Abstract
Objective:
A potential solution for islet transplantation and drug discovery vis-à-vis treating diabetes is the production of functional islets in a three-dimensional extracellular matrix. Although several scaffold materials have been reported as viable candidates, a clinically applicable one that is injectable and can maintain long-term functionality and survival of islet pancreatic beta-cells (β-cells) is far from being established.
Results:
In the current study, we evaluated a ready-to-use and injectable hydrogel’s impact on β-cells’ function and viability, both in vitro and in vivo. We found that β-cells in high concentration with hydrogels functionalized via Arg-Gly-Asp (RGD) demonstrated better viability and insulin secretory capacity in vitro. Moreover, it is a biocompatible hydrogel that can maintain β-cell proliferation and vascularization without stimulating inflammation after subcutaneous injection. Meanwhile, modifying the hydrogel with RGD can maintain β-cells’ secretion of insulin, regulating the blood glucose levels of mice with streptozotocin-induced diabetes.
Conclusions:
Thus, these preliminary results indicate that this RGD-modified hydrogel is a potential extracellular matrix for islet transplantation at extrahepatic sites, and they also provide a reference for future tissue engineering study.
Introduction
Islet transplantation is currently the only firmly established treatment for diabetes. However, the scarcity of human pancreas donors 1 and the short survival time of islet beta-cells (β-cells) after isolation not only limit islet transplantation in vivo, but also in vitro diabetes research.2,3 Islet damage and dysfunction begin with the breakdown of the extracellular matrix (ECM) during islet isolation;4–6 thus, many researchers have focused on attempting to build three-dimensional (3D) scaffolds to mimic the in vivo environment post-transplantation islets in order to enhance their functionality. 7 These 3D structures help to build cell-to-cell and cell-to-ECM interactions that are essential for cell differentiation, regeneration, and the prevention of apoptosis. 8 Although the normoglycemia and immunoprotective effects of encapsulation have been reported, islet survival times are still limited. 9 Many 3D scaffolds cannot be used in both in vitro and in vivo settings, meaning they cannot be used to conduct crossover studies, particularly in tissue engineering. 10 Furthermore, several researchers found that the 3D culture was inferior to a standard 2D tissue culture in terms of cell proliferation, causing a lack of mechanical strength, low-level adhesion of cells to the scaffold, and unintentional dissociation.11,12
To further promote cellular survival and function, different kinds of growth factors and functional biological ligands were composed in the scaffold to renovate cell-to-ECM connection. The essential connective molecules for islet function in the ECM are collagen and laminins.13–15 Collagen is selectively digested by collagenase, leading to actomyosin contraction, extensive fibrillogenesis, and uncontrolled cell migration.16,17 The most studied laminin is the Arg-Gly-Asp (RGD) peptide, which can also be found in many ECMs. Llacua found that a suitable concentration of RGD in alginate semi-permeable capsules can promote islet function in vitro. 4 Recently, Kim found that a short nanofiber polysaccharide soft hydrogel, VitroGel® 3D hydrogel (3D hydrogel), is more conducive to ovarian follicle nutrition and hormone exchange, morphology and oocyte maturation compared to 2D or alginate. 18 Furthermore, its body-temperature-induced coacervation properties made it suitable not only for in vitro study, but also for injection in vivo. 19 Although it is well behaved, to our knowledge, it must still be established whether it can support the survival and function of β-cells both in vitro and in vivo.
Thus, it is the first time to test the effect of VitroGel® 3D hydrogel on the β-TC6-cells which are widely used in diabetes drug discovery and mechanism research. In this study, we investigated the effects of different concentration of 3D hydrogel and clarify the effect of RGD modification on β-cells viability and functions. Their viability was measured by cell proliferation and survival ratio (in vitro) and tissue volume (in vivo). Their functions in blood glucose control and insulin secretion were also evaluated. These results will ultimately serve as a reference for the construction of 3D-β-cell-differentiation and -culture platforms, which will provide rigorously tested and functional models for future diabetes study about drug screening in vitro and the generation of functional islets in vivo, which will decrease reliance on cadaverous islets.
Materials and methods
Cell lines and cell culture
The mouse β islet cell line, β-TC-6, was supplied by FuHeng Cell Center (FH0387, Shanghai, China) and tested, shown to be free of mycoplasma and bacterial contamination. All cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) (Hyclone, Life Technologies, USA) with 20% FBS (10099-141, GIBCO, Australia), 0.1% 2-beta-mercaptoethanol (M3148, Sigma-Aldrich Corp, USA), and 1% penicillin/streptomycin (Biological Industries, Israel) and incubated in a 37°C CO2 incubator (ThermoFisher, USA). Half of the medium was changed every alternate day.
Extracellular matrix building
The VitroGel® 3D hydrogel (3D hydrogel) and RGD-VitroGel® 3D hydrogel (RGD-3D hydrogel) (TWG001/TWG002, The Well Bioscience Inc., NJ) were prepared according the VitroGel 3D Protocol Guidelines. 20 Briefly, the hydrogel was first diluted with deionized water at 1:0, 1:1, 1:2, or 1:3 (hydrogel: water, v/v) at room temperature. Then, the hydrogel dilutions were mixed with the complete growth media or cell suspensions at 4:1. Final cell concentration was 1 × 106 cells/mL. Next, a 100 μL mixture was gently added to the well of 96-well plate (TCPS, Costar) or a syringe. After 15 min stabilization at room temperature, a further 100 μL of complete growth medium was placed over the hydrogel or directly injected in vivo (Figure 1). Cells in the 2D culture were incubated under the same conditions. Microscope images of the cells were obtained with a Leica microscope (DMI3000, Leica, Germany).

Schematic of the cell coating with hydrogel and protocol for in vivo and in vitro study. In the control group, cells were cultured in 2D monolayers (2D group) or mixed with PBS for injection (control group). In 3D culture, cells were mixed with different concentrations of VitroGel® 3D (3D group) or RGD-VitroGel® 3D (RGD-3D group). Short nanofiber polysaccharides cross-linked to a network via ionic molecules in the cell culture medium within 15 min. The cells were then incorporated into the network. These cells were cultured in vitro or injected subcutaneously in the underarm for in vivo observation. RGD (green star), a short peptide of Arg-Gly-Asp.
Animal model and treatment
All mice (female C57BL/6, 8–12 weeks old) were purchased from SLAC Laboratory Animal Co., Ltd. (Shanghai, China). Our care and use of laboratory animals were according to stipulated guidelines and approved by the Ethics Committee of Xiamen Medical College (approval number: 201803070007). Diabetes was induced by a single intraperitoneal (IP) injection of streptozotocin (180 mg/kg, Sigma-Aldrich) according to a former protocol. 21 1 × 105 β-cells in 100 μl PBS (MeiLunBio, China), 3D hydrogel or RGD-3D hydrogel was subcutaneously injected under the arm. The mice were divided into three groups (control, 3D, and RGD-3D), with six mice per group (Figure 1). Glucose levels were followed up twice a week. β-cells were defined as functional if blood glucose levels measured less than 8.4 mM in two consecutive tests. The graft and serum of each mouse were harvested after the mice were sacrificed on Day 21. The size of the graft was measured by a Vernier scale.
Rheological analysis
To analyze the strength of the hydrogel, different ratios of hydrogel dilution were first prepared as 2.3 and incubated for 24 h in DMEM at 37°C in a CO2 incubator (ThermoFisher, USA). Then, the elastic modulus (G′) of the RGD-VitroGel® 3D was measured using a Kinexus DSR Rheometer (NETZSCH Ltd., Germany) on 20-mm parallels plate with a coarse surface at 0.1Hz at 37°C. The gap size was 500 μm and the control shear strain was 0.1%.
In vitro cell proliferation assay
Cell proliferation was detected with a Cell Counting Kit-8 (CCK-8, Dojindo, Japan) according to instructions. Briefly, after 3 d culture, 10μl CCK-8 was added to the medium. After a 1-h incubation, the OD values at 450 nm wavelength were measured via photometer (I2X, Molecular Devices, USA). The percentage of proliferation was calculated using following equation: proliferation ratio(%) = (ODt − ODb)/(OD2D − ODb) × 100%, where ODt, ODb, and OD2D denoted the OD values of the test, blank, and 2D culture samples, respectively.
Cell survival assay
Survival of the β-cells was assessed by trypan blue staining and flow cytometry (FCM). Cells were separated from the gel using VitroGel® cell recovery solution (MS03-100, TheWell Bioscience Inc., NJ) according to instructions. Cells were manually blind counted under the microscope after staining with 0.4% (w/v) trypan blue for 3 min; cells stained blue were counted as dead. The cells were then stained with an Annexin V-FITC/PI apoptosis detection kit (MA0220, MeiLunBio, China) and analyzed with a BD FACScan system (CytoFLEX LX, Beckman, Germany). The survival ratio is the percentage of Annexin V-FITC/PI double-negative cell.
Insulin secretion
β-cells were stimulated in DMEM containing 5.6 mM glucose (Hyclone Inc, USA) and DMEM supplemented with 16.7 mM glucose (Dalian Meilune Biotechnology Corporation, China) for 2 h successively. The culture supernatant was carefully collected separately. The in vitro glucose-stimulated insulin secretion (GSIS) and in-serum insulin secretion were measured using an insulin ELISA kit (rat/mouse insulin assay kit, Millipore, Billerica, MA) according to instructions. The stimulation index was calculated as the ratio of insulin released at high glucose to low glucose.
Intraperitoneal glucose tolerance test
The intraperitoneal glucose tolerance test (IPGTT) was performed on Day 14 after transplantation according to a former protocol. 21 Briefly, the mice were made to fast overnight and intraperitoneally injected with glucose 1 mg/g body weight (Dalian Meilun Biotechnology Corporation, China). The blood glucose levels were measured at 0, 15, 30, 60, and 120 min after injection.
Histopathology
Two-hundred microlitre hydrogel was injected subcutaneously. The area of skin injected with hydrogel was harvested and observed on Days 7, 14 and, 21 after injection. The hydrogel was fixed in 4% paraformaldehyde, embedded in paraffin, cut into 5 μm sections, and stained with hematoxylin-eosin. Images were captured by microscopy (Motic BA310; Motic Co. Ltd., Hong Kong, China). All results were evaluated blinded.
Statistical analysis
The data were analyzed using GraphPad Prism software (Inc., San Diego, CA). Data were expressed as mean ± SD. Variance was analyzed by one-way analysis. When multiple comparisons were made during the analyses, a Bonferroni correction was applied. Errors were SD of averaged results. p < 0.05 was considered a statistically significant difference (*), while p < 0.01(**) and p < 0.001 (***) indicated highly significant differences.
Results
The RGD-modified 3D hydrogel without dilution facilitate β-cell proliferation
The physical property and the chemical component of the ECM is very important for cell proliferation. According the instruction, different cell type prefer different strength of the hydrogel which can be adjust by dilution. To clarify which concentration of the hydrogel benefited β islet cell line growth, we diluted the hydrogel with deionized water at 1:0, 1:1, 1:2, or 1:3 (hydrogel: water, v/v). Similar as the instruction of the hydrogel, rheological analysis shows that the strength of the hydrogel could be adjusted from 10–4000 Pa of G’ with the concentration increases (Figure 2(a)). Then, we measured cell proliferation in different concentrations of hydrogel (Figure 2(b)). Results showed that the cell proliferation ratio in 3D hydrogel without dilution (1:0) was significantly higher than those of other groups (p < 0.001). This finding was the same in the 1:1 and 1:2 dilution groups, but higher than the 1:3 dilution group (p < 0.05). This trend was the same in the RGD-3D hydrogel. Overall, cell proliferation was significantly higher in the RGD-3D hydrogel than in the 3D hydrogel. A similar result was observed under the microscope (Figure 2(c)). On Day 1 after inoculation, cells adhered to the culture plates in the 2D culture, and cells maintained suspension in the 3D hydrogel and RGD-3D hydrogel. Three days later, cells covered 80% to 90% of the plate bottom in the 2D system but formed clusters in the 3D culture system. These clusters were larger and more numerous after RGD modification. Thus, the RGD-3D hydrogel without dilution is suitable for β-cell proliferation.
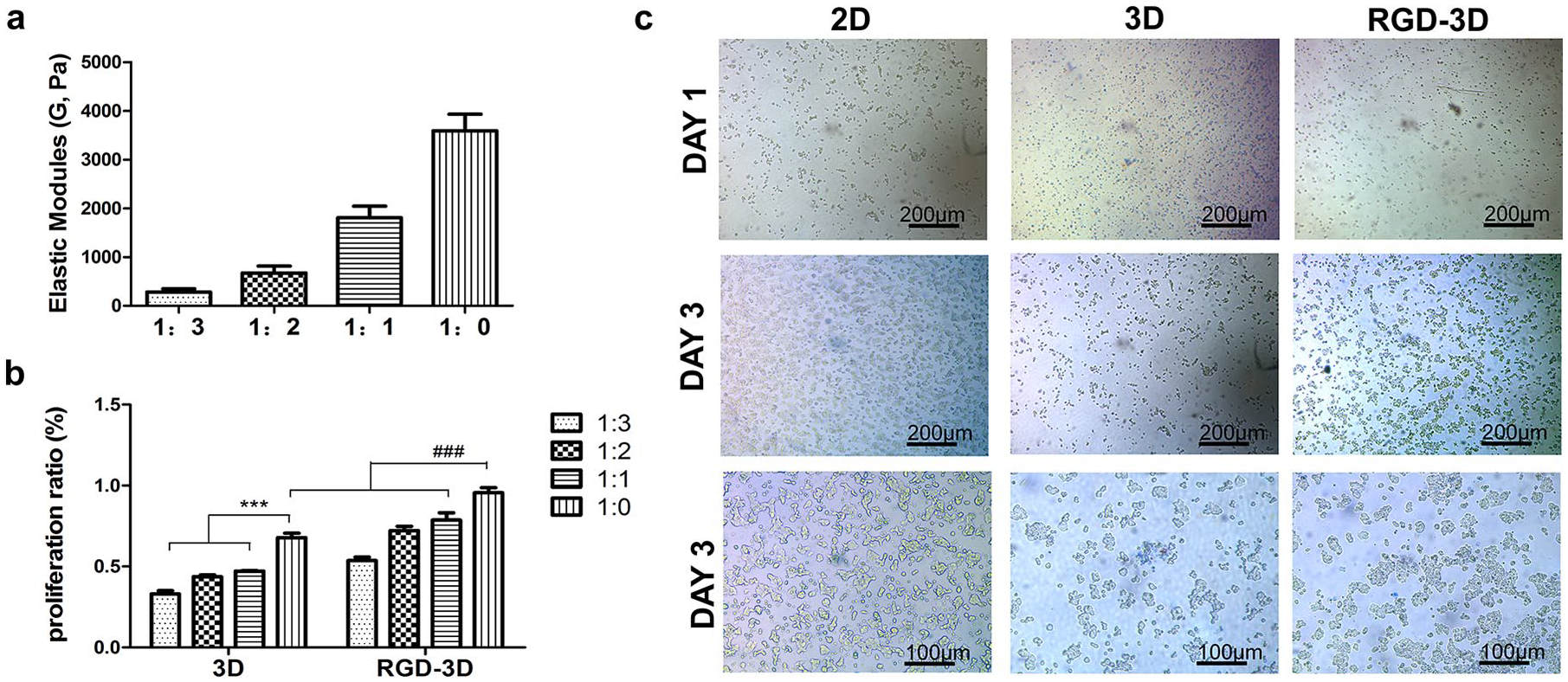
The effect of the hydrogel on β-cell proliferation. (a) The VitroGel elastic modules of the hydrogel in different concentrations. (b) The proliferation ratio of β-cells in different concentrations of hydrogels. (c) Cellular morphology in 2D or 3D hydrogel without dilution on Day 1 or 3 after inoculation.
RGD-modified hydrogel maintains β-cells viability
β-cells were dissociated from the hydrogel after 3 d culture to further demonstrate the effect of hydrogel on cell viability. Results showed that 3D-hydrogel incubation significantly lowered β-cells’ proliferation ability, while RGD-3D hydrogel maintained it (Figure 3(a)).

The effect of the hydrogel on β-cell survival. (a) Cell proliferation was detected using a CCK8 analysis kit. Triplicate well for each sample. (b) Cells on counting chamber by microscope after trypan blue staining. Surviving cells were not dyed. Scale bar, 50 μm. The total and live cells were counted and the death rate was calculated from four counting fields for each sample. (c) The histogram. (d) The dot-plot profiles of the percentage of surviving cells (quadrant Q4, Annexin V-FITC/PI double-negative cell) by FCM after Annexin V and PI staining.
The prevention of cell apoptosis was a possible mechanism for RGD-3D hydrogel’s support of cell proliferation. Therefore, we further examined the effect of the hydrogel on cell apoptosis. Observations from the trypan blue assay (Figure 3(b)) show that, without RGD (3D group), the total amount and percentage of cells exclusively dyed by trypan blue were lower than the control group and the RGD-3D group. Meanwhile, the FCM assay, after staining with Annexin-V-FITC/PI (Figure 3(c) and (d)) also shows that the rate of survival for cells in the RGD-3D group (91.13% ± 7.82%) is significantly higher than of those in the 3D group (68.68% ± 9.07%), and was similar with the control group (93.90% ± 6.35%). Thus, RGD is essential for maintaining β-cell viability by preventing cell apoptosis in this 3D culture system.
VitroGel® is a biocompatible scaffold
To evaluate the biocompatible of the hydrogel, 3D hydrogel and RGD-3D hydrogel was subcutaneously injected under the left and right arm area respectively (Figure 4(b)). After injection, hydrogel could be observed under the skin on Day 7 but was gradually diminished on Days 14 and 21 (Figure 4(a)). Pathological analysis showed that there were no lymphocytes infiltration in both 3D hydrogel and RGD-3D hydrogel. The fibrocytes migrate into the hydrogel and blood capillaries generate and pass through the hydrogel gradually. That process was more rapid and obvious in RGD -3D hydrogel (Figure 4(a)). Thus, both 3D hydrogel and RGD-3D hydrogel are biocompatible and RGD seems benefit tissue regeneration and revascularization.
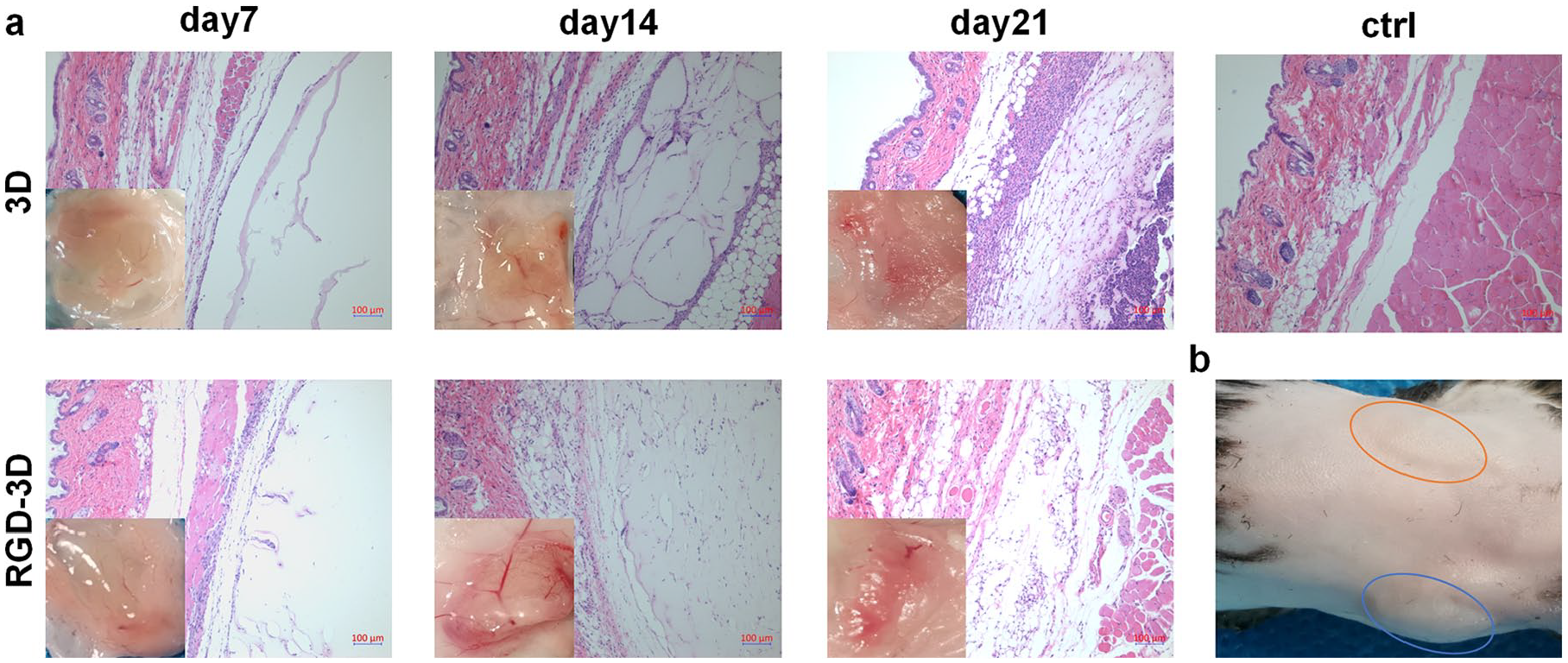
Biocompatibility of the hydrogel in vivo. (a) Pathological analysis of the skin by H&E staining on Days 7, 14 and 21 after subcutaneous hydrogel injection. Scale bar, 100 μm. (b) Representative images of subcutaneous injection location of 3D hydrogel (left, orange circle) and RGD-3D hydrogel (right, blue circle) on day 7. n = 3/group.
RGD-3D can promote β-cell growth and function in vivo
To further evaluate the effect of hydrogel on cell proliferation in vivo, β-cells in hydrogel were injected subcutaneously. Grafts were harvested and measured 21 days later. The volume of tissue development in the 3D group was dramatically restricted compared to the control group, while RGD modification maintained tumor formation compared to the control group (Figure 5(a) and (b)). Thus, RGD is important for β-cells growth in vivo.

Effect of the hydrogel on β-cell in vivo. β-cells in PBS (circle = control group), 3D hydrogel (square = 3D group) or RGD-3D hydrogel (regular triangle = RGD-3D group) were subcutaneously injected into mice with STZ-induced diabetes. (a) Tumor size on Day 21 after cell injection. (b) Histogram of tissue volume. (c)The blood glucose. (d) Body weight. (e) Insulin release level of the recipient. (f) Blood glucose levels curve. (g) Area under the curve (AUC) of IPGTT. (h) Stimulation indices in GSIS.
After β-cell injection, blood glucose levels gradually decreased in all groups (Figure 5(c)). Blood sugar in the RGD-3D group became normal after two weeks with no significant difference from the control. However, the 3D group maintained a high level. With the improvement of diabetes, body weight in the control and RGD-3D groups increased normally after injection without a significant difference between them (Figure 5(d)), which demonstrates the hydrogel’s non-toxicity. However, in the 3D group, bodyweight was maintained at a low level, as was the persistently high blood glucose level.
Additionally, β-cells in RGD-3D hydrogel maintained insulin secretion (Figure 5(e)) and they effectively regulated blood glucose levels during IPGTT (Figure 5(f) and (g)). Their insulin secretion was also sensitively adjusted by blood glucose stimulation (Figure 5(h)). However, this ability of the β-cells in the 3D group was relatively weaker. Overall, RGD was essential to maintaining the function of β-cells in the 3D hydrogel.
Discussion
Although the 2D culture can support β-cells growth, 22 it couldn’t maintain a natural space relationship with different cells in tissue and mimic ECM for tissue engineering study. However, different types of cells might prefer different cultural environments, and inappropriate 3D scaffolds couldn’t support cell growth well, sometimes even worse than 2D culture. Initially, Eric Simon found that electrospun polycarbonate scaffolds were inferior to standard 2D tissue culture, as it lacked mechanical strength. 12 Then, other researchers found short of growth factors and functional biological ligands can cause a low level of adhesion which is essential for some kinds of cells.13–15 Thus, many researchers still on their way to find and continually optimize the artificial substitute material for 3D culture. Herein, we aim to investigate which 3D hydrogel can support the survival and function of β-cells both in vitro and in vivo.
Given the rigid mechanical properties of scaffolds were essential for cell growth, 12 we adjust the strength of the hydrogel by different dilutions. Our data show that β-TC-6 cells preferred to grow in the 1:0 diluted hydrogel, which is even stiffer (Figure 2). Also, the cell proliferation ratio in RGD-3D hydrogel without dilution closely resembled that in the 2D-culture system. Thus, we surmise that RGD-3D hydrogel without dilution is an ideal environment for β-TC-6 cell proliferation.
Given that RGD is beneficial for cell growth, we suspected that it would also be essential for cell survival and function in this hydrogel. Our data suggest that RGD can dramatically increase the survival rate of β-TC-6 by preventing cell apoptosis (Figure 3(c) and (d)). Cells can also be easily harvested from RGD-3D hydrogel by centrifuge without needing enzymes to maintain viability (Figure 3(a)). This characteristic of the hydrogel is critical for tissue engineering. It can also facilitate the insulin secretion of β-TC-6 cells in GSIS in vitro (Figure 5(h)). This effect of RGD is consistent with Llacua’s findings in alginate-encapsulated human islets. 4 These beneficial effects are considered to be derived from indirect matrix restoration, as most of the β-cells were located inside of the islet and could not contact it. Here, in the β-cell injection model, we found that direct contact was especially important, which is also supported by the fact that RGD can interact with integrin in bone marrow cells 23 via the integrin–Akt pathway and facilitate cell adhesion and proliferation. Signal cascades were also involved in β-cell survival and insulin secretion.24,25 Overall, our findings suggest that RGD-3D hydrogel promotes β-cell survival and function in vitro.
Considering that the materials might have behaved differently in vivo and in vitro, 10 we further explored 3D hydrogel’s biocompatibility in vivo. We did not detect any toxic effects, as no bodyweight difference was observed after RGD-3D hydrogel injection as compared to the control group with a similar blood glucose level (Figure 5(c) and (d)). The hydrogel was observed after one week and absorbed gradually within three weeks (Figure 4(a)). This degradative property ensures temporary support for β-cells before new cell–cell and cell–ECM connections are made. 7 Dense capillaries are also important to pancreatic β-cells’ survival and functionality. 26 The short of these vessels hinders subcutaneous transplantation, however convenient it may be.27–29 We thus chose the underarm subcutaneous area, with its relative abundance of blood vessels, as the cell injection site. The results of H&E staining showed that both 3D and RGD-3D hydrogel was non-immunogenic, and a property of soft hydrogel was its cell and blood capillary permeability (Figure 4(a)). RGD-3D hydrogel showed better performance in tissue regeneration and revascularization. Consistent with Antonova’s discovery, RGD modification can improve the biocompatibility of small-diameter vascular grafts. 30 However, vascular density was still minimal. Further modification is necessary.
Although biocompatibility of the hydrogel has been observed, we were still curious as to whether the hydrogel would be effective in vivo. Consistent with the result in vitro, RGD is very important for β-cell growth and survival in vivo. As a classical model cell line, β-TC-6 cells not only can growth in 2D culture, but also have excellent tumorigenesis ability in vivo. Meanwhile, the effect of RGD-3D hydrogel is comparable to the control group. The reason might include RGD can help cell adherence, and do not hinder tissue regeneration and revascularization. Similar character of RGD was also found in tumor formation and migration. Lee et al. also noted a similar effect of RGD in an elastin-like polypeptide scaffold. 31 However, without RGD, 3D hydrogel couldn’t sustain the cell growth. In vitro, we found 3D gel can cause β-cell apoptosis and bad for the of cell masses formation (Figure 3). In vivo, the regeneration and revascularization ability seems weaker without RGD. A possible explanation is β-cells are adherent cells, while it might maintain suspension in the hole of 3D gel that will cause the cell short of growth and survival signal and short of nutrition which will impede cell growth and lead cell death.
The RGD-3D hydrogel not only support the cells’ growth, but also maintain their function. β-cells in hydrogel (RGD-3D group) can secrete a comparable amount of insulin to the control group (Figure 5(e)). However, without RGD (3D group), the small volume of the tissue limited its effect (Figure 5(a) and (b)), and caused uncontrollably high blood glucose (Figure 5(c)), low insulin secretion (Figure 5(e)), and slow bodyweight gain (Figure 5(c)). The IPGTT further confirms the effect of RGD in 3D hydrogel on the maintenance of β-cells’ function in blood glucose perception and regulation (Figure 5(f) and (g)). In conclusion, RGD-modified 3D hydrogel promotes β-cells’ function, likely via the prevention of cell apoptosis.
Conclusion
In summation, this is the first systematic evaluation of the effects of VitroGel® 3D hydrogel on β-cells. All results emphasize the role of RGD in β-cells’ survival and function, as well as the role it plays in providing a viable scaffold for β-cells vis-à-vis study in vitro and transplantation in vivo. Further evaluation and continuous optimization of its effects on islet and β-cell differentiation should be included in future efforts. Overall, RGD-VitroGel® 3D hydrogel is a promising platform for preclinical drug discovery and cell-based subcutaneous injection for treating diabetes.
Footnotes
Acknowledgements
We would like to thank Xiamen Medical College for supporting this study. Thanks Chang Gao, Junjie Xia and Wenhao Wang for their technical guidance.
Contributorship
Tianshu Lan researched literature, conceived the study, and wrote the first draft of the manuscript. Jingyi Guo, Xiaoming Bai and Zhimin Wei involved in protocol development, cell culture and data analysis. Zengjiong Huang, Guicheng Du, Guoliang Yan and Lebin Weng involved in Animal handling and feeding. Xue Yi was involved in gaining ethical approval and pathological analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by PhD start-up fund of Xiamen Medical College (Grant. 20170222), Key laboratory of functional and clinical translational medicine, Fujian province university (Grant. JNYLC1810) and Provincial Student Innovation and Entrepreneurship Program of Local Universities in 2018 (Grant. 201812631008).
Guarantor
Tianshu Lan.
