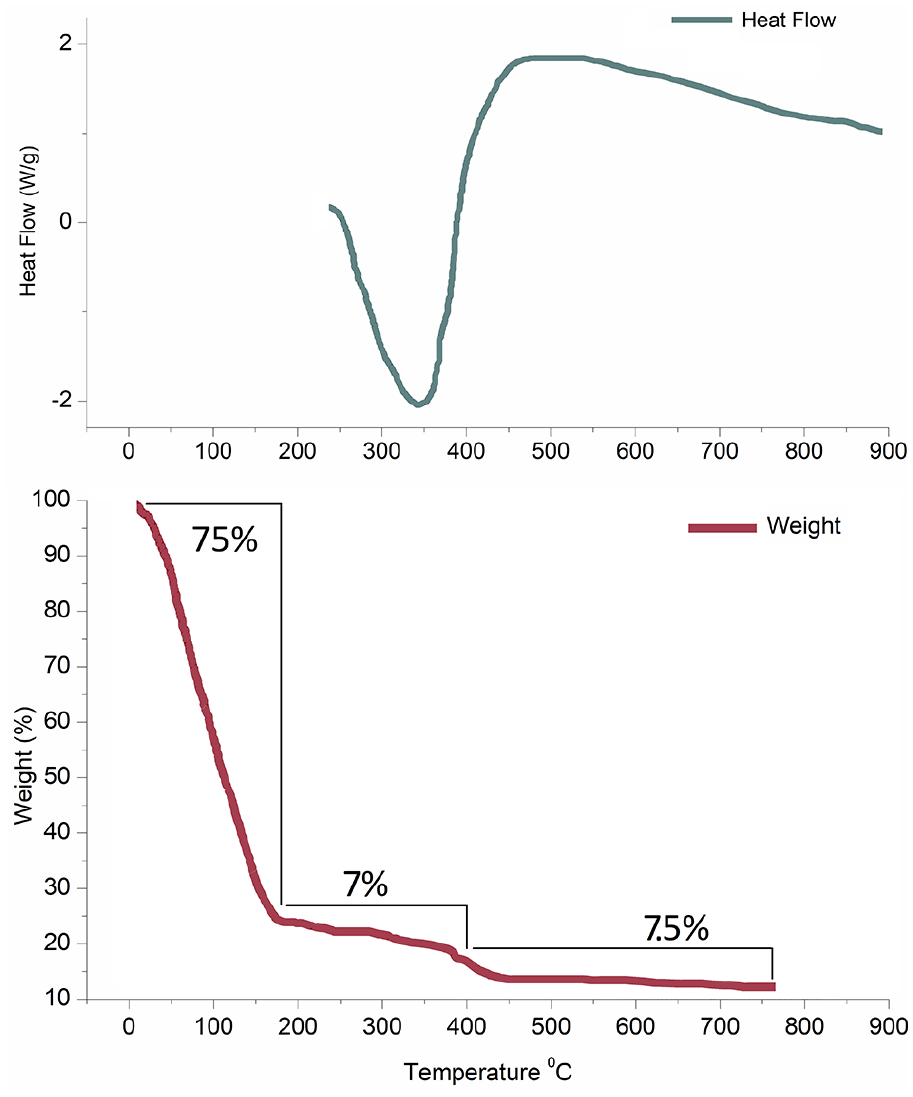
DSC and TGA plots of raw powder obtained from synthesized sol for coating.
Diffraction studies
Diffraction data shows distinct major peaks of 316L steel at 43° and 52° corresponding to the standard JCPDS data card. There has been a negligible effect on the relative peak intensity within all three coated samples. Apart from the major peaks, minor peaks of tetragonal zirconia corresponding to JCPDS Datacard 17-0923 were observed overall in all three coatings (Figure 2). No impurity phase was detected that confirms the applicability of Dysprosium in the phase formation of Zirconia coatings over the implant surface.
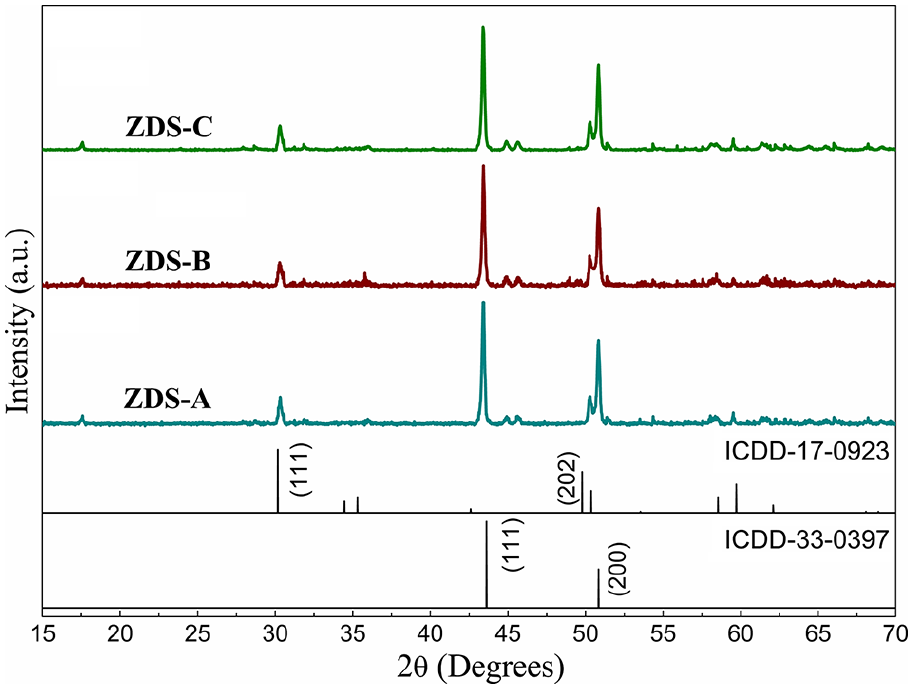
X-ray diffraction plots of Dysprosium stabilized tetragonal Zirconia coatings over 316L steel implants sintered at 600°C.
Morphological studies
SEM micrographs of the sample show even coatings with several layers visible atop the steel implants. Further magnification shows even but rough morphology thereby leading to an increase in the surface area of the sample. A deep dive into the magnification at 10 μm shows the granulated morphology of the zirconia coatings resembling the tetragonal structure (Figure 3a-c). It must be noted that multiple layering has left no space exposed for the steel surface forming an adherent physical blockage to the metal implants for body fluids to interact with.

SEM micrographs of Zirconia coatings (2000 rpm) over Steel implants sintered at 600°C.
Mechanical studies
The bonding strength of sintered samples shows an increase in bond strength with respect to an increase in rpm. Samples with 2000 rpm coatings showed a bonding strength of around 40.93 MPa. This elevated as the coating rpm was increased from 42.63 MPa in 3000 rpm to 48.42 MPa in 4000 rpm. Upon immersion in SBF for 5 days, the coatings lose mechanical stability by minor level and fall down to 47.6 MPa for 4000 rpm, 40.3 MPa for 3000 rpm, and 39.42 MPa for 2000 rpm (Figure 4). Further increase in the immersion time reduces the mechanical stability, but after 15 days, the samples start to gain stability and after 20 days of immersion, the samples have a stable mechanical strength of 46.2 MPa for 4000 rpm, 38.71 MPa for 3000 rpm, and 33.8 MPa for 2000 rpm. This reduction however hardly puts any impact on the mechanical stability as the coatings still have great adhesion to the implants.

Observed adhesion/ bonding strength of Zirconia coatings over implants in untreated sintered form and after immersion at different time intervals in SBF.
Electrochemical corrosion studies
Post TAFEL experiments, the data was plot as shown in Figure 5. Statistical analysis of the data using user defined “nonlinear least squares curve fit” with OriginLab Origin pro software, gave details about the ECorr (Corrosion potential), ICorr(corrosion current density) and ba and bc (anodic and cathodic Tafel constants) which is enough to deduce the corrosion resistance details if acquired by the coated samples as compared with the bare Diamond polished samples. 31 Table 1 shows the numeric values of all the parameters and shows a decrease in corrosion potential and an increase in Corrosion current as coating layers increase adding extra corrosion resistance. However, this trend gets a setback in form of cracks that are formed when number of layers are increased that are responsible for contact between body fluid and specimen surface. Similar effects due to cracks are also visible in changing values of Tafel constants ba and bc as well. This kind of change leads to the shift of graphs to a positive potential.

Potentiodynamic polarization plots of Dysprosium stabilized tetragonal zirconia coatings over 316L steel implants.
Electrochemical parameters attained from Tafel Plots.

Biocompatibility studies
Hemolytic assay
Test extracts obtained from subjecting the samples with physiological saline when treated show almost no effect on the blood samples. All the blood samples were bright red after an hour of incubation and there was hardly any visual evidence of hemolysis of the RBCs. Optical micrographs of the samples show no effect on the morphology of RBCs and all the blood cells were found to be intact in the images (Figure 6). OD values calculated against positive and negative controls show all the Z values below 2 that conclude the samples to be hemocompatible (Table 2).
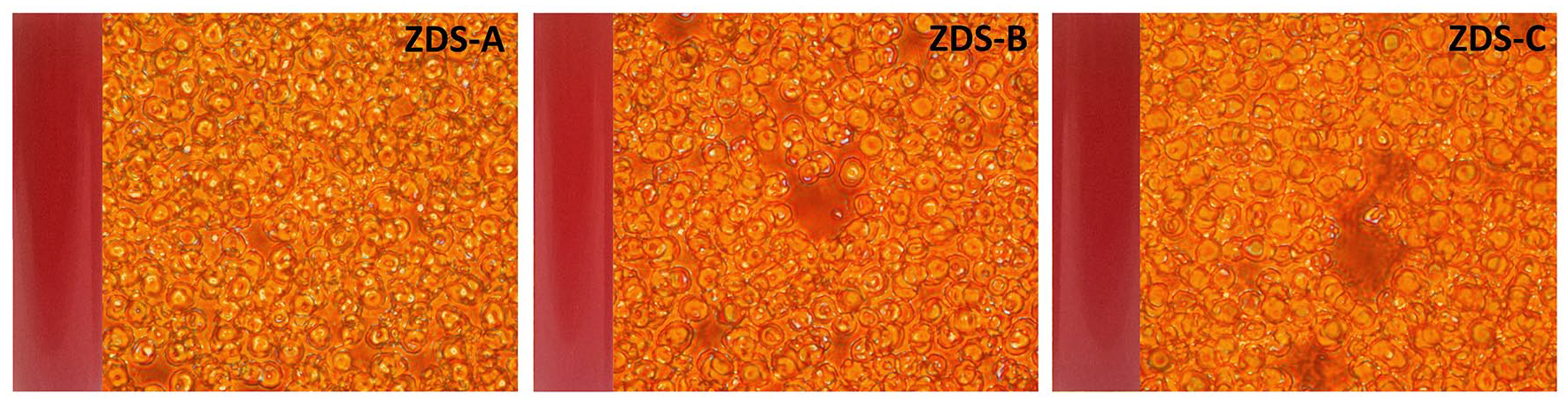
Optical images of blood solution treated with test extracts and red blood cells observed under 40× magnification.
Absorbance and hemolytic ratio of test extracts obtained from coated samples.

MTT assay
MTT assay shows all the samples having a negligible effect on the osteosarcoma cells over the test extracts. The bare samples however possibly with the leeching of component metal ions show a minor inhibitory effect over the cells. The OD values with respect to control show all the coated samples exhibiting cell viability above 97% (Figure 7(a)). The micrographs show a couple of cells deformed in the matrix (Figure 7(b)). Apart almost all the cells have maintained their structural integrity and are viable as we see them.

(a) Relative cell viability of Dysprosium stabilized Zirconia coatings over 316L steel implants calculated with test extracts. (b) Optical micrographs of MG-63 cells after MTT assay.
SBF immersion
SEM micrographs (Figure 8a-c) show amorphous deposition over the surface of coatings. An increase in magnification level shows clear apatite deposition over the granules. Due to SBF as the only source of calcium, very little apatite has been seeded over the surface of the zirconia granules.

SEM micrographs of SBF immersed tetragonal Zirconia coated Steel implants (2000 rpm) after 7 days of immersion.
Discussion
ELCA-D measurements of the coating thickness show the comparatively fair thickness of coatings. This corroborates with the three layers of coatings fabricated over the steel implants. The change in measurement area or point points standard deviation of the error around 0.07 which is evident in uniform coating overall. It has already been established that a uniform coating reduces the available surface area that reduces the corrosion level due to less surface interaction with the body fluids.
The TGA data and its weight loss are comparable with the heat loss and gain details corroborated by DTA data. The loss of water and moisture detailed as the first step of weight loss can be directly related to the negative differential of temperature pointing toward an endothermic reaction. The second step of TGA can be attributed to the debonding of water molecules from Zirconium hydroxide thereby leading to a little weight loss. This debonding leads to a positive temperature differential and hints at an exothermic reaction in the DTA curve. With a final weight loss, a little over 10% of sample weight is recorded as residue at the end of the TGA curve depicting the final step. This step can be attributed to the condensation of Zirconium hydroxide to zirconia. The exothermic reaction deduced from the DTA curve confirms the phase transformation of the tetragonal phase of Zirconia.
Diffraction data shows consistent 316L steel peaks in accordance with JCPDS Datacard (33-0397). Sintering in reducing atmosphere avoids any kind of oxidation that has maintained the relative peak intensity of all the three fabricated samples a far as diffraction peaks of steel are concerned. Apart, minor peaks of zirconia were indexed due to triple-layer coatings at designated 2-theta values (17-0923). 32 It must be noted that relative peak intensity was consistent overall that shows an identical fabrication environment has led to similar features of obtained coatings. Moreover, even close observation fails to index any trace of Dysprosium in the spectra that establish the fact that all of the stabilizers were utilized in the phase transformation of tetragonal zirconia.
The morphological features were rather absolute showing minor gaps over the coating surface which were covered by the inner layer of coatings visible with a minute inspection. These cracks are formed after drying due to loss of water forming a dense gel over the surface that when dried reduces in volume and leaves gaps or rather cracks. A triple-layer of coatings was fabricated to counter these gaps and obtain a perfect barrier over the implant surface. It must be noted that an increase in the number of layers will lead to loss of water from inner layers causing damage to outer layers as it tries to escape from inner layers forming cracks and, in some cases, even leading to detachment of coatings due to temperature gradient generated within inner layers between the implant itself due to evaporated water molecules. This phenomenon can occur during drying at room temperature or even during sintering. Close magnification leads to the visualization of characteristic granules of the tetragonal zirconia.
The mechanical strength of the coatings is inversely proportional to the spin speed as an increase releases a higher amount of fluids from the coating sol. This reduction in sol thereby accounts for the reduction in the thermal mismatch between surface and material affecting its adhesion. Close proximity and limited coating material also initiate a mechanical interlock-based bonding between the free surface ions and that of coatings that adds extra adhesion levels. The presence of fluid-based sol that supplies oxygen as well as H leads to its aggregation at the implant surface thereby leading to Hydrogen bond formation along with physical interlocking increasing the coating strength. The only factor of decrease in coating strength is residual stress which is directly related to thermal coefficient. Hence, with higher rpms, the amount of sol decreases as a result, during the drying process, there is a decline in thermal coefficient resulting in a reduction in residual stress thereby leading to an increase in adhesion of samples at 4000 rpm.
Only Tafel experiments were carried out to deduce the data in form of destructive mode of corrosion. Extracted electrokinetic parameters by extrapolation clearly point toward the increase in corrosion resistance of the steel implants. It can be seen from Table 1 that the lowest corrosion resistance was exhibited by the bare specimen at room temperature. The ECorr values of the sample increase with the application of a triple layer of the coating due to the presence of a barrier in form of zirconia over the implant surface. As expected, ICorr values of the samples decrease when compared to ECorr values that rise as rpm is increased at 3000 rpm but eventually return toward initial values of ZDS-A (2000 rpm) at 4000 rpm. 33
Corrosion current is defined as the intersection between anode and cathode linear extrapolation at ECorr. It is directly attributed to electrode potential and details the electrochemical behavior of the material better in our case the implants. Passivated current that is associated with Passivation current density is the recorded current that is generated while formation of a virtual layer at the surface of interaction of implant and fluid. Inspecting the curves of polarization shows an increase in passivity (IPass) and current density (ICorr) values when implants are coated with Zirconia films however, the Current density (ICorr) itself is strong enough to determine corrosion. 34 The decrease in breakdown potential values when coated can be attributed to the porosity and surface energy of the coatings. In simple terms, the corrosion rate of the material is directly proportional to corrosion current density and inversely to the potential and polarization resistance. 35 In our context, the presence of zirconia as a physical barrier with even crack-free coatings as obtained with 2000 rpm reduces the active flow of SBF to implant surface and removal of surface components. However, a partial indirect contact between the implant surface and SBF is established eventually as coated samples are introduced in SBF that affects the current density but active leeching out of metal implant components is restricted that reduces the levels of toxic components of alloy in extra cellular matrix which is favorable from the biocompatibility point of view. In other cases, where coatings were unstable, mostly due to thickness or cracks, preferential direct contact is established with implant surface thereby increasing the relative surface of interaction between SBF and implant affecting corrosion rate. Changes in current density however small represent the harshest destructive approach to corrosion measurement studies in vitro and will be scaled down in vivo but provide a miniaturized small-scale idea of the effects coatings generate and help us determine relatively optimized coating parameters. A lower corrosion rate means less leaching out of material in body fluid that might turn toxic and lead to adverse effects in the bone healing process.
The in vitro bioactivity of the samples was evaluated over RBCs, MG-63 osteosarcoma cell lines via MTT assay, and monitoring the bone apatite formation by immersion in SBF solution. Blood, being the most common body fluid interacting with the foreign material and responsible for most of the transportation in body physiology needs to be compatible with the implant surface now modified with Zirconia thin films. Hemolytic assay hardly showed a visible change in the color of blood treated with sample test extracts. Moreover, the OD values place all three samples well within the hemolytic limits of the Z value. Visualizing the RBCs treated with test extracts also shows perfectly healthy and unaffected cells. Hence, we can clearly state that the thin films coated over the steel implants are hemocompatible. 36
MG-63 cell lines are cytocompatible toward the bioinert ceramics in our case zirconia. However, the leaching of metal components from the implant surface leads to a little inhibitory effect on the cells. This has long been recorded and hence corrosion resistance of almost all the metal implants has been a prime focus of most of the researchers. These metal components leech out in the body fluids and not only interfere with the metabolism of bone formation but also interfere with the normal biochemistry of healthy cells adjoining the host tissue. An increased level of some of the leeched-out products also lads to toxic effects within the body system and often leads to surgical revisions.
The surface morphology of the implants is also an important factor in the regeneration of bone tissue. Mirror polished metal surgical implants offer relatively less surface area to interact with body fluids. Also, the surface tension leads to changes in contact angle that reduce interaction with body fluids. This might be applicable in reducing the relative surface corrosion but it must be noted that bone regeneration needs to interact with the implant surface. In our case, the relatively rough surface morphology of the implants and granulated zirconia coating as seen from SEM images favor bone regeneration in addition to corrosion resistance. The interaction with body fluids leads to the deposition of Calcium and phosphate groups over the surface of implants. With help of body enzymes, these calcium and phosphate groups form amorphous apatites that further crystallize to form new bone tissues. 37 These deposited Calcium and phosphate deposition was visible over the implant surface after 7 days of immersion in SBF solution as seen in Figure 7. These images confirm the applicability of Zirconia thin films in bone regeneration as they can eventually attract Calcium and phosphate groups that are crucial in bone remodeling.
Conclusion
Dysprosium addition in the zirconia system leads to stable tetragonal phase stabilization of zirconia coatings at room temperature. The coatings were uniform and were found to add an extra layer of corrosion resistance to the polished steel implant surface natively reducing the corrosion level by reducing surface area of contact with body fluids as compared to unpolished ones. Thermal analysis showed condensation of metal hydroxides to Zirconia with weight loss mostly in form of water. This loss of water formed several gaps in the uniform layer which were compensated with other layers visible in electron micrographs. Diffraction data confirmed the pure phase of coatings and involvement of dysprosium as hypothesized. Evaluation of corrosion resistance via electrochemical route proves better corrosion resistance and a reduction in implant leeching due to stable barrier formed by triple thin film layer of Zirconia over the implants evident from the decrease in Corrosion current values as compared to the bare sample. Taking account of electrochemical data and Mechanical stability, we can state that steel samples spin coated at 2000 rpm exhibit most favorable and stable coatings for surface modification of implants. On the biocompatibility front, samples showed a hemolysis ratio well below Z values of 2 and cell morphology of RBCs were found to be intact when treated with test extracts. The bare sample showed a little cytotoxic effect over MG-63 cells due to leeching of metal components of alloy which turned negligible when the implant was coated and cell viability was recorded well above 97% as compared to control. Above all, the promising bone-forming ability was observed when SEM images of ZDS-A samples, spin coated at 2000 rpm immersed in SBF showed coating surface capture and form amorphous apatite over it. It is remarkable that dysprosium addition hardly showed any effect in all of the studies and fulfilled its job of stabilizing the tetragonal phase successfully at room temperature. No matter how efficient, Spin coatings being a physical method does not match the efficiency of other coating methods such as Electrophoretic deposition and Plasma spray with their own set of issues. The above study was an initialization of attempts to study how Dysprosium stabilized Zirconia behaves in vitro. Further attempts to study the Genotoxicity, and enzymology-based evaluation of the coatings may open new doors for in vivo studies and finally clinical application. Dysprosium stabilized zirconia might be used to fabricate coatings by other means and on other biomaterials to add to knowledge about its vivid behavior in a variety of situations and related applications.
Footnotes
Acknowledgements
The Authors thank the host institute for the provision of a platform and arranging facilities to carry out the research work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


