Abstract
Background:
Titanium biomedical devices coated with strontium-doped calcium phosphate ceramics can support desirable bone regeneration through anabolic and anti-catabolic effects of strontium and the compositions close to that of natural mineral tissue.
Methods:
Strontium was doped into the calcium phosphate coating using the cyclic pre-calcification method on the anodized titanium plate. The effects of the different concentration of strontium in treatment solution and cycle numbers of the pre-calcification treatment on the biocompatibility were investigated in terms of the changes in morphology and chemical composition of coating, ion release pattern and cytocompatibility
Results:
At a high substitution ratio of strontium in the calcium phosphate coating, the size of precipitated particles was decreased and the solubility of the coating was increased. ASH55 group, which was coated by pre-calcification treatment of 20 cycles in coating solution with Sr:Ca molar ratio of 5:5, exhibited superior cellular attachment at 1 day and proliferation after 7 days of culturing in comparison with the non-doped surface and other doped surfaces.
Conclusion:
Sufficient strontium doping concentrations in calcium phosphate coating can enhance cell adhesion and proliferation on the titanium biomedical devices for bone regeneration.
Introduction
Recently, the incorporation of strontium (Sr) into calcium–phosphate (Ca-P) ceramics has been extensively investigated for biomedical applications, particularly for bone regeneration.1–3 Sr can simultaneously result in both stimulation of bone formation and inhibition of bone resorption like the effects of BMP-2 and bisphosphonates. 4 Therefore, the coating of Sr-doped Ca-P ceramics on titanium (Ti) substrates makes Ti biomedical devices, such as implants or guided bone regeneration (GBR) membranes, biocompatible.
The optimum concentration of Sr in biomaterials has been the subject of much debate, without a consensus. Different amounts of Sr for optimal bone regeneration were reported through various study models from
The mineral deposition method was used to prepare both hydroxyapatite (HA) and ion-doped HA (iHA) coatings on Ti substrates among numerous methodologies of Sr incorporation into Ca-P coatings.1,14,15 This wet chemical method is applicable to complex geometrical shapes, and is cost-effective. Moreover, the obtained HA coatings possessed Ca-deficient and nonstoichiometric structures with higher solubility, potentially leading to improved substrate bioactivity.
4
Thus, the local release of biologically active ions like Sr at the implant site was suggested to stimulate the specific cell responses and the osteoconductivity of materials
In our previous studies, Ca-P coatings on TiO2 nanotubular arrays formed by anodization of Ti, which were prepared using cyclic pre-calcification and heat (APH) treatment,16,17 exhibited high bioactivity and good integration with bone tissue. Thus, in this study, Sr ions were added during cyclic pre-calcification to produce Sr-doped Ca-P coatings on Ti substrates. Additionally, to optimize the extent of Sr doping, various doping ratios and treatment cycle numbers were investigated in terms of the changes in morphology, bioceramic coating components, ion release pattern, bioactivity, and cytocompatibility
Materials and methods
Sample preparation
Pure Ti plates were prepared for APH treatment as described in our previous study. 16 Pure Ti plates (1 cm × 1 cm, 1 mm thick) were anodized in a glycerol–distilled water (DW)–NH4F (79, 20, and 1 wt %, respectively) electrolyte at 20 V for 1 h to form TiO2 nanotubes. Cyclic pre-calcification was subsequently performed by sequentially exposing anodized specimens to 50 mM NaH2PO4 (at 80 °C) and 10 mM Ca(OH)2 (at 100 °C) solutions for 1 min/cycle. This process was repeated for 10, 20, and 30 cycles. Samples were washed with DW to remove the residual coating solution and annealed at 500 °C for 2 h after pre-calcification. Sr was doped into Ca-P coatings by replacing Ca(OH)2 with Ca(OH)2/Sr(OH)2 mixtures of different compositions, as shown in Table 1, maintaining a total (Ca2+ + Sr2+) concentration of 10 mM. Sr-doped samples were named to reflect the used Ca:Sr ratios, i.e., ASH19, ASH37, ASH46, and ASH55. The non-doped sample (APH) was used as a control to evaluate the effects of different doping ratios.
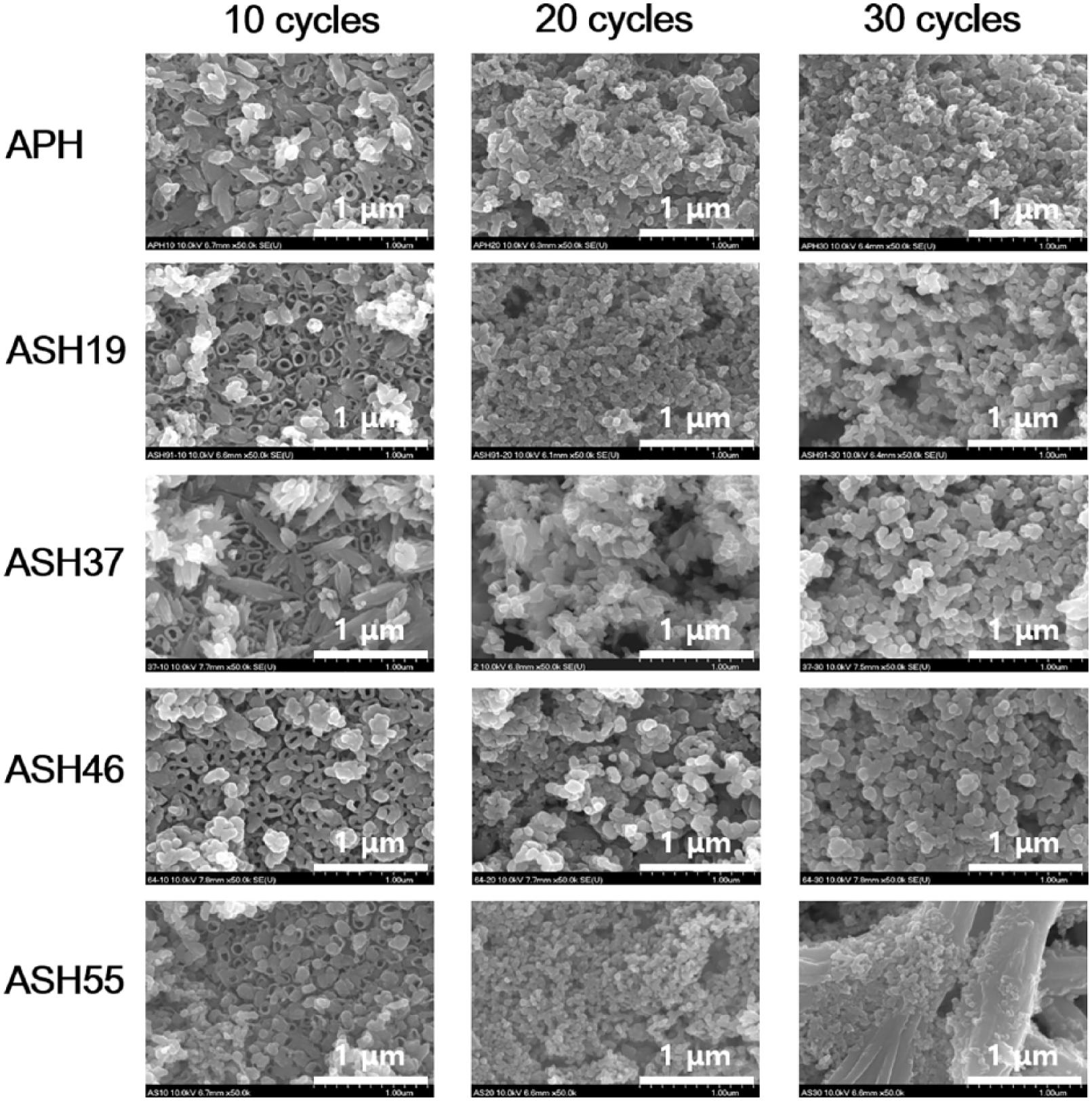
Concentrations of solutions used for cyclic pre-calcification and expected Sr/(Ca + Sr) molar ratios of treated samples.
Surface characterization
The morphologies and chemical compositions of sample surfaces were characterized by field emission scanning electron microscopy (FE-SEM, in KBSI Jeonju, S-4700, Hitachi, Japan) coupled with energy dispersive X-ray analysis (EDS).
Bioactivity test
Sr-doped and non-doped samples were immersed in simulated body fluid (SBF) solution for 1 day to assess their initial apatite formation ability. The SBF solution, exhibiting ionic concentrations similar to those of human blood plasma, was prepared by mixing 0.185 g L−1 CaCl2·2H2O, 0.35 g L−1 NaHCO3, 0.09767 g L−1 MgSO4 in Hank’s balanced solution (H2387, Sigma Chemical Co., USA) in which 0.4 g L−1 KCl, 0.06 g L−1 KH2PO4, 8.0 g L−1 NaCl, 0.04788 g L−1 Na2HPO4, 1.0 g L−1
Ion release test
A Ca- and P-free tris(hydroxymethyl)aminomethane-HCl (Tris-HCl) buffer solution was used for the ion release test. Processed samples were placed in separate plastic containers containing 10 mL of 0.1 M Tris-HCl buffer with pH 7.4 at 36.5 °C (standard ISO 10993-14), followed by shaking at 120 r/min and 37 °C. Sampling of solutions was performed by taking out 1 mL aliquots and replacing them with the same volume of fresh solution at the following time intervals: 0.5, 1, 2, 3, 4, 7, and 14 days. Control samples containing solely the 0.1 M Tris-HCl buffer were used to exclude any influence of the test process on the final results. All measurements were performed in triplicate. Solutions were filtered using sterile filters to ensure separation of any residual solids. Spectroscopic elemental analyses were performed using inductively coupled plasma mass spectrometry (ICP-MS, Agilent 7500A, Agilent Technologies, USA).
Cytocompatibility
Mouse osteoblast-like cells (MC3T3-E1, American Type Culture Collection) were cultured in Minimum Essential Medium-
Cell attachment
MC3T3-E1 cells with a density of 2 × 104 cell cm−2 were seeded on the samples. After 24 h culturing, non-adherent cells were removed by rinsing with PBS, whereas attached cells were fixed with 4% paraformaldehyde (MP Biomedicals LLC, Inc., OH, USA) for 30 min, washed with PBS, and permeabilized with 0.25% Triton X-100 in PBS (PBST) for 15 min. Non-specific binding sites were blocked with 1% bovine serum albumin (BSA) in PBST. Subsequently, samples were subjected to 30-min incubation with 500 μL of rhodamine-phalloidin (Cytoskeleton Inc., USA) at room temperature in the dark. Finally, cell nuclei were stained by 1 min incubation with 0.1 μg mL−1 4′,6-diamidino-2-phenylindole (DAPI, Enzo Life Science, Inc., NY, USA) followed by three PBS rinses. Cell morphologies were examined by confocal laser scanning microscopy (CLSM; LSM 510 Meta, Zeiss, Germany) at a magnification of ×630.
Cell proliferation
Cell viability was evaluated by measuring dehydrogenase activity in cell suspensions with a cell counting kit (CCK-8, Enzo Life Sciences, Inc., NY, USA) utilizing a highly water-soluble tetrazolium salt (WST-8). 2-(2-Methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium monosodium salt produces a water-soluble formazan dye upon reduction in the presence of an electron carrier, e.g., it is reduced by mitochondrial dehydrogenases in cells to a yellow-colored product (formazan) that is soluble in the tissue culture medium, with its amount being directly proportional to the number of living cells.
Cell seeding was achieved by incubating samples in a 24-well plate with 1 mL containing 2 × 104 MC3T3-E1 cells. The plates was kept in a humidified incubator under an atmosphere of 5% CO2 at 37 °C for 2 and 7 days. The culture medium was refreshed every two days. After incubation, microplate wells containing samples and cells were treated with 1 mL of CCK-8 solution (prepared by mixing with α-MEM in a volume ratio of 1:10) and incubated once more for 1.5 h as described above to allow the reduction of WST-8 into yellow-colored formazan. Finally, 200 μL aliquots taken from these wells were transferred to a 96-well plate (three wells per sample) and characterized using a microplate reader (Molecular Devices, CA, USA) at 450 nm. Cell proliferation was evaluated by comparing the absorbances of sample solutions to that of the control well containing CCK-8. The results obtained for WST-8 are reported as optical density (OD).
Statistical analysis
Statistical analyses were carried out using SPSS software version 12.0 (SPSS Inc., Chicago, IL, USA). Data were collected for three independent experiments and expressed as the mean ± SD (SD: standard deviation). Differences among groups were analyzed by one-way analysis of variance (ANOVA) using Tukey’s
Results and discussion
Sample surface characterization
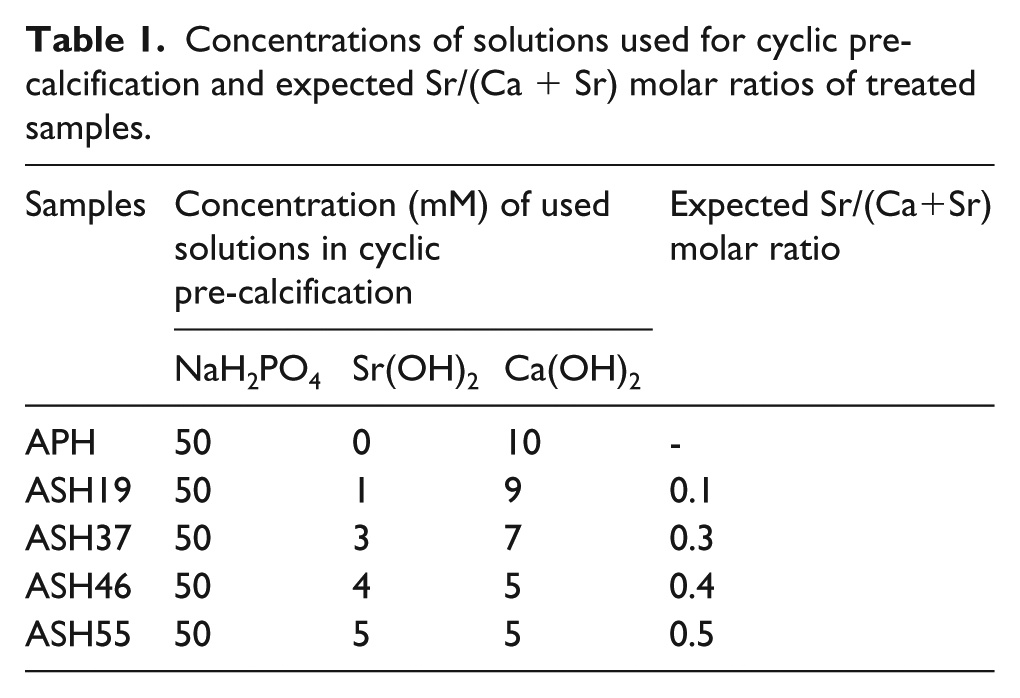
Figure 1 shows FE-SEM images of Ti surfaces after 10, 20, and 30 pre-calcification cycles with and without Sr doping. The number of cycles strongly affected the surface morphology of Ca-P coating formed on all anodized samples. The Ca-P coating layer composed of nanosized granules was partially formed on the anodized surface after 10 cycles of all groups, but granular coating layer covered the whole surface of all groups after 20 and 30 cycles. Apparently, the increased exposure time at pre-calcification solution led to increased amounts of Ca2+/Sr2+ and H2PO4− ions adsorbed onto the TiO2 nanotube surface via Coulombic interactions (16).
Field emission scanning electron microscopy (FE-SEM) images (top view, ×50.000) showing morphologies of different treated Ti surfaces.
Considering the different extents of initial Sr doping, ASH19, ASH37, and ASH46 groups did not exhibit large differences in the morphology and size of particles precipitated on anodized Ti substrates, as compared to the non-doped APH group. However, a noticeable decrease of granule size was observed for ASH55. In particular, after 30 cycles, large needle-like crystals with attached small granules formed on the ASH55 group. These crystals were characterized by EDS, which revealed the presence of Sr (41.96 wt %) as the major component (Figure 2).

Sr-containing crystals formed on ASH55 after 30 cycles. (a) Field emission scanning electron microscopy (FE-SEM) image (1000×). (b) Energy dispersive X-ray (EDS) detection at spectrum 2 point.
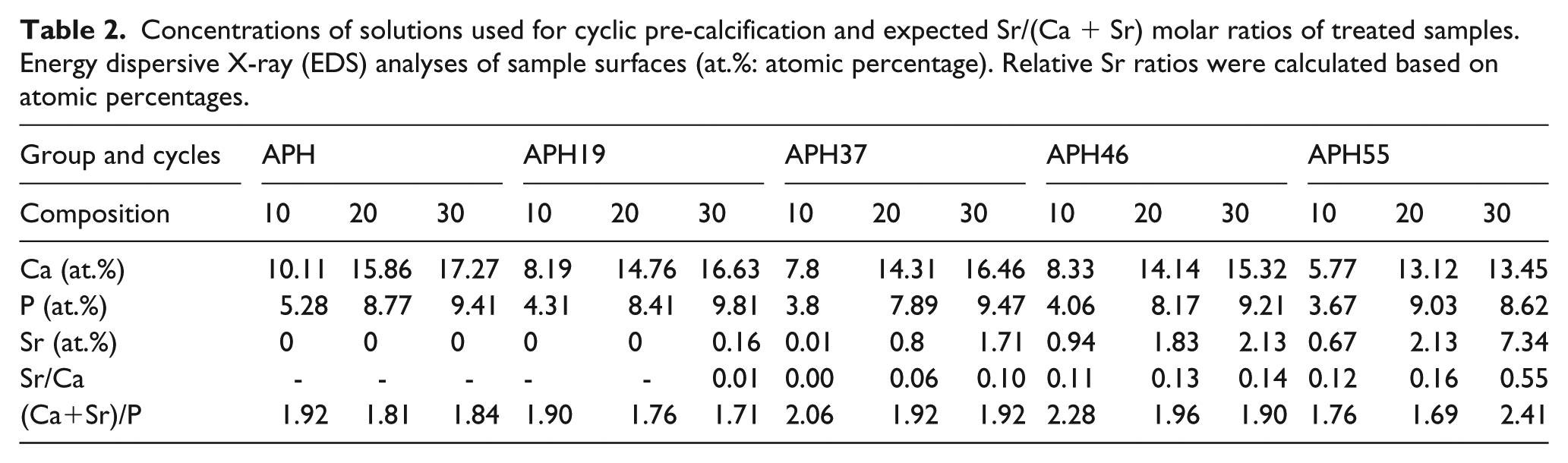
The amounts of Ca, P, and Sr (at.%) on treated surfaces were determined by EDS. The median values were chosen after analyzing two points for three samples for each group, as summarized in Table 2. Judging from the atomic percentages of Ca, P, and Sr, ion precipitation increased with increasing number of pre-calcification cycles. While the concentration of P was independent of the treatment method, the concentration of Ca on the non-doped surface (APH) was higher than that on Sr-doped surfaces (ASH19, ASH37, ASH46, ASH55) at the same number of cycles. On the ASH19 surface, no Sr were detected after 10 and 20 pre-calcification cycles, but Sr of very low level being observed after 30 cycles. The amount of Sr and Sr/(Ca + Sr) ratios increased with increasing the concentration of Sr(OH)2 in solution used for doping, and the (Ca + Sr)/P ratios of all surfaces exceeded the typical value of HA (1.67). These changes in element-to-Ca ratios indicated that Sr2+ ions in solution were incorporated into the Ca-P crystal lattice by occupying the sites of Ca2+, in accordance with earlier reports.10,19 Moreover, the total amount of ions incorporated into the mineral film by biomineralization increased with increasing ion concentration of the solution.10,15,19 In a previous study, it was reported that replacement of Ca by Sr through micro-arc treatment (MAO) also showed that increased Sr content in the electrolyte bath resulted in a greater extent of Sr substitution at Ca sites within the HA phase, which facilitate the formation of Sr-HA coatings where Sr was fully solid soluble in the HA phase. 20
Concentrations of solutions used for cyclic pre-calcification and expected Sr/(Ca + Sr) molar ratios of treated samples. Energy dispersive X-ray (EDS) analyses of sample surfaces (at.%: atomic percentage). Relative Sr ratios were calculated based on atomic percentages.
It was identified that the amount of Sr incorporated into the Ca-P structure could change the morphology of the coating, particularly for the heavily doped ASH55 group. This corresponds with the results obtained by Oliveira et al. for doping Sr into the apatite coating on the surface of Ti6Al4V alloy via biomimetic synthesis. 1 Insignificant apatite morphology changes were observed for small degrees of Sr substitution. However, when the concentration of Sr in solution increased to 1.0 and 1.5 mM, the size of apatite crystals was remarkably decreased. Li et al. also proved that incorporation of Sr at levels below 1.5% may not change the chemical composition and lattice structure of HA, 21 whereas a dramatic reduction of crystallinity was observed for 15% Sr-HA. Thus, the effect of Sr doping doses on Ca-P structure depends on the degree of substitution. A similar study for magnesium alloys also revealed a marked decrease of particle size with increasing the concentration of Sr2+. 22 At high doping level, the presence of Sr inhibits the growth of apatite crystals, which leading to the formation of a nano-apatite structure and reducing crystallinity.1,10,21 This observation is possibly related to the replacement of Ca2+ by the larger Sr2+ and agrees with earlier studies on the effect of Sr on Ca-P crystallinity.1,10,19 Thus, the decrease of precipitated particle size observed in the ASH55 group at over 20 cycles was attributed to the high degree of Sr substitution (>0.14). In particular, at 30 cycles, excessive doses of Sr resulted in the formation of big Sr-containing crystals on sample surface.
Bioactivity
Figure 3 shows that morphologies of all sample surfaces changed after immersion for 1 day in SBF. Precipitated particles were replaced by very large aggregates of nanosize crystals after immersion test. The crystals of APH and ASH19 groups had flake-like shapes, but those of ASH37, ASH46, and ASH55 groups had form of spongy in which thickness of crystals was thinner than crystals of APH and ASH19.
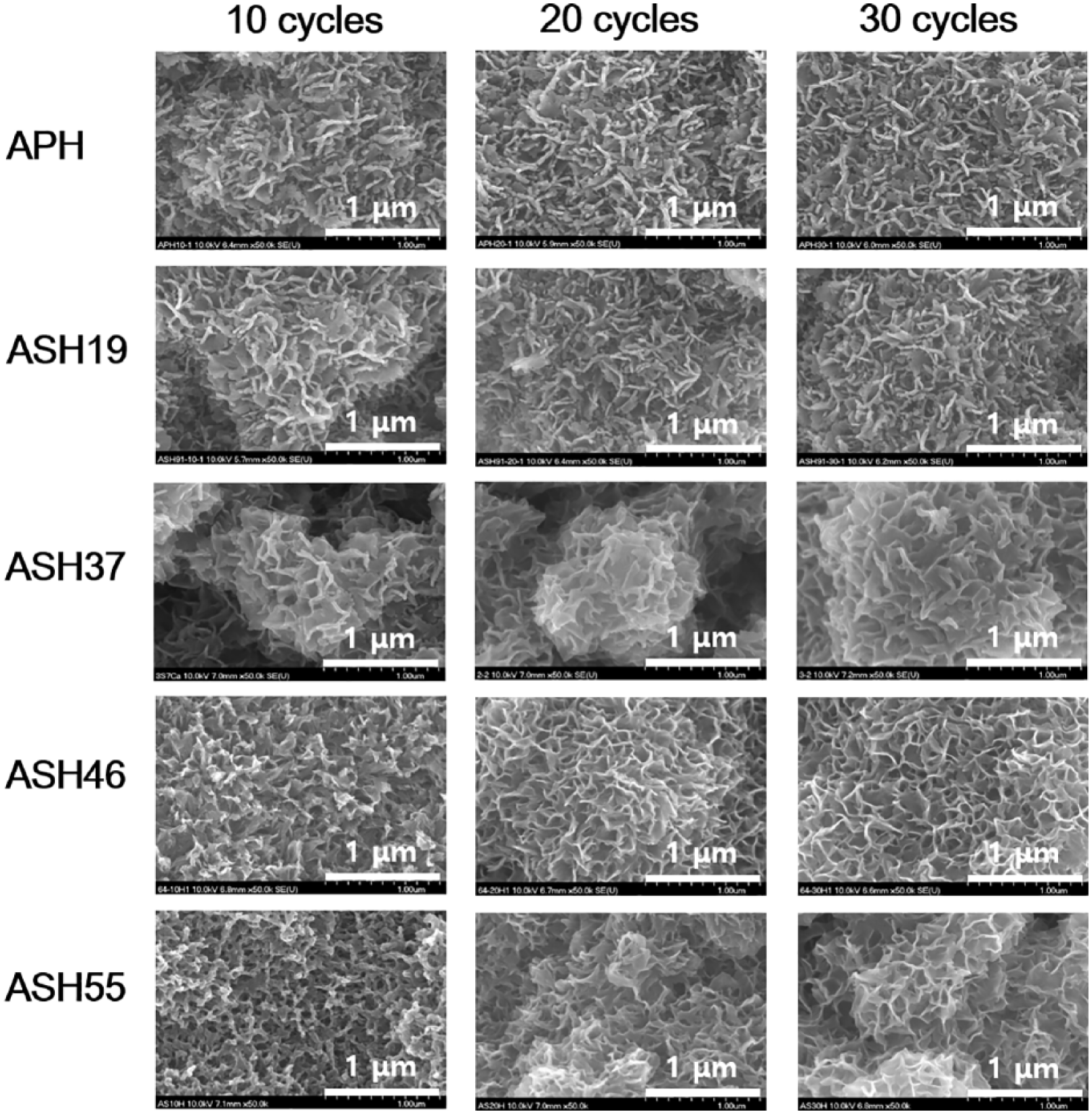
Field emission scanning electron microscopy (FE-SEM) images (top view, ×50,000) showing morphologies of different treated Ti surfaces after 1-day SBF immersion.
Table 3 summarizes the chemical composition of precipitates formed on the surfaces after immersion in SBF. All surfaces exhibited increased Ca and P atomic percentages and decreased amounts of Sr after immersion in SBF for 1 day SBF, which was resulted from the release of Ca, P and Sr from the coating and precipitation of Ca and P from the SBF solution.23,24
Energy dispersive X-ray (EDS) analyses of sample surfaces after 1-day SBF immersion (at.%: atomic percentage). Relative Sr ratios were calculated based on atomic percentages.
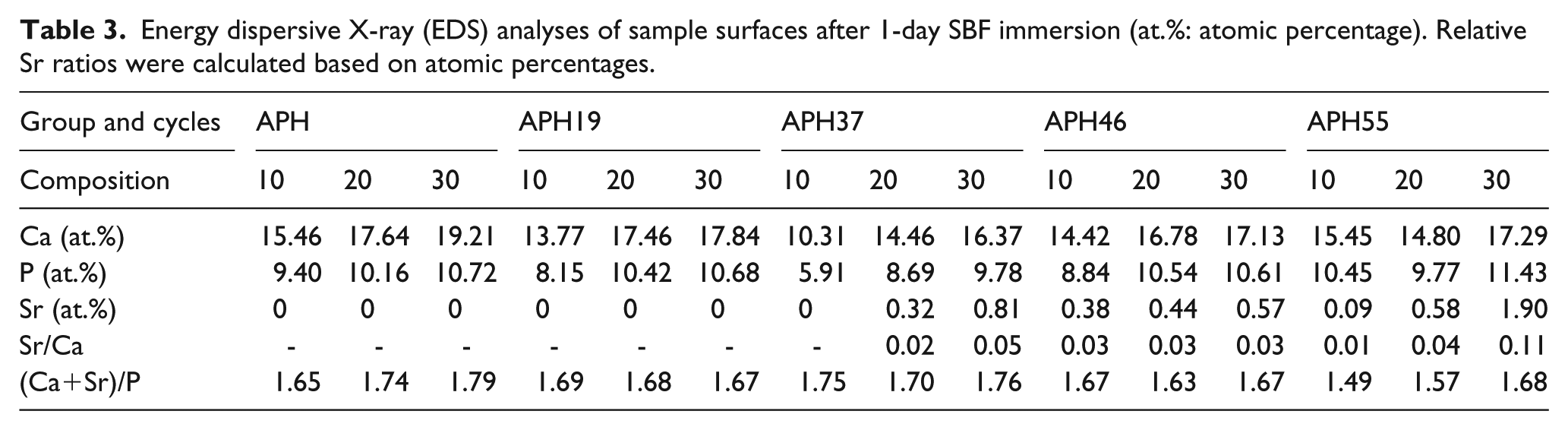
The surfaces of all samples were covered by a bioactive layer containing higher amounts of Ca and P than before immersion, and (Ca + Sr)/P ratios of all groups became closer to the typical ratio of HA (1.67), resulting in good bioactivity in SBF solution in all cases. 3 Although the effect of Sr doping on the bioactivity of the Ca-P coating was not obvious, small morphological changes of apatite precipitates were observed in ASH groups in comparison with the APH group. Lindahl et al. revealed that Sr influenced the biological formation of apatite in SBF at early stages. 23 Also, Christoffersen et al. reported that more apatite layer was formed on the surface of Sr-HA with Sr content below 10 mol.% than on pure HA in SBF, 9 which could be caused by higher dissolution rate of Sr-containing ceramics.
Based on the results for surface characterization and immersion test, 20 cycles for pre-calcification was selected, and ASH19 group was excluded due to unsuccessful Sr doping to reduce the number of groups for ion release test and cytocompatibility tests, since 10 cycles for pre-calcification was insufficient to cover the whole nanotubular surface, and excessive Sr accumulation on the ASH55 surface was observed at 30 cycles, resulting in the formation of large crystals.
Rate of ion release from coatings
The decrease of apatite crystallinity due to ion doping is expected to affect the release profile of the incorporated additive and the solubility of the recipient itself, 4 influencing their biological effects. Thus, in this study, the leaching of Sr and Ca ions from ASH samples were evaluated in a Ca- and P-free Tris-HCl buffer.
In the Tris-HCl solution, the release profiles of Ca2+ and Sr2+ were not affected by other ions, as displayed in Figure 4. The release profiles of Ca ions from various Sr-doped samples were similar, which was much higher than those of the APH group. Following the release of Ca ions, a rapid release of Sr ions occurred within 4 days, and the concentration of Sr was stabilized by day 14 for all investigated ASH groups. The lowest Sr2+ release was observed for ASH37 followed by ASH46, and the highest value was observed for ASH55. The concentrations of released Sr ions ranged from 251.67 ppb (ASH37 after 14 days) to 1603.67 ppb (ASH55 after 0.5 day), agreeing with the optimized range of strontium ranelate concentrations (210.7–21,070 ppb) recommended by Sila-Asna et al. to induce early osteoblast differentiation and enhance rapid bone formation. 7 The Sr/Ca ratios of Sr-doped groups in Tris-HCl were significant higher than those in coatings. In addition, the Sr/Ca ratio of ASH55 was different from those of ASH37 and ASH46 groups. During the release test, the Sr/Ca ratio of the ASH55 group lied above the 1.0 line, whereas that of other groups was below this line, but Sr/Ca ratios released from all ASH groups remained steady over the whole period of ion release test. This phenomenon results in the positive effects of Sr in Sr-HA materials. 11
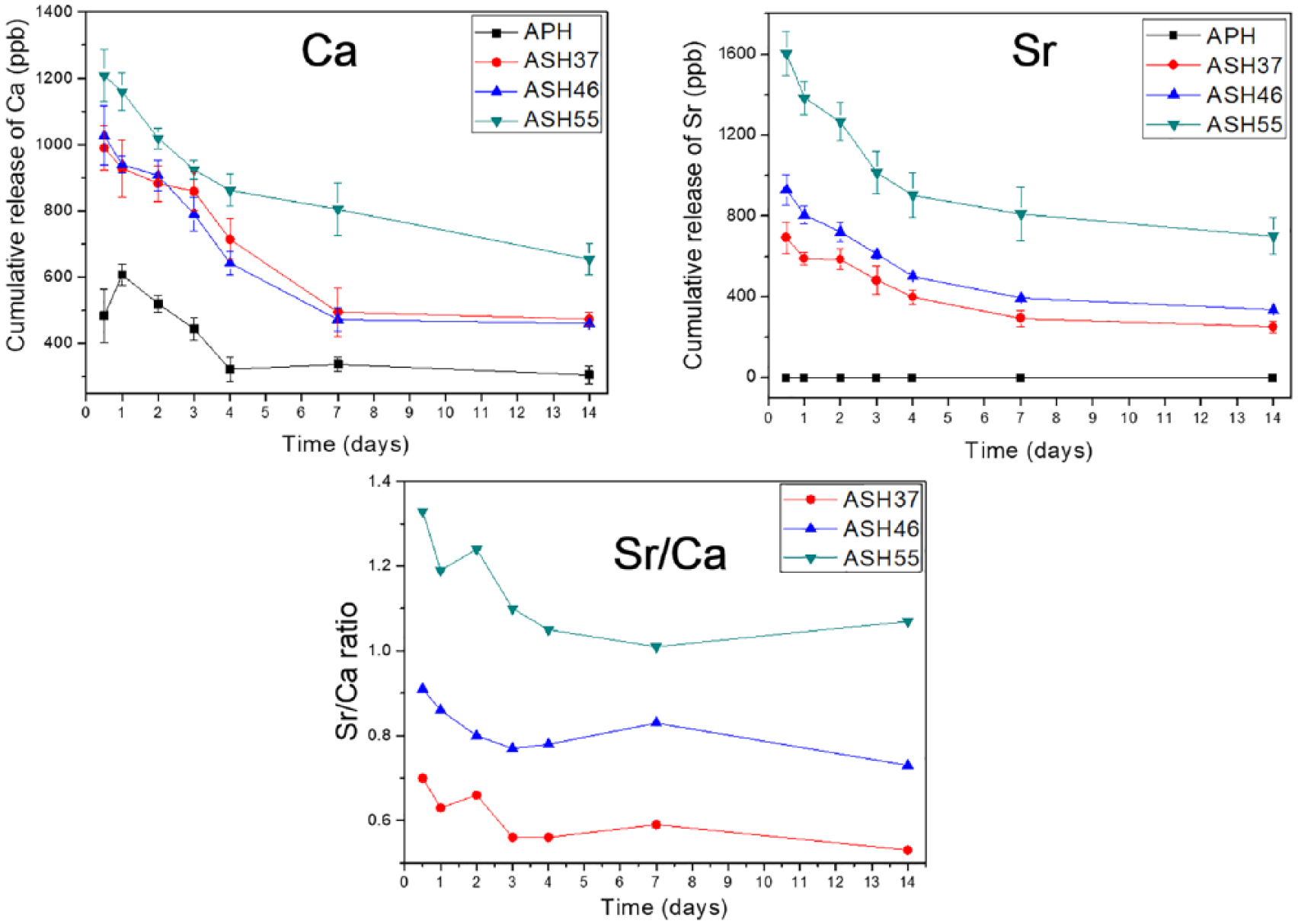
Cumulative release profiles of Ca2+ and Sr2+, and Sr/Ca ratios during release from treated samples in Tris-HCl solution.
The degree of ion release depends on the initial doping amount, which was the highest in the ASH55 group. Apparently, the presence of Sr increased the coating solubility due to the destabilization of crystallinity caused by the replacement of Ca by Sr.12,25 Christoffersen et al. also showed that the solubility of apatites containing 1–10 mol.% Sr2+ increases with increasing Sr content. 9 Thus, Sr-doped coatings are more soluble than non-doped ones due to the deformed lattice of the Ca-deficient HA structure, as reported in previous studies.4,26
Unlike the dissolution of bioactive coatings in SBF, where concurrent remineralization occurred, no interfering precipitation of new HA was observed in Tris-HCl, allowing the real solubility of coatings to be seen. Nanocrystalline apatites formed during biomimetic growth are known to be highly soluble due to containing loosely bonded ions. 24 Thus, the mechanism of ion release from biomimetic coatings was controlled by a combination of Fickian diffusion and coating dissolution, according to the analysis of Lindahl et al. using the Korsmeyer–Peppas model. 4
Cytocompatibility
It is important to investigate the effect of different concentrations of Sr2+ on cytocompatibility since it was identified that the metabolic activity of osteoblast-like cells could be enhanced by a dose-dependent method through various
For
Cell morphology
To investigate whether Sr substitution affects initial cellular attachment and morphology, cells incubated for 1 day were observed by fluorescence microscopy (Figure 5). Cells were stained with phalloidin to visualize actin (red) and DAPI to visualize nuclei (blue), exhibiting polygonal configurations with numerous lamellipodia (white asterisk) and filopodia (white arrow) extensions on all samples. However, osteoblasts on the ASH55 surface were even more flattened, wider spread, and exhibited better cell-cell connections than those on other surfaces. In previous study, significantly enhanced early-stage osteoblast responses (4 and 24 h) were observed on Sr-HA coatings with various Sr contents, as compared with those observed for raw Ti and non-Sr HA coatings. 20 Moreover, Bracci et al. reported that osteoblasts on a Sr-CaP coating on Ti appeared more flattened and better spread across the surface, forming an extracellular matrix, also mentioning the deposition of mineralized granules after 7-day culturing. 29 These studies support the current finding that early cellular adhesion is promoted by ASH55 group samples subjected to 20 pre-calcification cycles.
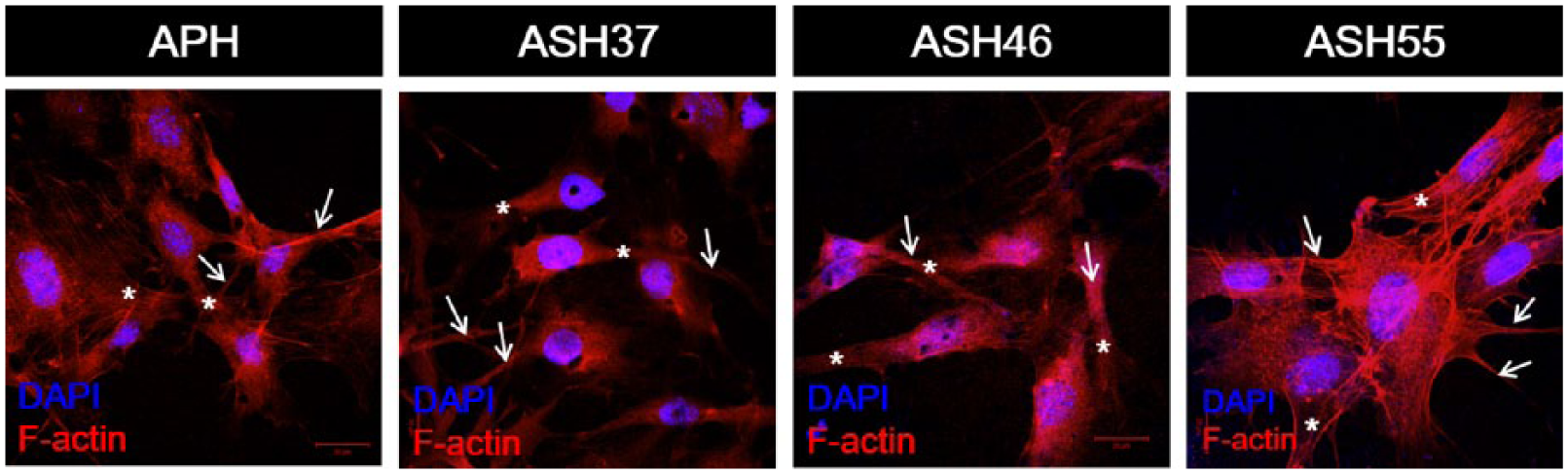
Cell morphology at 1 day, nuclei stained by DAPI, actin stained by phalloidin (×630). White arrows: filopodia, white asterisk: lamellipodia.
Cell proliferation
Although superior cellular activity was observed on the ASH55 surface, a quantitative cell proliferation test should be performed to clarify the effect of Sr doping on cellular behavior. As seen from Figure 6, all samples showed good cell proliferation during culturing. At 2 days, cells on treated Ti samples showed lower viabilities than those on a cell culture disc (control). However, ASH55 samples exhibited the highest cell viability at 7 days, which exceeded that of the control and APH samples (
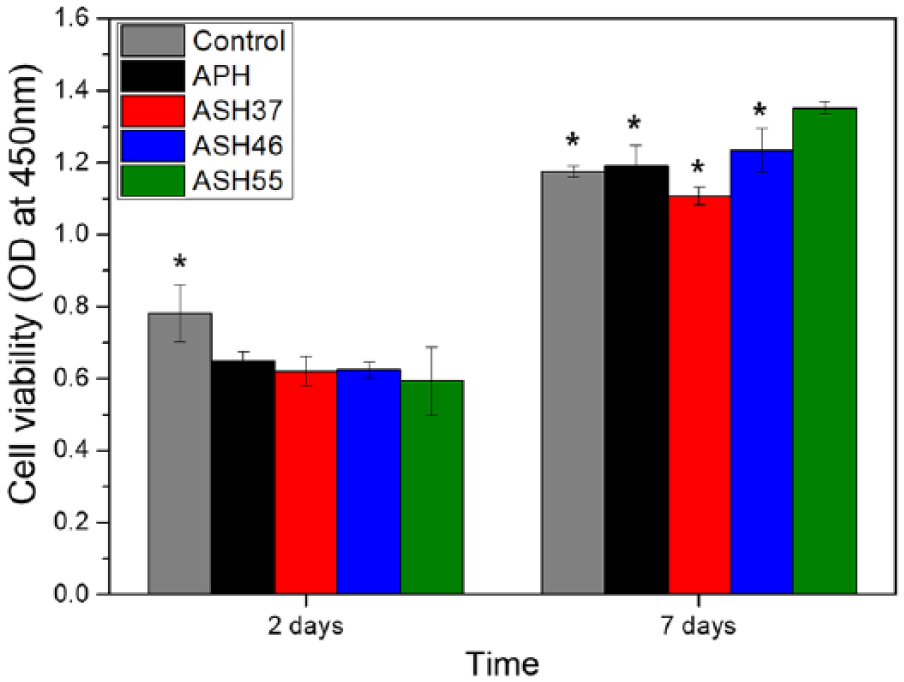
Cell proliferation (WST-8 assay) after 2 and 7 days of culturing. Significant differences from the ASH55 group (
Conclusions
In this study, Ca in cyclically pre-calcified surfaces was substituted by Sr using four different doping ratios and three different numbers of cycles. The morphology of these coatings changed, and a size decrease of precipitated particles was observed at high doping ratios. The solubility of coatings increased with increasing Sr content. Osteoblast responses were investigated
Footnotes
Acknowledgements
Thuy-Duong Thi Nguyen and Yong-seok Jang contributed equally to this work and are considered as joint first authors.
Declaration of Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) funded by the Korea government (MSIP) (grant number 2016R1A2B4011697).
