Abstract
Background:
Human umbilical cord mesenchymal stem cells (HuMSCs) injected directly have been proven effective for improving chronic wounds. However, HuMSCs largely die within 14 days. The aim of study is to establish a cellularly modified gelatin sponge and investigate its characteristics and clinical potential.
Methods:
HuMSCs were isolated, expanded and seeded in a poly-L-lysine (PLL)-coated gelatin sponge. Fabricated gelatin sponges were estimated through observation of morphological surface and ultrastructure, following confirmed by histology method. Supernatants were collected at different times for enzyme-linked immunosorbent assays (ELISAs) to measure growth factors. The cell embedded gelatin sponges were implanted subcutaneously on the backs of mice and the samples were harvested and studied histologically.
Results:
HuMSCs gradually modified the gelatin sponge by depositing collagen and hyaluronic acid, and degrading the structure of gelatin, resulting in a dense, and elastic structure. Compared with cells cultured in monolayer, the levels of growth factors increased remarkably when HuMSCs were cultivated in the gelatin sponge. Upon subcutaneous implantation in the backs of mice, the cellularized gelatin sponges persisted for up to 2 months and eventually integrated into the host tissue, while blank gelatin sponges degraded completely by the end of the second month.
Conclusion:
Gelatin sponge is a clinically accessible scaffold for HuMSCs implantation to maintain short-term survival of the cells and high-level production of growth factors, which demonstrates good clinical potential for enhancing wound healing.
Introduction
Cells therapy, which involves transfer progenitor/stem cells into human body through local or systemic delivery, offers a novel approach to cure chronic diseases such as diabetes, 1 arthritis, 2 and chronic wounds. 3 Among the different kinds of stem cells currently identified, mesenchymal stem cells (MSCs) are an optimal choice for treating chronic wounds, considering that MSCs take part in wound healing by enhancing extracellular matrix (ECM) production and secreting growth factors. 4 A variety of preclinical and clinical studies have shown MSCs therapy to be useful for treating non-healing wound. MSCs have several advantageous properties, including ease of isolation from adult or fetal tissue sources (e.g. bone marrow, adipose tissue, and umbilical cord), a large capability of being expanded in vitro, immunosuppressive activity and differentiation multipotency.5,6 Among MSCs, human umbilical cord-derived MSCs have been increasingly noticed for their advantages of acquisition by non-invasive harvesting, no ethical problems, low immunogenicity, noncontroversial nature, and excellent expandability. 7
Chronic wounds, exhibit common features, including excessive levels of pro-inflammatory cytokines, proteases, reactive oxygen species (ROS), and senescent cells, as well as persistent infection. 8 Persistent inflammation is a hallmark of chronic non-healing wounds, and chronic wounds might be trapped in inflammatory state that fails to progress. 9 Although inflammatory cells can produce growth factors that are crucial for new tissue formation, they also secrete numerous toxic mediators, including proteases and ROS that are harmful to the surrounding tissues, making the microenvironment extremely harsh for engrafted cells. 10 Locally-injected MSCs are also known to be largely lost soon after transplantation due to cellular damage caused by enzymatic treatment during harvesting 11 and the lack of cell-ECM attachment. 12
ECM can promote cell density and cell survival at the transplantation site, probably by preserving the cell-cell junctions and cell surface proteins that are disrupted by enzymatic treatment, so an ECM can assist cells in promoting recovery of tissue function, tissue regeneration, and therapeutic efficacy compared with single cell suspensions.13–15 By mimicking the features of an ECM, numerous biocompatible biomaterials are being developed for dermal filling or wound healing care, and are also used to enhance delivery efficiency and therapeutic efficacy in forms of cell sheets, cell-loaded hydrogels and composite scaffolds.16,17 However, the fabricated scaffolds using chemical cross-linking agents are mainly associated with toxicity due to aldehyde residues that cause damage to the targeted organ.18,19
The ideal scaffold should be stable, inexpensive, readily available, and have excellent biocompatibility and the ability to be loaded with viable cells, and finally it should not be harmful to the targeted tissue. Gelatin sponges are biocompatible and biodegradable biomaterials that have proven beneficial clinical properties. The porous structure of the gelatin sponge can be conducive to cell attachment. As a commercial product, gelatin sponges are also inexpensive and readily available. Given these advantages, we chose gelatin sponges as the scaffold to prepare cellularized biomaterials in this study.
To improve HuMSCs adhesion to the gelatin sponges, the scaffolds were coated with PLL before seeding cells. The study investigated PLL which are positively charged polyelectrolyte chains that help MSCs adhere to the scaffold by exploiting the negative charge of the cell membrane surface. 20 It is expected that HuMSCs could modify the biodegradable materials to provide an optimal microenvironment to maintain cell survival and functioning, which would be confirmed in the in vitro and in vivo experiments.
Materials and methods
Isolation and culture of HuMSCs from human umbilical cords
The ethical permission was approved in accordance with the Ethics Committee of Shantou University Medical College (SUMC, Shantou, China). With a previously signed informed consent form, umbilical cords from full-term (38–40 weeks) healthy pregnancies (excluding patients with HIV, hepatitis, and syphilis) through cesarean section were obtained from the Department of Obstetrics and Gynecology, the Second Affiliated Hospital of SUMC. Primary HuMSCs were isolated, cultured, and characterized in the laboratory of the research center. Briefly, umbilical cords were harvested under sterile conditions and washed in phosphate-buffered saline (PBS). Wharton’s jelly was exposed and isolated from the dissected umbilical cords. Then Wharton’s jelly pieces were cut into pieces about 1 mm2 and attached to 10 cm plates (JET BIOFIL, China). The prepared high glucose Dulbecco’s modified Eagle’s medium (DMEM, Gibco, Thermo Fisher Scientific, Inc., USA) containing 2% fetal bovine serum (Gibco, USA), 1% penicillin and streptomycin solution (Beyotime, China), basic fibroblast growth factor (bFGF, Zhuhai Essex Bio-Pharmaceutical Co., Ltd., China), transferrin (Sigma, Sigma-Aldrich, Co., USA), insulin (Sigma, Sigma-Aldrich, Co., USA), and selenium acid (Aldrich, Sigma-Aldrich, Co., USA) was added to the plates and the tissue pieces were incubated in a humidified CO2 incubator at 37°C. The culture medium was replaced after HuMSCs grew out of the explants after 5–7 days. When the cells reached confluence, the cells were passaged. Cells of the third passage were used for the following studies.
Preparation of gelatin sponge seeded with HuMSCs (GS-MSC)
Gelatin sponges (Xiang’en, Jiangxi Xiang’en Medical Technology Development Co., Ltd.,China) were cut into 20 mm ×20 mm ×10 mm pieces and immersed into 0.75 mg/L PLL solution (Beyotime, China) overnight at room temperature. The gelatin sponges were washed three times with PBS and placed in six-well plate, prewetted with culture medium prior to cell seeding. Each gelatin sponge block was seeded with 1.5 mL of HuMSCs suspension (3 × 106 cells/mL) and incubated at 5% CO2 and 37°C for 30 min. Medium for the multilayer culture was prepared by adding 1 µL/mL ascorbic acid (50 µg/mL). After incubating cells with gelatin sponges for 30 min, each sponge was immersed in 6 mL of prepared culture medium. The culture medium was changed every 2 days throughout the duration of the experiments. The morphological changes of GS-MSCs at Day 14 and Day 28 were captured by a digital camera (EOS 6D, Canon, Japan). The GS-MSCs were harvested for histological and ultrastructural studies and for implantation into mice.
Histological study of GS-MSCs
The GS-MSCs were collected at Day 14 and Day 28 and fixed with 4% paraformaldehyde overnight. After GS-MSCs were paraffin-embedded and sectioned at 4 μm, the sections were rehydrated, dewaxed and stained with hematoxylin-eosin (HE), Van gieson (VG), and Alcian blue (AB) stains according to respective protocols. HuMSCs embedded in the gelatin sponge could be stained with HE. Collagen and hyaluronic acid produced by HuMSCs were visualized through VG and AB staining. Images were captured with a fluorescence microscope (Axio Imager, Zeiss, Germany).
Scanning electron microscopy
The ultrastructure of GS-MSCs was observed by SEM. The preparation of samples was in accordance with procedures reported, 21 with minor modifications. Briefly, GS-MSC samples were harvested on Day 14 and Day 28, and fixed with 2.5% glutaraldehyde for 3 h at 4°C, and then washed three times with PBS, each for 15 min. The samples were dehydrated in a graded series of ethanol for 10 min and air-dried at room temperature. Then they were coated with gold and observed with a Gemini 300 field emission scanning electron microscope (ZEISS, Germany).
Evaluation of growth factor levels (ELISA assay)
Collected culture supernatants were divided into two groups: one group where supernatants were collected from monolayer HuMSCs (cultivated on Day 3 and Day 5) and the cells were counted, and the other group where GS-MSCs supernatants were collected on Day 3, 7, 14, 21, and 28. One day before supernatant collection, the culture medium was changed to serum-free medium. Supernatants were centrifuged at 3000 rpm for 10 min to remove floating cells and cell debris, and then aliquoted for ELISA analysis. The secreted levels of vascular endothelial growth factor (VEGF), keratinocyte growth factor (KGF), hepatocyte growth factor (HGF) in the supernatants were quantified by ELISA kits (R&D systems, USA) according to the manufacturer’s protocol. The data expressed were compiled from three independent assays, with each performed in duplicate, and growth factor levels were calculated from four parameter logistic curves.
Subcutaneous implantation of GS-MSCs
Male Kunming mice, 7–8 weeks of age and weighing about 30 g, were purchased from the Laboratory Animal Center of SUMC. We used male mice to exclude possible hormone effects on graft taking and cell functioning. Twenty mice were divided randomly into two groups. Mice in one group were implanted with a blank gelatin sponge on one side and a GS-MSC (14 days after cell loading) on the other side, and mice were subsequently housed for 1 month. In the other group, mice were similarly transplanted but subsequently housed for 2 months. The implantation areas of their backs were depilated under anesthesia by intraperitoneal injection of pentobarbital (40 mg/kg) and sterilized with Betadine. Blank sponges soaked with culture medium and GS-MSCs were implanted subcutaneously respectively on each side of the back of the mice. After being implanted for 1 and 2 months, the mice were euthanized, and samples were harvested and analyzed histologically. Paraffin-embedded specimens were prepared as above. The samples were stained with HE, VG, and AB and were observed under the microscope.
Statistical analysis
The data are presented as the mean ± standard deviation (SD), and comparisons between groups were analyzed with one-way ANOVA. The comparisons among collections of growth factors were presented. A p < 0.05 was considered statistically significant.
Results
Morphological changes of gelatin sponges
The gelatin sponge was highly porous originally (Figure 1(a)). After cell seeding, the sponge surface became smooth. GS-MSCs resembled a rubbery jelly with smooth surface after culture for 1 month. GS-MSCs did not change much in size by Day 14 (Figure 1(b)) while shrinking to 3/4 of the original by Day 28 (Figure 1(c)).

Morphological surface of blank gelatin sponge and GS-MSCs (Day 14 and Day 28): (a) blank gelatin sponge and magnification of highly porous gelation sponge (insert); length: 2 cm, (b) GS-MSCs harvested at Day 14; length: 2 cm, and (c) GS-MSCs harvested at Day 28; length: 1.5 cm.
Histology of GS-MSCs in vitro
Blank gelatin sponges and GS-MSCs cultivated for 14 and 28 days were analyzed by HE, VG, and AB staining. In blank gelatin sponges, gelatin fibers were loosely and sporadically arranged without cells, collagen fibers, and hyaluronic acid (Figure 2(a)–(c)). HuMSCs, collagen fibers and hyaluronic acid were located throughout the surface area of the GS-MSCs on Day 14 (Figure 2(d)–(f)). The filamentous collagen fibers were stained red with VG stain (Figure 2(e) and (h), black arrows) and hyaluronic acid were stained blue with AB stain (Figure 2(f) and (i), red arrows). HuMSCs gradually migrated into the innermost part and its secretions distributed around the cells. Along with the remarkable increase in HuMSCs, collagen fibers and hyaluronic acid were densely distributed in the superficial regions of GS-MSCs (Figure 2(g)–(i)).

Histological assessment of blank gelatin sponge and GS-MSCs (Day 14 and Day 28). Gelatin sponge without HuMSCs (a), collagen fibers (b) and hyaluronic acid (c). HuMSCs were stained purple with HE staining (d and g). Collagen fibers were stained red with VG staining (e and h; black arrows). Hyaluronic acid were stained blue with AB staining (f and i; red arrows). (d and f) Showed the changes of GS-MSCs at Day 14. (g–i) Showed the changes of GS-MSCs at Day 28. HuMSCs, collagen fibers, and hyaluronic acid of GS-MSCs at Day 28 distributed more than GS-MSCs at Day 14. Bars:100 μm (a–i).
Scanning electron microscopy
The original gelatin sponge displayed a porous structure with thin, sharp gelatin fiber walls (Figure 3(a)). After cell seeding and culture for 14 days, SEM showed that cells had adhered to the porous cavities of gelatin sponge (Figure 3(b) and (c)). With the proliferation of HuMSCs, the original gelatin fibers (Figure 3(d), blue arrow) were covered with massive ECM matrix composed of collagen fibers and hyaluronic acid by Day 28. Some of HuMSCs still adhered to chambers of gelatin sponges, but most of them distributed throughout ECM matrix (Figure 3(e)).

Ultrastructure observation of a blank gelatin sponge and GS-MSC (Day 14 and Day 28): (a) blank gelatin sponge with sharp fiber, (b) gelatin sponge after seeding with HuMSCs for 14 days, (c) magnification of B, HuMSCs adhered to the porous cavity of gelatin sponge, and (d) gelatin sponge after seeding with HuMSCs for 28 days. Original gelatin fibers (blue arrow) were covered with cells and ECM matrix. (d) HuMSCs and ECM matrix combined together. Bars: 20 μm (a, b, d), 10 μm (c and e).
Increased levels of HuMSCs-secreted growth factors at different times in the GS-MSC group
Supernatants from monolayer cultures and GS-MSC cultures were collected and measured for growth factors. Since the cell number and amount of culture medium for monolayer and GS-MSC cultivation were different, the value of growth factor concentrations was adjusted to be pg/mL/million cells. The secretion levels of HGF, VEGF-α, and KGF released by GS-MSCs were increased and maintained at a higher level compared with those from monolayer HuMSCs (Figure 4). Levels of growth factors were enhanced obviously on GS-MSCs groups by Day 3 and significant differences (p < 0.05) between monolayer HuMSCs and GS-MSCs were observed. The secretion of three growth factors was not significantly increased in monolayer groups (p > 0.05). In GS-MSC groups, VEGF-α secreted highest among the three growth factors. The secretion of VEGF-α increased consecutively in 1 week and then the level was sustained for 1 month. Secretion of HGF peaked on Day 3 and decreased slowly to the same level as monolayer groups from Day 21. Secretion of KGF rose considerably and peaked on Day 7, then gradually decreased but still maintained higher levels compared with monolayer groups.

Growth factor assessment of monolayer groups and GS-MSCs groups. The supernatants of monolayer groups were collected at Day 3 and Day 5. The supernatants of GS-MSCs groups were collected at Day 3, 7, 14, 21, and 28. (a–c) Showed the secretion trend of VEGF-α, HGF, and KGF. monolayer d5 versus GS-MSCs groups.
Histological analysis of gelatin sponges and GS-MSCs following in vivo implantation
After being implanted with blank gelatin sponges and GS-MSCs for 1 and 2 months, mice were euthanized, and the sponges were harvested and analyzed histologically. By 1 month, the implanted blank gelatin sponges became transparent and small (Figure 5(a), yellow arrow). GS-MSCs also shrunk in size but blood vessels appeared around the implanted GS-MSCs (Figure 5(a), green arrow). By the first month, the blank sponges were deeply infiltrated by host cells and the structure of gelatin fibers was loose (Figure 5(c), black arrow). After GS-MSCs had been implanted for 1 month, the original gelatin fibers had already started dissolving and were being replaced with densely deposited collagen and hyaluronic acid (Figure 5(d), green arrow). Such an ECM matrix, mainly composed of collagen and hyaluronic acid, may repress inflammatory cell invasion (Figure 5(d), blue arrow). By 2 months, the implanted blank sponges had already disappeared, but GS-MSCs were still present, although shrinked to tissue mass (Figure 5(b), red arrow). Similarly, blank sponges could not be identified, but GS-MSCs were still present and had become integrated into the surrounding tissue (Figure 5(e), blue arrow).
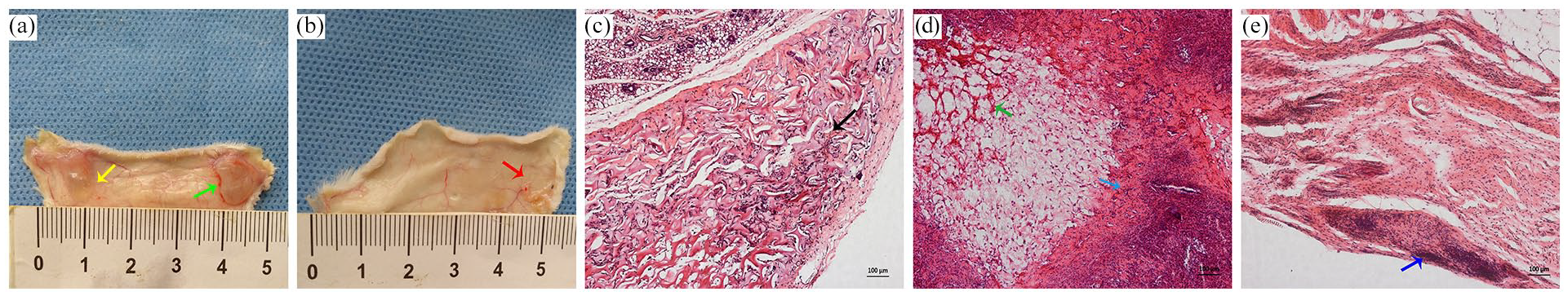
Gross view and histological assessment of blank gelatin sponges and GS-MSCs in vivo. The samples were harvested at 1 month and 2 months. By 1 month, implanted blank gelatin sponges became transparent and small (a, yellow arrow) and the implanted GS-MSCs can cause microangiogenesis (a, green arrow). By 2 month, GS-MSCs shrinked to tissue mass (b, red arrow) and blank gelatin sponges have degraded. Histological result: By 1 month, original gelatin fibers of GS-MSCs were being replaced with ECM (d, green arrow). GS-MSCs may resist immune cell infiltration (d, blue arrow) compared with blank gelatin sponge (c, black arrow). GS-MSCs transplanted for 2 months had still not been completely degraded (e, blue arrow). Bars: 100 μm (c–e).
Discussion
Studies have shown that MSCs exert therapeutic effects mainly through secretion of trophic mediators, rather than directly differentiate to repair injured tissues. 22 MSCs secrete numerous mediators, including growth factors, cytokines, and chemokines. Certain growth factors are known to be involved in wound healing. VEGF is a key component in the development of blood vessels, that can reestablish the blood supply for wound healing.23,24 Angiogenesis is so important that it can provide fibroblasts with a sufficient nutrient supply during the proliferation phase of wound healing. 25 Without adequate microvascular vessels, the wound closure may develop into a chronic non-healing wound. HGF is another important growth factor that can help reduce scar formation through inhibiting myofibroblastic differentiation of fibroblast 26 and reform the harsh environment in non-healing wounds. 27 KGFs have been shown to promote cellular proliferation, ECM formation and angiogenesis 28 and help reepithelialization and epidermal homeostasis of the wounds. 29 In our study, HuMSCs cultured in the gelatin sponge effectively promote three growth factors which are critical to wound healing process. To fill GS-MSCs in the wounds of patients may be effective therapy clinically.
Despite MSCs playing a critical role in wound healing by paracrine mechanisms, a remaining challenge lies in how to maintain a lasting therapeutic effect. Over 90% of MSCs grafted through simple injection can die within 14 days.30,31 Ischemia, anoikis, and lack of adherence to surrounding ECM may be major causes for the death of the grafted cells.32,33 For chronic wounds, the inflammatory environment is extremely harsh for MSCs survival. 34 With a profound loss of cells, therapeutic effects of MSCs will be diminished.
Preconditioning of MSCs is essential to enhance the tissue repair potential of MSCs. Effective means for enhancing growth factor production include hypoxic culture or cultivation in a 3-D scaffold.35,36 An established 3-D structure could provide a stable scaffold where embedded MSCs could keep in constant contact with the surrounding ECM and the cells could elude detrimental factors in the host environment. In our previous work, a 3-D platelet poor plasma (PPP) gel was used as a temporary scaffold, which facilitated HuMSCs survival, produced higher levels of growth factors and repaired skin wounds faster than traditional injections of cell suspensions. 37 However, PPP gels had to be extracted and processed from autologous blood, and PPP gels might undergo fibrinolysis within several days. Consistent with our previous study, HuMSCs cultivated in a gelatin sponge, produced remarkably higher levels of growth factors than monolayer cultures. Although the levels of growth factors varied during the 28-day period, they were maintained at relatively high level, especially VEGF. This finding suggests that GS-MSCs might keep releasing growth factors to enhance wound repair when being implanted in a host. In our vivo study, GS-MSCs were retained for at least 2 months while the pure gelatin sponges completely degraded. GS-MSCs degraded slowly, which allowed the integration of host cells and further deposition of ECM. The distinct ECM proteins deposited from serum onto scaffold may mediate additional adhesion of MSCs to these polymers. 38 HuMSCs and ECM matrix mutually promote each other. HuMSCs secrete hyaluronic acid, collagen fiber and enzymes for ECM formation, a process that could continually modify the surrounding tissue to aid wound healing. Our results show an MSC-cellularized gelatin sponge might be a good bioactive cell-matrix complex for enhancing wound healing.
Nevertheless, we failed to mark the HuMSCs in the scaffold. We tried to label HuMSCs with Dil staining prior to implanting into the gelatin sponge. However, cell proliferation was suppressed and cells did not survive in the gelatin sponge in vitro beyond 14 days. Therefore, we could not track the fate of the implanted HuMSCs and differentiation of the cells in the gelatin sponge after implantation. In further experiments, a better labeling procedure should be adopted that could stain the cells for a longer period without adverse effects on cell survival.
Conclusions
In conclusion, the gelatin sponge is a clinically and economically available scaffold for HuMSCs embedding. The cellularly modified gelatin sponge could enable prolonged retention of HuMSCs, allowing the HuMSCs exert their effects through paracrine mechanisms involving continuous release of growth factors, followed by integration into the host environment. The potential for enhancing wound repair in vitro and in vivo will be examined in further experiments involving chronic wound animal models.
Footnotes
Acknowledgements
We would like to thank the Department of Obstetrics and Gynecology, the Second Affiliated Hospital of SUMC for their assistance in obtaining umbilical cords.
Author Contributions
LY conceived of the project, designed the study and revised the manuscript. DY performed most of the research, data analysis and manuscript writing. SW participated in the vivo experiments and collected the data. BZ and CH contributed to data analysis. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by the National Natural Science Foundation of China (grant NO. 81272116), Grant for Key Disciplinary Project of Clinical Medicine under the Guangdong High-level University Development Program and Shantou Medical and Health Science and Technology Project:2020(58)-25.
Guarantor
LY
