Abstract
Brain injury is a common cause for physical and emotional effects to the large number of populations. Moreover, glioblastoma is the tumor in brain with no possible treatment leading to death. The blood–brain barrier’s makes the treatment more difficult by preventing the drugs to reach central nervous system. Paclitaxel (PTX) encapsulated Poly (lactic-co-glycolic acid) (PLGA) nanoparticles (NPs), PTX-PLGA-NPs were developed using emulsification method. The PTX-PLGA-NPs were characterized using Malvern Zetasizer and Scanning Electron Microscopy and were evaluated for their cytotoxicity in U87MG cells. PTX-PLGA-NPs were prepared using single emulsion method having size of 154 ± 22.19 nm with zeta potential of –23.7 mV. The PTX-PLGA-NPs were spherical in shape and have dose dependent cytotoxicity on U87MG cells. The PTX was released from the particles with initial burst release followed by sustained release pattern. The biodistribution was studied in mice with glioblastoma model using 125I radiolabeled PTX-PLGA-NPs and anti-glioblastoma was studied with PTX-PLGA-NPs. The biodistribution studies revealed PTX-PLGA-NPs after intranasal administration resulted in higher in vivo uptake with high anti-glioblastoma efficacy. The results suggest that PTX-PLGA-NPs administered through intranasal route have potential in the treatment of glioblastoma.
Introduction
A large number of human populations suffer from brain injuries which effect physical as well as emotional sentiments of patients and their relatives. 1 In order to cure these injuries, the options of diagnosis and therapy are limited due to complex brain structure and blood brain barrier (BBB). Hence this remains a challenging situation for physicians and researchers especially after traumatic or ischemic brain injuries. Glioblastoma is the tumor causing severe damage to the brain resulting in almost 1 year survival of the patient. 2 The characteristics of glioblastoma includes poor prognosis and present treatment including has radiotherapy, chemotherapy and surgery does not improve the situation. 3 The residual cells after the glioblastoma treatment, including surgery, invades parenchyma making it difficult to completely eradicate glioblastoma and results in recurrence. 4 The BBB is a preventive barrier consists of proteins junction at endothelial cells of brain, which mainly involved in prevention of blood component transport containing drugs into central nervous system (CNS). 5 Neurovascular unit (NVU) consist of glial cells, neurons and endothelial cells. 6 BBB is a component of this extensive network. Any disturbance of NVU due to primary or secondary injury may disturb the BBB. 7 Further on the disturbance of BBB accelerate there is a development of neurodegeneration via reduction of amyloid-β clearance and also neurotoxic molecules leakage. 8
Paclitaxel (PTX) is the drug which is used as one of the most common chemotherapeutic agents to treat wide range of cancer including lung and breast. However, P-glycoprotein (P-gp) mediated efflux reduces the PTX absorption in the tumor cells.9,10 Nanoparticles (NP) are ideal vehicle for administration of drugs into the brain by crossing biological barriers. 11 The NPs encapsulated drug is one of the effect approaches to enhance bioavailability, site-specific delivery and minimize deleterious effects of therapeutic drugs. 12 Here we utilize Poly (lactic-coglycolic acid) (PLGA) copolymer for NPs synthesis. PLGA, being a FDA approved polymer with its wide use in encapsulating and delivering of hydrophobic drugs including PTX offers the high internalization of drug in the tumor cells owing to the phagocytosis or endocytosis mechanism. 13 They have excellent biodegradability and biocompatibility. 14 The poor permeability of NPs in the BBB hinders the delivery of NPs in central nervous system and is challenging situation. The previous studies focused on direct drug delivery into brain tissue or cerebrospinal fluid reservoirs. 15
In last decades intranasal (IN) drug delivery has gained major interest in research by scientists and physicians in clinics. 16 The nasal path epitomizes a non-invasive administration of effective pharmaceutical elements for CNS systemic action. 17 Due to the fact that epithelium of nasal cavity is a big barrier, the tightness of junction mucosa of nasal route is low mainly due to leaky nature of epithelial tissue. Although the mucosa is extensively vascularized but leaky epithelium and lamina propri provide an excellent surface area for drug administration. 18
The absorption of molecules directly through olfactory pathways via nasal cavity enhances the performance of pharmacokinetic due to direct entrance in the brain. 19 The active pharmaceutical elements absorbed in the large surface area of nasal mucosa, which provide local and systemic effect and can be administrated to target the brain directly. The drug administration by nasal pathway is advantageous over intravenous and oral administration, because the nasal route is self-administration, non-invasiveness, shortest duration to onset of effect and enhanced bioavailability due to absence of hepatic metabolism.19,20 Moreover, crossing the BBB may potentially enhanced central nervous system (CNS) availability of the therapeutic drug.
Keeping in mind the above-mentioned studies, we design a protocol and develop PTX-loaded PLGA nanoparticles for the IN administration. The design of delivery system and its mechanism is presented as Figure 1.

The schematic representation of particle administered via intranasal route for brain injury.
Materials and methods
Materials
PLGA having molecular weight of ~ 30,000 was purchased from Shandong Institute of Medical Instrument, Shandong, China). PTX was obtained from Hefei Bo Mei Biotechnology Co., Ltd. China. Penicillin-streptomycin solution, Roswell Park Memorials Institute (RPMI)-1640 medium, Trypsin-EDTA solution and Fetal bovine serum (FBS) was purchased from Sigma Aldrich, USA. [3–(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide] cell viability/cytotoxicity assay kit (MTT) was obtained from Sangon Biotech (Shanghai, China). Triple distilled water from Milli-Q Plus System (Millipore Corporation, Bedford, USA) was used.
Methods
Synthesis of PLGA- PTX nanoparticles
In order to encapsulate hydrophobic compound PTX in PLGA, single emulsion –solvent evaporation method was used to prepare PTX laden PLGA-NPs. 21 Briefly, in 2 mL of ethyl acetate, 200 mg of PLGA was dissolved along with 20 mg PTX and kept for 30 min under stirring. Aqueous solution of 5% w/v of PVA (4 mL) act as surfactant and NPs mixture was added drop wise at intermittent vortex. Following the complete mixing of PLGA- PTX, it is sonicated for another 1 min to form clear emulsion. The mixture is added in 0.3% w/v aqueous solution of PVA (100 mL) followed by 3 h magnetic stirring. The PVA stabilized the emulsion and formed uniformed size particles. The pure NPs obtained after centrifugation at speed of 6000 rpm for 20 min and washed with ultrapure water three times. The collected pure particles are freeze dried and placed in freezer for further studies. The synthesis parameters were optimized by repeating the steps involved in fabrication process. The dispersion of NPs in water was analyzed at an equivalent concentration of PTX (10mg/mL).
Physiochemical properties of the nanoparticles and PTX Content
PTX laden PLGA-NPs were dispersed in Milli Q water for analyzing their physiochemical properties. The PTX laden PLGA-NPs size of and morphology were characterized using Malvern zetasizer and electron microscopy. The surface charge of PTX laden PLGA-NPs was also analyzed using the Zetasizer of Malvern Instruments. The surface morphology was studied using Scanning Electron Microscopy (SEM).
The pure freeze dried 5 mg NPs was thoroughly dissolved in DCM (1 mL). To extract PTX from NPs, 3 mL of water is added. The organic phase is evaporated under continuous stirring at 1500 rpm for 1 h at room temperature. To get rid of polymer residue, aqueous solution is filtered. The spectroscopic analysis was performed at 273 nm to calculate the content of PTX in NPs. The drug loading is calculated as content of PTX that is, expressed in milligram/100 mg of NPs and encapsulation efficiency was evaluated as drug encapsulated percentage to the initially added drug during preparation.
Entrapment efficiency (%) = weight of PTX in PTX-PLGA-NPs /weight of PTX initially taken × 100
Loading efficiency (%) = weight of PTX in PTX-PLGA/ weight of PTX × 100
In Vitro drug release analysis
The in vitro studies of PTX release from NPs were performed by using membrane diffusion method. 60 mg of purified drug loaded NPs was suspended in 1mL of water and carefully placed in 60 kDa dialysis bag. These designed dialysis bag loaded with formulation is permeable to PTX but prevent NPs release and suspended in 25 mL (pH 7.4). The mixture is gently shaken with help of magnetic stirrer at speed of 100 rpm. After set time interval 2.5 mL of sample is withdrawn from receiving phase and replaced by fresh medium in order to avoid sink condition. The collected sample was carefully analyzed by spectroscopic method at 273 nm, in order to find the drug content in the sample. The release experiments were performed trice and mean value is calculated.
Cytotoxicity studies
The toxicity and permeability of fabricated PTX-PLGA-NPs were studied on U87MG, human glioblastoma cells. Eagle’s minimal essential medium with 10% foetal bovine serum was used to grow cells. The effect and toxicity of PTX-PLGA-NPs along with free PTX and blank NPs was studied by 3-(4,5-dimethyltiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay method. The cells were treated at an equivalent concentration of PTX (0.01 µM, 0.1 µM, 0.5 µM, and 1 µM) for 24 h followed by addition of MTT solution (0.5 mg/mL). The cells were incubated for additional 4 h and the insoluble formazan crystals were dissolved by adding 100 µL of DMSO. The optical density of the plate was measured at 570 nm using ELISA plate reader.
In vitro permeability of PTX
A vertical Franz-diffusion cell system was used to study in vitro permeability. Donor phase consist of 5 mg/mL of PTX, that was placed on synthetic membrane. The membrane was impregnated in isopropyl myristate, 1.8 cm2 of membrane area was involved in effective diffusion. The PBS is applied as acceptor phase. The stirring was adjusted at 100 RPM speed. At set time period 0.8 mL of sample is collected from acceptor phase and replaced by fresh medium. Spectroscopic analysis was used to calculate the concentration of PTX.
Cell uptake studies
U87MG cells were employed for the cell uptake studies for PTX-PLGA-NPs. Firstly, the 1 × 105 cells were seeded in a glass bottom dish and incubated in an incubator for 24 h at 37°C. Subsequently, the cells were treated with Nile red tagged PTX-PLGA-NPs and free Nile red for 6 h. Afterwards, the cells were washed with PBS and the nucleus was stained with DAPI (0.5 μg/mL). The cells were imaged by a confocal microscope after several washings with PBS.
In-vivo studies
We performed the animal studies after the approval of local ethical committee of Chongqing Emergency Medical Center under the protocol number: CEMC/185-0/2019. The animals were treated according to the protocol provided by the local ethical committee.
Bio-distribution test of PTX-PLGA-NPs
The biodistribution studies of PTX-PLGA-NPs was evaluated by radiolabelling it with 125I by the Iodogen. The unloaded residues were removed using size exclusion chromatography.
The male mice (Balb/c) were numbed with 5% chloral hydrate and placed on a stereotaxic apparatus. U87MG cells (1 × 106 cells/5 ml) were used to established glioblastoma model. The cells were injected into right brain of the mice and monitored under standard condition for 7 days. The treatment was started as the size of brain tumor reaches 0.4–0.6 cm in diameter.
The 125I labeled PTX-PLGA-NPs was administered via IN route and also injected via tail vein into tumor bearing mice at concentration of ∼35 μCi per mouse. The mice were sacrificed after 2 h and 24 h post injection. The injected dose per gram was counted in organs and blood with the help of gamma counter. These experiments were repeated thrice and final data was presented as mean ± standard deviation (SD).
Anti-glioblastoma study
The tumor model was stabled by using above protocol. The studies were started when the tumor volume reached approximately 70–100 mm3. The mice were randomly distributed in 3 groups (n = 6) and administered with 10 mg/kg body weight dose of PTX in PTX-PLGA-NPs via IN route and by intravenous administration, and PBS via tail vein injection. The dose (7.5 mg/kg body weight) was injected of day 1 and day 7. The tumor volume was monitored for 12 days and the mice were sacrificed at the end of the study.
Statistical analysis
The significance difference in the results were measures using Statistical analysis software Prism 5.0. Differences among the groups were analyzed using one-way ANOVA and the p < 0.05 was set as level of significance.
Result and discussion
Synthesis and characterization of PLGA- PTX nanoparticles
We have successfully used single emulsion method for the development of PTX-PLGA-NPs. 22 We have used PLGA to prepare the NPs as its offers various advantages over other polymers. PLGA is biocompatible and has been approved by US-FDA also it is used clinically. The process was optimized to obtain the homogenous NPs with maximum encapsulation efficiency. The size of PTX-PLGA-NPs was found to be 154 ± 22.19 nm as measured using Malvern Zetasizer and SEM. The size and surface morphology of the particles were further confirmed by using SEM, which revealed the smooth surface and homogeneity of the particles. Figure 2 shows the size measured by DLS and surface morphology of the particles observed through SEM. The surface charge on the particles was found to be –23.7 ± 2.71 mV as measured by Malvern zetasizer. The encapsulation efficiency of PTX laden PLGA-NPs were found to be 79.41%, however the loading efficiency was found to be 6.48 %. The results of physicochemical characterization have been illustrated in Table 1.

The DLS size of PTX-PLGA-NPs and SEM image of PTX-PLGA-NPs.
Physiochemical characterization of PTX-PLGA-NPs.

In vitro release of PTX from NPs
The in-vitro release study was carried out for 24 h using a dialysis bag method. The results revealed that the free PTX could be released from the dialysis membrane at much higher rate than compare to the PTX loaded in PTX-PLGA-NPs. However, initial burst release was observed from the PTX-PLGA-NPs which might be attributed to the PTX present at the surface of NPs. However, the PTX firmly associated with the NPs showed a sustain release pattern. The ability of PLGA to sustain the release of the drug improve the therapeutic effect of anticancer agents and minimize the side effect. Also, the thickness of PLGA could determine the release kinetics.
The diffusion of free PTX revealed the contrary results and was released within 8 h of the experiment, whereas only 53% of PTX was release from PTX-PLGA-NPs at the particular time. However, complete release was obtained after 72 h. The results have been illustrated in Figure 3.

The percentage cumulative release of PTX-PLGA-NPs at pH 7.4 and 37°C.
In vitro cytotoxic
The cytotoxic effect of the developed PTX-PLGA-NPs was evaluated in U87MG cells. 23 The cells were treated with free PTX and PTX-PLGA-NPs at equivalent concentration and the cytotoxicity was measured. The results of cytotoxicity were compared with free PTX and untreated cell designated as control. The results revealed that the free PTX and PTX-PLGA-NPs cause dose dependent cytotoxicity to the cells as compared to the control. However, PTX-PLGA-NPs causes significantly high cytotoxicity to the cells in comparison to free PTX. PTX triggers apoptosis and it acts by stabilizing the microtubules at the time of disassembling of chromosomes and inhibits the progression of mitosis. The PLGA-NPs alone does not cause any cytotoxicity and were nontoxic to the cells, revealing their biocompatibility as illustrated in Figure 4.
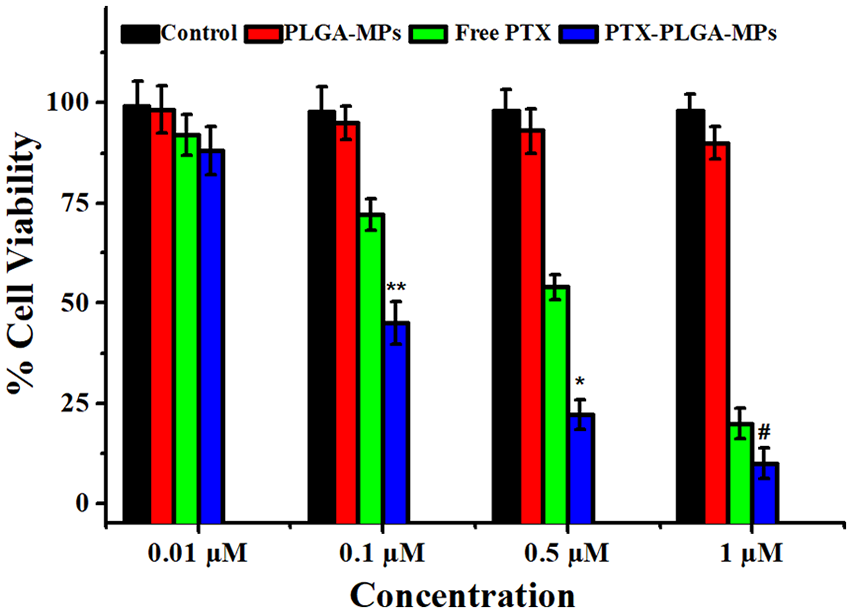
The percentage cell viability of PTX-PLGA-NPs after 24 h of incubation with the cells.
In vitro permeability assay
The Franz cell diffusion system was used to analyze the PTX diffused from PTX-PLGA-NPs and the diffusion was compared with free PTX. 24 The diffusion study was carried out for 12 h. As shown in Figure 5 it was clearly observed that the diffusion of PTX-PLGA-NPs through the synthetic membrane was higher at all-time points. On the contrary, the diffusion of free PTX was slower in comparison to PTX-PLGA-NPs.

The percentage amount of PTX diffused from PTX-PLGA-NPs and free PTX.
Cellular uptake
The cellular uptake studies conducted on U87MG cells revealed that the Nile red tagged PTX-PLGA-NPs could be engulfed by the cells significantly more than plain Nile red. 25 The results obtained after 4 h of incubation of Nile red tagged PTX-PLGA-NPs and plain Nile red has been represented in Figure 6. The higher uptake of PTX-PLGA-NPs was clearly observed as high fluorescence in the cells around the DAPI stained nucleus. The results might be attributed to the higher engulfment of PTX-PLGA-NPs in comparison to plain Nile red by means of endocytosis.

U87MG cell uptake of 100 μg/mL Nile red-loaded PLGA-NPs and free Nile red.
Bio-distribution assay
The tissue distribution analysis for U87MG glioblastoma tumor mice model has been summarized in Figure 7. The results illustrated the comparison of PTX-PLGA-NPs injected through IV and the PTX-PLGA-NPs administered via IN route. The NPs administered through IV resulted in a high concentration in blood at the initial 2 h, which decreases significantly at 24 with the distribution in organs with limited amount present in tumor. The PTX has a short half-life when administered systemically. Therefore, high dose of PTX is required during systemic treatment, which results in high accumulation of drug in non-targeted organs and severe toxicity. However, the NPs administered through IN route allowed the higher uptake of NPs in tumor of the brain. Also a significant decrease in ID%/g value from 1.50 ± 0.078 to 0.45 ± 0.078 were observed which indicated in vivo uptake of U87MG. 26

Bio-distribution of PTX-PLGA-NPs radiolabeled with 125I in mice with tumors: (a) PTX-PLGA-NPs administered through IN and (b) PTX-PLGA-NPs administered through IV.
The ID%/g of blood sample of both PTX-PLGA-NPs after IV and IN administration indicated a decrease in value from 2 h to 24 h. However, the radioactivity of tumor tissue didn’t indicate any significant decrease. The ratio of ID%/g value between blood and tumor increased for 24 h while the ratio of intravenously injected PTX-PLGA-NPs decreased then PTX-PLGA-NPs administered IN. The finding proved a higher accumulation PTX-PLGA-NPs into the tumor, which might be due to enhanced permeability of PTX via IN administration.
Antitumor effect
The studies confirmed that IN delivery of PTX-PLGA-NPs revealed excellent glioblastoma growth inhibition as compared to control group and IV administered PTX-PLGA-NPs. 27 As shown in Figure 8, PTX-PLGA-NPs administrated IN indicated strongest anti-glioblastoma efficacy as compared to other groups, this might be due to neovasculature and U87MG glioblastoma cells specific delivery of PTX. The size of tumor after the treatment with PTX-PLGA-NPs (IN administered) revealed significant decrease p < 0.001 and p < 0.05 in comparison with PBS group and PTX-PLGA-NPs (IV administered) respectively). The results might also be attributed to the restricted penetration of the drugs in the brain, when administered systemically due to the presence of BBB. To achieve the desirable concentration of the drug in the malignant tumor of the brain, high systemic dose would be required, which could result in severe cytotoxicity of the non-targeted organs. The use of targeting agent might facilitate the penetration of the drug in brain tumor. However, the alternative route of administration, IN could minimize the systemic toxicity, while significantly improving the drug delivery to the brain. The delivery through IN bypass the BBB and results in better accumulation of drug in brain and inhibition of tumor. The PTX-PLGA-NPs administered via IV was less effective than IN PTX-PLGA-NPs which might be because of the presence of BBB.

Anti- glioblastoma efficacy of PTX-PLGA-NPs post IV and IN administration.
Conclusion
The study focuses on the treatment efficacy of PTX-PLGA-NPs administered by IN in brain injury caused by the tumor. We have developed homogeneously distributed spherical PTX-PLGA-NPs using single emulsion method. The non-toxic PTX-PLGA-NPs were capable of releasing the drug in sustain manner. The IN delivery of PTX-PLGA-NPs revealed enhanced delivery of drug in tumor, when compared to IV delivered PTX-PLGA-NPs in which the concentration of the drug was significantly high in non-targeted organs and have limited drug penetration in CNS. The IN delivery of PTX-PLGA-NPs significantly inhibit the glioblastoma as it overcomes the limitation of systemic administration, which is hindered via BBB. We conclude that the IN delivery of the PTX-PLGA-NPs have significant potential in inhibition of brain tumor.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Foundation of the Program of Technology of Education Committee of Chongqing (No. KJQN201800122) and the Program of Technology of Science and Technology Committee of Yuzhong District in Chongqing (No. 20180131).
