Abstract
Motivation
A series of Eu2+ (0.0025≤ × ≤0.025) activated Sr2SiO4:xEu2+ (SSO:xEu2+) phosphors were synthesized via a sol-gel method. The phosphors were characterized by x-ray diffraction (XRD), scanning electron microscopy (SEM) and photoluminescence (PL) spectroscopy. The differences between
Results
The energy gap of
Introduction
Increasing attentions have been paid to the white light emitting diodes (LEDs) due to their high efficiency, long life times, low contamination to environment, and absence of mercury. LEDs have been regarded as the new generation of illumination (1-2-3). The most commonly used method to produce white light is a combination of blue chip with yellow emitting phosphor materials. However, the low color rendering limited its applications in many fields. To solve this problem, one approach is to add some red phosphors into the yellow phosphors; while the other approach is to combine an ultraviolet (UV) Indium Gallium Nitride (InGaN) chip with red, green and blue multiphase phosphors (4). The latter model white LEDs can offer high color rendering index (
Especially, Eu2+-activated SSO phosphor contains two phases, α’-SSO and β-SSO and they can emit green light (5). JK Park prepared Eu2+ activated Sr2SiO4 yellow phosphor by a solid-state method for the first time (6). XY Sun synthesized green yellow emitting
Experimental procedure
Sample preparation
Powder samples of Sr2-
XRD analysis
SEM images
Luminescent properties of the samples
The structural and phase composition of the prepared powder samples were characterized by x-ray diffusion (XRD) using Cu Kα1 radiation (Bruker D8) at 40 kV and 40 mA. Photoluminescence (PL) and photoluminescence excitation (PLE) spectra of the samples were measured by a fluorescence photospectrometer (Hitachi F-7000) with a 150 W Xe lamp. The SEM images were taken by a JSM-7500F Field emission scanning electron microscope (FESEM). All above measurements were performed at room temperature except the temperature spectra.
Results and discussions
Figure 1 shows the XRD patterns of the SSO:x%Eu2+ phosphors with different concentrations of Eu2+ ions (
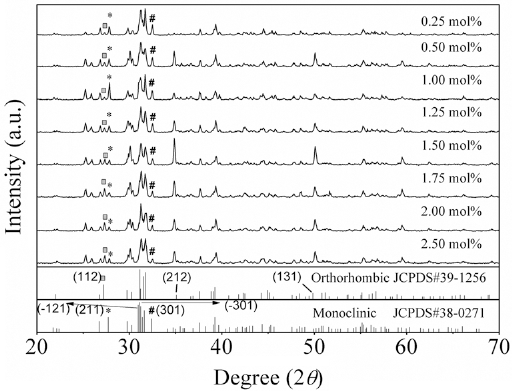
(Color online) x-ray diffusion (XRD) patterns of Sr2-
First principle calculation was implemented in the software CASTEP (11). A Vanderbilt-type ultra-soft pseudopotential formalism and the exchange-correlation function based on the generalized gradient approximation (GGA) in the scheme of Perdew-Burke-Eruzerhof (PBE) were used in the calculations of energy band structure and density of state. As seen in the energy band structures of both SSO phases, they displayed an indirect optical band gap, and the gap between the lowest energy level of the conduction band and the highest energy of the valence band were about 4.489 and 4.106 eV, respectively (Fig. 2A and 2C). According to the report of Xiaocheng Zeng [Qu et al (12)], the electron-occupied 4f energy levels of Eu located within the band gap of the host, while the empty 5d levels were just above the conduction band minimum (CBM), which indicated that the electronic transition between 4f and 5d levels of Eu induced the luminescence. Thus, SSO belonged to the materials with large band gaps that are usually good hosts for various luminescent ions. The valence band was about 3.1 eV by the calculation of the absorption spectra (See Supplementary Figure 2, available online as supplementary material at www.jab-fm.com). The error existed between the calculated value and the theoretical value.
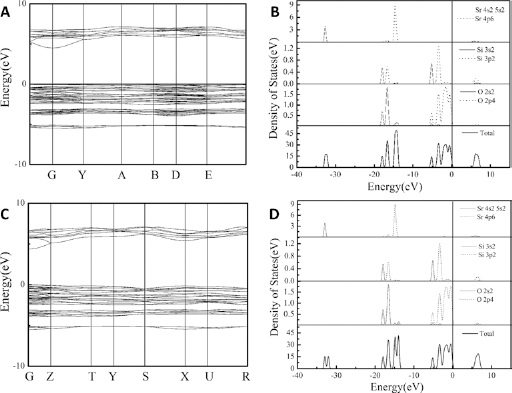
Energy band structures (
Both SSO samples showed similar total and partial densities of states. The top of the valence band is dominated by Sr4p, O2p and Si3p states, while the conduction band is mainly composed of O 2p and Si 3p states (Fig. 2B and 2D).
The SEM patterns of the SSO:xEu2+ phosphor with different Eu2+ ions (
The excitation spectra of SSO:0.01Eu2+ phosphor monitor for 532 nm and 464 nm were shown in Figure 3A. The excitation spectra of SSO:0.01Eu2+ phosphor showed that the excitation peaks monitoring for 464 nm locate at 288 and 309 nm, while that for 532 nm located at 274, 310 and 365 nm. The excitations were attributed to the dipole allowed 4
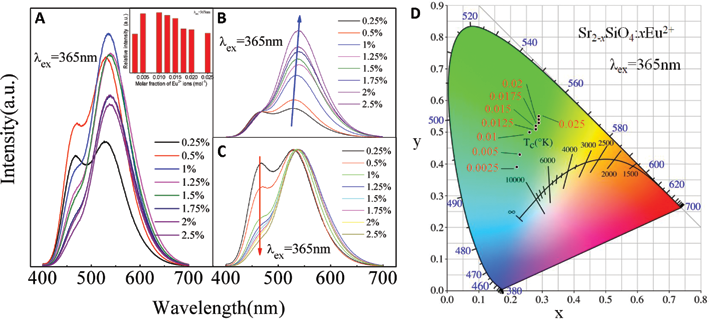
(
An empirical formula (Eq. 1) fitting the position in energy of the lower d-band edge for Eu2+ or Ce3+ ions was given by Van Uitert (14).
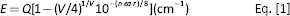
where
The Eu2+ substitution for Sr(I) causes the longer wavelength (532 nm) and Sr(II) attributed to the shorter one (464 nm). The populations of Sr(I) and Sr(II) are almost equal. When the crystal phase changes from
The inset of Figure 3A showed the relationship between emission spectra of SSO:xEu2+ (
The critical energy transfer distance between ions can be estimated by Blasse's equation (18):

where
The Commission Internationale de L'Eclairage (CIE) chromaticity coordinates and CIE chromaticity diagram for the SSO:xEu2+ phosphors with Eu2+ concentration, were shown in Figure 3D. It showed that the CIE chromaticity coordinates (
The thermal stability was important for phosphor to be used in the pc-white LEDs. The emission intensity of SSO:0.01Eu2+ phosphor at various temperatures from 50oC to 150oC were measured and the emission spectra are presented in Figure 4. It can be seen that the emission intensity of SSO:Eu2+ phosphor was 60% of its original values as the temperature increased to 423
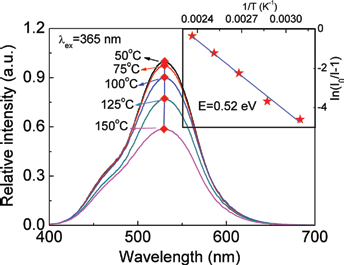
Emission spectra of Sr1.995SiO4:0.005Eu2+ at different temperatures from 50oC to 150oC under 365 nm excitation. The inset plots fitted the activation energy for thermal quenching of SSO:
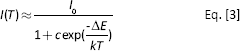
where
The inset of Figure 4 plotted the In[(
Conclusions
In summary, SSO:xEu2+ phosphors with different Eu2+ concentrations have been synthesized by the sol-gel method. There are two different phases for SSO, orthorhombic
Footnotes
Financial support: This work is financially supported by the National Science Foundation of China (No.61205180), Natural Science Foundation of Hebei Province (Grants No. E2012201087), the first batch of young talent support plan of Hebei Province, the distinguished young scholars of Hebei University (2012JQ01) and Natural Science Foundation of Hebei University (Grants No. 3333112). We also appreciate the financial support from the Midwest universities comprehensive strength promotion project.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
