Abstract
A novel graphene oxide (GO)-based carrier was fabricated for the controlled release of Benazepril (BENA). Freeze dried samples of GO–BENA carrier were prepared for controlled drug release at different pHs (pH = 2, 7, and 10) and release kinetics indicate BENA desorption from GO is by Fickian diffusion. The BENA yield from the carrier amounted to ~55% of the adsorbed material in a strongly acidic medium after 50 h. Binding fractions of BENA to 10 mg/L GO was determined for different solution concentrations of the drug. In vitro assays of cell proliferation (WST-1 kit), cell structural integrity (LDH kit) and flow cytometric indicators of necrosis in three different cell lines (CACO-2, SGC-7901, and primary mouse hepatic fibroblast) all demonstrated that the GO carrier had a good biocompatibility. The pH-dependent release sensitivity of the GO-based carrier suggests that it is a potential candidate for use in the controlled release of drugs in the acidic environment of the stomach.
Introduction
Nanomaterial-based carriers have been studied as transport vehicles for various materials, such as drugs and genes, due to their capacity to increase local access to cellular targets and enhance their distribution and availability.1–4 One important consideration in designing such carriers is to select an appropriate nanoparticle (NP) carrier that will attach to the transported substance, then retain and slowly release the target substance at a cellular locus where its effectiveness is optimal. 5
BENA is a type of broad-spectrum antihypertension medication that, after cleavage of an ester group in the liver, becomes an angiotensin-converting enzyme (ACE) inhibitor. However, due to poor water solubility, BENA has poor absorption and cellular uptake. Only 37% of the physiological dose is found to be transported systemically, as most of this is consumed by conversion or excretion in vivo. Due to its short retention and release times, BENA requires repeated dosing. This inconvenience plays a critical role in the clinical application of the drug. As a result, the development of new sustained-release carriers could offer important benefits in increasing the utilization rate of BENA and reducing the required frequency of drug administration.
Graphene oxide (GO) is a remarkable modification of graphene. Graphene oxide comprises monomolecular sheets of oxidized graphene, having high specific surface area that is decorated with disordered hydroxyl groups, carboxyl groups, and epoxide bridges on its surface and edges, which greatly enhance its water-solubility and biocompatibility. The skeleton of benzene rings offers large numbers of potential π–π binding sites for aromatic drugs. In addition, surface oxygen atoms are available to form hydrogen bonds with H donors on the drug. This combination contributes to making GO a potential drug carrier with promising applications. GO can be used directly for drug transport and extended release without requiring any chemical modification.6–9 Liu et al. have investigated the low-molecular-weight heparin (LHT7) and DOX loaded with GO. 10 Bramini et al. have constructed an engineer multifunctional nanoparticles (NPs) which was able to cross the blood–brain barrier to reach neural cells. 11 Moreover, covalently and non-covalently functionalized GO has enhanced water solubility and has shown huge potential as a delivery system for countless medicines.12–15 Yang et al. and Li et al. make use of a simple and effective chemical precipitation method to get a superparamagnetic GO–Fe3O4 nanohybrid.16,17 Ma et al. have synthesized a multifunctional superparamagnetic graphene oxide–iron oxide hybrid nanocomposite. 18 Additionally, some associated delivery systems based on multi-functionalized GO and superparamagnetic iron/cobalt oxide nanoparticles have been demonstrated for use in magnetic targeting. 19 According to several studies, GO remains unstable in terms of biological affinity. However, GO showed good biocompatibility under certain conditions.20,21 Andreeva et al. have synthesized the graphene oxide/polysaccharide and proved the biocompatibility of the complex. 22 di Luca et al. have fabricated one kind of electro-responsive graphene oxide hydrogels for skin bandages and proved that was biosecurity. 23 Santos et al. have built the nanographene oxide linked to porphyrins and evaluated the evaluation. 24 Other studies have also shown that GO has good biocompatibility.25,26
We have formulated BENA as a novel drug sustained-release system by loading the ACE inhibitor into GO nanoparticles while in a colloidal suspension. In order to demonstrate the good biocompatibility of GO as drug carrier under the experimental conditions, we examined the cell structural integrity, characteristics of cell proliferation, and apoptosis/necrosis.
Methods
Fabrication of the GO–BENA carrier
In a typical experiment, separate GO and BENA concentrations were prepared in ultrapure water, then mixed in equal volumes and slowly shaken overnight to form a GO–BENA carrier complex. The mixed solutions were centrifuged at 8000 rpm for 30 min to remove free BENA and purify the bound complexes by sedimentation. Solvents were removed by freeze-drying. The effect of different ratios of starting reagents on the composition of the final mixtures was studied by varying the initial concentration of BENA from 5 to 50 mg/L (the saturation concentration is 30 mg/L), while maintaining the carrier concentration at 10 mg/L. The GO–BENA composites were then sedimented, freeze-dried (Thermo Heto PowerDry LL3000, USA) and stored in vacuum until use.
Release of BENA
The GO–BENA carrier complex was dried using the freeze-drying method and the drug release kinetics in neutral water (pH = 7), ionized HCl (pH = 2), and NaOH (pH = 10) solutions that were carried out with composite samples. A final drug concentration of 30 mg/L was achieved by adding the drug–carrier mixtures to 10 mL of water, HCl solution, or NaOH solutions in plastic tubes. The solutions were left undisturbed in the dark for varying elapsed times and then spectral absorbances were measured. The amount of released GO–BENA was determined by UV absorbance. The wavelength used for BENA determination is 240 nm. The cumulative amount of released drug as a percentage of the initial concentration was calculated from a standard curve. The release profiles were obtained based on the results of three parallel experiments at each pH.
Test of biocompatibility
The cells were seeded in 96-well flat-bottomed microtiter plates (5 × 103 cells in 200 µL per well) for 36 h and treated with different concentrations of GO, GO–BENA and distilled water (Laboratory homemade). The percentage of living cells was determined using the Lactate Dehydrogenase (LDH) Cytotoxicity Assay Kit and WST-1 Cell proliferation and cytotoxicity assay kit (Beyotime Institute of Biotechnology, Jiangsu, China). Assays were performed in accordance with the manufacturer’s instructions. The absorbance at 450 nm was monitored based on the absorbance maximum of the formazan dye and the reference wavelength was set at 630 nm. The data was measured using an automated microplate reader (Bio-Rad 5 Model 550, Bio-Rad, Hercules, CA, USA). Cytotoxicity percentage(LDH kit) was determined by the equation: Cytotoxicity percentage(%)=(Absorbance of treated samples − Absorbance of control group)/(Maximum enzyme activity absorbance − Absorbance of control group). Cell viability percentage (WST-1 kit) was determined by the mean optical density (OD) of one experimental group divided by the mean OD of the untreated group × 100%.
Flow cytometry
The cells suspensions of the colon adenocarcinoma line were harvested for flow cytometric analysis (FCM). Cells were cultured in 6-well plates with 2 × 105 cells per well and incubated with different concentrations of experiment material for 36 h to allow for cell uptake. The following experimental materials were used for FCM: GO, GO–BENA, and distilled water. Apoptotic cell identification was achieved using the Annexin V-FITC apoptosis detection kit (C1063, Beyotime, China). Flow cytometry data were collected with a BD LSRFortessa cell analyzer (BD Biosciences, Franklin Lakes, NJ, USA) and analyzed by FlowJo software (Tree Star, Ashland, OR, USA). Each experiment was repeated three times.
Results
Characterization of BENA–GO conjugates
The GO–BENA carrier was successfully constructed. The characterization of GO–BENA conjugates showed that the BENA was successfully loaded onto GO (Figures 1–3). Figure 1(a) shows the relationship between the combination fraction for each added amount of drug. As shown in Figure 1(b), the major peak of GO occurs at ~11.78° and the major peak of GO–BENA is ~6.56°. These peaks correspond to major layer spacing of 3.77 and 6.74 Å before and after loading, respectively. Direct evidence for BENA loading on GO appears in its infrared spectra (Figure 1(c)). Figure 1(d) shows the Raman spectra of GO, BENA, and GO–BENA. Figure 2 shows the XPS data recorded for GO, GO–BENA, and BENA with a focus on the O, C, and N-characteristic regions. Sharp peaks located at around 284 and 289 eV correspond to C–C in aromatic rings and C–OH, C=O, O=C–OH in hydroxyl and epoxy groups, respectively. 27 Evidence of such a complex formation is shown in Figure 3(a)–(h), that is, to be contrasted with the morphology of different imaging material, showing the GO and the GO–BENA microstructure.

(a) GO loaded with BENA binding curve. (b) XRD spectra of GO and GO–BENA. (c) Infrared spectra. (d) Raman spectra of lyophilized GO and GO–BENA.
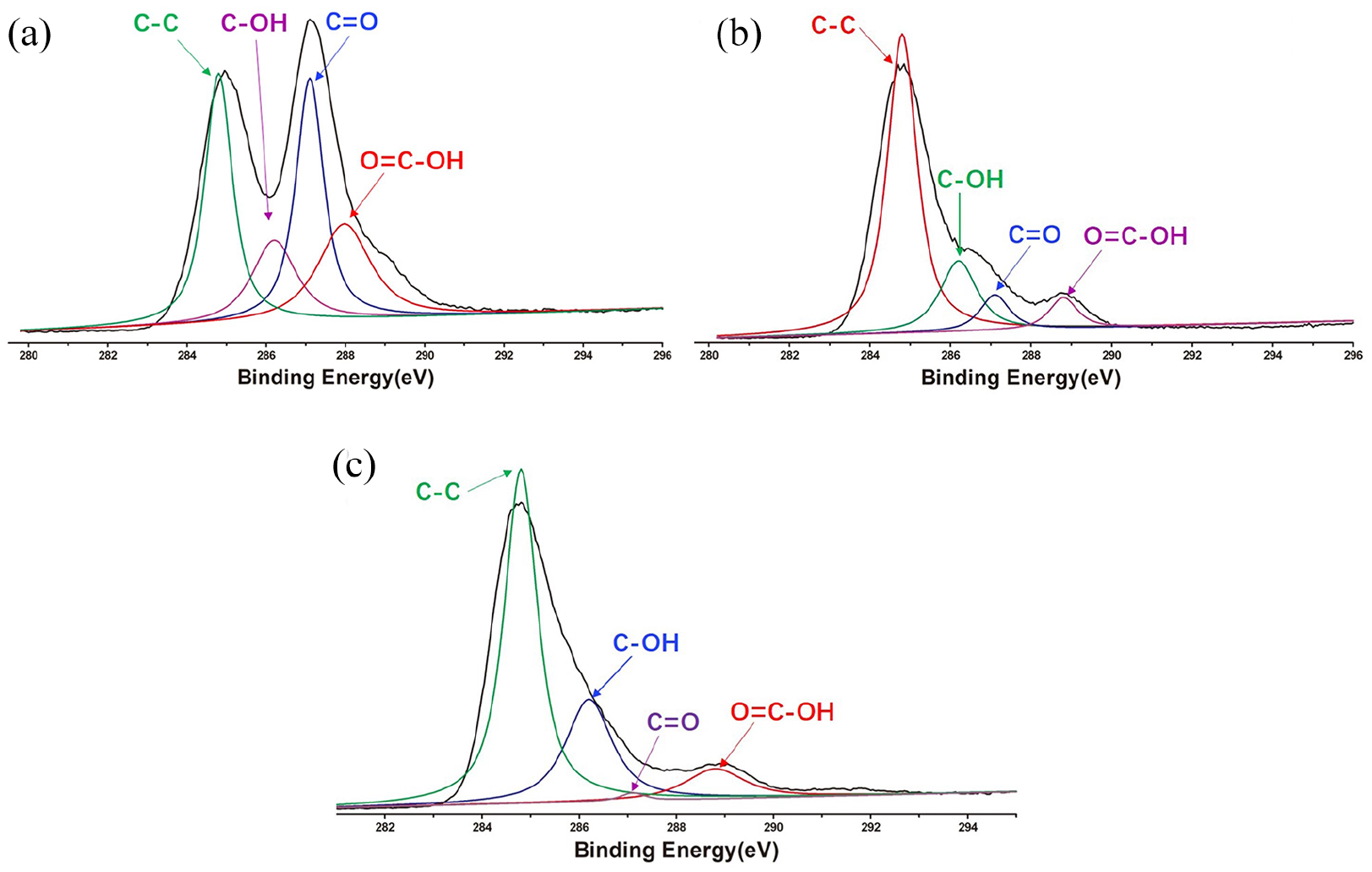
XPS spectra of GO (a), GO–BENA (b), and BENA (c).

TEM analysis of GO (a) and GO–BENA (b) Scale bar: 0.2 μm. SEM images of lyophilized GO (c) and GO–BENA (d–f). Scale bar: 1 μm. AFM images of GO (g) and GO–BENA (h).
Drug release process
Figure 4 shows the release kinetics of drug-loaded complexes under dialysis at different pH values. The first 10 h represents the rapid release phase, accounting for 65% to 75% of the total release yield. Then, the release of the drug gradually slows down until it reaches a plateau in about 24 h. The time course of BENA release under different pH environments is similar and varies only with respect to the plateau levels reached.

Yields of drug released from freeze-dried carrier at different pH values. The data points are averages of three parallel experiments; error bars represent the standard deviations.
Test of biocompatibility
To examine the effects of GO and the GO–BENA on cell structural damage, we challenged in vitro cultures of different kinds of cells (CACO-2, SGC-7901, and primary mouse hepatic fibroblast) with varying dosages of the GO and to the GO–BENA. The different kit (WST-1 kit, LDH kit, and Flow cytometry) was used to test their effects on cytotoxicity. As shown in Figure 5 and Supplemental Figure S3, the incubation of GO at different concentrations of carrier alone and carrier plus cargo produced graded changes in cell proliferation, as compared with the control group. Both GO and GO–BENA showed good biocompatibility.

Effects of different concentrations (1, 5, 10, 20, and 50 mg/L) of GO and GO–BENA on different kinds of cells (a, CACO-2, b, SGC-7901, and c, primary mouse hepatic fibroblast) proliferation (WST-1) and structural integrity (LDH kit). Deionized water (DIW) was used for the control group.
Discussion
Preparation and characterization of GO–BENA carrier
As shown in Figure 1(a), when the concentration of BENA increased from the initial 5 mg/L to 25 mg/L, the drug loading increased dramatically from 0.09 to 1.13 mg/mg. This indicates that when the concentration of BENA was at a low level, the drug loading of BENA on GO increased significantly with an increase in BENA concentration. As previously stated, GO possesses many surface sites for BENA binding. As the free concentration of BENA increases, the available ligands on the surface of GO gradually diminish. As a result, the drug loading gradually decreases with increasing concentration of BENA in the range between 25 and 30 mg/L. When free BENA concentration increases from 30 to 50 mg/L, drug loading increased only from 1.38 to 1.40 mg/mg. Therefore, when the concentration of GO is maintained at 10 mg/L, the effective saturated load capacity of BENA is 1.38 mg/mg GO. Accordingly, we used this concentration ratio in all formulations in which BENA and GO solutions were mixed, centrifuged, and freeze-dried for further characterization.
The result of XRD suggests that the addition of BENA increased the distance between the major crystallographic planes of the composite by ~3 Å and also demonstrates that the loading position of BENA is located on the GO surface. As shown of the infrared spectra results, the signature shift confirms that the superposition of GO and BENA signals were not simply due to the existence of a physical mixture. The complex structure is made possible by formation of hydrogen bond interactions. Raman spectroscopy is an effective measure to take for detection of the regions in carbonaceous structures, therefore make us to comment on the skeletal changes of the GO and GO–BENA. The GO lattice exhibits a G peak at 1588 per cm, which is generally assigned to C=C stretching vibrations. Because of the vigorous oxidation of GO, the D peak appears at 1366 per cm. When BENA was attached to GO, the D peak and G peak were moved to 1358 and 1599 per cm, respectively. Besides, the increase of ID/IG was demonstrated that the BENA was successful attached to GO. Furthermore, the results of Zeta potential (Supplemental Figure S2) have also proved the GO was loaded with BENA.
The specific types and contents of various functional groups on the surface of three samples(GO, BENA, and GO–BENA) can be obtained by integrating each fitting peak. The carbonyl group of GO (C=O) is the dominant group in its functional group, followed by the alcohol hydroxyl group and carboxyl group. The abundant oxygen-containing groups in GO make it have good hydrophilicity and extensive reactivity, which is the basic condition for its application in drug delivery. According to the changes of various oxygen-containing groups in the three samples, we can conclude that GO and BENA combine to form the drug-carrying complex.
The existence of multiple –OH and –COOH functionalities on GO suggest the possibility of forming hydrogen bonds with the –NH and –COOH groups on BENA, strengthening the complex structure and enhancing its rigidity. Although the hydrogen bond is rather weak, it can be further weakened under changes of pH. This suggests the possibility of selective stripping of BENA from the GO surface. This possibility becomes the basis of utilizing GO as a carrier that is intrinsically equipped to enhance drug release under pH change.
According to the spatial conformation of BENA (Supplemental Figure S1), its two benzene rings should be located on different planes. Allowing for the strong π–π interaction of GO and BENA, it should therefore not be sterically possible for the two benzene rings of the same BENA molecule to be adsorbed on the same graphene oxide sheet. Therefore, models of stacking infer that the rings should contact GO monolayers localized on different planes. The more twisted and rougher microstructures in Figure 3(d)–(f), as compared with the smoother and less undulating microstructures in Figure 3(c), suggest that interactions between each of the two benzene rings of BENA and the GO surfaces distort the planarity of GO further from its original smoothness. The transmission electron microcopy (TEM) images (Figure 3(a)) show that the nanosheet was smooth and no other impurities. However, the Figure 3(b) indicates that there are smaller nanocrystals attached to form nanosheet. Atomic force microscopy (AFM) was used to image the micromorphology of the carrier complex (Figure 3(g) and (h)). As shown in Figure 3(g), the GO monolayer appears as a smooth surface, and in Figure 3(h), several BENA particles appear in the GO surface.
Controlled release of GO–BENA
The curves in Figure 4 demonstrate that release slows from 10 to 24 h, and after 24 h is basically complete. BENA releases the greatest yield in the pH = 2 environment, up to 58% of the total adsorbed drug. In the pH = 7 environment, the carrier releases 42% of its original bound cargo, and only 28% in the pH = 10 environment. The carrier functions as a better drug release agent under acidic conditions. In the human body, acidity of gastric fluid is approximately pH = 2. Under such conditions, the release yield is maximal and continuous for more than 24 h.
Based on the above results, in a neutral environment, –OH and –COOH in GO are capable of forming four types of hydrogen bonds with the –COOH and –NH– functionalities in BENA. In an acidic environment, –NH– forms (–NH2–)+ with H+ in solution, leaving only one hydrogen bonding group (–COOH) per mole of BENA. This reduces the possibilities hydrogen bonding to only two groups per drug molecule. Considering that free H+ in the solvent also competes for the bonding group, further reducing the number of potential hydrogen bonds, it is easy to rationalize arguments that the effective hydrogen bonding holding the complex together is significantly weaker under acidic conditions than under a neutral pH. Upon rapid lowering of pH, BENA proceeds to peel off from the surface of GO, thus achieving environmentally controlled cargo release. In alkaline environments, –COOH exists as –COO−, so only –OH in GO can form hydrogen bonds. Although BENA loses one bonding group, the steric rigidity of BENA is reduced due to the hydrolysis of the ester bond, enabling greater flexibility of motion for the benzene rings that increases the likelihood of creating additional π–π bonds. Under alkaline conditions, Figure 4 shows that GO binds even more tightly with BENA, consequently releasing even less of the drug. In an alkaline environment, drug release is poor, presumably due to the presence of π–π conjugation holding the complex together.
To better understand the mechanism of BENA release in GO, we fitted the cumulative release data to the following empirical equation:

In this formula,
Effects of GO and GO–BENA on cell proliferation and cell structural integrity
The WST-1 assay can measure the reduction of WST-1 dye depending upon the activity of mitochondrial dehydrogenase enzyme. Therefore, the experiment could show whether the substance caused damage to the structure of the cell membrane. As the concentrations of GO increased, cell proliferation became more inhibited, in a dose-dependent manner. Different types of cells have the same trend. When the carrier concentration reached 50 mg/L, cell proliferation became further inhibited. However, at the optimal release concentration, the compound of GO and BENA had only a minor effect on Caco-2 cell proliferation. As the result of the test it is proved that GO had no significant effect on cell proliferation before or after binding to BENA. To test the effects of GO and GO–BENA on the structural integrity of cells, we assayed for LDH leakage across the plasma membrane. With increasing GO concentrations, the LDH leakage across cells increased. Different types of cells have the same trend. As shown in Figure 5, when the GO concentration was raised to 5 mg/L, membrane damage grew significantly. When GO concentration reached 50 mg/L, membrane destruction further intensified. Nevertheless, the negative effects of GO and drug load on cell membrane integrity remained within a small range. Furthermore, according to the result (Supplemental Figure S3), treatment with GO at different concentrations and GO–BENA only slightly increased the number of advanced apoptotic/necrotic cells, while it showed no impact on early apoptotic cells.
Conclusion
In summary, we have fabricated a novel graphene oxide-based carrier with drug molecules. BENA hydrochloride was utilized as a model drug and was added to GO to form a compound by supramolecular fabrication. The resulting compound had pH-sensitive performance and the samples had different drug release profiles in neutral, alkaline and acidic media. The samples were found to be capable of controlled release of drugs in the strongly acidic environment of the stomach. Meanwhile, the drug carrier in this paper had good biological compatibility. We have successfully obtained pH-sensitive graphene oxide–BENA supramolecular carriers as biosafety controlled release system. We supposed that this biosafety controlled drug release system of GO-based carriers could be appropriate for many other hydrophilic drugs with N-containing and/or O-containing functional groups, such as nucleosides, peptides, and so on.
Supplemental Material
sj-pdf-1-jbf-10.1177_2280800020963471 – Supplemental material for Fabrication of pH-sensitive graphene oxide–Benazepril carrier as biosafety controlled release systems
Supplemental material, sj-pdf-1-jbf-10.1177_2280800020963471 for Fabrication of pH-sensitive graphene oxide–Benazepril carrier as biosafety controlled release systems by Tianxiong Liu, Yujiao Li, Xianzhe Chen, Xiaoming Zhao, Jianfang Wang and Dongyi Zhang in Journal of Applied Biomaterials & Functional Materials
Footnotes
Acknowledgements
The authors would like to thank college students’ scientific research platform of Xiangya Medical College, Central South University for their generous help in flow cytometry analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (61376125).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
