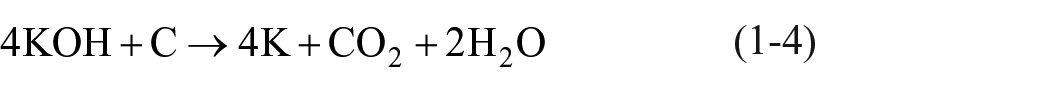Abstract
The electro-Fenton (EF) process is one of the advanced oxidation processes (AOPs). Graphite felt is widely used as an cathode material for the EF process, and its performance can be improved by surface modification. Active carbon nanotubes (ACNTs) have more oxygen-containing functional groups and better electrochemical properties compared to Multi-wall carbon nanotubes (MWCNTs). In this study, graphite felt was used as the substrate, and composite cathodes were prepared by surface treatment using MWCNT, graphene, and ACNTs. Rhodamine B (RhB) dye decolorization tests were then conducted to investigate the degradation performance of the EF system with different cathodes. The results showed that based on the micromorphology of ACNT, the tubular form of MWCNT was activated into a GR-like flake structure, it was also found that the strength of the oxygen-containing functional groups of ACNT improved significantly. The activated MWCNT/C cathode exhibited a 60-min decolorization rate of 77.28% compared to the unactivated MWCNT/C cathode, whereas the decolorization rate of the ACNT/C cathode increased to 85.01% after activation, which was close to that of the GR/C cathode at 88.55%. In summary, the ACNT/C cathode exhibited degradation efficiency comparable to that of the GR/C cathode.
Introduction
In the electro-Fenton (EF) system designed and developed by Oturan,
1
the EF process involved differed from previous processes. In this process, ferric ions [
Carbon nanotubes (CNT) and graphene (GR) are allotropic materials with properties such as high specific surface area, excellent conductivity, high mechanical strength, and high ductility8,9; therefore, they are the ideal cathode materials. Zhang et al. 10 synthesized the CNTs and performed cyclic voltammetry tests. The results showed that H2O2 output from the CNT cathode increased by 11.38 mg/L than graphite cathode, indicating that the application of CNTs on cathode can improve performance to good effect. Mousset et al. 11 used graphene to modify carbon cloth and then performed cyclic voltammetry tests, which showed a 10-fold increase in the active surface area of the graphene-modified carbon cloth. Another experiment showed that the use of CNT- and graphene-modified carbon felt cathodes can improve the treatment efficiency of an EF system compared to Reactive Black 5 (RB5). Based on the findings of this experiment, graphene-modified carbon felt cathodes could increase treatment efficiency by approximately 14% compared to CNT-modified carbon felt cathode. 12 However, it is not easy to mass-produce high-quality graphene at the moment and doing so will increase the cost of EF systems.
In recent years, scholars have studied the surface modification of graphite fibers and nanotubes, and found that the structure of carbonaceous materials can be effectively modified by alkaline compounds, such as potassium hydroxide (KOH) and sodium hydroxide (NaOH), thereby significantly increasing their specific surface area.13,14 Raymundo-Piñero et al. 15 found that a better activation effect could be achieved using KOH compared to NaOH. In the activation mechanism, K2CO3, H2, and potassium are formed when carbonaceous materials and KOH are heated in an inert gas atmosphere at a temperature higher than 700°C, and the potassium thus formed is embedded into the carbon substrates. This phenomenon leads to the widening of the spaces between layers of carbon atoms and an increase in pore volume. 16 The experiment conducted by Niu et al. 17 showed that the specific surface area of KOH-activated multi-wall carbon nanotubes (MWCNTs) significantly increased by 12.8 times compared to unactivated MWCNTs. Wang et al. 18 activated graphite felt using KOH and then used it for degrading dimethyl phthalate (DMP) through the EF process. Based on the findings of their study, the reaction rate of activated graphite felt cathode could be increased by 10 times compared to unactivated graphite felt cathode. The reactions involving KOH-activated MWCNTs are expressed in equations (1-4) through (1-6). 19
Based on the literature cited above, this study carried out the surface modification of graphite felt substrates by coating equal amounts of MWCNTs, GR, and ACNTs onto these substrates in order to investigate the treatment efficiency of different composite cathodes in the EF process, with the aim of improving the electrochemical properties of MWCNTs through the activation of MWCNTs using KOH, in the hopes of preparing ACNT materials whose performance is comparable to that of graphene.
Experiment methods
Activated carbon nanotubes
Multi-wall carbon nanotubes (MWCNTs, Cheng En Material Tech. Co., Ltd., Taiwan) were mixed with potassium hydroxide (KOH, Nihon Shiyaku Industries, Japan) based on a weight ratio of 1:4. Next, KOH powder and MWCNTs were mixed thoroughly and placed in a vacuum oven and baked at 110°C for 12 h in order to remove residual moisture. After that, the powder mixture was put in a porcelain combustion boat and placed in a horizontal tube furnace. The powder mixture was heated to 800°C at a rate of 10°C per min and kept at a constant temperature for 1 h. The horizontal tube furnace was vacuumed before heating. The activation process was carried out in a chamber filled with nitrogen.
The activated MWCNT powder was put into a deionized aqueous solution and neutralized using a 1.0 mol/L hydrochloric acid (HCl, Nihon Shiyaku Industries, Japan) solution to remove residual KOH in the MWCNT powder after the activation process. After that, the ACNTs were collected on the bottom of the centrifuge tube using a centrifuge, where the centrifuge speed was set to 3000 rpm in order to wash and separate the ACNTs. Next, the centrifuged ACNT powder was placed in a crucible, and then placed in a vacuum oven to be dried at a temperature of 200°C for 24 h to complete the activation process for the ACNTs.
Preparation of cathodes
The substrates used in this study were pieces of graphite felt (CeTech Co., Ltd., Taiwan) with dimensions of 50 × 15 × 7 mm (L × W × T). Pre-treatment was conducted before the experiment to remove impurities in the graphite felt. The graphite felt was immersed in a mixture containing 450 mL of deionized water and 50 mL of hydrogen peroxide (35% H2O2) (Nihon Shiyaku Industries, Japan). The mixture was then heated to 90°C in a water bath for 3 h. Next, the graphite felt was removed and placed in 500 mL of distilled water and shake-washed for 30 min using an ultrasonic cleaner (DC-200H, Delta Ultrasonic Co., Ltd., South Korea). After that, the graphite felt was placed in a vacuum oven and baked at a temperature of 200°C for 4 h to evaporate any residual H2O2 and water in the graphite felt, completing the pre-treatment of the graphite felt.
In this study, the cathodes applied in the EF system were prepared using the dip coating method. First, 0.025g of MWCNT, GR (Yancheng Zengcai Technology Co., Ltd., China), or ACNTs were weighed using a precision micro-balance (GR-200, A&D, Japan), and added to the solvent N-methyl-2-pyrrolidone (NMP) (C5H9NO, Nihon Shiyaku Industries, Ltd., Japan). Next, the mixture was stirred for 30 min using an electromagnetic heating stirrer (MS-H-Pro, Dragon Lab, China), and ultrasonically heated and shaken for 15 min to prepare a slurry. Following this, the graphite felt was dipped in the slurry and repeated five times, and the mixture was placed in the vacuum oven and baked at a temperature of 200°C for 2 h, so that the powder firmly adhered to the graphite felt. The mixture was removed after the furnace had cooled to room temperature to complete the preparation of the composite cathodes.
Characterization of cathode
Before conducting the experiment, the composite cathodes were plated for 1 min to improve the conductivity of the samples. Images were captured by secondary electron imaging (SEI) using a scanning electron microscope (SEM, JSM-IT100, JEOL Ltd., Japan) to observe the surface topography of the composite cathodes.
The MWCNTs, GR, and ACNT powder were scanned with a Fourier-transform infrared (FT-IR) spectrometer (NICOLET iS10, Thermo Fisher, USA), using the OMSIC Specta software, at wavenumbers between 650 and 4000 cm-1. The functional groups of these materials were thus compared using infrared spectroscopy.
Analysis of electrochemical properties
In this study, the response current was measured using linear sweep voltammetry (LSV) to evaluate the current response of cathodes modified by different materials. Measurements for this experiment were performed using an electrochemical workstation (ECW-5600, Jiehan, Taiwan). The field was applied using a tripolar electrode system, in which the cathode (Ca) was the substrate and modified electrode, the counter electrode (CE) was a platinum (Pt) plate (Anatech Co., Ltd., Taiwan), and the reference electrode (RE) was a silver/silver chloride (SCSE) electrode (Anatech Co., Ltd., Taiwan). An aqueous solution of 0.1 M potassium nitrate (KNO3) (Acros Organics, Belgium) was formulated and used as an electrolyte. Before measurement, pure-oxygen aeration was performed for 10 min at a flow rate of 150 sccm equipped with gas flow meter (5850E, Brooks, USA). The experiment was performed from 0 to −1 V using the electrochemical workstation at a scan rate of 10 mV/s. To evaluate the ability to generate the H2O2 for each cathode further, 5 wt% titanium (IV) sulfate (Ti(SO4)2) was mixed with electrolyte, and electrogeneration process was performed at potential of −0.6 V versus SCSE for 30 min. The visible-light spectrophotometer was used to measure the absorbance for the specimen mixed with Ti(SO4)2 and H2O2 at 410 nm, and the concentration of H2O2 was transferred from absorbance by calibration curve. 20
In addition, to calculate the active surface area of the cathodes, the cyclic voltammetry (CV) was performed at different potential. A tripolar electrode system was adopted for this test. The experiment was performed with an aqueous solution containing 0.01 M potassium ferricyanide (K3[Fe(CN)6]) (Nihon Shiyaku Industries, Japan) and 1 M potassium chloride (KCl) (Nihon Shiyaku Industries, Japan). Potential was scanned from 1 to −1 V using EC Ware software on the electrochemical workstation at a scan rate of 10 mV/s.
RhB dye degradation test in the electro-fenton system
In this study, Rhodamine B (C28H31ClN2O3) (RhB, Sigma-Aldrich Corp, USA) was used as the detection reagent. During the test, a constant potential was supplied by the electrochemical workstation (ECW-5600, Jiehan, Taiwan). Light absorbance was measured using a visible light spectrophotometer (SH-U880, Shishin Technology Co., Ltd., Taiwan) at the RhB characteristic wavelength of 550 nm. The formula for converting light absorbance to decolorization rate is shown in equation (2-1), where A0 denotes the initial light absorbance of the solution and A denotes the light absorbance of the solution after reaction.
The TOC of the samples is measured through the wet oxidation method using a TOC analyzer (Phoenix 8000, Teledyne Tekmar, USA) together with the TOC Talk/Phoenix 8000 software, in order to compare changes in the TOC of different composite cathodes in the solution before and after reaction. The formula for converting TOC is shown in equation (2-2), in which A0 denotes the initial TOC of the solution and c denotes the TOC of the solution after reaction.
The field of the EF system in this study was applied with a tripolar electrode system, where the Ca was the substrate and modified electrode, the CE was a platinum (Pt) plate, and the RE was a silver/silver chloride (SCSE) electrode. In addition, a previous study indicated that an electric potential of −0.6 V versus SCSE was the optimal cathode potential for carbonaceous materials in the EF process 11 ; therefore, the tests in this study were conducted using an electric potential of −0.6 V versus SCSE. A 0.1 M potassium nitrate (KNO3) aqueous solution was added into the reaction tank and counter-electrode tank as the electrolyte required for the reaction system. In addition, 20 ppm of ferrous sulfate (FeSO4‧7H2O) (Nihon Shiyaku Industries, Japan) was added to provide the ferrous catalyst required for the reaction, and 5 ppm of RhB dye was used as the degradation reagent in the system. During electrogeneration process, oxygen was supplied to the reaction solution using a pump. In the process, the reaction solution was stirred using an electromagnetic heating stirrer at 600 rpm. Figure 1 shows a diagram of the EF system setup.

Electro-Fenton system setup.
Results and discussion
Characterization of cathode
Figure 2(a) shows an SEM image of the micromorphology of an unmodified graphite felt substrate at a magnification of 5000 times, which shows that impurities in the graphite felt fiber were removed after pre-treating the graphite felt. It can also be observed that its surface is smooth and flat. In contrast, Figure 2(b) and (c) show SEM images of a MWCNT/C cathode and a GR/C cathode at a magnification of 10,000 times, respectively. It can be observed from these images that MWCNTs and graphene flakes were successfully adhered to the surface of the graphite felt after applying the dip coating method. It is expected that this will improve the conductivity and specific surface area of the graphic felt cathode. Figure 2(d) shows an SEM image of the microtexture of an ACNT/C cathode at a magnification of 30,000 times. It can be seen from this image that many grooves have formed on the surface of the MWCNTs after activation, causing the surface of the MWCNTs to become rough. It can also be observed that the MWCNTs have been transformed from a tubular form into a graphene-like flake form. Zhang and Chen 21 noted that a graphene layer with defects or distortion can be obtained from the surface of MWCNTs due to the chemical reaction between KOH and carbon. This indicates that the powder mixture of MWCNTs and KOH can effectively activate the surface of the MWCNT walls when heated at a temperature of 800°C for 1 h. In summary, the MWCNTs exhibit a graphene-like flake morphology after activation, which facilitates the increase in specific surface area.

Surface morphology of graphite felt composite cathodes prepared by different treatments: (a) unmodified graphite felt cathode, (b) MWCNT/C cathode, (c) GR/C cathode, and (d) ACNT/C cathode.
The functional groups of a material affect its chemical properties. It can be seen from Figure 3 that extensional vibrations attributed to the O–H bond were observed at wavenumbers of 3475and 2912 cm−1. 22 Extensional vibrations attributed to the C=O bond at a wavenumber of 1680 cm−1, 23 extensional vibrations attributed to the C–O bond at a wavenumber of 1418 cm−1, 24 and internal vibrations attributed to the C–H bond at a wavenumber of 1310 cm−1 were also observed. 25 The intensity of the peak related to the oxygen-containing functional groups of the ACNTs after activation is higher than that of unactivated MWCNTs, indicating that the composition of the MWCNTs was successfully improved by chemical activation. The study conducted by Dai et al. 26 showed that the activation of CNTs by KOH can enhance the intensities of peaks related to functional groups, which is consistent with the findings of this study, thus verifying that oxygen-containing functional groups were successfully introduced into MWCNT through activation. Luo et al. 27 noted that oxygen-containing functional groups contribute to an increase in specific surface area, porosity, and H+ adsorption capacity. In summary, mixing MWCNTs and KOH at a weight ratio of 1:4 followed by high-temperature activation at 800°C can promote the formation of high quantities of oxygen-containing functional groups on the surfaces of MWCNTs.

Infrared spectrum of unactivated CNT, graphene, and ACNT powder.
Analysis of electrochemical properties
Figure 4(a) shows the LSV curve for each cathode. At the optimum cathode potential of carbonaceous materials of −0.6 V versus SCSE, the response currents of each cathode were found to be as follows: −4.60 mA (graphite felt substrate), −7.07 mA (MWCNT/C cathode), −11.00 mA (GR/C cathode), and −8.46 mA (ACNT/C cathode). The magnitude of the response current is an important indicator for the rate of formation of hydrogen peroxide (H2O2) and hydroxyl radicals (•OH) in EF reactions, it implied that the response current of the graphite felt cathode improved significantly after surface modification, which is consistent with the results obtained in previous experiments. 12 To evaluate the ability to generate H2O2 for each cathode, the colorimetry method was performed to measure concentration of H2O2 after electrogeneration process by cathode for 30 min, the result was showed in Figure 4(b), and the concentration of H2O2 ranked differently graphite felt modified by variety type of material: GR/C (13.6 ± 0.68 mg/L) > ACNT/C (11.56 ± 0.58 mg/L) > MWCNT/C (8.84 ± 0.44 mg/L) > graphite felt substrate (5.12 ± 0.31 mg/L). After modification, the production of H2O2 for graphite felt substrate can be significantly increased, Among then, GR/C cathode generated the highest concentration of H2O2, The magnitude of the response current is positively correlated with the degradation efficiency of the system; that is, a larger response current has a strengthening effect on the EF system, 28 which proved the result from LSV experiment; ACNT/C cathode enhanced 31% more H2O2 than CNT/C cathode. In addition to the reason that specific surface area is increased, the experiment in Section 3.1 showed that strong C=O peaks could be detected from the surface of the CNTs after activation, which is conducive to the activity of oxygen reduction reactions (ORR). 11 However, the ACNT/C cathode still showed a lower response current than the GR/C cathode, which can be attributed to the fact that although the activation of MWCNTs can increase their specific surface area and intensity of oxygen functional group, it also destroys the structure of the CNTs, thus the conductivity of CNTs cannot be increased simultaneously. 13 Therefore, the GR/C cathode prepared using graphene with high conductivity still showed the highest response current at potential of −0.6 V versus SCSE. In summary, the ACNT/C cathode prepared using ACNTs showed improved specific surface area and electrochemical performance.

(a) Linear sweep voltammetry curve unmodified graphite felt cathode, MWCNT/C cathode, GR/C cathode, and ACNT/C cathode and (b) the concentration of H2O2 for each cathode by electrogeneration process after 30 min.
The efficiency of the electrochemical reactions affects the degradation ability of the EF system, which can be estimated from the active surface area of the cathode. In this study, scanning was performed at different potential using CV, in order to compare the active surface area of different composite cathode. With regard to the active surface area, the peak current of the system was obtained from the potential-current curves measured in this study, and then calculated using the Randles–Sevcik equation, as shown in equation (3-1). 29 Table 1 shows the relevant parameters for this equation.
Parameters related to the Randles-Sevcik equation.

Figure 5 shows the CV curves for each cathode. Based on calculations using the Randles–Sevcik equation, the active surface areas of each cathode were determined as follows: 43.69 cm2 (graphite felt substrate), 60.58 cm2 (MWCNT/C cathode), 83.09 cm2 (GR/C cathode), and 69.57 cm2 (ACNT/C cathode). Sirés et al. 30 reported that a larger active surface area can improve the reduction ability of ferrous ions in EF reactions, where the larger the active surface area of the cathode, the better the reaction efficiency of the EF system. The active surface area of the ACNT/C cathode prepared by the activation process was increased compared to the MWCNT/C cathode. He et al. 14 conducted an experiment using a platinum catalyst supported on MWCNTs (MWCNT/Pt), and found that the active surface area of MWCNTs/Pt activated by KOH at temperatures of 700°C, 800°C, and 900°C can be increased compared to unactivated-MWCNTs/Pt. This is because the aqueous solution can come closer to the surface of the cathode after the surfaces of the MWCNTs are modified by oxygen-containing groups, which facilitates the electrochemical reaction.

Cycle voltammetry curves of the unmodified graphite felt cathode, MWCNT/C cathode, GR/C cathode, and ACNT/C cathode.
In summary, the GR/C cathode was found to have the highest active surface area, and the results obtained by LSV exhibit the same trend as well. Compared to the MWCNT/C cathode, the active surface area of the ACNT/C cathode were increased by 14.8%, respectively, which facilitated an increase in the rate of H2O2 production and the reduction ability of iron ions, thereby increasing the performance of the system.
Analysis of degradation efficiency in the electro-fenton process
Figure 6 shows the results of the 60-min decolorization test conducted using the EF process with the MWCNT/C cathode, the GR/C cathode, and the ACNT/C cathode, where the decolorization rates of these cathodes were 77.28%, 88.55%, and 85.01%, respectively. The decolorization rates of these cathodes increased significantly compared to the decolorization rate of the unmodified graphite felt substrate, which was 50.28%. To ensure the stability of the modified cathode, The EF treatment was repeated three times using the same cathode and RhB decolorization yield after 1 h, the result are shown in Figure 7(a) to (d), respectively. After three times repeats, the decolorization efficiency of RhB in EF with GR/C cathode and ACNT/C cathode remained above 80% after 1 h, and CNT/C cathode remained above 70%, which implies that after modification, the performance and stability of graphite felt can be improved. In particular, the decolorization rate of the ACNT/C cathode increased by about 8% compared to that of the MWCNT/C cathode, and even approached to those of the GR/C cathode. The decolorization rates of each cathode at the 180th min were as follows: 75.42% (graphite felt substrate), 97.37% (MWCNT/C cathode), 97.82% (GR/C cathode), and 97.99% (ACNT/C cathode). With the prolong, of EF reaction time, the concentration of residual dye decreased gradually, which resulted in a decrease in the frequency of collisions between hydroxyl radicals and RhB molecules, causing the dye removal rate to decrease with increasing electrolysis time. 31

Decolorization rate for RhB dyes treated by the EF system with an unmodified graphite felt cathode, MWCNT/C cathode, GR/C cathode, and ACNT/C cathode.
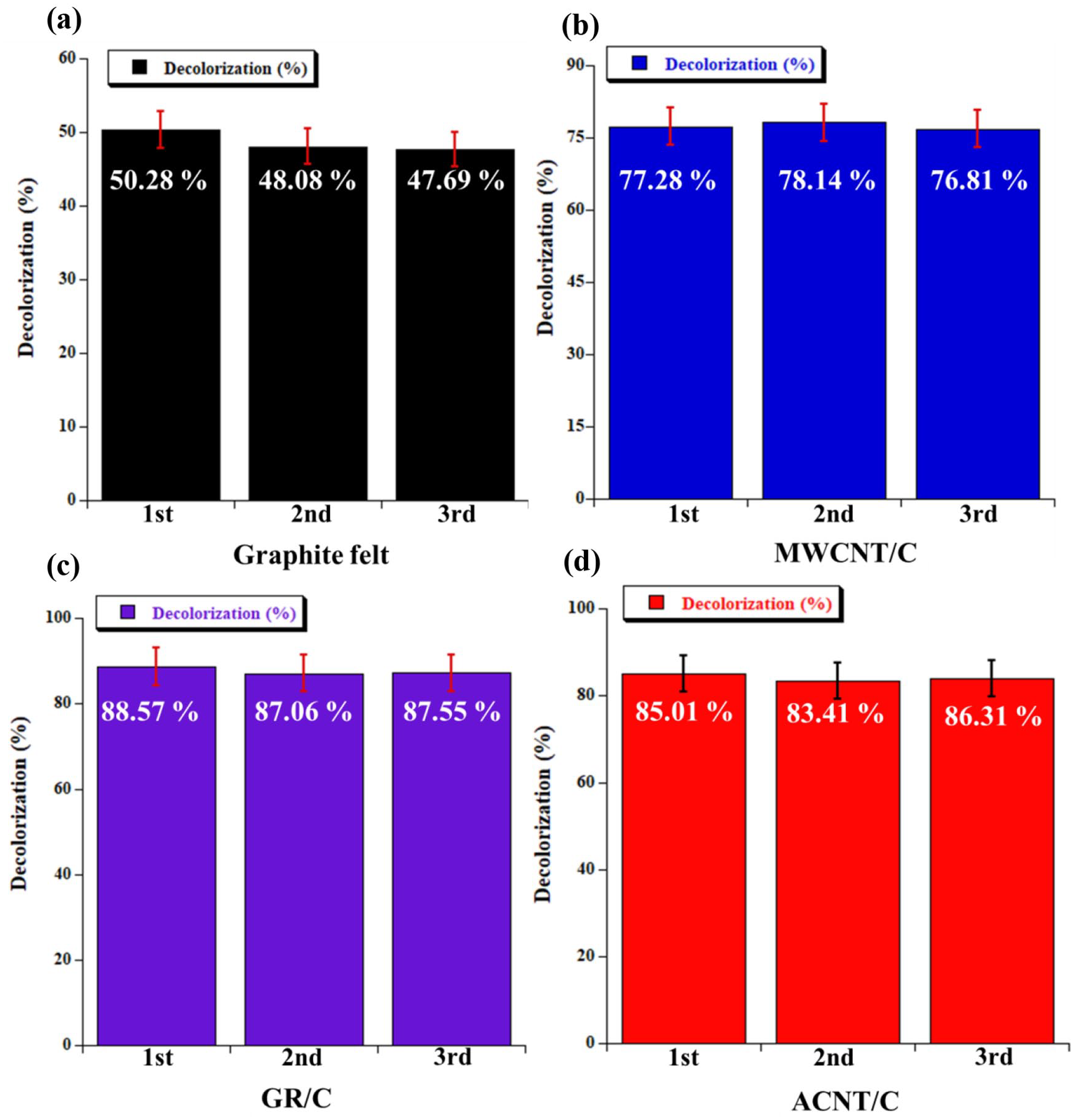
Decolorization rate for RhB dyes treated by the EF system for 1 h after 3 times experiment: (a) graphite felt cathode, (b) MWCNT/C cathode, (c) GR/C cathode, and (d) ACNT/C cathode.
Hydroxyl radicals (•OH) generated from EF reactions can promote the degradation of RhB organic carbon dye by decomposing its structure into inorganic substances, such as CO2 and H2O, as well as reduce the TOC of the system. Therefore, this study employed the wet oxidation method to analyze the TOC of the system. Figure 8 shows the TOC removal rate curves of different composite cathodes, the GR/C cathode exhibited the highest TOC removal rate at 26.21%, followed by the ACNT/C cathode (24.68%), MWCNT/C cathode (22.91%), and lastly the graphite felt substrate (12.89%). The results showed that the TOC removal rate of the system is positively correlated with decolorization rate. Despite having significant decolorization effects, it is not easy to completely degrade organic carbon within a short period of time.

TOC removal rate for RhB dyes treated by the EF system with unmodified graphite felt cathode, MWCNT/C cathode, GR/C cathode, and ACNT/C cathode.
The result showed that GR/C cathode had the best effect on organic carbon decomposition in the system, while the TOC removal rate of the ACNT/C cathode increased compared to that of the MWCNT/C cathode. This phenomenon can be attributed to the benefits brought about by the increase in oxygen-containing functional groups, which promote the ORR occurred on the surface of cathode. Zhao et al. 32 indicated that a high TOC removal rate is related to the capacity for H2O2 formation, which facilitates the production of more hydroxyl radicals (•OH). Li et al. 33 noted that increasing the specific surface area of the cathode can improve the efficiency of the EF reaction. As shown by the LSV data in Section 3.2, it can confirm that the response current measured by LSV experiment is positively correlated with the decolorization and TOC removal rate. Although it was shown in Sections 3.2 that the GR/C cathode exhibited the highest response current and active surface area, it can be seen in this section that the RhB decolorization rate of the ACNT/C cathode approached to that of the GR/C cathode. This can be attributed to the fact that the surface of the ACNT/C cathode after activation became rich in oxygen-containing functional groups compared to the GR/C and MWCNT/C cathodes; thus, not only can this improve ORR in the cathode, it also increases the organic dye adsorption capacity of the ACNT/C cathode. 34
In summary, the ACNT/C cathode after activation improved compared to that of the MWCNT/C cathode, which shows that activation facilitates the generation of more hydroxyl radicals (•OH), thus improving the efficiency of pollutant decomposition. The decolorization rate of the ACNT/C cathode are similar to the results obtained for the GR/C cathode, which demonstrates that ACNTs have the potential to replace graphene in the EF system.
Conclusion
In this study, MWCNTs were successfully activated into a GR-like flake structure by KOH, which facilitated the increase in specific surface area; and the intensity of oxygen-containing functional groups of ACNTs increased compared to that of unactivated MWCNTs, confirming that oxygen-containing functional groups were introduced onto the surface of MWCNTs through activation. The concentration of H2O2 for the ACNT/C cathode increased by 31% compared to the MWCNT/C cathode. In addition, the active surface area of the ACNT/C cathode were increased by 14.8%. With regard to the analysis of EF decolorization performance, it was shown that the decolorization rates achieved with each cathode at the 60th min were as follows: 50.28% (graphite felt substrate), 77.28% (MWCNT/C cathode), 88.55% (GR/C cathode), and 85.01% (ACNT/C cathode). Both the ACNT/C and GR/C cathodes exhibited similar decolorization effects, indicating that the activation of MWCNTs by KOH could improve their decolorization rate. After MWCNTs are activated by KOH at a temperature of 800°C for 1 h, the electrochemical properties listed above can be effectively improved, thus allowing them to achieve a similar degradation ability as the GR/C cathode in the EF system, and showing the potential of ACNTs to replace graphene in terms of chemical properties. This study reports the successful preparation of an ACNT/graphite felt (ACNT/C) cathode, which can potentially be used for developing cathode materials in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We sincerely appreciate the financial supported by the Ministry of Science and Technology, R.O.C. through the grant MOST 108-2221-E-197-014.